Paramyl Osteo
Brand Information
| Brand name | Paramyl Osteo |
| Active ingredient | Paracetamol |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Paramyl Osteo.
Full CMI
1. Why am I using Paramyl Osteo?
Paramyl Osteo contains the active ingredient paracetamol. Paramyl Osteo is an analgesic and works to stop the pain messages from getting through to the brain.
Paramyl Osteo provides effective relief of persistent pain associated with osteoarthritis for up to 8 hours.
2. What should I know before I use Paramyl Osteo?
Warnings
Do not use Paramyl Osteo if:
- you are allergic to paracetamol, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you are taking other prescription or non-prescription medicines containing paracetamol to treat pain, fever, symptoms of cold and flu, or to aid sleep.
Check with your doctor or pharmacist if you:
- have or have had liver or kidney disease
- are underweight or malnourished
- regularly drink alcohol
You may need to avoid using this product altogether or limit the amount of paracetamol that you take. - have a severe infection, are severely malnourished or are a chronic, heavy alcohol user as this may increase the risk of metabolic acidosis.
Signs of metabolic acidosis include deep, rapid, difficult breathing, feeling sick (nausea), being sick (vomiting), loss of appetite. Contact a doctor immediately if you get a combination of these symptoms.
Please see your doctor if your symptoms do not improve.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
Paracetamol may be used during pregnancy and if you are breastfeeding but you should always consult your doctor first. Consider taking the lowest effective dose for the shortest period of time.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Paramyl Osteo and affect each other.
- Warfarin, a medicine used to prevent blood clots
- Metoclopramide, a medicine used to control nausea and vomiting
- Medicines used to treat epilepsy or fits
- Propantheline
- Certain medicines used to treat depression or to control pain
- Chloramphenicol, an antibiotic used to treat ear and eye infections.
- Alcohol
- Probenecid, a medicine used to treat gout or sometimes given with an antibiotic
- Cholestyramine, a medicine used to treat high cholesterol levels in the blood.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Paramyl Osteo.
4. How do I use Paramyl Osteo?
How much to take / use
Adults and children aged 12 years and over: Take 2 tablets three times a day, every six to eight hours as needed. Do not take more than 6 tablets in 24 hours.
Not recommended for children under 12 years.
- Follow the instructions provided with the medicine.
- Do not exceed the recommended dosage.
- Use the smallest dose that you need to treat your symptoms.
When to take / use Paramyl Osteo
- Paramyl Osteo should only be used for a few days at a time in adults or no more than 48 hours for children aged 12 – 17 years
- unless your doctor tells you to take it for longer.
- Use for the shortest period of time necessary.
How to take Paramyl Osteo
- Swallow the tablets whole with water or other fluid.
- Do not crush or chew the tablets.
- They can be taken with or without food.
- Try to space the doses at equal intervals throughout the day.
If you use too much Paramyl Osteo
If you think that you have used too much Paramyl Osteo, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning because of the risk of liver failure.
5. What should I know while using Paramyl Osteo?
Things you should do
Remind any doctor, dentist, pharmacist or other health professionals you visit that you are using Paramyl Osteo.
Talk to your pharmacist or doctor if your symptoms do not improve. They will assess your condition and decide if you should continue to take the medicine.
Things you should not do
- Do not use for more than a few days at a time in adults or more than 48 hours for children aged 12 – 17 years unless your doctor tells you to.
- Do not take more than the recommended dose unless your doctor tells you to.
- Do not use Paramyl Osteo to treat any other complaints unless your pharmacist or doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Paramyl Osteo affects you.
Drinking alcohol
Tell your doctor or pharmacist if you drink alcohol.
Only drink small quantities of alcohol (beer, wine or spirits) while using Paramyl Osteo. Drinking large quantities of alcohol may increase the risk of liver side effects.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
Keep your medicine in the original pack until it is time to take it.
Store below 30°C.
Store it in a cool, dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions.
Very rare side effects
| Very rare side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What Paramyl Osteo contains
| Active ingredient (main ingredient) | Paracetamol |
| Other ingredients (inactive ingredients) | Hypromellose Maize starch Povidone Croscarmellose sodium Magnesium stearate Titanium dioxide Macrogol 400 |
Do not take this medicine if you are allergic to any of these ingredients.
What Paramyl Osteo looks like
Paramyl Osteo is a white to off-white, capsule-shaped, biconvex tablet, plain on both sides
(AUST R 459289).
