Pharmacy Action Heartburn Relief
Brand Information
| Brand name | Pharmacy Action Heartburn Relief |
| Active ingredient | Pantoprazole |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Pharmacy Action Heartburn Relief.
Summary CMI
Pharmacy Action Heartburn Relief
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I taking Pharmacy Action Heartburn Relief?
Pharmacy Action Heartburn Relief contains the active ingredient pantoprazole sodium sesquihydrate. Pharmacy Action Heartburn Relief is used for lasting symptomatic relief of frequent heartburn and stomach acid complaints due to gastro-oesophageal reflux disease (GORD).
For more information, see Section 1. Why am I taking Pharmacy Action Heartburn Relief? in the full CMI.
2. What should I know before I use Pharmacy Action Heartburn Relief?
Do not take Pharmacy Action Heartburn Relief if you have ever had an allergic reaction to pantoprazole or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Pharmacy Action Heartburn Relief? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Pharmacy Action Heartburn Relief and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I take Pharmacy Action Heartburn Relief?
- Take one (1) Pharmacy Action Heartburn Relief enteric coated tablet everyday (once every 24 hours).
- If you are not sure, ask your pharmacist or doctor how to take your medicine.
More instructions can be found in Section 4. How do I take Pharmacy Action Heartburn Relief? in the full CMI.
5. What should I know while taking Pharmacy Action Heartburn Relief?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while taking Pharmacy Action Heartburn Relief? in the full CMI.
6. Are there any side effects?
Pharmacy Action Heartburn Relief may have some common side effects like headache, dizziness, diarrhoea, constipation, dry mouth, blurred vision, etc.
Serious side effects include unusual tiredness or weakness, nausea, vomiting, chest pain, fast heartbeat, loss of appetite, increased or decreased need to urinate, high blood pressure, water retention, swelling, etc.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I taking Pharmacy Action Heartburn Relief?
Pharmacy Action Heartburn Relief contains the active ingredient pantoprazole sodium sesquihydrate.
Pharmacy Action Heartburn Relief belongs to a group of medicines called proton pump inhibitors (PPls). It works by decreasing the amount of acid the stomach makes to give relief from the symptoms.
Pharmacy Action Heartburn Relief is used for lasting symptomatic relief of frequent heartburn and stomach acid complaints due to gastro-oesophageal reflux disease (GORD).
These symptoms can be caused by "washing back" (reflux) of food and acid from the stomach into the food pipe, also known as the oesophagus.
Reflux can cause a burning sensation in the chest rising up to the throat, also known as heartburn.
Frequent heartburn is when you have heartburn for two or more days a week. Heartburn that occurs frequently is a typical symptom of GORD.
Pharmacy Action Heartburn Relief is not the right medicine for you if you suffer heartburn only occasionally (one episode of heartburn a week or less), or if you want immediate relief of heartburn.
Pharmacy Action Heartburn Relief will start to suppress acid within a few hours; however it is not intended to provide instant symptom relief. For effective, lasting relief, you should take a full course of Pharmacy Action Heartburn Relief (see ‘How to take Pharmacy Action Heartburn Relief’ below).
Pharmacy Action Heartburn Relief is not addictive.
Pharmacy Action Heartburn Relief is for use by adults over 18 years of age.
2. What should I know before I use Pharmacy Action Heartburn Relief?
Warnings
Do not use Pharmacy Action Heartburn Relief if:
- you are allergic to pantoprazole, or any other PPI (such as omeprazole, rabeprazole or lansoprazole) or any of the ingredients listed at the end of this leaflet. Some symptoms of an allergic reaction include skin rash, itching, shortness of breath or swelling of the face, lips or tongue, which may cause difficulty in swallowing or breathing.
- you have severe liver disease or cirrhosis.
- you have recently had trouble swallowing, pain when swallowing, persistent vomiting or experienced unintended weight loss.
- have recently vomited blood, had black stools or notice blood in your stools.
- are taking atazanavir or nelfinavir (anti-viral medications).
- are pregnant, intend to become pregnant, are breastfeeding or wish to start breastfeeding.
Always check the ingredients to make sure you can use this medicine.
