Pariet 10 Tablets
Brand Information
| Brand name | Pariet 10 Tablets |
| Active ingredient | Rabeprazole sodium |
| Schedule | S2 | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Pariet 10 Tablets.
Summary CMI
PARIET® 10 Tablets
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using PARIET 10?
PARIET 10 contains the active ingredient rabeprazole sodium. PARIET 10 is used for symptomatic relief of heartburn and stomach acid complaints due to gastro-oesophageal reflux.
For more information, see Section 1. Why am I using PARIET 10? in the full CMI.
2. What should I know before I use PARIET 10?
Do not use if you have ever had an allergic reaction to rabeprazole sodium or any of the ingredients listed at the end of the CMI.
Talk to your doctor or pharmacist if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use PARIET 10? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with PARIET 10 and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use PARIET 10?
- The usual dose is one tablet per day, to be taken at the same time each day, for at least 7 days and up to 14 days.
- PARIET 10 should be swallowed whole, with a glass of water or other liquid.
More instructions can be found in Section 4. How do I use PARIET 10? in the full CMI.
5. What should I know while using PARIET 10?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using PARIET 10? in the full CMI.
6. Are there any side effects?
PARIET 10 is usually well tolerated but tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking PARIET 10. The common side effects include headache, dizziness, diarrhoea, nausea, abdominal pain, wind, vomiting, constipation, runny or blocked nose, sore throat, cough, non-specific pain, back pain, muscle pain, rash, muscle weakness, physical weakness or lack of energy, flu-like syndrome, infection, insomnia, and chest pain. The serious side effects include allergic or hypersensitivity reactions; frequent infections; urinary tract infection, vision or taste disturbance, depression, feeling dizzy, faint, lightheaded or weak (hypotension), pain or indigestion, begin to vomit blood or food, pass black (blood-stained) stools, symptoms of bowel inflammation such as severe (watery or bloody) diarrhoea, fever, abdominal pain or tenderness.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
PARIET® 10 Tablets
Active ingredient(s): rabeprazole sodium
Consumer Medicine Information (CMI)
This leaflet provides important information about using PARIET 10. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using PARIET 10.
Where to find information in this leaflet:
1. Why am I using PARIET 10?
2. What should I know before I use PARIET 10?
3. What if I am taking other medicines?
4. How do I use PARIET 10?
5. What should I know while using PARIET 10?
6. Are there any side effects?
7. Product details
1. Why am I using PARIET 10?
PARIET 10 contains the active ingredient rabeprazole sodium. PARIET 10 belongs to a group of medicines called proton pump inhibitors (PPIs). PARIET 10 works by decreasing the amount of acid the stomach makes to give relief from the symptoms. Your food will still be digested in the same way.
PARIET 10 is used for symptomatic relief of heartburn and stomach acid complaints due to gastro-oesophageal reflux caused by "washing back" of food and acid from the stomach into the food pipe, also known as the oesophagus.
Reflux can cause a burning sensation in the chest, rising up to the throat, also known as heartburn. Frequent heartburn is when you have heartburn for two or more days a week. Heartburn that occurs frequently is a typical symptom of gastro-oesophageal reflux disease (GORD).
PARIET 10 will start to suppress acid within a few hours; however it will not give instant symptom relief. You may need to take PARIET 10 for a few days before experiencing the full effect.
There is no evidence that PARIET 10 is addictive.
2. What should I know before I use PARIET 10?
Warnings
Do not use PARIET 10 if:
- you are allergic to rabeprazole sodium, other PPIs (omeprazole, esomeprazole, lansoprazole, pantoprazole), or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine.
Check with your doctor or pharmacist if you:
- have now, or have had in the past, liver problems
- take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
Children and adolescents
PARIET 10 is not recommended for use in children under 18 years of age. Safety and effectiveness of PARIET 10 in children has not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with PARIET 10 and affect how it works.
- clopidogrel, an antiplatelet medicine.
- cyclosporin, a medicine used to treat several conditions including prevention of graft rejection following kidney, liver or heart transplantation; severe, active rheumatoid arthritis; severe skin diseases; kidney disease where other treatments have failed.
- methotrexate, a medicine used to treat some kinds of cancer. It is also used to treat psoriasis (skin disease) and rheumatoid arthritis.
