Pentasa Sachets
Brand Information
| Brand name | Pentasa Sachets |
| Active ingredient | Mesalazine |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Pentasa Sachets.
Summary CMI
PENTASA® sachets
Consumer Medicine Information (CMI) summary
The full CMI (see from next page) has more details. If you are worried about taking this medicine, speak to your doctor or pharmacist.
1. Why am I taking PENTASA?
PENTASA contains the active ingredient mesalazine. PENTASA is taken to treat Crohn's disease and ulcerative colitis: diseases associated with inflammation, ulcers and sores in the bowel causing bleeding, stomach pain, and diarrhoea.
For more information, see Section 1. Why am I taking PENTASA? in the full CMI (see from next page), starting from page 2.
2. What should I know before I take PENTASA?
Do not take PENTASA if you have ever had an allergic reaction to mesalazine or any of the ingredients listed at the end of the CMI (Section 7 Product details).
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I take PENTASA? in the full CMI, starting from page 2.
3. What if I am taking other medicines?
Care may be needed when you are also taking other medicines.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI, starting from page 2.
4. How do I take PENTASA?
- Tear or cut to open the sachet along the dotted line.
- Empty the content of the sachet (granules) onto the tongue and wash down with water or juice to swallow without chewing or crushing the granules.
- Alternatively, mix the entire content of the sachet (granules) with yoghurt and consume immediately without chewing or crushing the granules. More instructions can be found in Section 4. How do I take PENTASA? in the full CMI, starting from page 2.
5. What should I know while taking PENTASA?
| Things you should do | Tell any doctor, dentist or pharmacist you visit that you are taking PENTASA Have all blood tests recommended by your doctor If you become pregnant while taking this medicine, tell your doctor immediately. |
| Things you should not do | Do not stop taking this medicine or change the dosage without checking with your doctor Do not take PENTASA to treat any other complaints unless your doctor tells you to Do not give your medicine to anyone else, even if they have the same condition as you. |
| Driving or using machines | PENTASA is not expected to affect your ability to drive a car or operate machinery. |
| Looking after your medicine | Keep your PENTASA in a cool dry place where the temperature stays below 25°C. |
For more information, see Section 5. What should I know while taking PENTASA? in the full CMI, starting from page 2.
6. Are there any side effects?
All medicines can have side effects. Side effects reported from PENTASA can be minor and temporary. However, some side effects may need medical attention.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI, starting from page 2.
Full CMI
PENTASA® sachets
Active ingredient: mesalazine
Consumer Medicine Information (CMI)
This leaflet provides important information about taking PENTASA.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about taking PENTASA.
Where to find information in this leaflet:
1. Why am I taking PENTASA?
2. What should I know before I take PENTASA?
3. What if I am taking other medicines?
4. How do I take PENTASA?
5. What should I know while taking PENTASA?
6. Are there any side effects?
7. Product details
1. Why am I taking PENTASA?
PENTASA contains the active ingredient, mesalazine. PENTASA is an anti-inflammatory agent, similar to aspirin, and is thought to work by reducing inflammation in the bowel.
PENTASA is taken to treat Crohn's disease and ulcerative colitis, diseases associated with inflammation, ulcers and sores in the bowel causing bleeding, stomach pain, and diarrhoea.
2. What should I know before I take PENTASA?
Warnings
Do not take PENTASA if:
- you are allergic to mesalazine, sulfasalazine, aspirin-like medicines or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can take this medicine
- you have a severe kidney or liver problem
- the expiry date printed on the pack has passed
- the packaging is torn or shows signs of tampering.
Your doctor should make you aware of the signs and symptoms that may indicate allergic reaction when taking PENTASA.
Check with your doctor if you:
- have a known allergy to PENTASA, sulfasalazine or aspirin-like medicines, or any of the ingredients listed at the end of this leaflet
- take any medicines for any other condition
- have a kidney or liver problem
- have a bleeding disorder
- have a history of asthma.
During treatment, you may be at risk of developing certain side effects.
It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor can discuss with you the risks and benefits involved with taking PENTASA. PENTASA should be taken with caution during pregnancy and breast-feeding and only if the potential benefits outweigh the possible risks in the opinion of the doctor. The underlying condition itself (inflammatory bowel disease) may increase risks for the pregnancy outcome.
Children
Do not give PENTASA to a child 12 years old or under.
The safety and effectiveness of PENTASA in this age group have not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
There is no information available on interactions between PENTASA and other medicines. However, mesalazine belongs to a group of medicines called salicylates that may interfere with some medicines.