Tell your doctor if you:
- have any other medical conditions like:
- jaundice, liver problems or anaemia;
- a feeling of weakness or you look pale;
- previously taken heartburn/indigestion medications continuously for 4 or more weeks;
- persisting heartburn symptoms despite taking Pharmacy Action Heartburn Relief (or other similar medicines) continuously for 2 weeks, or your symptoms have recently changed;
- been told by your doctor that you have a stomach ulcer, or gastrointestinal surgery is planned;
- heartburn/indigestion symptoms for the first time and you are over 40 years of age;
- a scheduled endoscopy (investigation of your stomach lining performed by a specialist);
- any allergies to any medicines or other substances, such as foods, preservatives or dyes. - have take any medicines for any other condition
Your pharmacist or doctor can provide specific advice on whether you should take Pharmacy Action Heartburn Relief.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Pharmacy Action Heartburn Relief and affect how it works. These include:
- warfarin, phenprocoumon – medicines used to prevent blood clots (blood thinners);
- atazanavir, nelfinavir – medicines used to treat viral infections such as HIV;
- ketoconazole, itraconazole, posaconazole – medicines used to treat fungal infections;
- methotrexate – a medicine used to treat arthritis and some types of cancer;
- erlotinib or related medicines used to treat cancer;
- tacrolimus, mycophenolate mofetil – medicines used to suppress the immune system;
- fluvoxamine – a medicine used to treat anxiety and depression.
You may need different amounts of your medicine, or you may need to take different medicines. Your doctor will advise you.
Your pharmacist or doctor has more information on medicines to be careful with or to avoid while taking Pharmacy Action Heartburn Relief. Check with them if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Pharmacy Action Heartburn Relief.
4. How do I take Pharmacy Action Heartburn Relief?
The directions for use are included on the Medicine Information panel on the pack.
If you do not understand the instructions on the pack, ask your pharmacist or doctor for help.
Follow all directions given to you by your pharmacist or doctor. They may differ from the information contained in this leaflet.
Swallow your tablets whole with a little water with or without food.
Do not crush or chew the tablets. Pharmacy Action Heartburn Relief tablets have a special coating to protect them from the acidic contents of your stomach. For Pharmacy Action Heartburn Relief to work effectively, this coating must not be broken.
How long to take
For effective, lasting relief, take Pharmacy Action Heartburn Relief strictly according to the directions on your pack.
Pharmacy Action Heartburn Relief tablets should be taken for at least 7 days, and up to 14 days.
If you purchased the pack containing 7 days' supply and you need to take it for longer than 7 days, ask your pharmacist for advice.
Do not take beyond 14 days without consulting your doctor.
Pharmacy Action Heartburn Relief is not intended to provide instant relief from your heartburn/indigestion symptoms.
If you forget to take Pharmacy Action Heartburn Relief
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking it as you would normally.
Do not take a double dose to make up for the dose you missed. This may increase the chance of you getting an unwanted side effect. If you are not sure what to do, ask your pharmacist or doctor. If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you use too much Pharmacy Action Heartburn Relief
Immediately telephone your doctor or Poisons Information Centre for advice (13 11 26) or go to Accident and Emergency at your nearest hospital, if you think that you or anyone else may have taken too much Pharmacy Action Heartburn Relief.
You may need urgent medical attention. Keep telephone numbers for these places handy.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while taking Pharmacy Action Heartburn Relief?
Things you should do
Call your doctor if:
- you do not feel better while taking Pharmacy Action Heartburn Relief.
- your symptoms recur within 2 weeks of completing the course
- you are about to have any blood tests, tell your doctor that you are taking this medicine. It may interfere with the results of some tests.
- your doctor if your reflux symptoms return after you stop taking this medicine. The symptoms of reflux may return after stopping this medicine suddenly, especially if you have taken it for a while.
Things to be careful of
Some self-help measures suggested below may help your condition. Talk to your doctor or pharmacist about these measures and for more information.
Aspirin and many other medicines used to treat arthritis, period pain, headaches: These medicines may irritate the stomach and may make your heartburn/indigestion symptoms worse. Your doctor or pharmacist may suggest other medicines you can take.
Caffeine: You may be advised to limit the number of drinks which contain caffeine, such as coffee, tea, cocoa and cola drinks, because they contain ingredients that may irritate your stomach.
Eating habits: Eat smaller, more frequent meals. Eat slowly and chew your food carefully. Try not to rush at mealtimes.
Smoking: It is advisable that you stop smoking or at least cut down.
Weight: You may be advised that losing some weight will help your reflux/indigestion symptoms.
Driving or using machines
Be careful before you drive or use any machines or tools.
Pharmacy Action Heartburn Relief may cause dizziness and visual disturbances. Do not drive or operate machinery while taking Pharmacy Action Heartburn Relief.
