Panamax
Brand Information
| Brand name | Panamax |
| Active ingredient | Paracetamol |
| Schedule | S2 | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Panamax.
Summary CMI
Panamax®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Panamax?
Panamax contains the active ingredient paracetamol. Panamax is used to relieve pain and fever in adults (including arthritis, headache and muscular pain) and children (including teething, earache and immunisation). For more information, see Section 1. Why am I using Panamax? in the full CMI.
2. What should I know before I use Panamax?
Do not use if you have ever had an allergic reaction to Panamax or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. For more information, see Section 2. What should I know before I use Panamax? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Panamax and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Panamax?
- The label on the pack or bottle will tell you how to take your medicine and how often. If you are unsure about the directions ask your pharmacist or doctor.
- The dose will be different depending upon which product you are taking and whether you are an adult or child.
More instructions can be found in Section 4. How do I use Panamax? in the full CMI.
5. What should I know while using Panamax?
| Things you should do |
|
| Things you should not do |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Panamax? in the full CMI.
6. Are there any side effects?
- Less serious side effects include nausea and stomach pain.
- Tell your doctor or pharmacist immediately if you notice any of the following side effects shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue, or other parts of the body, rash, itching or hives on the skin. They may be the signs of an allergic reaction.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using Panamax?
Panamax contains the active ingredient paracetamol. Panamax is an analgesic.
Panamax is used to relieve pain and fever in adults (including arthritis, headache and muscular pain) and children (including teething, earache and immunisation).
Panamax works to stop the pain messages from getting through to the brain. Panamax also acts in the brain to reduce fever.
2. What should I know before I use Panamax?
Warnings
Do not use Panamax if:
- you are allergic to paracetamol, or any of the ingredients listed at the end of this leaflet.
The symptoms of an allergic reaction may include:
- shortness of breath wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
- Always check the ingredients to make sure you can use this medicine.
- Do not take this medicine if you have liver or kidney problems.
- Do not take Panamax if you have glucose-6-phosphate-dehydrogenase deficiency (an enzyme deficiency).
- Do not use Panamax if the packaging is torn or shows signs of tampering.
- Do not use Panamax after the expiry date (EXP) printed on the pack or bottle.
- If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your pharmacist or doctor.
Check with your doctor or pharmacist if you:
- have allergies to any ingredients listed under Product Details at the end of this leaflet
- have allergies to aspirin or any other NSAID medicine
- have liver or kidney problems
- have malnutrition and other sources of low glutathione reserves
- severe renal insufficiency and sepsis
- chronic alcohol use including recent cessation of alcohol intake
- have Gilbert's syndrome
- have acute breathing difficulties such as bronchitis, unstable asthma or emphysema
- drink large quantities of alcohol
- take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant or if you are breastfeeding or intend to breastfeed.
Panamax may be used during pregnancy and if you are breastfeeding. However, it is recommended that non-drug therapy such as rest and massage be tried first. If Panamax is needed during pregnancy, it should be used at the lowest effective dose for the shortest possible time and at the lowest possible frequency.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Panamax and affect how it works.
These include:
- Any medicines which thin the blood, for example warfarin
- Medicines to treat epilepsy
- Metoclopramide or domperidone, medicines used to control nausea and vomiting
- Propantheline, a drug used to treat stomach ulcers
- Other pain relief medication
- Alcohol and medicines containing alcohol (ethanol) eg, some cough syrups
- Medicines used to treat depression
- Medicines used to aid sleep
- Chloramphenicol, an antibiotic used to treat ear and eye infections
- Flucloxacillin, zidovudine and rifampicin, drugs used to treat infections
- Probenecid, a drug used to treat high uric acid levels in the blood associated with gout
- Cholestyramine, a drug used to reduce blood cholesterol
- Chelating resin
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Panamax.
4. How do I use Panamax?
How much to take
There are three different Panamax products:
- Panamax Tablets
- Panamax Elixir
- Panamax 240 Elixir
The dose will be different depending upon which product you are taking.
