Patent Blue V
Brand Information
| Brand name | Patent Blue V |
| Active ingredient | Patent blue V |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Patent Blue V.
Summary CMI
PATENT BLUE V
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about receiving this medicine, speak to your doctor or pharmacist.
1. Why am I given PATENT BLUE V?
PATENT BLUE V contains the active ingredient patent blue V sodium salt. PATENT BLUE V is used to reveal vessels of your body in order to improve the visualisation of certain vessels. PATENT BLUE V is used to prepare for a radiological examination or as a direct aid to diagnosis of some diseases.
For more information, see Section 1. Why am I given PATENT BLUE V? in the full CMI.
2. What should I know before I am given PATENT BLUE V?
You must not be given PATENT BLUE V if you are allergic to the active ingredient (patent blue), to another triphenylmethane dye or any of the other ingredients of Patent Blue V listed at the end of the CMI.
You must not be given PATENT BLUE V if you are pregnant.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am given PATENT BLUE V? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with PATENT BLUE V and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How am I given PATENT BLUE V?
PATENT BLUE V will be diluted with suitable fluids and injected by a doctor under the skin (subcutaneous route).
More instructions can be found in Section 4. How am I given PATENT BLUE V? in the full CMI.
5. What should I know while receiving PATENT BLUE V?
| Things you should do | Tell your doctor if:
|
| Looking after your medicine | The clinic will store PATENT BLUE V as recommended by the manufacturer. |
For more information, see Section 5. What should I know while receiving PATENT BLUE V? in the full CMI.
6. Are there any side effects?
Serious side effects can include sudden swelling of the face, neck pr other parts of the body; breathing difficulties with a cough (bronchospasm). Rare but more serious allergic reactions include drop in blood pressure that can be life-threatening (allergic shock), and sudden fainting.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I given PATENT BLUE V?
PATENT BLUE V is a sterile dye solution for injection. It belongs to the group of triphenylmethane dyes used to reveal vessels of your body in order to improve the visualisation of certain vessels.
PATENT BLUE V is used by your doctor to prepare for a radiological examination or as a direct aid to diagnosis of some diseases.
2. What should I know before I am given PATENT BLUE V?
Warnings
You must not be given PATENT BLUE V if:
- You are allergic to the active ingredient (patent blue), to another triphenylmethane dye or any of the other ingredients of Patent Blue V listed at the end of the CMI.
- You are pregnant.
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have any other medical conditions
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
You must not be given PATENT BLUE V if you are pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with PATENT BLUE V and affect how it works. These include medicines used to treat high blood pressure, such as beta-blocker or ACE inhibitor agents.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect PATENT BLUE V.
4. How am I given PATENT BLUE V?
How much is given
- PATENT BLUE V will be given to you in a hospital or clinic.
- The doctor will determine the dose that you will receive and will supervise the injection.
When will PATENT BLUE V be given?
- PATENT BLUE V will be diluted with suitable fluids and then administered to you under the skin before your radiological examination.
If you are given too much PATENT BLUE V
It is highly unlikely that you will receive too much PATENT BLUE V as it will be given to you by a trained healthcare professional.
If you think that you have been given too much PATENT BLUE V, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while receiving PATENT BLUE V?
Things you should do
Follow carefully the directions given to you by your doctor or other medical staff.
Call your doctor straight away if you notice any of the following:
- Swelling of the face, neck or other parts of the body
- Lightheadedness (low blood pressure)
- Breathing difficulties
- Skin rash and itching
Each of the signs above may be a warning of the start of a shock.
Looking after your medicine
The clinic will store PATENT BLUE V as recommended by the manufacturer.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergic reactions such as:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is only available at radiological examination clinics.
What PATENT BLUE V contains
| Active ingredient (main ingredient) | Patent blue V sodium salt |
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What PATENT BLUE V looks like
PATENT BLUE V is a sterile, concentrated deep blue solution.
PATENT BLUE V is available in packs of 5 clear glass ampoules of 2 mL each.
AUST R 27539
Who distributes PATENT BLUE V
Guerbet Australia Pty Ltd
166 Epping Road, Level 2
Lane Cove, NSW, 2066
Australia.
This leaflet was prepared in December 2025.
Brand Information
| Brand name | Patent Blue V |
| Active ingredient | Patent blue V |
| Schedule | Unscheduled |
MIMS Revision Date: 01 January 2022
1 Name of Medicine
Patent Blue V.
2 Qualitative and Quantitative Composition
Patent Blue V injection solution contains 25 mg/mL (2.5% w/v) of Patent Blue V (sodium salt) as the active ingredient.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection.
Patent Blue V injection solution is a deep blue sterile, isotonic solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Colours the lymph vessels so that they can then be injected with an X-ray contrast medium.
4.2 Dose and Method of Administration
For subcutaneous use only.
2 mL of Patent Blue V injection solution is diluted with an equal volume of sterile normal saline (an alternative method advocates dilution with 1% lignocaine hydrochloride solution) and 0.5 mL of diluted dye is injected into the subcutaneous tissue of each interdigital web space.
When the lower extremities are being examined, 1 mL is also injected below the tip of each malleolus. The lymphatics should then be visible through the skin and it is important that there should be no accidental spillage of the dye.
Patent Blue V may be used for children at the surgeon's discretion.
After the administration of Patent Blue V, the patient must be monitored for at least 30 minutes.
Note. After injection of the dye a bluish coloration of the skin occurs, which normally disappears within 24 to 48 hours. More persistent coloration which should not be confused with cyanosis, may occasionally be seen in cases of lymphatic stasis or circulatory disorder, this persistence may be avoided by using the minimum quantity of dye. An area of blue coloration can persist around the injection site for 8 to 10 days.