- digoxin, a medicine used to treat heart problems.
- ketoconazole, a medicine used to treat fungal infections.
- atazanavir, a medicine used to treat viral infections.
- mycophenolate mofetil, a medicine used to prevent organ rejection following kidney, liver or heart transplants.
- clarithromycin, a medicine used to treat infections.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect PARIET 10.
4. How do I use PARIET 10?
How much to take
- The usual dose is one tablet per day.
- Follow the instructions provided with the medicine.
- Do not exceed the recommended dosage.
When to take PARIET 10
- PARIET 10 should be taken at the same time each day.
- Take one tablet daily for at least 7 days and up to 14 days. You should not take PARIET 10 for more than 14 days unless directed by a doctor. If symptoms persist or recur within 14 days of completing the course, consult a doctor. Further examination may be required.
- If you forget to take your tablet, take it as soon as you remember and then continue to take it as you would normally. However, if it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Do not take a double dose to make up for the dose you missed. If you are not sure what to do, check with your doctor or pharmacist. If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
How to take PARIET 10
- PARIET 10 should be swallowed whole, with a glass of water or other liquid.
- Do NOT crush or chew the tablets. They have a special coating, which protects them from the acid in your stomach. If the coating is broken by chewing, the tablets may not work.
- It does not matter if you take PARIET 10 with food or on an empty stomach.
If you use too much PARIET 10
If you think that you have used too much PARIET 10, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using PARIET 10?
Things you should do
- Tell your doctor if you need to have a specific blood test (Chromogranin A) while you are taking PARIET 10. It may affect the results of this test.
Remind any doctor, dentist or pharmacist you visit that you are using PARIET 10.
Things you should not do
- Do not use PARIET 10 to treat any other complaints unless your doctor says to.
- Do not give this medicine to anyone else, even if they have the same symptoms as you.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how PARIET 10 affects you.
PARIET 10 may cause dizziness in some people.
Looking after your medicine
- Do not take PARIET 10 tablets out of the blister pack until it is time to take them. PARIET 10 tablets are packaged in a double-sided aluminium blister strip. If you take them out of the blister pack they may not keep well.
- Keep PARIET 10 tablets in a cool dry place where the temperature is below 25°C. Do not keep PARIET 10 in the refrigerator or freezer.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres (1.5 m) above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions.
Less serious side effects
| Less serious side effects | What to do |
Brain or nervous system related:
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergic or hypersensitivity reactions:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription. PARIET 10 is only available from your pharmacist or doctor.
What PARIET 10 contains
| Active ingredient (main ingredient) | rabeprazole sodium |
| Other ingredients (inactive ingredients) | mannitol, magnesium oxide, hyprolose, magnesium stearate, ethylcellulose, hypromellose phthalate, diacetylated monoglycerides, purified talc, titanium dioxide, carnauba wax, red iron oxide and printed with gray ink (Edible Ink Gray F6) |
Do not take this medicine if you are allergic to any of these ingredients.
The tablets do not contain lactose or gluten.
What PARIET 10 looks like
PARIET 10 tablets are pink and have "E241" in black ink on one side.
Each PARIET 10 tablet contains 10 mg rabeprazole sodium (Aust R 170438).
The tablets are available in blister packs of 7 and 14 tablets.
Not all pack sizes may be marketed.
Who distributes PARIET 10
Janssen-Cilag Pty Ltd
17 Khartoum Road,
Macquarie Park,
NSW 2113,
Australia
Telephone: Toll Free 1800 226 334
This leaflet was prepared in January 2026.
PARIET® 10 is a trademark of Eisai Ltd.
Brand Information
| Brand name | Pariet 10 Tablets |
| Active ingredient | Rabeprazole sodium |
| Schedule | S2 | S3 |
MIMS Revision Date: 01 March 2026
1 Name of Medicine
Rabeprazole sodium.
2 Qualitative and Quantitative Composition
Pariet 10 is available as enteric coated tablets containing 10 mg rabeprazole sodium (equivalent to 9.42 mg rabeprazole).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Pariet 10 tablets are pink, biconvex tablets, marked with "ε 241" in black ink on one side.
4 Clinical Particulars
4.1 Therapeutic Indications
Pariet 10 is indicated for:
Symptomatic relief of heartburn and other symptoms of gastroesophageal reflux disease.