Panamax Tablets
The usual dose is:
Adults:
- 1 to 2 tablets.
- This dosage may be repeated every 4 to 6 hours if necessary.
- You should not take more than 8 tablets in 24 hours.
Children (7-12 years):
- Half (1/2) to 1 tablet.
- This dosage may be repeated every 4 to 6 hours if necessary.
- Children should not take more than 4 tablets in 24 hours.
Panamax tablets are not recommended for children under 7 years.
Swallow tablets whole with a little water or other liquid.
Panamax Elixir
The usual dose is:
Infants:
1 to 3 months (4 - 6 kg): 2 to 4 mL;
3 to 6 months (6 - 8 kg): 4 to 5 mL;
6 to 12 months (8 - 10 kg): 5 to 6 mL.
Children:
1 to 2 years (10 - 12 kg): 6 to 8 mL;
2 to 4 years (12 - 16 kg): 8 to 10 mL;
4 to 6 years (16 - 20 kg):10 to 13 mL;
6 to 8 years (20 - 25 kg): 13 to 16 mL;
8 to 10 years (25 - 32 kg): 16 to 20 mL;
10 to 12 years (32 - 41 kg): 20 to 26 mL.
This dosage may be repeated every 4 to 6 hours if necessary.
Do not give more than 4 doses in 24 hours.
Panamax Elixir is not recommended for infants under 1 month.
It can be given in water.
Panamax 240 Elixir
Children:
5 to 6 years (18 - 20 kg): 6 mL;
6 to 8 years (20 to 25 kg): 6 to 8 mL;
8 to 10 years (25 - 32 kg): 8 to 10 mL;
10 to 12 years (32 - 41 kg): 10 to 12 mL.
Do not give more than 4 doses in 24 hours.
Adults:
10 to 20 mL
This dosage may be repeated every 4 to 6 hours if necessary up to 4 times in 24 hours.
Do not take more than 80 mL per day.
Panamax 240 Elixir is not recommended for children under 5 years of age.
When to take Panamax
- The label on the pack or bottle will tell you how to take your medicine and how often. If you are unsure about the directions ask your pharmacist or doctor.
It can be given in water if necessary.
Your doctor may ask you to take a different dose. If so, you should follow the doctor's instructions.
If you are unsure what dose to take ask your pharmacist.
If you forget to use Panamax
If you miss a dose, take it as soon as you remember.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you use too much Panamax
If you think that you have used too much Panamax, you may need urgent medical attention.
If you take too much Panamax you may feel nauseous, dizzy or sweaty.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Panamax?
Things you should do
Take Panamax exactly as directed.
Remind any doctor, dentist or pharmacist you visit that you are using Panamax.
Things you should not do
- Do not take more than the recommended dose unless your doctor tells you to.
Children:
- Do not give Panamax for more than 48 hours unless a doctor has told you to.
Adults:
- Do not take for more than a few days at a time unless your doctor tells you to.
Drinking alcohol
Tell your doctor if you drink alcohol.
Only drink small quantities of alcohol (beer, wine or spirits) while taking paracetamol.
Drinking large quantities of alcohol while taking paracetamol may increase the risk of liver side effects.
Looking after your medicine
- Keep your tablets in the blister pack until it is time to take them.
- If you take the tablets out of the box or the blister pack they will not keep well.
- Keep bottles tightly closed, and ensure that the child resistant cap is engaged after use.
- Store below 30°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions.
Less serious side effects
| Less serious side effects | What to do |
General
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Skin related
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What Panamax tablets contain
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Panamax tablets do not contain gluten, lactose, sucrose, tartrazine or any azo dyes.
What Panamax Elixir (100 mL) contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
What Panamax 240 Elixir (200 mL) contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What Panamax looks like
Panamax comes as a white tablet, marked PANAMAX.
A box may contain 50 or 100 tablets.
Panamax is also available as an Elixir (100 mL) or 240 Elixir (200 mL). A clear light red coloured syrup liquid.