Medicines that may have their effects increased if used with PENTASA include:
- anti-coagulants which are used to thin blood or stop blood clots (e.g. warfarin). When PENTASA is used with these medicines, it may increase the medicine's effects on thinning your blood
- sulphonylureas which are used to lower blood sugar and treat diabetes (e.g. glibenclamide, glipizide). When PENTASA is used with these medicines, it may increase the medicine's effect on lowering your blood sugar
- methotrexate which is used to treat some kinds of cancers, some skin conditions, and rheumatoid arthritis. When PENTASA is used with this medicine, it may increase the possibility of the medicine producing toxic effects on your body
- glucocorticoids which are used to treat inflammation, swelling or allergies (e.g. prednisolone). When PENTASA is used with these medicines, it may increase the undesirable gastric effects caused by these medicines.
Medicines that may have their effects decreased if used with PENTASA include:
- probenicid or sulfinpyrazone which are used to treat gout. When PENTASA is used with these medicines it may decrease the effectiveness of the medicine
- spirinolactone or furosemide, which are used to increase the amount of urine produced and to lower blood pressure. When PENTASA is used with these medicines it may decrease the effectiveness of the medicine
- rifampicin which is used to treat tuberculosis. When PENTASA is used with this medicine it may decrease the effectiveness of the medicine.
Medicines that may have a greater likelihood of reducing bone marrow activity if used with PENTASA include:
- azathioprine which is used to suppress the immune system
- mercaptopurine and tioguanine, used to treat leukaemia.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect PENTASA.
4. How do I take PENTASA?
This medicine helps to control your condition, but does not cure it. It is important to keep taking your medicine even if you feel well.
How much to take
- Recommended doses for various conditions are presented below.
- Your doctor will prescribe a dose based on your individual needs, and this may differ from these recommendations.
- Follow the instructions provided by your doctor and use PENTASA until your doctor tells you to stop.
Ulcerative Colitis
- For active disease (flare-up of symptoms) for ulcerative colitis, take up to 4 g PENTASA once a day or in divided doses.
- For maintenance (during remission) for ulcerative colitis, take up to 2 g PENTASA once a day or in divided doses.
Crohn's disease
- For active disease (flare-up of symptoms) for Crohn's disease, take up to 4 g PENTASA daily in divided doses.
- For maintenance (during remission) for Crohn's disease, take up to 4 g PENTASA daily in divided doses.
When to take PENTASA
- Take your medicine at the same time each day. This will help you remember when to take it.
- You can take PENTASA with or without food
- Continue taking your medicine for as long as your doctor tells you.
How to take PENTASA
- Tear or cut to open the sachet along the dotted line
- Empty the content of the sachet (granules) onto the tongue and wash down with water or juice to swallow without chewing or crushing the granules.
- Alternatively, mix the entire content of the sachet (granules) with yoghurt and consume immediately without chewing or crushing the granules.

Scan the QR code with your smartphone or tablet to access a video on administering PENTASA® Enema. Select the PENTASA® link on the website, enter the AUST R number from the PENTASA® box or Product details of this document, and scroll to resources. Alternatively, visit patients.ferring.com.au/gastroenterology/
Access available only within Australia.
If you forget to take PENTASA
PENTASA should be taken regularly at the same time each day.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
- If you are not sure what to do, ask your doctor or pharmacist.
- If you have trouble remembering to take PENTASA, ask your pharmacist for some hints.
If you take too much PENTASA
If you think that you have taken too much PENTASA, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Accident and Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while taking PENTASA?
Things you should do
Have all blood tests recommended by your doctor
- PENTASA may cause kidney, liver or blood problems in a few people. You should have regular blood tests to check your kidney function.
- Kidney stones may develop while taking PENTASA. Symptoms may include pain in the sides of the abdomen and blood in the urine.
- Take care to drink plenty of fluids while you are being treated with PENTASA.
If you are going to have surgery
- Tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking this medicine, tell your doctor immediately.
Remind any doctor, dentist or pharmacist you visit that you are taking PENTASA.
Things you should not do
- Do not stop taking this medicine or change the dosage suddenly without checking with your doctor. If you stop taking it suddenly, your condition may worsen.
- Do not take PENTASA to treat any other complaints unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
Driving or using machines
PENTASA is not expected to affect your ability to drive a car or operate machinery.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
- Keep your PENTASA in the packaging until it is time to take your next dose.
- If you take PENTASA out of the packaging it may not keep well.
- Keep your PENTASA in a cool dry place (temperature below 25°C), away from moisture, heat or sunlight.
Do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to take this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not take this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. Most side effects reported by patients after taking PENTASA (mesalazine) are minor and temporary. However, some side effects may need medical attention.
See the information below and, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Common side effects (affect more than 1 in 100 users):
| These side effects are not usually serious but can become serious Speak to your doctor if you have any of these side effects |
Serious side effects
| Serious side effects | What to do |
Rare side effects (affect less than 1 in 1,000 users):
| Call your doctor straight away, if you notice any of these serious side effects |
Allergic reaction (very rare – affect less than 1 in 10,000 users):
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What PENTASA contains
| Active ingredient (main ingredient) | mesalazine |
| Other ingredients (inactive ingredients) | povidone ethylcellulose |
Do not take this medicine if you are allergic to any of these ingredients.