4.2 Dose and Method of Administration
Adults 18 years of age and over. The recommended dose is one tablet per day to be taken at the same time each day (to facilitate treatment compliance) for at least 7 days and up to 14 days. If symptom control has not been achieved after two weeks of continuous treatment with Pariet 10, patients should be referred to their doctor. Pariet 10 tablets should not be chewed or crushed, but should be swallowed whole. Pariet 10 tablets were taken with or without food in the pivotal clinical trials.
Use in children under 18 years of age. Pariet 10 is not recommended for use in children as there is no experience of its use in this group.
Use in elderly patients. No dosage adjustment is necessary in elderly patients.
Use in patients with renal impairment. No dosage adjustment is necessary for patients with renal impairment.
There are no data on the use of rabeprazole in combination with antibiotic regimens in patients with renal impairment.
Use in patients with hepatic impairment. Patients with mild to moderate hepatic impairment experience higher exposure to rabeprazole sodium at a given dose than do healthy patients. Caution should be exercised in patients with severe hepatic impairment (see Section 4.4 Special Warnings and Precautions for Use).
There are no data on the use of rabeprazole in combination with antibiotic regimens in patients with hepatic impairment.
4.3 Contraindications
Pariet 10 is contraindicated in patients with known hypersensitivity to rabeprazole sodium, proton pump inhibitors, or any ingredient of this product.
4.4 Special Warnings and Precautions for Use
Symptomatic response to therapy with Pariet 10 does not preclude the presence of gastric malignancy, therefore, the possibility of malignancy should be excluded prior to commencing treatment with Pariet 10.
Patients on Pariet 10 should be further reviewed and/or investigated if symptoms persist or recur within 2 weeks of completing the course.
Patients should be referred to their doctor for review if:
they have unintentional weight loss, anaemia, gastrointestinal bleeding, dysphagia, persistent vomiting or vomiting with blood, malaena, gastric ulcer is suspected or present or gastrointestinal surgery, as treatment with pantoprazole may alleviate symptoms and delay diagnosis. In these cases, malignancy should be excluded;
they have had to take other medication for indigestion or heartburn continuously for four or more weeks in order to control their symptoms;
they are being treated for symptomatic GORD and require Pariet 10 for more than 14 days;
they have any other significant medical condition.
Acute tubulointerstitial nephritis. Acute tubulointerstitial nephritis (TIN) has been observed in patients taking proton pump inhibitors (PPIs) including rabeprazole sodium. Acute tubulointerstitial nephritis may occur at any point during PPI therapy and is generally attributed to an idiopathic hypersensitivity reaction. Acute tubulointerstitial nephritis can progress to renal failure. Discontinue rabeprazole sodium if acute tubulointerstitial nephritis develops.
Cyanocobalamin (vitamin B12) deficiency. Daily treatment with acid suppressing medicines over a long period of time (e.g. longer than 3 years) may lead to malabsorption of cyanocobalamin (vitamin B12) caused by hypo- or achlorhydria.
Hypomagnesaemia. Hypomagnesaemia, symptomatic and asymptomatic, has been reported rarely in patients treated with PPIs. Serious adverse events include tetany, arrhythmias, and seizures. In most patients, treatment of hypomagnesaemia required magnesium replacement and discontinuation of the PPI.
For patients expected to be on prolonged treatment or who take PPIs with medications such as digoxin or drugs that may cause hypomagnesaemia (e.g. diuretics), health care professionals may consider monitoring magnesium levels prior to initiation of PPI treatment and then periodically while treatment continues (see Section 4.8 Adverse Effects (Undesirable Effects)).
Hypomagnesaemia may lead to hypocalcaemia and/or hypokalaemia (see Section 4.8 Adverse Effects (Undesirable Effects)).
Fractures. Observational studies suggest that proton pump inhibitor (PPI) therapy may be associated with an increased risk for osteoporosis related fractures of the hip, wrist, or spine. The risk of fracture was increased in patients who received high dose, and long-term PPI therapy (a year or longer).
Concomitant use of rabeprazole with methotrexate. Literature suggests that concomitant use of PPIs with methotrexate (primarily at high dose; see methotrexate prescribing information) may elevate and prolong serum levels of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities. In such high doses methotrexate administration, a temporary withdrawal of the PPI may be considered in some patients.