Drinking alcohol
Tell your doctor if you drink alcohol.
Looking after your medicine
Follow the instructions on the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink; or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground, is a good place to store medicines.
When to discard your medicine
If you stop taking Pharmacy Action Heartburn Relief or the tablets have passed their expiry date, ask your pharmacist what to do with any tablets that are left over.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Digestion related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
These are the more common side effects of Pharmacy Action Heartburn Relief. It helps most people with heartburn/indigestion, but it has been uncommonly associated with such unwanted side effects.
Serious side effects
| Serious side effects | What to do |
Skin related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
The above list includes serious side effects that may require medical attention. Serious side effects are rare or very rare.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed above may occur in some people.
Ask your doctor or pharmacist if you do not understand some of the information in this list.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Pharmacy Action Heartburn Relief contains
| Active ingredient (main ingredient) | Pantoprazole (as sodium sesquihydrate) |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | Pharmacy Action Heartburn Relief tablets do not contain gluten, lactose, sucrose, tartrazine or other azo dyes. |
Do not take this medicine if you are allergic to any of these ingredients.
What Pharmacy Action Heartburn Relief looks like
Pharmacy Action Heartburn Relief is available as 20 mg tablets. The tablets have an acid-resistant coating called an enteric coating.
The 20 mg tablets are yellow to pale yellow, oval-shaped, biconvex, enteric-coated tablets plain on both sides. AUST R 303946
Pharmacy Action Heartburn Relief tablets are available in blister packs containing 7 and 14 tablets*.
* Not all pack sizes may be marketed.
Sponsor
Generic Health Pty Ltd
Suite 2, Level 2
19-23 Prospect Street Box Hill,
VIC, 3128 Australia
 +61 3 9809 7900
+61 3 9809 7900
This leaflet was prepared in July 2025.
Brand Information
| Brand name | Pharmacy Action Heartburn Relief |
| Active ingredient | Pantoprazole |
| Schedule | S3 |
MIMS Revision Date: 01 March 2026
1 Name of Medicine
Pantoprazole (as sodium sesquihydrate).
2 Qualitative and Quantitative Composition
Each Pharmacy Action Heartburn Relief 20 mg enteric coated tablet contains 22.7 mg pantoprazole sodium sesquihydrate equivalent to 20 mg of pantoprazole.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Pharmacy Action Heartburn Relief 20 mg tablets are yellow to pale yellow, oval shaped, biconvex, enteric-coated tablets, plain on both sides.
4 Clinical Particulars
4.1 Therapeutic Indications
Pharmacy Action Heartburn Relief is indicated for symptomatic relief of heartburn, acid regurgitation and other symptoms associated with gastro-oesophageal reflux disease (GORD).
4.2 Dose and Method of Administration
Pharmacy Action Heartburn Relief is indicated for use in adults 18 years of age and over.
Pharmacy Action Heartburn Relief tablets should not be chewed or crushed but swallowed whole with a little water.
Symptomatic GORD. The recommended dosage is one Pharmacy Action Heartburn Relief 20 mg tablet per day for at least 7 days, and up to 14 days. If symptom control has not been achieved after two weeks of continuous treatment with Pharmacy Action Heartburn Relief 20 mg tablet per day, patients should be referred to their doctor.
Use in children. There are limited data currently available on the use of pantoprazole in children. Pharmacy Action Heartburn Relief is not recommended for use in children and adolescents under 18 years of age.
Use in the elderly. No dose adjustment is necessary in elderly patients.
Impaired renal function. No dose adjustment is required when pantoprazole is administered to patients with impaired renal function.
Impaired hepatic function. Pantoprazole is contraindicated in patients with cirrhosis or severe liver disease (see Section 4.3 Contraindications). No dose adjustment is required when pantoprazole is administered to patients with milder forms of impaired liver function.
4.3 Contraindications
Known hypersensitivity to pantoprazole, substituted benzimidazoles or any other components of the formulation; or in cases of cirrhosis or severe liver disease.
Pantoprazole, like other proton pump inhibitors, should not be co-administered with HIV protease inhibitors, such as atazanavir or nelfinavir (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
4.4 Special Warnings and Precautions for Use
Patients should be referred to their doctor for review if:
they have unintentional weight loss, anaemia, gastrointestinal bleeding, dysphagia, persistent vomiting or vomiting with blood, malaena, gastric ulcer is suspected or present or gastrointestinal surgery, as treatment with pantoprazole may alleviate symptoms and delay diagnosis. In these cases, malignancy should be excluded;
they have had to take other medication for indigestion or heartburn continuously for four or more weeks in order to control their symptoms;
they are being treated for symptomatic GORD and require Pharmacy Action Heartburn Relief for more than 14 days;
they have jaundice or severe hepatic impairment (e.g. cirrhosis); or
they have any other significant medical condition.