This medicine does not contain sucrose, gluten, tartrazine or any other azo dyes.
What PENTASA looks like
PENTASA Sachets contain short and cylindrical (or tube) shaped granules that are white-grey to pale white brown in colour.
PENTASA 1 g Sachets are supplied in packs of 30, 100 and 120. (AUST R 161063)
PENTASA 2 g Sachets are supplied in packs of 15 and 60. (AUST R 161064)
PENTASA 4 g Sachets are supplied in packs of 8 and 30. (AUST R 216259)
Not all pack sizes are distributed in Australia.
Who distributes PENTASA
PENTASA Sachets are supplied in Australia by:
Ferring Pharmaceuticals Pty Ltd
Suite 2, Level 1, Building 1
20 Bridge Street
Pymble, NSW 2073, Australia.
PENTASA® is a registered trademark of Ferring B.V.
® = Registered trademark
This leaflet was prepared in October 2025
AU-PA-2100053_v4.0
Brand Information
| Brand name | Pentasa Sachets |
| Active ingredient | Mesalazine |
| Schedule | S4 |
MIMS Revision Date: 01 December 2025
1 Name of Medicine
Mesalazine.
2 Qualitative and Quantitative Composition
Pentasa prolonged release tablets contain either 0.5 g or 1 g mesalazine as the active ingredient, and the following inactive excipients: magnesium stearate, purified talc, povidone, ethylcellulose, microcrystalline cellulose.
Pentasa prolonged release granules in sachets contain either 1 g, 2 g or 4 g mesalazine as the active ingredient, and the following inactive excipients: ethylcellulose, povidone.
3 Pharmaceutical Form
Pentasa 0.5 g prolonged release tablets are presented as white grey to pale brown, speckled round tablets with break mark and embossing: 500 mg on one side, PENTASA on the other side.
Pentasa 1 g prolonged release tablets are presented as white-grey to pale brown, speckled, oval tablets. Embossing on both sides: PENTASA.
Pentasa 1 g, 2 g, and 4 g prolonged release granules are presented as cylindrical shaped granules that are white-grey to pale white brown in colour.
4 Clinical Particulars
4.1 Therapeutic Indications
Treatment of mild to moderate ulcerative colitis and Crohn's disease and maintenance of remission.
4.2 Dose and Method of Administration
Ulcerative colitis. Treatment of active disease. Adults. Individual dosage, up to 4 g given once daily or in divided doses.
Maintenance treatment. Adults. 2 g once daily or individual dosage, starting with 1.5-2 g daily in divided doses.
Crohn's disease. Treatment of active disease. Adults. Individual dosage, up to 4 g daily in divided doses.
Maintenance treatment. Adults. Individual dosage, up to 4 g daily in divided doses.
Method of administration. Do not crush or chew the tablets or granules.
Tablets. To facilitate swallowing, the tablets may be dispersed in 50 mL of cold water. Stir and drink immediately.
Sachets. The content of the sachet should be emptied onto the tongue and washed down with some water or juice.
Alternatively, the entire content of the sachet can be taken with yoghurt and consumed immediately.
4.3 Contraindications
Hypersensitivity to mesalazine or any of the excipients or salicylates.
Severe liver or renal impairment.
4.4 Special Warnings and Precautions for Use
Most patients who are intolerant or hypersensitive to sulfasalazine are able to take Pentasa without the risk of similar reactions. However, caution is recommended when treating patients allergic to sulfasalazine because of the risk of allergy to salicylates (also see Section 4.3 Contraindications). Severe cutaneous adverse reactions (SCARs), including drug reaction with eosinophilia and systematic symptoms (DRESS). Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), have been reported in association with mesalazine treatment.
Treatment should be discontinued immediately in cases of acute intolerance reactions such as nausea, exacerbation of diarrhoea, abdominal cramps, acute abdominal pain, fever, severe headache, and/or the first appearance of signs and symptoms of severe skin reactions, such as skin rash, mucosal lesions, or any other signs of hypersensitivity.
Mesalazine-induced cardiac hypersensitivity reactions (myocarditis and pericarditis) have been reported rarely.
Serious blood dyscrasias have been reported rarely with mesalazine. Haematological investigations should be performed if the patient develops unexplained bleeding, bruising, purpura, anaemia, fever or sore throat. Treatment should be stopped if there is suspicion or evidence of blood dyscrasia (also see Section 4.8 Adverse Effects (Undesirable Effects)). Also, a blood test for differential blood count is recommended prior to and during treatment, at the discretion of the treating physician. As stated in Section 4.5 Interactions with Other Medicines and Other Forms of Interactions, concomitant treatment with mesalazine can increase the risk of blood dyscrasia in patients receiving azathioprine, or 6-mercaptopurine or tioguanine. Treatment should be discontinued on suspicion or evidence of these adverse reactions (also see Section 4.8 Adverse Effects (Undesirable Effects)).