Clostridium difficile. Treatment with proton pump inhibitors may possibly increase the risk of gastrointestinal infections such as Clostridium difficile.
Subacute cutaneous lupus erythematosus. Subacute cutaneous lupus erythematosus (SCLE) has been reported with the use of PPIs. If lesions occur, especially in sun-exposed areas of the skin, and if accompanied by arthralgia, the patient should seek medical help promptly and the health care professional should consider stopping rabeprazole. The occurrence of SCLE with previous PPI treatment may increase the risk of SCLE with other PPIs.
Fundic gland polyps. As with other PPIs, long-term use of rabeprazole is associated with an increased risk of fundic gland polyps (see Section 4.8 Adverse Effects (Undesirable Effects), Post-marketing data). Most fundic gland polyps are asymptomatic. Patients with large or ulcerated polyps may be at risk of gastrointestinal bleeding or small intestinal blockage. Use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated.
Use in hepatic impairment. No dosage adjustment is necessary for patients with hepatic impairment. While no evidence of significant drug related safety problems was observed in patients with hepatic impairment, it is advised to exercise caution when treatment with Pariet 10 is first initiated in patients with severe hepatic dysfunction (see Section 4.2 Dose and Method of Administration). Patients should be referred to their doctor for review if they have severe hepatic impairment (e.g. cirrhosis).
Use in renal impairment. No data available.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. PPI-induced decreases in gastric acidity may lead to increases in serum chromogranin A (CgA) levels, which may lead to erroneous interpretations of laboratory results in investigations for neuroendocrine tumors. To avoid this interference, temporarily stop tradename treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Effect of rabeprazole sodium on other drugs - demonstrated interactions. In vitro studies with human liver microsomes indicated that rabeprazole sodium is metabolised by isoenzymes of CYP450 (CYP2C19 and CYP3A4).
Patients may need to be monitored when the following drugs are taken together with Pariet 10:
Clopidogrel. Clopidogrel is metabolised to its active metabolite by CYP2C19. Inhibition of CYP2C19 by rabeprazole would be expected to result in reduced drug levels of the active metabolite of clopidogrel and a reduction in its antiplatelet activity and therefore its clinical efficacy. Concomitant use of rabeprazole with clopidogrel should be discouraged.
Cyclosporin. In vitro incubations employing human liver microsomes indicated that rabeprazole inhibited cyclosporin metabolism with an IC50 of 62 micromolar, a concentration that is over 50 times higher than the Cmax in healthy volunteers following 14 days dosing with 20 mg rabeprazole. Although in vitro studies may not always be predictive of an in vivo status these findings indicate that no interaction is expected between rabeprazole and cyclosporin.
Methotrexate. Case reports, published population pharmacokinetic studies, and retrospective analyses suggest that concomitant administration of PPIs and methotrexate (primarily at high dose; see methotrexate prescribing information) may elevate and prolong serum levels of methotrexate and/or its metabolite hydroxymethotrexate. However, no formal drug interaction studies of methotrexate with PPIs have been conducted.
Digoxin. A 22% increase in trough digoxin levels was observed in normal subjects given both drugs concomitantly.
Ketoconazole. A 33% decrease in ketoconazole levels was observed in normal subjects given both drugs concomitantly.
Atazanavir. Coadministration of atazanavir with other proton pump inhibitors resulted in a substantial reduction in atazanavir exposure. The absorption of atazanavir is pH dependent. Therefore, Pariet 10 should not be coadministered with atazanavir.
Mycophenolate mofetil. Coadministration of proton pump inhibitors with mycophenolate mofetil in healthy and transplant patients has been reported to reduce the exposure to the active metabolite, mycophenolic acid. This is possibly due to a decrease in mycophenolate mofetil solubility at an increased gastric pH. The clinical relevance of reduced mycophenolic acid exposure on organ rejection has not been established in transplant patients receiving proton pump inhibitors and mycophenolate mofetil. Use rabeprazole sodium with caution in transplant patients receiving mycophenolate mofetil.
Effect of rabeprazole sodium on other drugs - theoretical interactions. Rabeprazole sodium produces sustained inhibition of gastric acid secretion. An interaction with compounds whose absorption depends on gastric pH may occur due to the magnitude of acid suppression seen with rabeprazole sodium.