Who distributes Paramyl Osteo
SANDOZ Pty Ltd
North Sydney, NSW 2060, AUSTRALIA
Tel 1800 726 369
This leaflet was prepared on 13th August 2024
Brand Information
| Brand name | Paramyl Osteo |
| Active ingredient | Paracetamol |
| Schedule | S3 |
MIMS Revision Date: 01 April 2025
1 Name of Medicine
Paracetamol.
2 Qualitative and Quantitative Composition
Active ingredient. Paracetamol 665 mg/tablet.
Excipients. For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
White to off-white, capsule shaped, biconvex tablets, plain on both sides.
4 Clinical Particulars
4.1 Therapeutic Indications
For the relief of persistent pain associated with osteoarthritis for up to 8 hours.
4.2 Dose and Method of Administration
Dose. Adults and children aged 12 years and over. Swallow 2 tablets with water or other fluid, three times a day, every 6 to 8 hours as required. Swallow whole, do not crush or chew.
Maximum of 6 tablets in 24 hours.
Do not use for more than a few days at a time in adults except on medical advice.
Should not be used for more than 48 hours for children aged 12 - 17 years, except on medical advice.
Children under 12 years. Not recommended for children under the age of 12 years.
Method of administration. Take with water or other fluid. Can be taken with or without food.
The 3 doses should be equally spaced throughout the day. The tablets must not be crushed.
Do not exceed the stated dose.
The lowest dose necessary to achieve efficacy should be used for the shortest duration of treatment. Should not be used with other paracetamol-containing products.
Minimum dosing interval. 6 hours.
Maximum daily dose for children 12 years of age to adults. 4000 mg.
4.3 Contraindications
Contraindicated in patients with a previous history of hypersensitivity to paracetamol or to any of the excipients.
4.4 Special Warnings and Precautions for Use
Identified precautions. Contains paracetamol. Do not use with any other paracetamol- containing products. The concomitant use with other products containing paracetamol may lead to an overdose.
Paracetamol overdose may cause liver failure which may require liver transplant or lead to death. If symptoms persist, medical advice must be sought.
Keep out of sight and reach of children.
Use in hepatic impairment. Paracetamol should be used with caution in patients with impaired liver function: Underlying liver disease increases the risk of paracetamol-related liver damage.
Patients who have been diagnosed with liver impairment must seek medical advice before taking this medication.
Cases of hepatic dysfunction/failure have been reported in patients with depleted glutathione levels, such as those who are severely malnourished, anorexic, have a low body mass index, are chronic heavy users of alcohol or have sepsis.
In patients with glutathione depleted states the use of paracetamol may increase the risk of metabolic acidosis.
Use in renal impairment. Paracetamol should be used with caution in patients with impaired kidney function: Administration of paracetamol to patients with moderate to severe renal impairment may result in accumulation of paracetamol conjugates.
Patients who have been diagnosed with kidney impairment must seek medical advice before taking this medication.
Use in the elderly. No data available.
Paediatric use. Not recommended for children under 12 years of age.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The following interactions with paracetamol have been noted:
The anticoagulant effect of warfarin and other coumarins may be enhanced by prolonged regular daily use of paracetamol with increased risk of bleeding; occasional doses have no significant effect. Anticoagulant dosage may require reduction if paracetamol and anticoagulants are taken for a prolonged period of time.
Paracetamol absorption is increased by substances that increase gastric emptying, e.g. metoclopramide.
Paracetamol absorption is decreased by substances that decrease gastric emptying, e.g. propantheline, antidepressants with anticholinergic properties, and narcotic analgesics.
Paracetamol may increase chloramphenicol concentrations.
The risk of paracetamol toxicity may be increased in patients receiving other potentially hepatotoxic drugs or drugs that induce liver microsomal enzymes such as alcohol and anticonvulsant agents.
Paracetamol excretion may be affected and plasma concentrations altered when given with probenecid.
Cholestyramine reduces the absorption of paracetamol if given within 1 hour of paracetamol.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Paracetamol has been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the foetus having been observed.
As with the use of any medicine during pregnancy, pregnant women should seek medical advice before taking paracetamol. The lowest effective dose and shortest duration of treatment should be considered.
Use in lactation. Paracetamol is excreted in breast milk. Human studies with paracetamol have not identified any risk to lactation or the breast-fed offspring. These results are based on immediate release preparations of paracetamol. There is no data available on the excretion of sustained-release paracetamol preparations in breast milk. However, it is not expected that Paramyl Osteo would provide any increase in the excretion of paracetamol in breast milk as this product is designed to maintain rather than increase plasma paracetamol concentrations compared to immediate release preparations. Maternal ingestion of paracetamol in usual analgesic doses does not appear to present a risk to the breastfed infant.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Adverse events from historical clinical trial data are both infrequent and from small patient exposure. Accordingly, events reported from extensive post-marketing experience at therapeutic/labelled doses and considered attributable are tabulated below by System Organ Class and frequency.