Patients with inflammatory bowel disease are at risk of developing nephrolithiasis. Cases of nephrolithiasis have been reported with the use of mesalazine including kidney stones composed entirely of mesalazine. It is recommended to ensure adequate fluid intake during treatment.
Idiopathic intracranial hypertension (pseudotumor cerebri) has been reported in patients receiving mesalazine. Patients should be warned for signs and symptoms of idiopathic intracranial hypertension, including severe or recurrent headache, visual disturbances or tinnitus. If idiopathic intracranial hypertension occurs, discontinuation of mesalazine should be considered.
Use in hepatic impairment. Caution is recommended in patients with impaired liver function (also see Section 4.3 Contraindications). Liver function parameters like ALT or AST should be assessed prior to and during treatment, at the discretion of the treating physician.
Mesalazine may produce red-brown urine discolouration after contact with sodium hypochlorite bleach (e.g. in toilets cleaned with sodium hypochlorite contained in certain bleaches).
Use in renal impairment. Mesalazine is not recommended for use in patients with renal impairment (also see Section 4.3 Contraindications). Renal function should be monitored regularly in all patients (e.g. serum creatinine, urinalysis for protein) especially during the initial phase of treatment. Mesalazine-induced nephrotoxicity should be suspected in patients developing renal dysfunction during treatment. Mesalazine treatment should be discontinued immediately if renal function deteriorates. The concurrent use of other known nephrotoxic agents should increase monitoring frequency of renal function.
Use in the elderly. Age related factors (such as altered renal and hepatic function as described above and polypharmacy) should be taken into consideration.
Paediatric use. Pentasa should not be used in children 12 years of age and under, as there is limited experience with this age group.
Effects on laboratory tests. Use of mesalazine may lead to spuriously elevated test results when measuring urinary normetanephrine by liquid chromatography with electrochemical detection, because of the similarity in the chromatograms of normetanephrine and mesalazine's main metabolite, N-acetyl-5-aminosalicylic acid. An alternative selective assay for normetanephrine should be considered.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Whilst there are no data on interactions between Pentasa and other medicines, in common with other salicylates, interactions may occur during concomitant administration of mesalazine and the following medicines:
Coumarin type anticoagulants (e.g. warfarin sodium). Possible potentiation of the anticoagulant effect (increasing the risk of gastrointestinal haemorrhage).
Glucocorticoids. Possible increase in undesirable gastric effects.
Sulfonylureas. Possible increase in the blood glucose lowering effects.
Methotrexate. Possible increase in toxic potential of methotrexate.
Probenecid or sulfinpyrazone. Possible attenuation of the uricosuric effects.
Spironolactone or furosemide (frusemide). Possible attenuation of the diuretic effects.
Rifampicin. Possible attenuation of the tuberculostatic effects.
Combination therapy with Pentasa and azathioprine, or 6-mercaptopurine or tioguanine have in several studies shown a higher frequency of myelosuppressive effects, and an interaction seems to exist. However, the mechanism behind the interaction is not fully established. Regular monitoring of white blood cells is recommended and the dosage regime of the thiopurines should be adjusted accordingly.
The concomitant use of mesalazine with other known nephrotoxic agents, such as non-steroidal anti-inflammatory drugs (NSAIDS) and azathioprine, may increase the risk of renal reactions.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Oral administration of mesalazine at doses up to 400 mg/kg/day to male rats prior to mating and female rats from prior to mating through gestation and lactation did not affect fertility or elicit embryofetal toxicity.
Use in pregnancy. (Category C)
Oral administration of mesalazine during organogenesis in rats and rabbits at respective doses up to 1000 and 800 mg/kg/day was associated with concomitant embryofetal toxicity and maternotoxicity. At a dose of 1000 mg/kg/day in rats, fetuses showed enlarged brain ventricles. Nonembryofetal toxic and nonmaternotoxic dosages were 500 and 400 mg/kg/day in rats and rabbits, respectively.
Mesalazine is known to cross the placental barrier and its concentration in umbilical cord plasma is lower than the concentration in maternal plasma. The metabolite acetyl-mesalazine is found at similar concentrations in umbilical cord and maternal plasma. There are no adequate and well controlled studies of Pentasa use in pregnant women. Limited published human data on mesalazine show no increase in the overall rate of congenital malformations. Some data show an increased rate of preterm birth, stillbirth, and low birthweight; however, these adverse pregnancy outcomes are also associated with active inflammatory bowel disease.
Blood disorders (pancytopenia, leucopenia, thrombocytopenia, anaemia) have been reported in newborns of mothers being treated with Pentasa.
NSAIDS inhibit prostaglandin synthesis and, when given during the latter part of pregnancy, may cause closure of the fetal ductus arteriosus, fetal renal impairment, inhibition of platelet aggregation, and delay labour and birth. Continuous treatment with NSAIDS during the last trimester of pregnancy should only be given on sound indications. During the last few days before expected birth, agents with an inhibitory effect on prostaglandin synthesis should be avoided.