Clostridium difficile. PPI therapy may be associated with an increased risk of Clostridium difficile infection.
Pantoprazole, like all proton pump inhibitors, might be expected to increase the counts of bacteria normally present in the upper gastrointestinal tract. Treatment with pantoprazole may lead to a slightly increased risk of gastrointestinal infections caused by bacteria such as Salmonella, Campylobacter and Clostridium difficile.
Influence on vitamin B12 absorption. Pantoprazole, as all acid blocking medicines, may reduce the absorption of cyanocobalamin (vitamin B12) due to hypo- or achlorhydria. This should be considered in patients with reduced body stores or risk factors for reduced vitamin B12 absorption such as the elderly and in patients with Zollinger-Ellison Syndrome and other pathological hypersecretory conditions or if respective clinical symptoms are observed. Rare cases of cyanocobalamin deficiency following acid blocking therapy have been reported.
Bone fracture. PPI therapy may be associated with an increased risk for osteoporosis related fractures of the hip, wrist, or spine. The risk of fracture was increased in patients who received high doses; defined as multiple daily doses, and long-term PPI therapy (a year or longer).
Acute interstitial nephritis. Acute interstitial nephritis has been observed in patients taking PPIs including pantoprazole. Acute interstitial nephritis may occur at any point during PPI therapy and is generally associated to an idiopathic hypersensitivity reaction. Discontinue pantoprazole if acute interstitial nephritis develops.
Hypomagnesaemia. Hypomagnesaemia has been rarely reported in patients treated with PPIs for at least three months (in most cases after a year of therapy). Serious consequences of hypomagnesaemia include tetany, arrhythmia, and seizure. Hypomagnesaemia may lead to hypocalcaemia and/or hypokalaemia (see Section 4.8 Adverse Effects (Undesirable Effects)).
Severe cutaneous adverse reactions. Severe cutaneous adverse reactions, including erythema multiforme, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) have been reported in association with the use of PPIs (see Section 4.8 Adverse Effects (Undesirable Effects)). Discontinue pantoprazole at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation.
Subacute cutaneous lupus erythematosus (SCLE). Proton pump inhibitors are associated in rare cases with the occurrence of subacute cutaneous lupus erythematosus (SCLE). If lesions occur, especially in sun exposed areas of the skin, and if accompanied by arthralgia, the patient should seek medical help promptly and the healthcare professional should consider stopping the product.
General toxicity. Gastrointestinal system. Treatment with pantoprazole causes dose-dependent hypergastrinaemia as a result of inhibition of gastric acid secretion. Gastrin has a trophic effect on the gastric mucosa and increases in gastric weight have been observed in rats and dogs to be dependent upon both dose and duration of treatment. Accompanying histopathological changes in the gastric mucosa were increased height, dilatation of fundic glands, chief cell hyperplasia and/or atrophy and parietal cell hyperplasia or vacuolation/degeneration. Increased density of enterochromaffin-like (ECL) cells was observed after 12 months treatment at dose levels from 5 mg/kg/day in rats and 2.5 mg/kg/day in dogs; all changes were reversible after various recovery periods. Since these gastric effects are a consequence of the pharmacological effect of acid secretion inhibition, no-effect doses were not established in all instances.
Although rats might be more susceptible to this effect than other species because of their high ECL cell density and sensitivity to gastrin, ECL cell hyperplasia occurs in other species, including mice and dogs, and has been observed in one of two clinical trials in which ECL cell density was measured (a 2-fold increase was observed in study RR126/97 after up to 5 years of treatment with regular and high doses, but no increase was observed in study RR125/97). No dysplastic or neoplastic changes were observed in gastric endocrine cells in either study.
Ocular toxicity and dermal phototoxicity/sensitivity. Studies have shown that pantoprazole is retained in low levels in the eyes and skin of pigmented rats. It is likely that the retention reflects a reversible association with melanin. Animal studies investigating the potential for phototoxicity/photosensitivity have not been conducted. A 2-week dog study, conducted specifically to investigate the effects on the eye and ear, did not reveal any changes relating to pantoprazole treatment, but the doses chosen were relatively low (40 and 160 mg (about 4 and 15 mg/kg) orally and 60 mg (about 6 mg/kg) IV). No ophthalmological changes or changes in electroretinographs were observed in cynomolgus monkeys at IV doses of up to 15 mg/kg/day for 4 weeks.