Effect of rabeprazole sodium on other drugs - potential interactions that have been excluded. Studies in healthy subjects have shown that rabeprazole sodium does not have clinically significant interactions with other drugs metabolised by the CYP450 system. These studies included the drugs warfarin and theophylline (as single oral doses), phenytoin (as a single intravenous dose with supplemental oral dosing), diazepam (as a single intravenous dose) and amoxycillin (as single and multiple oral doses).
Taking Pariet 10 with antacids produces no clinically relevant changes in plasma rabeprazole sodium concentrations.
Plasma concentrations of rabeprazole and the active metabolite of clarithromycin are increased by 24% and 50% respectively during concomitant administration. This is considered to be a useful interaction during H. pylori eradication.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Rabeprazole at intravenous doses up to 30 mg/kg/day (plasma AUC of 8.8 microgram.hr/mL, about 10 times the human exposure at 20 mg/day) was found to have no effect on fertility and reproductive performance of male and female rats.
Use in pregnancy. (Category B1)
Teratology studies have been performed in rats at intravenous doses up to 50 mg/kg/day (plasma AUC of 11.8 microgram.hr/mL, about 13 or 6.5 times the human exposure at 20 mg/day and 40 mg/day respectively), and rabbits at intravenous doses up to 30 mg/kg/day (plasma AUC of 7.3 microgram.hr/mL, about 8 or 4 times the human exposure at 20 mg/day and 40 mg/day respectively) and have revealed no evidence of impaired fertility or harm to the foetus due to rabeprazole. There are no adequate and well controlled studies in pregnant women and post-marketing experience is very limited. Rabeprazole sodium should be used in pregnancy only if the potential benefit justifies the potential risk to the foetus.
Use in lactation. Following intravenous administration of 14C-labelled rabeprazole to lactating rats, radioactivity in milk reached levels that were about 2 to 7-fold higher than levels in the blood. Administration of rabeprazole to rats in gestation and during lactation at doses of 400 mg/kg/day (about 195 or 85 times a 20 mg or 40 mg human dose based on mg/m2) resulted in decreases in bodyweight gain of the pups.
It is not known whether rabeprazole sodium is excreted in human breast milk and there are no studies in lactating women. Since many drugs are excreted in milk and because of the potential for adverse reactions to nursing infants from rabeprazole sodium, a decision should be made to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Throughout this section, adverse reactions are presented. Adverse reactions are adverse events that were considered to be reasonably associated with the use of rabeprazole based on the comprehensive assessment of the available adverse event information. A causal relationship with rabeprazole cannot be reliably established in individual cases. Further, because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Clinical trials. Rabeprazole was generally well tolerated during clinical trials. The observed side effects have generally been mild or moderate and transient in nature. In the majority of cases, the incidence of the adverse events in the rabeprazole treatment group was equal to or less than that observed in the placebo control treatment group.
Only headaches, diarrhoea, abdominal pain, asthenia, flatulence, rash and dry mouth have been associated with the use of rabeprazole.
The adverse events, which may or may not be causally related to rabeprazole, reported in clinical trials are listed below in descending order of frequency.
Common (≥ 1% and < 10%). Nervous system. Headache, dizziness.
Gastrointestinal. Diarrhoea, nausea, abdominal pain, flatulence, vomiting, constipation.
Respiratory. Rhinitis, pharyngitis, cough.
Musculoskeletal. Non-specific pain, back pain, myalgia.
Skin. Rash.
Other. Asthenia, flu-like syndrome, infection, insomnia, chest pain.
Uncommon (≥ 0.1% and < 1%). Gastrointestinal. Dyspepsia, eructation, dry mouth.
Respiratory. Sinusitis, bronchitis.
Musculoskeletal. Arthralgia, leg cramps.
Urinary. Urinary tract infection.
Other. Fever, nervousness, somnolence, chills, peripheral oedema.
Rare (≥ 0.01% and < 0.1%). Gastrointestinal. Anorexia, gastritis, weight gain, stomatitis.
Skin. Pruritus, sweating.
Special senses. Vision or taste disturbances.
Haematologic. Leucocytosis.
Other. Depression.