Data on 165 women exposed to mesalazine during pregnancy were prospectively collected and pregnancy outcome was compared with that of a control group. The investigators concluded that mesalazine does not represent a major teratogenic risk, as the reported rate of major malformations was within the expected baseline risk of the general population.
Pentasa should be used with caution during pregnancy only if the potential benefits outweigh the possible hazards in the opinion of the physician. The underlying condition itself (inflammatory bowel disease/ IBD) may increase risks for the pregnancy outcome.
Use in lactation. Mesalazine is excreted in breast milk. The concentration is lower than in maternal blood, whereas the metabolite acetyl-mesalazine appears in similar or increased concentrations.
In rats, oral administration of mesalazine during late gestation and lactation at doses of 400 and 800 mg/kg/day was associated with maternotoxicity and toxicity in offspring; a dose of 200 mg/kg/day was devoid of toxicity in either generation.
As data are very limited, Pentasa should be used with caution during lactation and only if the potential benefits outweigh the possible hazards in the opinion of the physician. Hypersensitivity reactions, like diarrhoea, in the nursing infant cannot be excluded.
4.7 Effects on Ability to Drive and Use Machines
Treatment with Pentasa is unlikely to affect the ability to drive and/or use machines.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile. Hypersensitivity reactions and drug fever may occasionally occur. Severe cutaneous adverse reactions (SCARs), such as drug reaction with eosinophilia and systemic symptoms (DRESS), Stevens Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), have been reported in association with mesalazine treatment (see Section 4.4 Special Warnings and Precautions for Use). Mesalazine may be associated with an exacerbation of the symptoms of colitis in those patients who have previously had such problems with sulfasalazine.
Table 1 represents the frequency of adverse effects based on clinical trials and reports from postmarketing surveillance for all formulations of Pentasa, including orals.
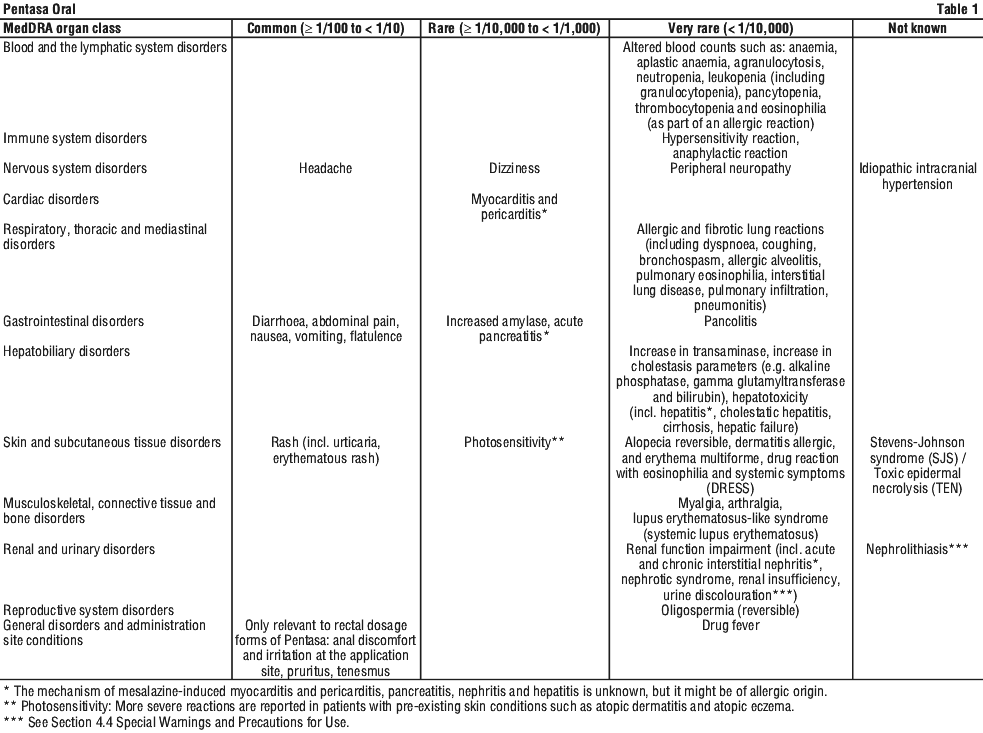
In the MOTUS study the incidences of nausea were 4.9% and 2.0% in the 4 g once daily and 2 g twice daily groups, respectively. All events of nausea were mild to moderate in intensity, and resolved; none led to withdrawal from the trial.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Acute experience in animals. Single oral doses of mesalazine up to 5 g/kg in pigs or a single intravenous dose of mesalazine at 920 mg/kg in rats were not lethal.