Use in the elderly. See Section 4.2 Dose and Method of Administration, Use in the elderly; Section 4.4 Special Warnings and Precautions for Use, Influence on vitamin B12 absorption; Section 5.2 Pharmacokinetic Properties, Special populations.
Paediatric use. To date there has been limited experience with treatment in children.
Effects on laboratory tests. Increased Chromogranin A (CgA) level may interfere with investigations for neuroendocrine tumours. To avoid this interference, proton pump inhibitor treatment should be stopped 14 days before CgA measurements.
Patients should consult their doctor before taking this product if they are due to have an endoscopy.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Pantoprazole is metabolised in the liver via the cytochrome P450 enzyme system. A study using human liver microsomes suggested that the P450 enzymes CYP2C19 and CYP3A4 are involved in its metabolism. In addition, CYP2D6 and CYP2C9-10 were implicated in another study. An interaction of pantoprazole with other drugs or compounds which are metabolised using the same enzyme system cannot be excluded. However, no clinically significant interactions were observed in specific tests with a number of such drugs or compounds, namely carbamazepine, caffeine, diazepam, diclofenac, digoxin, ethanol, glibenclamide, metoprolol, naproxen, nifedipine, phenytoin, piroxicam, theophylline and the low dose oral contraceptive, Triphasil (levonorgestrel and ethinylestradiol). There was also no interaction with a concomitantly administered antacid (aluminium hydroxide and magnesium hydroxide).
Treatment of dogs with IV famotidine shortened the duration of the pH elevation effect of pantoprazole.
Four crossover pharmacokinetic studies designed to examine any interactions between pantoprazole and the drugs clarithromycin, amoxicillin and metronidazole, conducted in 66 healthy volunteers, showed no interactions.
Drugs with pH dependent absorption pharmacokinetics. As with all acid suppressant medications, the absorption of drugs whose bioavailability is pH dependent (e.g. ketoconazole, itraconazole, posaconazole, erlotinib), might be altered due to the decrease in gastric acidity.
HIV protease inhibitors. It has been shown that co-administration of atazanavir 300 mg/ritonavir 100 mg with omeprazole (40 mg once daily) or atazanavir 400 mg with lansoprazole (60 mg single dose) to healthy volunteers resulted in a substantial reduction in the bioavailability of atazanavir. The absorption of atazanavir is pH dependent. Therefore, proton pump inhibitors, including pantoprazole, should not be co-administered with HIV protease inhibitors for which absorption is dependent on acidic intragastric pH, such as atazanavir or nelfinavir (see Section 4.3 Contraindications).
Mycophenolate mofetil. Co-administration of PPIs in healthy subjects and in transplant patients receiving mycophenolate mofetil has been reported to reduce the exposure to the active metabolite, mycophenolic acid. This is possibly due to a decrease in mycophenolate mofetil solubility at an increased gastric pH. The clinical relevance of reduced mycophenolic acid exposure on organ rejection has not been established in transplant patients receiving PPIs and mycophenolate mofetil. Use pantoprazole with caution in transplant patients receiving mycophenolate mofetil.
Methotrexate. Concomitant use with methotrexate (primarily at high dose), may elevate and prolong serum levels of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities.
Drugs that inhibit or induce CYP2C19 (tacrolimus, fluvoxamine). Concomitant administration of pantoprazole and tacrolimus may increase whole blood levels of tacrolimus, especially in transplant patients who are intermediate or poor metabolisers of CYP2C19. Inhibitors of CYP2C19, such as fluvoxamine, would likely increase the systemic exposure of pantoprazole.