Post-marketing data. In addition to the adverse reactions reported during clinical studies and listed above, the following adverse reactions have been reported during post-marketing experience.
Erythema and rarely bullous reactions, urticarial skin eruptions and acute systemic allergic reactions, for example facial swelling, hypotension and dyspnoea have been reported in patients treated with rabeprazole. These usually resolved after discontinuation of therapy.
Erythema multiforme, tubulointerstitial nephritis (with possible progression to renal failure), gynaecomastia, myalgia and potential allergic reactions including anaphylactic reactions have been reported rarely. Blood dyscrasia including thrombocytopenia, neutropenia, leukopenia, pancytopenia, agranulocytosis and bicytopenia have been reported rarely. Hypomagnesaemia has also been reported rarely. Hypocalcaemia and/or hypokalaemia have been reported, which may be related to the occurrence of hypomagnesaemia (see Section 4.4 Special Warnings and Precautions for Use).
There have also been reports of increased hepatic enzymes and serious hepatic dysfunction such as hepatitis and jaundice. Rare reports of hepatic encephalopathy have been received in patients with underlying cirrhosis.
There have been very rare reports of toxic epidermal necrolysis (TEN), Stevens-Johnson syndrome and bullous rashes including subacute cutaneous lupus erythematosus.
There have been post-marketing reports of bone fractures and post-marketing reports of subacute cutaneous lupus erythematosus (SCLE) and fundic gland polyps (see Section 4.4 Special Warnings and Precautions for Use).
Gastrointestinal disorders. Frequency not known: withdrawal of long-term PPI therapy can lead to aggravation of acid-related symptoms and may result in rebound acid hypersecretion.
Metabolism and nutrition disorders. Hyponatraemia.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Experience to date with deliberate or accidental overdose is limited. The maximum established exposure has not exceeded 60 mg twice daily, or 160 mg once daily. Effects are generally minimal, representative of the known adverse event profile, and reversible without further medical intervention. No specific antidote is known. Rabeprazole sodium is extensively protein bound and is therefore not readily dialysable. As in any case of overdose, treatment should be symptomatic and general supportive measures should be used.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Rabeprazole sodium is a substituted benzimidazole and belongs to the class of proton pump inhibitors.
Rabeprazole sodium suppresses gastric acid secretion by the specific inhibition of the H+/K+-ATPase enzyme (proton pump) at the secretory surface of the gastric parietal cell thereby blocking the final step of acid production. This effect is dose related and leads to inhibition of both basal and stimulated acid secretion irrespective of the stimulus. Animal studies indicate that after administration, rabeprazole sodium rapidly disappears from both the plasma and gastric mucosa.
Mechanism of action. Anti-secretory activity. Oral administration of a 20 mg dose of Pariet provides rapid and effective reduction of gastric acid secretion. The onset of the antisecretory effect occurs within one hour with the maximum effect occurring within two to four hours. Inhibition of basal and food stimulated acid secretion 23 hours after the first dose of rabeprazole sodium is 69% and 82% respectively, and the duration of inhibition lasts up to 48 hours. The duration of pharmacodynamic action is much longer than the pharmacokinetic half-life (approximately one hour) would predict. This effect is probably due to the prolonged binding of rabeprazole sodium to the parietal H+/K+ ATPase enzyme. The inhibitory effect of rabeprazole sodium on acid secretion increases slightly with repeated once daily dosing, achieving steady-state inhibition after three days. When the drug is discontinued, secretory activity normalises over 2 to 3 days.
Serum gastrin effects. In clinical studies, patients were treated once daily with 10 or 20 mg rabeprazole sodium for up to 12 months' duration. Serum gastrin levels increased during the first 2 to 8 weeks reflecting the inhibitory effects on acid secretion. Gastrin values returned to pretreatment levels, usually within 1 to 2 weeks after discontinuation of therapy. In a maintenance study, which was subsequently extended up to 5 years' duration, serum gastrin levels were only modestly raised in most patients.
Enterochromaffin-like (ECL) cell effects. Increased serum gastrin secondary to antisecretory agents stimulates proliferation of gastric ECL cells which, over time, may result in ECL cell hyperplasia in rats and mice and gastric carcinoids in rats, especially females (see Section 5.3 Preclinical Safety Data).