Human experience. There is limited clinical experience with overdose of Pentasa. Since Pentasa is an aminosalicylate, symptoms of salicylate toxicity may occur. Symptoms of mild salicylate intoxication include nausea, vomiting, tinnitus or dizziness. Symptoms of more severe salicylate intoxication include hyperthermia, dehydration, disturbance of electrolyte balance and blood pH, seizures, dysrhythmias, coagulopathy, renal failure and coma.
There have been reports of patients taking daily doses of 8 g for a month without any adverse events.
Management of overdose in humans. There is no specific antidote. As Pentasa is an aminosalicylate, conventional therapy for salicylate toxicity may be beneficial. General supportive and symptomatic measures are recommended. Steps to prevent further gastrointestinal tract absorption may be appropriate. Fluid and electrolyte imbalance should be corrected by the administration of appropriate intravenous therapy. Renal function should be closely monitored.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
Pharmacotherapeutic group: intestinal anti-inflammatory agents (A07 EC02).
5.1 Pharmacodynamic Properties
Mechanism of action. It has been established that mesalazine is the active component of sulfasalazine, which is used for the treatment of ulcerative colitis and Crohn's disease. Based on clinical results, the therapeutic value of mesalazine after oral as well as rectal administration appears to be due to a local effect on the inflamed intestinal tissue, rather than to systemic effects. There is information suggesting that the severity of colonic inflammation in ulcerative colitis patients treated with mesalazine is inversely correlated with mucosal concentrations of mesalazine.
Increased leukocyte migration, abnormal cytokine production, increased production of arachidonic acid metabolites, particularly leukotriene B4 and increased free radical formation in the inflamed intestinal tissue, are all present in patients with inflammatory bowel disease. The mechanisms of action of mesalazine are not fully understood, although mechanisms such as activation of the γ-form of peroxisome proliferator activated receptors (PPAR-γ) and inhibition of nuclear factor kappa B (NF-κB) in the intestinal mucosa have been implicated. Mesalazine has in vitro pharmacological effects that inhibit leukocyte chemotaxis, decrease cytokine production, scavenge for free radicals and also reduce leukotriene production via inhibition of the lipo-oxygenase pathway. Prostaglandin production is reduced via inhibition of the cyclo-oxygenase pathway. It is currently unknown which, if any, of these mechanisms play a predominant role in the clinical efficacy of mesalazine.
Observed effects of mesalazine in experimental models show downregulation of both inflammation dependent and non-inflammation dependent signalling pathways involved in the development of colitis associated colorectal cancer (CRC).
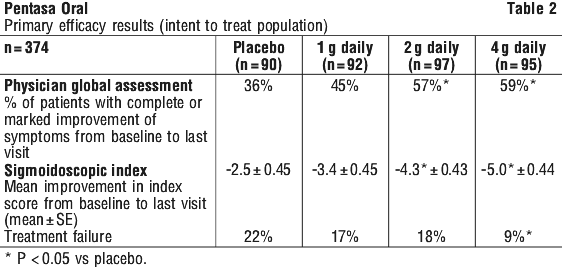
Clinical trials. Ulcerative colitis: treatment of active disease. In a placebo controlled, double blind, randomised study of 374 patients aged 18 and over, with active mild to moderate ulcerative colitis, patients were treated with either placebo, or Pentasa 1 g, 2 g or 4 g daily for 8 weeks, given orally as 250 mg slow release capsules. Three primary efficacy parameters were assessed at baseline and weeks 1, 4 and 8 (see Table 2).
Physician global assessment; an investigator rating of the patient's improvement of symptoms since baseline, based on a scale of 1 to 6 (e.g. 1 = complete relief of symptoms/ 6 = worsening in symptoms).
Sigmoidoscopic index (SI); an evaluation of the presence/ severity of erythema, friability, granularity/ ulceration, mucopus, and the appearance of mucosal vascular pattern, each assigned a value between 0 (normal) to 3 (severe) and totalled to provide an overall index score of between 0-15.
Treatment failure; which were those patients who were not receiving therapeutic benefit, defined as an increase of 5 points in SI and a worsening or no improvement in any symptoms (including trips to the toilet).
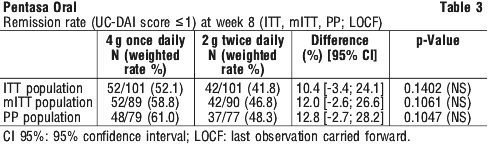
The primary efficacy endpoint was the percentage of patients in clinical and endoscopic remission after 8 weeks of treatment, defined as an Ulcerative Colitis Disease Activity (UC-DAI) score ≤ 1. The statistical analysis was based on the intention to treat (ITT), modified ITT (mITT), and per protocol (PP) analyses with equal importance; remission rates are presented in Table 3.