AUST R 15490 (Tablets)
AUST R 13750 (Elixir)
AUST R 49623 (240 Elixir)
Who distributes Panamax
sanofi-aventis australia pty ltd
12-24 Talavera Road
Macquarie Park NSW 2113
This leaflet was prepared in August 2025.
panamax-ccdsv5-cmiv13-25aug25
Brand Information
| Brand name | Panamax |
| Active ingredient | Paracetamol |
| Schedule | S2 | S3 |
MIMS Revision Date: 01 October 2025
1 Name of Medicine
Paracetamol.
2 Qualitative and Quantitative Composition
Panamax Tablets. Each tablet contains paracetamol 500 mg.
Excipients with known effect. Potassium sorbate.
Panamax Elixir. Each 5 mL contains paracetamol 120 mg.
Excipients with known effect. Benzoic acid, potassium sorbate, saccharin sodium.
Panamax 240 Elixir. Each 5 mL contains paracetamol 240 mg.
Excipients with known effect. Benzoic acid, potassium sorbate, saccharin sodium, sorbitol solution (70 per cent) (non-crystallising).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Panamax Tablets. Flat, round, scored, white tablet with bevelled edges, front face marking 'PANAMAX' with break line on the reverse.
Panamax Elixir. Clear, red coloured syrup liquid with a fruity odour and taste.
Panamax 240 Elixir. Clear light red coloured syrupy liquid.
4 Clinical Particulars
4.1 Therapeutic Indications
Relief of pain and discomfort in arthritis, headache, muscular and neuralgic conditions. Reduces fever. Panamax is useful as an analgesic for patients with dyspepsia, ulcers or gout.
4.2 Dose and Method of Administration
Panamax Tablets. Children. 7 to 12 years. 250 to 500 mg (1/2 to 1 tablet) every four to six hours (maximum 4 tablets per day). Take with water.
Adults. 500 mg to 1 g (1 to 2 tablets) every four to six hours (maximum 8 tablets per day). Take with water.
Panamax Elixir. Administer in water or fruit juice at 4 to 6 hourly intervals.
Infants. 1 to 3 months (4-6 kg). 2 to 4 mL.
3 to 6 months (6-8 kg). 4 to 5 mL.
6 to 12 months (8-10 kg). 5 to 6 mL.
Children. 1 to 2 years (10-12 kg). 6 to 8 mL.
2 to 4 years (12-16 kg). 8 to 10 mL.
4 to 6 years (16-20 kg). 10 to 13 mL.
6 to 8 years (20-25 kg). 13 to 16 mL.
8 to 10 years (25-32 kg). 16 to 20 mL.
10 to 12 years (32-41 kg). 20 to 26 mL.
Administration to infants under 1 month is not recommended.
Recommended dosages are based on 15 mg of paracetamol per kg of bodyweight.
Panamax 240 Elixir. Administer in water or fruit juice if necessary.
Children. 5 to 6 years (18-20 kg). 6 mL.
6 to 8 years (20-25 kg). 6 to 8 mL.
8 to 10 years (25-32 kg). 8 to 10 mL.
10 to 12 years (32-41 kg). 10 to 12 mL.
Adults. 10 to 20 mL (maximum 80 mL per day).
If necessary repeat 4 to 6 hourly up to 4 times in 24 hours.
Panamax 240 Elixir is not recommended for children under 5 years of age.
4.3 Contraindications
Panamax is contraindicated in patients who are hypersensitive to paracetamol or to any other component of the Panamax formulations. It must not be used in patients with known glucose-6-phosphate dehydrogenase deficiency. Paracetamol should not be used in patients with severe hepatocellular insufficiency. Paracetamol should not be used in patients with active alcoholism as chronic excessive alcohol ingestion predisposes patients to paracetamol hepatotoxicity.