In over 400 patients treated with Pariet (10 or 20 mg/day) for up to one year, the incidence of ECL cell hyperplasia increased with time and dose, which is consistent with the pharmacological action of the proton pump inhibitor. No patient developed the adenomatoid, dysplastic or neoplastic changes of ECL cells in the gastric mucosa. No patient developed the carcinoid tumours observed in rats.
Clinical trials. Symptomatic gastroesophageal reflux disease (GORD). On-demand treatment was assessed in a European multicentre, double blind placebo-controlled randomised withdrawal study (n = 418) in endoscopically negative patients.
Following an acute open label phase, patients were randomised to receive rabeprazole 10 mg or placebo taken once daily, when required, over a six month period. Efficacy of rabeprazole 10 mg on-demand, in patients with complete heartburn relief at baseline was primarily evaluated by the unwillingness to continue the trial because of inadequate heartburn control. Overall, the proportion of patients discontinuing due to inadequate heartburn control was significantly higher for placebo (20%) compared to rabeprazole (6%) (p < 0.00001).
Patients were instructed to take study drug until they had experienced a full 24 hours free of heartburn, most patients in the rabeprazole group had maximum episode duration of 4 days or less. In addition, antacid use was about 2-fold higher in the placebo group than in the rabeprazole group (p = 0.0011). Treatment failure was associated with an increased antacid consumption.
The efficacy of rabeprazole 10 mg or 20 mg daily versus placebo was assessed in a randomised, double blind, parallel group study (n = 199) over a four week interval in subjects with moderately severe gastroesophageal reflux disease and grade 0 or 1 oesophagitis at endoscopy. The primary efficacy variable was defined as the time in days for subjects to achieve their first 24 hour interval without heartburn. Results showed that the average times were 6.541 +/- 0.923 days for rabeprazole 10 mg, 10 +/- 1.258 days for rabeprazole 20 mg and 16.347 +/- 1.105 days for placebo. There was no significant difference between the rabeprazole groups.
5.2 Pharmacokinetic Properties
Absorption. Pariet 10 tablets are enteric coated to allow rabeprazole sodium, which is acid labile, to pass through the stomach intact. Absorption is rapid, with peak plasma levels of rabeprazole sodium occurring approximately 3.5 hours after a 20 mg dose. Peak plasma concentrations (Cmax) of rabeprazole sodium and AUC are linear over the dose range of 10 mg to 40 mg.
Absolute bioavailability of an oral 20 mg dose (compared to intravenous administration) is about 52%, largely due to presystemic metabolism. Additionally, the bioavailability does not appear to increase with repeat administration. In healthy subjects, the plasma half-life is approximately one hour (range 0.7 to 1.5 hours) and the total body clearance is estimated to be 283 ± 98 mL/min.
Distribution. Rabeprazole sodium is approximately 97% bound to human plasma proteins. After intravenous administration the volume of distribution is 0.34 L/kg.
Metabolism. Rabeprazole sodium is metabolised through the cytochrome P450 (CYP450) hepatic drug metabolism system (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). In humans, the thioether (M1) and carboxylic acid (M6) are the main plasma metabolites with the sulphone (M2), desmethyl thioether (M4) and mercapturic acid conjugate (M5) minor metabolites observed at lower levels. Only the desmethyl metabolite (M3) has a small amount of antisecretory activity, but its presence in plasma is minimal.
Excretion. Following a single 20 mg 14C-labelled oral dose of rabeprazole sodium, no unchanged drug was excreted in the urine. Approximately 90% of the dose was eliminated in urine mainly as the two metabolites: a mercapturic acid conjugate (M5) and a carboxylic acid (M6), plus two unknown metabolites also found in the species used in the toxicology studies. The remainder of the dose was recovered in faeces. Total recovery was 99.8%. This suggests low biliary excretion of the metabolites; with biotransformation and urinary excretion of water soluble metabolites as the primary route of elimination.
Special populations. Renal disease. In patients with stable, endstage, renal failure requiring maintenance haemodialysis (creatinine clearance ≤ 5 mL/min/1.73 m2), the pharmacokinetics of rabeprazole sodium was very similar to that in healthy volunteers.
Hepatic disease. In a single dose study of 10 patients with chronic mild to moderate compensated cirrhosis of the liver who were administered a 20 mg dose of rabeprazole, AUC0-24 was approximately doubled, the elimination half-life was 2 to 3-fold higher, and total body clearance was decreased to less than half compared to values in healthy men.