The following convention has been utilised for the classification of undesirable effects: very common (≥ 1/10), common (≥ 1/100, < 1/10), uncommon (≥ 1/1,000, < 1/100), rare (≥ 1/10,000, < 1/1,000), very rare (< 1/10,000), not known (cannot be estimated from available data).
Adverse event frequencies have been estimated from spontaneous reports received through post-marketing data. See Table 1.

4.9 Overdose
Poisons information centre. If an overdose is taken or suspected, contact the Poisons Information Centre immediately on 131 126 (Australia). The patient should go to the nearest hospital straight away. This should be done even if they feel well because of the risk of delayed, serious liver damage (see Section 4.8 Adverse Effects (Undesirable Effects)).
Because Paramyl Osteo is a sustained-release formulation of paracetamol, absorption will be prolonged in overdose. It is recommended that for the management of overdose, where Paramyl Osteo is suspected, that an additional plasma paracetamol level be obtained 4-6 hours after the initial measurement. If either level is above or close to the treatment line on the paracetamol overdose nomogram, administration of antidote would be indicated.
Treatment. Paracetamol overdose may cause liver failure which may require liver transplant or lead to death. Acute pancreatitis has been observed, usually with hepatic dysfunction and liver toxicity.
Immediate medical management is required in the event of an overdose, even if the symptoms of overdose are not present.
Administration of N-acetylcysteine may be required.
In cases of overdose, methods of reducing absorption of ingested drug are important. Activated charcoal may reduce absorption of the medicine if given within one hour after oral ingestion. In patients who are not fully conscious or have impaired gag reflex, consideration should be given to administering activated charcoal via a nasogastric tube, once the airway is protected.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Paracetamol is a para-aminophenol derivative that exhibits analgesic and anti-pyretic activity. Its mechanism of action is believed to include inhibition of prostaglandin synthesis, primarily within the central nervous system. It does not possess anti-inflammatory activity. It provides relief from mild to moderate pain and fever.
The sustained release of paracetamol provides pain relief, which may last up to 8 hours.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Paracetamol is rapidly and almost completely absorbed from the gastrointestinal tract. Food intake delays paracetamol absorption.
Paramyl Osteo is formulated to provide a sustained-release dose of paracetamol.
Coadministration of Paramyl Osteo with food leads to a small increase in bioavailability and a small delay in Tmax.
When administered under repeat dose conditions according to the recommended dosage of two tablets every 6 to 8 hours, Paramyl Osteo tablets achieve mean plasma concentrations above the minimum therapeutic level for analgesia of 4 microgram/mL throughout the dosage interval.
Distribution. Paracetamol is distributed into most body tissues. Binding to the plasma proteins is minimal at therapeutic concentrations but increases with increasing doses.
Metabolism. Paracetamol is metabolised extensively in the liver and excreted in the urine mainly as inactive glucuronide and sulphate conjugates.
The metabolites of paracetamol include a minor hydroxylated intermediate which has hepatotoxic activity. This intermediate metabolite is detoxified by conjugation with glutathione. However, it can accumulate following paracetamol overdosage (more than 150 mg/kg or 10 g total paracetamol ingested) and, if left untreated, can cause irreversible liver damage.
Paracetamol is metabolised differently by premature infants, newborns, infants and young children compared to adults, the sulfate conjugate being predominant.1
Excretion. Paracetamol is excreted in the urine mainly as the glucuronide and sulfate conjugates. Less than 5% is excreted unchanged. Approximately 85% of a dose of paracetamol is excreted in urine as free and conjugated paracetamol within 24 hours of ingestion. Administration of paracetamol to patients with moderate to severe renal impairment may result in accumulation of paracetamol conjugates.2 The elimination half-life varies from one to three hours.
1 Core Paracetamol PI.
2 American Hospital Formulary Service Drug Information 2012.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Hypromellose, maize starch, povidone, croscarmellose sodium, magnesium stearate, titanium oxide, macrogol 400.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Blister pack of 96 tablets. Bottle of 96 tablet.
Not all presentations may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
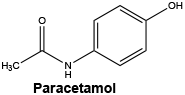
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S3 - Pharmacist Only Medicine.
Date of First Approval
13 August 2024
Date of Revision
13 August 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.