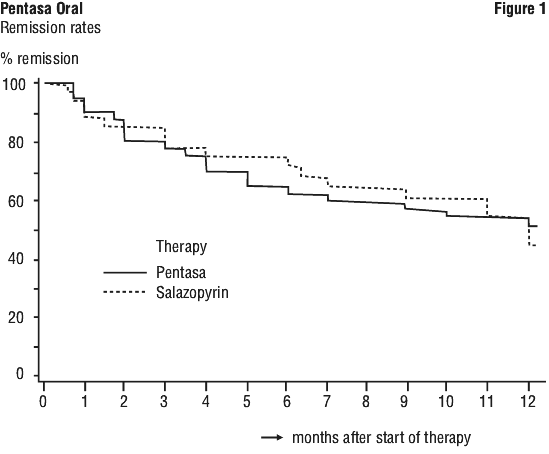
Ulcerative colitis: maintenance of remission. A double blind, double dummy, randomised study comparing Pentasa 1.5 g daily (administered as two 250 mg slow release tablets, three times a day) with sulfasalazine 3 g daily (administered as two 500 mg enteric coated tablets, three times a day) treatment for 12 months, was conducted in 75 patients aged 18 years and over, with ulcerative colitis, who had been in remission for between 1 month and 5 years and had not taken steroids (either orally or as an enema) or azathioprine during at least 1 month before entry. Patients were assessed clinically, endoscopically and histologically before, and 3, 6, 9 and 12 months after the start of treatment. Endoscopy examined mucosal colour, vessel pattern, granularity, presence of valves, distention, polypoid structures, ulcers, spontaneous haemorrhage, and mucopurulent covering, and a wipe test was performed to determine friability. Endoscopy was scored as normal, mild, moderate, severe abnormality or very severe abnormality. Histological assessment was made on the basis of biopsy examination for oedema and haemorrhage in the mucosa and submucosa, for quality and quantity of mucosal cellular infiltrate, and for epithelial architecture of the crypts and was scored as normal, little inflammation, medium inflammation, severe inflammation, or UC in remission. Patients were assessed immediately if symptoms developed or if side effects occurred.
Patients were considered to have remained in remission if all data obtained at each visit were assessed as 'normal' or 'in remission'.
The data of 41 patients treated with Pentasa and 34 patients treated with sulfasalazine were included in life table analysis for calculating remission rates (see Figure 1). No significant differences between the two treatments were revealed (chi2 = 0.14, df = 1, p > 0.70). The final remission rates were 54% for Pentasa and 46% for sulfasalazine, with 95% confidence intervals of 38%-69% for Pentasa and 26%-64% for sulfasalazine. The difference is 8% in favour of Pentasa, with a 95% confidence interval of -16% to 31%.
The risk of colorectal cancer (CRC) is increased in ulcerative colitis, especially in patients with extensive disease, with a disease course > 8 years, with a first degree family history of CRC, or with comorbid primary sclerosing cholangitis. The risk for colitis associated CRC has been estimated to be 2% at 10 years, 8% at 20 years, and 18% at 30 years after onset of ulcerative colitis (see Section 5.1 Pharmacodynamic Properties).
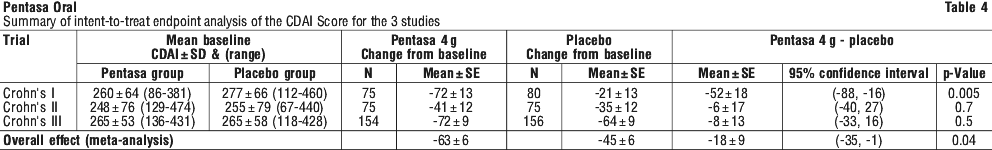
Crohn's disease: treatment of active disease. A meta-analysis was conducted of three double blind, placebo controlled, randomised, multicentre studies in 615 patients aged 18 and over, of whom 304 were treated with up to 4 g/day Pentasa administered as oral capsules and 311 were treated with placebo, for mild to moderate Crohn's disease for a period of 16 weeks (see Table 4).
The primary efficacy variable used in these trials was the Crohn's Disease Activity Index (CDAI), which included the following components: sum of liquid/ very soft stools (per 7 days), sum of abdominal pain rating (per 7 days), sum of general wellbeing ratings (per 7 days), use of loperamide or codeine, bodyweight, haematocrit, abdominal mass, sum of symptoms.
Crohn's disease: maintenance of remission. In a randomised, double blind, placebo controlled study conducted in 293 patients aged 18 years and over, with Crohn's disease in remission, a daily 3 g dose of Pentasa was administered as 250 mg capsules for a period of up to 48 weeks (with assessments at baseline and weeks 4, 12, 24, 36, 48). Relapse was defined as a Crohn's Activity Index of > 150, with at least a 60 point increase over baseline.
246 patients completed a minimum of 4 weeks treatment. Of these, thirty of the 118 patients (25%) who received Pentasa had a relapse compared with 47 of 128 (36%) receiving placebo (p = 0.056).
5.2 Pharmacokinetic Properties
The therapeutic activity of mesalazine appears to depend on local contact of the drug with the diseased area of the intestinal mucosa.