Coumarin anticoagulants (phenprocoumon or warfarin). Coadministration of pantoprazole with warfarin or phenprocoumon did not affect the pharmacokinetics of warfarin, phenprocoumon or international normalised ratio (INR). However, there have been reports of increased INR and prothrombin time in patients receiving PPIs and warfarin or phenprocoumon concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding, and even death. Therefore, in patients being treated with coumarin anticoagulants (e.g. warfarin or phenprocoumon), monitoring of prothrombin time/ INR is recommended after initiation, termination or during irregular use of pantoprazole.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B3)
Teratological studies in rats and rabbits gave no evidence of a teratogenic potential for pantoprazole. In oral rat studies, dose dependent toxic effects were observed on foetuses and pups: increased pre- and postnatal deaths at 450 mg/kg/day, reduced foetal weight (greater than or equal to 150 mg/kg/day) and delayed skeletal ossification and reduced pup growth (greater than or equal to 15 mg/kg/day). For the latter, a no effect dose was not established. Doses of 450 mg/kg/day were maternotoxic and may have been associated with dystocia and incomplete parturition. Penetration of the placenta was investigated in the rat and was found to increase with advanced gestation. As a result, concentrations of pantoprazole in the foetus are increased shortly before birth regardless of the route of administration.
The significance of these findings in humans is unknown. As there is no information on the safety of the drug during pregnancy in women, pantoprazole should not be used during pregnancy unless the benefit clearly outweighs the potential risk to the foetus.
Use in lactation. A peri/post-natal study in rats found that treatment with pantoprazole at doses of 10 mg/kg/day or greater decreased pup growth. A transient effect on one of a series of development tests (startle response) was only evident in the 30 mg/kg/day group at an age when male and female offspring showed lower body weights, paralleled with lower brain weight, than the controls. The significance of these findings for humans is unknown, and there is currently no information on the safety of pantoprazole during breast feeding in humans. Excretion into human milk has been reported. Therefore, pantoprazole should only be used during lactation if the benefits clearly outweigh the risks.
4.7 Effects on Ability to Drive and Use Machines
Pantoprazole does not exert its pharmacological action centrally, therefore it is not expected to adversely affect the ability to drive or use machines, however, adverse drug reactions such as dizziness and visual disturbances may occur (see Section 4.8 Adverse Effects (Undesirable Effects)). If affected, patients should not drive or operate machines.
4.8 Adverse Effects (Undesirable Effects)
Pantoprazole tablets are well tolerated. Most of the adverse reactions seen with treatment were of mild or moderate intensity in clinical trials and post-marketing surveillance. The following adverse reactions have been reported in patients receiving pantoprazole.
Adverse reactions within each body system are listed in descending order of frequency: very common: ≥ 10%; common: ≥ 1% and < 10%; uncommon: ≥ 0.1% and < 1%; rare: ≥ 0.01% and < 0.1%; very rare: < 0.01%; not known: cannot be estimated from the available data.
These include the following:
General disorders and administration site conditions. Uncommon: fatigue, malaise, asthenia and increased sweating.
Rare: fever, peripheral oedema, and increased body temperature.
Very rare: flushing, substernal chest pain and hot flushes.
Cardiovascular disorders, general. Rare: hypertension.
Very rare: circulatory collapse.
Nervous system disorders. Uncommon: headaches, dizziness.
Rare: taste disorders, metallic taste.
Very rare: reduced movement and speech disorder, changes to the senses of smell and taste.
Gastrointestinal system disorders. Uncommon: diarrhoea, nausea, vomiting, abdominal distension and bloating, constipation, dry mouth, abdominal pain and discomfort.
Rare: rectal disorder and colonic polyp.
Very rare: faecal discolouration and increased saliva.
Not known: flatulence, severe eructation, withdrawal of long-term PPI therapy can lead to aggravation of acid-related symptoms and may result in rebound acid hypersecretion.
Hearing and vestibular disorders. Very rare: tinnitus.
Immune system disorders. Rare: hypersensitivity (including anaphylactic reactions and anaphylactic shock).
Hepatobiliary disorders. Uncommon: liver enzymes increased.
Rare: bilirubin increased.
Very rare: hepatocellular failure, cholestatic hepatitis and jaundice.
Not known: hepatocellular injury.
The occurrence of severe hepatocellular damage leading to jaundice or hepatic failure having a temporal relationship to the intake of pantoprazole has been reported with a frequency of approximately one in a million patients.
Metabolic and nutritional disorders. Rare: hypertriglyceridaemia. and lipid increases (triglycerides, cholesterol), weight changes.
Not known: hyponatraemia, hypomagnesaemia, hypocalcaemia, hypokalaemia (hypocalcaemia and/or hypokalaemia may be related to the occurrence of hypomagnesaemia (see Section 4.4 Special Warnings and Precautions for Use).
Musculoskeletal and connective tissue disorders. Rare: myalgia and arthralgia.
Very rare: pain including skeletal pain.
Not known: fracture of wrist, hip and spine.