4.3 Contraindications
Hypersensitivity to Patent Blue V, triphenylmethane dyes or any of the excipients listed (see Section 6.1).
Pregnancy.
4.4 Special Warnings and Precautions for Use
There is a risk of allergic reactions regardless of the administration route or dose. Patent Blue V may cause minor or major immediate allergic reactions that may be life-threatening or even fatal (anaphylactic shock). They are often unpredictable, but they occur more frequently in patients with a history of hypersensitivity reactions to Patent Blue V or related triphenylmethane dyes contained in medicine, food and cosmetics. The indication should be very carefully assessed in these predisposed patients. In such circumstances it is advisable to administer a corticosteroid or H1-type antihistamine premedication. The preventative effect of premedication with corticosteroids and H1-type antihistamines is not entirely dependable, as only a partial (though significant) inhibition of mediators involved in anaphylactoid reactions can be attained.
The risk of a major reaction implies that emergency measures must be immediately available especially in patients on beta blockers in whom adrenaline and vascular perfusion would be insufficiently effective. Therefore, Patent Blue V must only be administered in hospital or a clinic capable of adequate treatment.
It would also seem prudent to test for hypersensitivity by injecting a very small volume of the solution and waiting a few moments to ascertain if there is a reaction.
Before administration of Patent Blue V:
Identify patients at risk by enquiring if there is a previous history of allergy or intolerance to Patent Blue V or related triphenylmethane dyes.
Insert an indwelling venous catheter.
Throughout the examination, maintain:
Medical monitoring.
An indwelling intravenous catheter.
Medicines and equipment for resuscitation readily available.
After administration of Patent Blue V, the patient must be monitored for at least 30 minutes.
Use in the elderly. No data available.
Paediatric use. Patent Blue V may be used for children at the surgeon's discretion.
Effects on laboratory tests. The measurement of partial oxygen pressure measured by spectrophotometry may show a transient false decrease of 5 to 10% below baseline values, during an examination with Patent Blue V. When in doubt, arterial blood gases should be checked. The value of serum methaemoglobin measured by the same spectrophotometric method may be falsely increased.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Beta blockers, vasoactive substances, angiotensin-converting enzyme inhibitors, angiotensin receptor antagonists.
These medicinal products reduce the efficacy of cardiovascular compensation mechanisms for haemodynamic disorders. The physician must be aware of this before injecting Patent Blue V and emergency measures must be available.
The value of partial oxygen pressure measured by spectrophotometry may show a transient false decrease of 5 to 10% below baseline values during examinations with Patent Blue. When in doubt, it is advisable to check by arterial blood gas analysis. The value of serum methaemoglobin measured by the same spectrophotometric method may be falsely increased.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. No reliable animal teratogenesis data are available. The currently available relevant data are not sufficient to evaluate a possible malformative or foetotoxic effect of Patent Blue V when it is administered during pregnancy. Consequently, the use of this medicinal product is contraindicated during pregnancy.
Use in lactation. It is not known whether Patent Blue V is excreted in breast milk.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Patent Blue V solution can provoke allergic reactions of varying degrees of severity. These reactions are rare and can be rapidly controlled with a corticosteroid. They occur immediately or a few minutes to several hours after injection of the dye.
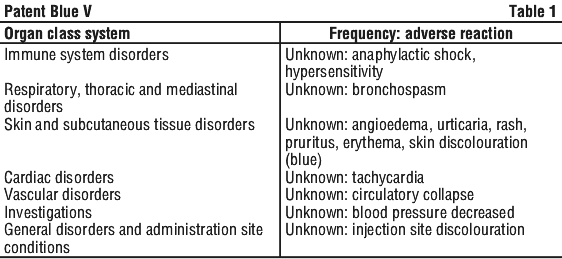
Immediate hypersensitivity reactions are possible. These reactions may involve one or more effects, occurring concomitantly or successively, and usually including cutaneous, respiratory and/or cardiovascular manifestations, each of which can be a warning sign of incipient shock and, in very rare instances, can even prove fatal. The most frequently reported effects in a context of hypersensitivity reaction include rash, pruritus, erythema, urticaria, angioedema (such as face oedema or laryngeal oedema), bronchospasm, tachycardia, blood pressure decreased, and circulatory collapse.
A bluish colouring of the integuments is observed after the injection, which disappears within 24 to 48 hours. In patients with lymph stasis or circulatory disorders, the colouring may last longer.
Nausea, hypotension and generalised muscle tremors have also been reported; laryngeal spasm, if it occurs, requires the use of a muscle relaxant and intubation. Rare but more serious allergic reactions include circulatory failure with a state of shock, dyspnoea and oedema of the glottis.
Post-marketing experience. The adverse effects are presented in Table 1, by system organ class and by frequency using the following categories: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1000 to < 1/100), rare (≥ 1/10,000 to < 1/100), very rare (< 1/10,000), not known (cannot be estimated from the available data).
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: lymphotropic dye.
The lymphotropic property of Patent Blue V 2.5% w/w results in the staining of local lymph vessels after subcutaneous injection.
Mechanism of action. No data available.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Carcinogenicity. No data available.
Genotoxicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium chloride, dibasic sodium phosphate dodecahydrate, water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
3 years.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
Patent Blue V is packed in type I clear glass ampoules of 2 mL volume fill.
Available in packs of 5.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. Patent Blue V (sodium salt) is N-(4-((4-(diethylamino)phenyl) (5-hydroxy-2,4-disulfophenyl) methylene)-2,5-cyclohexadien-1-ylidene) -N-ethylethanaminium, hydroxide, sodium salt.
CAS number. 20262-76-4.
7 Medicine Schedule (Poisons Standard)
Not scheduled.
Date of First Approval
21 October 1991
Date of Revision
18 November 2021
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.