Pentasa prolonged release granules and tablets consist of ethylcellulose coated microgranules of mesalazine. Following administration of the tablets and granules, and the disintegration of the tablets, mesalazine is continuously released from the individual microgranules throughout the gastrointestinal tract in any enteral pH conditions.
The microgranules enter the duodenum within an hour of administration, independent of food coadministration. The average small intestinal transit time is approximately 3-4 hours in healthy volunteers.
Absorption. Based on urinary recovery in healthy volunteers, 30-50% of the ingested dose is absorbed following oral administration, predominantly from the small intestine. Mesalazine is detectable in plasma 15 minutes after administration.
Maximum plasma concentrations are seen 1-6 hours post-dose. Dosage regimens of 2 g twice daily and 4 g once daily of mesalazine result in comparable systemic exposures (AUC) over 24 hours and indicate the continuous release of mesalazine from the formulation over the treatment period. Steady-state is reached after a treatment period of 5 days following oral administration (see Table 5).
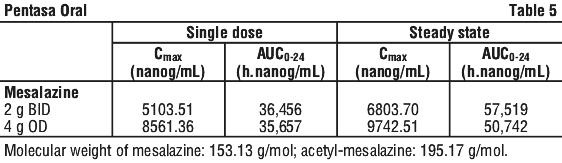
The transit and release of mesalazine after oral administration are independent of food coadministration, whereas the systemic absorption will be reduced.
Distribution. Protein binding of mesalazine is approximately 50% and of acetyl-mesalazine about 80%.
Metabolism. Mesalazine is metabolised both presystemically by the intestinal mucosa and systemically in the liver to N-acetyl mesalazine (acetyl-mesalazine). Some acetylation also occurs through the action of colonic bacteria. The acetylation seems to be independent of the acetylator phenotype of the patient. The metabolic ratio of acetyl-mesalazine to mesalazine in plasma after oral administration ranges from 3.5 to 1.3 after daily doses of 500 mg x 3 and 2 g x 3, respectively, implying a dose dependent acetylation which may be subject to saturation.
Acetyl-mesalazine is thought to be clinically, as well as toxicologically, inactive but this still remains to be confirmed.
Excretion. After intravenous administration, the plasma half-life of mesalazine is approximately 40 minutes and for acetyl-mesalazine approximately 80 minutes. Due to the continuous release of mesalazine from Pentasa throughout the gastrointestinal tract, the elimination half-life cannot be determined after oral administration. However, steady-state is reached after a treatment period of 5 days following oral administration.
Both substances are excreted in the urine and faeces. The urinary excretion consists mainly of acetyl-mesalazine and the faecal excretion consists mainly of mesalazine.
Characteristics in patients. The delivery of mesalazine to the intestinal mucosa after oral administration is only slightly affected by pathophysiologic changes, such as diarrhoea and increased bowel acidity observed during active inflammatory bowel disease. A reduction in systemic absorption to 20-25% of the daily dose has been observed in patients with accelerated intestinal transit. Likewise, a corresponding increase in faecal excretion has been seen.
In patients with impaired liver and kidney function, the resultant decrease in the rate of elimination and increased systemic concentration of mesalazine may constitute an increased risk of nephrotoxic adverse reactions.
5.3 Preclinical Safety Data
Genotoxicity. Mesalazine was negative in bacterial assays of gene mutation and in a mouse micronucleus test.
Carcinogenicity. There is no evidence of carcinogenicity in mice or rats treated with mesalazine in the diet at respective doses up to 2500 and 800 mg/kg/day for two years. These doses were associated with plasma concentrations of mesalazine and its metabolite N-acetyl-5-aminosalicylic acid of 7-fold (mice) and 3-fold (rats) the peak plasma concentrations of these compounds at the maximal recommended human dose of the granules and the tablets.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
Prolonged release tablets: 3 years.
Prolonged release granules: 2 years.
6.4 Special Precautions for Storage
Store below 25°C. Keep in original container.
6.5 Nature and Contents of Container
Pentasa prolonged release tablets. Pentasa 0.5 g tablets are supplied in blister packs of 30 and 100 tablets.
Pentasa 1 g tablets are supplied in blister packs of 20, 60 and 120 tablets.
Pentasa prolonged release granules. Pentasa 1 g granules are supplied in packs of 30, 50, 100, 120 and 150 sachets.
Pentasa 2 g granules are supplied in packs of 10, 15 and 60 sachets.
Pentasa 4 g granules are supplied in packs of 8 and 30 sachets.
Not all pack sizes are being distributed in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
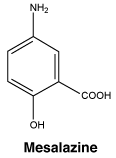
Synonyms: 5-aminosalicylic acid; 5-amino 2-hydroxybenzoic acid; mesalamine.
CAS number. 89-57-6.
7 Medicine Schedule (Poisons Standard)
(S4) Prescription Only Medicine.
Date of Revision
14 October 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.