Renal and urinary disorders. Very rare: interstitial nephritis.
Platelet, bleeding, clotting disorders. Very rare: increased coagulation time.
Blood and lymphatic system disorders. Rare: anaemia, agranulocytosis.
Very rare: leucopenia, thrombocytopenia, pancytopenia.
Psychiatric disorders. Uncommon: sleep disorders.
Rare: depression, hallucination, disorientation and confusion, especially in predisposed patients, as well as the aggravation of these symptoms in case of pre-existence.
Very rare: anxiety.
Resistance mechanism disorders. Rare: sepsis.
Respiratory system disorders. Very rare: dyspnoea.
Reproductive system and breast disorders. Rare: gynaecomastia.
Skin and subcutaneous tissue disorders. Uncommon: pruritus, rash, exanthema/ eruption.
Rare: angioedema and urticaria.
Very rare: severe skin reactions such as Stevens-Johnson syndrome, toxic epidermal necrolysis, erythema multiforme, Lyell syndrome and photosensitivity.
Not known: subacute cutaneous lupus erythematosus, drug reaction with eosinophilia and systemic symptoms (DRESS). acute generalised exanthematous pustulosis.
Eye disorders. Uncommon: disturbances in vision (blurred vision).
Very rare: conjunctivitis.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There are no known symptoms of overdosage in humans. In individual cases, 240 mg was administered i.v or p.o. and was well tolerated. As pantoprazole is extensively protein bound, it is not readily dialyzable. As in any case of overdosage, treatment should be symptomatic and supportive measures should be utilised.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Pantoprazole is a proton pump inhibitor (PPI). It inhibits specifically and dose-proportionately H+/K+-ATPase, the enzyme which is responsible for gastric acid secretion in the parietal cells of the stomach.
The substance is a substituted benzimidazole which accumulates in the acidic environment of the parietal cells after absorption. There, it is converted into the active form, a cyclic sulfenamide which binds to the H+/K+-ATPase, thus inhibiting the proton pump and causing potent and long lasting suppression of basal and stimulated gastric acid secretion. As pantoprazole acts distal to the receptor level it can influence gastric acid secretion irrespective of the nature of the stimulus (acetylcholine, histamine, gastrin).
Pantoprazole's selectivity is due to the fact that it only exerts its full effect in a strongly acidic environment (pH < 3), remaining mostly inactive at higher pH values. As a result, its complete pharmacological, and thus therapeutic, effect can only be achieved in the acid secretory parietal cells. By means of a feedback mechanism this effect is diminished at the same rate as acid secretion is inhibited.
As with other proton pump inhibitors and H2 receptor inhibitors, treatment with pantoprazole causes a reduced acidity in the stomach and thereby an increase in gastrin in proportion to the reduction in acidity. The increase in gastrin is reversible.
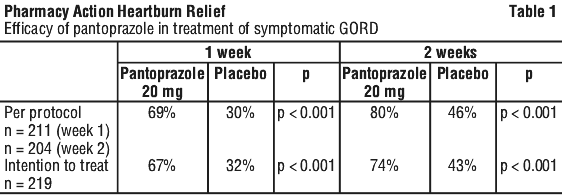
Clinical trials. Treatment of symptomatic reflux (GORD). The relief of symptoms of reflux in patients who showed no oesophageal lesions on endoscopy has been shown in the following double blind, multi-centre, placebo-controlled study (245/98) using pantoprazole 20 mg once daily. Overall, 219 patients were enrolled into the study. Each patient was to have a normal oesophagus as assessed by endoscopy and to have suffered from at least one episode of heartburn of at least moderate intensity on all three days prior to inclusion into the study. Additionally, patients were to have a history of reflux symptoms (heartburn, acid eructation, pain on swallowing) for at least three months prior to entry into the study. Efficacy of pantoprazole 20 mg is shown in Table 1.
5.2 Pharmacokinetic Properties
Absorption. Pantoprazole is rapidly absorbed and the maximal plasma concentration appears after one single oral dose. After single and multiple oral doses, the median time to reach maximum serum concentrations was approximately 2.5 hours, with a Cmax of 1.2 microgram/mL. Terminal half-life is approximately 1 hour.
Pharmacokinetics do not vary after single or repeated administration. The plasma kinetics of pantoprazole are linear (in the dose range of 10 to 80 mg) after both oral and intravenous administration.