4.4 Special Warnings and Precautions for Use
This medication may be dangerous when used in large amounts or for long periods. Hepatotoxicity may occur with paracetamol even at therapeutic doses, after short treatment duration and in patients without pre-existing liver dysfunction. Hepatotoxicity may develop following as little as 10 to 15 g of paracetamol and hepatic failure is known to occur occasionally with the long-term use of paracetamol.
To avoid the risk of overdose. Check that paracetamol is absent from the composition of other medicinal products taken concomitantly.
Patients with known analgesic intolerance or known bronchial asthma must only use Panamax after having consulted a physician (hypersensitivity reactions including bronchospasm possible).
Caution is advised in patients with underlying sensitivity to aspirin and/or to nonsteroidal anti-inflammatory drugs (NSAIDs).
Severe cutaneous adverse reactions (SCARs). Life threatening cutaneous reactions, Stevens-Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN) have been reported with the use of paracetamol. Patients should be advised of the signs and symptoms and monitored closely for skin reactions. If symptoms or signs of SJS and TEN (e.g. progressive skin rash often with blisters or mucosal lesions) occur, patients should stop paracetamol treatment immediately and seek medical advice.
Paracetamol should be used upon medical advice in patients with: mild-to-moderate hepatocellular insufficiency; severe renal insufficiency and sepsis; chronic alcohol use including recent cessation of alcohol intake; malnutrition and other sources of low glutathione reserves; glucose-6-phosphate-dehydrogenase deficiency; Gilbert's syndrome.
Cases of high anion gap metabolic acidosis (HAGMA) due to pyroglutamic acidosis have been reported in patients with severe illness and/or pre-disposing factors (see above) who were treated with paracetamol at therapeutic dose for a prolonged period or combination of paracetamol and flucloxacillin. Symptoms of HAGMA may include serious breathing difficulties with deep rapid breathing, drowsiness, nausea and vomiting. Prompt discontinuation of paracetamol and close monitoring is recommended if symptoms of HAGMA appear. The measurement of urinary 5-oxoproline may be useful to identify pyroglutamic acidosis as underlying cause of HAGMA in patients with multiple risk factors.
Use in hepatic impairment. Panamax should not be administered to patients with severe hepatocellular insufficiency (see Section 4.3 Contraindications).
Paracetamol should be used with caution in patients with mild-to-moderate hepatocellular insufficiency. These patients must seek medical advice before taking this medication. Underlying liver disease increases the risk of paracetamol-related liver damage.
Use in renal impairment. Panamax should be used with caution in patients with impaired kidney function: Administration of paracetamol to patients with moderate to severe renal impairment may result in accumulation of paracetamol conjugates. Patients who have been diagnosed with kidney impairment must seek medical advice before taking this medication.
Use in the elderly. No data available.
Paediatric use. Panamax Tablet is not recommended for children under 7 years of age.
Panamax Elixir is not recommended for infants under 1 month of age.
Panamax 240 Elixir is not recommended for children under 5 years of age.
Effect on laboratory tests. Uric acid and blood glucose. Intake of paracetamol may affect the laboratory determination of uric acid by phosphotungstic acid and of blood glucose by glucose oxidase-peroxidase.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Paracetamol may increase the risk of bleeding in patients taking warfarin and other antivitamin K. Anticoagulant dosage may require reduction and patients should be monitored for appropriate coagulation and bleeding complications.
Paracetamol absorption is increased by drugs which increase gastric emptying, e.g. metoclopramide and domperidone and decreased by drugs which decrease gastric emptying, e.g. propantheline, antidepressants with anticholinergic properties, narcotic analgesics.
Paracetamol may increase chloramphenicol concentrations by slowing down excretion, entailing the risk of increased toxicity. The risk of paracetamol toxicity may be increased in patients receiving other potentially hepatotoxic drugs or drugs that induce liver microsomal enzymes, such as antiepileptics (such as phenobarbital, phenytoin, carbamazepine, topiramate), barbiturates, hypnotics, rifampicin and alcohol.
Paracetamol excretion may be affected and plasma concentrations altered when given probenecid.