In a multiple dose study of 12 patients with mild to moderate hepatic impairment administered 20 mg rabeprazole once daily for eight days, AUC0-∞ and Cmax values increased approximately 30% compared to values in healthy age and gender matched subjects. These increases were not statistically significant.
No information exists on rabeprazole disposition in patients with severe hepatic impairment. Please see Section 4.2 Dose and Method of Administration for information on dosage adjustments in patients with hepatic impairment.
Geriatrics. Elimination of rabeprazole sodium was decreased in the elderly. Following 7 days of daily dosing with 20 mg of rabeprazole sodium, the AUC approximately doubled and the Cmax increased by 60% as compared to young healthy volunteers. However, there was no evidence of rabeprazole sodium accumulation.
5.3 Preclinical Safety Data
Note. In the following section, the relative exposure levels in animals have been calculated using a human dose of 20 mg/day.
Genotoxicity. Rabeprazole was positive in assays for gene mutations (the AMES test, forward gene mutation tests in Chinese hamster ovary cells (CHO/HGPRT) and mouse lymphoma cells (L5178Y/TK+/-). Its demethylated-metabolite was also positive in the AMES test. Rabeprazole was negative in assays for chromosomal damage (the in vitro Chinese hamster lung cell chromosome aberration test, the in vivo mouse micronucleus test), and in vitro and ex vivo rat hepatocyte unscheduled DNA synthesis (UDS) tests.
Carcinogenicity. In an 88/104 week carcinogenicity study in CD-1 mice, rabeprazole at oral doses up to 100 mg/kg/day did not produce any increased tumour occurrence. The highest tested dose produced a systemic exposure to rabeprazole (AUC) of 1.40 microgram.hr/mL which is 1.6 times the human exposure at 20 mg/day.
In a 104 week carcinogenicity study in SD rats, males were treated with oral doses of 5, 15, 30 and 60 mg/kg/day and females with 5, 15, 30, 60 and 120 mg/kg/day. Rabeprazole produced gastric enterochromaffin-like (ECL) cell hyperplasia in male and female rats and ECL cell carcinoid tumours in female rats at all doses. The lowest dose (5 mg/kg/day) produced a systemic exposure to rabeprazole (AUC) of about 0.1 microgram.hr/mL which is about 0.1 times the human exposure at 20 mg/day. In male rats, no treatment related tumours were observed at doses up to 60 mg/kg/day producing a rabeprazole plasma exposure (AUC) of about 0.2 microgram.hr/mL (0.2 times the human exposure at 20 mg/day).
6 Pharmaceutical Particulars
6.1 List of Excipients
Pariet 10 tablets contain the inactive ingredients mannitol, magnesium oxide, hyprolose, magnesium stearate, ethylcellulose, hypromellose phthalate, diacetylated monoglycerides, purified talc, titanium dioxide and carnauba wax, iron oxide red and Edible Ink Gray F6.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine. (See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Pariet 10 tablets should be stored below 25°C. Do not refrigerate.
6.5 Nature and Contents of Container
Pariet 10 tablets are presented in blister packs of 7 and 14 tablets. Tablets are presented in an aluminium/aluminium blister.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
The chemical name for rabeprazole sodium is (±) 2-[{4-(3-methoxypropoxy)-3-methylpyridin-2-yl}-methylsulphinyl]-1H-benzimidazole sodium. Rabeprazole has one chiral centre and is a racemate of two enantiomers.
Its solubility in water is pH dependent, being very soluble in water at pH 9 to 11, and only slightly soluble in water at pH 8. It is very soluble in methanol, freely soluble in dichloromethane and practically insoluble in hexane.
Chemical structure.
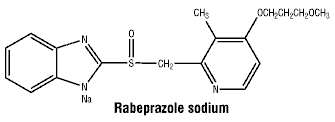
MW: 381.43.
CAS number. CAS-117976-89-3 (rabeprazole).
CAS-117976-90-6 (rabeprazole sodium).
7 Medicine Schedule (Poisons Standard)
S3 - Pharmacist Only Medicine.
Date of First Approval
01 April 2010
Date of Revision
22 January 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.