Cholestyramine reduces the absorption of paracetamol if given within 1 hour of paracetamol. Chelating resin can decrease the intestinal absorption of paracetamol and potentially decrease its efficacy if taken simultaneously. In general, there must be an interval of more than 2 hours between taking the resin and taking paracetamol, if possible.
Coadministration of flucloxacillin with paracetamol may lead to high anion gap metabolic acidosis due to pyroglutamic acidosis, particularly in patients with risk factors (see Section 4.4 Special Warnings and Precautions for Use).
When used concurrently with zidovudine, an increased tendency for neutropenia may develop. Combination of Panamax and zidovudine should be avoided.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Drugs which have been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the foetus having been observed. Paracetamol can be used during pregnancy if clinically needed however it should be used at the lowest effective dose for the shortest possible time and at the lowest possible frequency.
Paracetamol can cross the placenta; however, no teratogenic effects have been observed in rats or mice after doses of up to 250 mg/kg.
A woman in the third trimester of pregnancy ingested 22.5 g paracetamol; early treatment with oral acetylcysteine resulted in a good outcome for both mother and foetus.
Use in lactation. Paracetamol is excreted in breast milk. The amount available for ingestion by the infant has been reported variously as less than 0.1% of a single 500 mg dose, and as 0.04 to 0.23% of a single 650 mg dose. Maternal ingestion of paracetamol in usual analgesic doses does not appear to present a risk to the nursing infant.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Reports of adverse reactions are rare. Although the following reactions have been reported: dyspepsia, sweating, erythema, urticaria, anaphylactic shock, angioneurotic oedema, difficulty breathing, drop in blood pressure, nausea, allergic reactions such as skin rashes, hypersensitivity reactions and haematological reactions, including thrombocytopenia, leukopenia, neutropenia, agranulocytosis and pancytopenia. Bronchospasm may be triggered in patients having a tendency of analgesic asthma. Toxic epidermal necrolysis (TEN), Stevens-Johnson syndrome (SJS), acute generalised exanthematous pustulosis, fixed drug eruption (see Section 4.4 Special Warnings and Precautions for Use) and cytolytic hepatitis, which may lead to acute hepatic failure, have also been reported. Overdosage with paracetamol if left untreated can result in severe, sometimes fatal liver damage and rarely, acute renal tubular necrosis.
Haemolytic anaemia, particularly in patients with underlying glucose 6-phosphate-dehydrogenase deficiency has been reported. Kounis syndrome has been reported, as has high anion gap metabolic acidosis due to pyroglutamic acidosis in patients with pre-disposing factors (see Section 4.4 Special Warnings and Precautions for Use).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Elderly persons, small children, patients with liver disorders, chronic alcohol consumption or chronic malnutrition, as well as patients concomitantly treated with enzyme-inducing drugs are at an increased risk of intoxication, including fatal outcome.
Symptoms. Toxic symptoms include vomiting, abdominal pain, hypotension and sweating. Nausea, vomiting, anorexia, pallor and abdominal pain generally appear during the first 24 hours of overdosage with paracetamol. Overdosage with paracetamol may cause hepatic cytolysis which can lead to hepatocellular insufficiency, gastrointestinal bleeding, metabolic acidosis, encephalopathy, disseminated intravascular coagulation, coma and death. Increased levels of hepatic transaminases, lactate dehydrogenase and bilirubin with a reduction in prothrombin level can appear 12 to 48 hours after acute overdosage. Overdosage can also lead to pancreatitis, acute renal failure and pancytopenia. The most serious adverse effect of acute overdosage of paracetamol is a dose dependent, potentially fatal hepatic necrosis. In adults, hepatotoxicity may occur after ingestion of a single dose of 12 g (24 tablets) of paracetamol; a dose of 25 g (50 tablets) or more is potentially fatal. Symptoms during the first 2 days of acute poisoning by paracetamol do not reflect the potential seriousness of the intoxication. Major manifestations of liver failure such as jaundice, hypoglycaemia and metabolic acidosis may take at least 3 days to develop.
Treatment. Despite lack of significant early symptoms, patients should be referred to hospital urgently for immediate medical attention.
Determinations of the plasma concentration of paracetamol are recommended.
Plasma concentration of paracetamol should be measured at 4 hours or later after ingestion (earlier concentrations are unreliable).
Where paracetamol intoxication is suspected, intravenous administration of SH group donators such as acetylcysteine within the first 10 hours after ingestion is indicated. Although acetylcysteine is most effective if initiated within this period, it can still offer some degree of protection if given as late as 48 hours after ingestion; in this case it is taken for longer.
If the history suggests that 12 g paracetamol or more has been ingested, administer one of the following antidotes.
Acetylcysteine 20% i.v. Administer intravenously, 20% acetylcysteine immediately without waiting for positive urine test or plasma level results. For dosage instructions refer to the acetylcysteine 20% i.v. product information.
Oral methionine. For dosage instructions refer to the methionine product information.
Further measures will depend on the severity, nature and course of clinical symptoms of intoxication and should follow standard intensive care protocols.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Paracetamol has analgesic and antipyretic effects.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. After oral administration, paracetamol is absorbed rapidly and completely from the small intestine; peak plasma levels occur 10 to 60 minutes after oral administration. Food intake delays paracetamol absorption.
Distribution. Paracetamol is uniformly distributed throughout most body fluids; the apparent volume of distribution is 1 to 1.2 L/kg. Paracetamol can cross the placenta and is excreted in breast milk. Plasma protein binding is negligible at usual therapeutic concentrations but increases with increasing concentrations.
Metabolism. Paracetamol is metabolised by the hepatic microsomal enzyme system. In adults at therapeutic doses, paracetamol is mainly conjugated with glucuronide (45-55%) or sulfate (20-30%). A minor proportion (less than 20%) is metabolised to catechol derivatives and mercapturic acid compounds via oxidation. Paracetamol is metabolised differently by infants and children compared to adults, the sulfate conjugate being predominant.
Excretion. Paracetamol is excreted in the urine mainly as the glucuronide and sulfate conjugates. Less than 5% is excreted as unchanged paracetamol. 85-90% of the administered dose is eliminated in the urine within 24 hours of ingestion.
The elimination half-life is about 1 to 4 hours.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Panamax Tablets. The inactive ingredients are: maize starch, purified talc, pregelatinised maize starch, povidone, stearic acid and potassium sorbate.
Panamax Elixir. The inactive ingredients are: macrogol 1500, propylene glycol, glycerol, tutti frutti flavour, raspberry flavour, benzoic acid, potassium sorbate, Lycasin, saccharin sodium, ponceau SX and purified water.
Panamax 240 Elixir. The inactive ingredients are: macrogol 1500, allura red AC, propylene glycol, glycerol, saccharin sodium, sorbitol solution (70 per cent) (non-crystallising), benzoic acid, potassium sorbate, raspberry flavour, imitation candied sugar and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Panamax Tablet. Blister packs of 50 and 100 tablets.
Panamax Elixir. 100 mL.
Panamax 240 Elixir. 200 mL.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Paracetamol is a white or almost white, crystalline powder. It is sparingly soluble in water, freely soluble in alcohol and very slightly soluble in methylene chloride. It has a melting point between 168°C and 172°C.
Chemical structure.
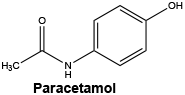
Chemical formula: C8H9NO2.
Chemical name: N-(4-Hydroxyphenyl) acetamide.
CAS number. 103-90-2.
7 Medicine Schedule (Poisons Standard)
Panamax Tablet. Pharmacy Medicine (Schedule 2) pack size: 50.
Pharmacist Only Medicine (Schedule 3) pack size: 100.
Panamax Elixir and Panamax 240 Elixir. Pharmacy Medicine (Schedule 2).
Date of Revision
25 August 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.