Pantoprazole is completely absorbed after oral administration. The absolute bioavailability of the tablet is approximately 77%. Concomitant intake of food had no influence on AUC, maximum serum concentrations and thus bioavailability.
Distribution. The serum protein binding of pantoprazole is approximately 98%. Volume of distribution is approximately 0.15 L/kg and clearance is approximately 0.1 L/h/kg.
Metabolism. Pantoprazole is metabolised in the liver via the cytochrome P450 enzyme system.
Excretion. Pantoprazole is rapidly eliminated from serum and is almost exclusively metabolised in the liver. Renal elimination represents the most important route of excretion (approximately 80%) for the metabolites of pantoprazole, the rest are excreted with the faeces. The main metabolite in both the serum and urine is desmethyl-pantoprazole which is conjugated with the sulphate. The half-life of the main metabolites (approximately 1.5 hours) is not much longer than that of pantoprazole.
In studies in healthy volunteers, 2% of subjects showed a slower elimination of pantoprazole from serum/ plasma, with an increase in terminal elimination half-life of up to 10 h. Patients with a half-life of greater than 3.5 h and with an apparent clearance of less than 2 L/h/kg are considered to be slow metabolisers of pantoprazole.
Special populations. After a single 20 mg tablet, AUC increased 3-fold in patients with mild hepatic impairment and 5-fold in patients with severe hepatic impairment compared with healthy controls. Mean elimination half-life was 3.3 h in mild hepatic impairment and 6.0 h in severe hepatic impairment compared with 1.1 h in controls. The maximum serum concentration only increased slightly by a factor of 1.3 compared with healthy subjects.
In patients with renal impairment (including those undergoing dialysis) no dose reduction is required. Although the main metabolite is moderately increased, there is no accumulation. The half-life of pantoprazole is as short as in healthy subjects. Pantoprazole is poorly dialysable.
The slight increase in AUC and Cmax in elderly volunteers compared with their younger counterparts is also not clinically relevant.
5.3 Preclinical Safety Data
Genotoxicity. A number of in vitro and in vivo genotoxicity assays covering mutagenicity, clastogenicity and DNA damage end points were conducted on pantoprazole and the results were generally negative. Exposures achieved in the in vivo tests in mice and rats were well in excess of exposures expected clinically. However, pantoprazole was clearly positive in carefully conducted cytogenetic assays in human lymphocytes in vitro, both in the presence and absence of metabolic activation. Omeprazole was also positive in a comparable test conducted in the same laboratory, suggesting a possible class effect. A minute amount of radioactivity was bound to rat hepatic DNA after treatment with 200 mg/kg/day pantoprazole for 14 days. However, no distinct DNA-adduct has been detected.
Pantoprazole was found to be negative in the following studies: in vivo chromosome aberration assay in rat and bone marrow (126E/95), mouse lymphoma test (222E/95) and a gene mutation test in Chinese hamster ovary cells (in vitro) (188E/95). In addition, toxicokinetic studies were conducted in rats at the doses used in the bone marrow assay (50 to 1200 mg/kg) (56E/96) and in mice at the high dose from the earlier micronucleus test (710 mg/kg) (89E/96). In both species, pantoprazole exposure was high with the AUCs being 26 to 30 times higher in the rat or mouse respectively, than humans using the 20 mg tablet.
Carcinogenicity. A two year oral carcinogenicity study in Sprague Dawley rats at doses up to 200 mg/kg/day showed gastric carcinoids after pantoprazole treatment at doses greater than 0.5 mg/kg/day in females and greater than 5 mg/kg/day in males, with none observed in controls. The development of gastric tumours is attributed to chronic elevation of serum gastrin levels with associated histopathological changes in the gastrointestinal system.
In both male and female rats, the development of hepatocellular adenomas was increased at doses greater than 5 mg/kg/day and the development of hepatocellular carcinomas was increased at doses greater than 50 mg/kg/day. Hepatocellular tumours, which were also observed in female mice at oral doses greater than 25 mg/kg/day, may be associated with pantoprazole-induced increases in hepatic enzyme activity.
Treatment with pantoprazole at doses greater than 50 mg/kg/day also increased the development of thyroid follicular cell adenomas in male and female rats. Several studies in rats were conducted to investigate the effect of pantoprazole on the thyroid, the results of which suggested that the effect may be secondary to the induction of enzymes in the liver.
In a more recent carcinogenicity study, Fischer rats were studied using lower doses (5, 15 and 50 mg/kg,). Gastric carcinoids were detected at all doses in females and at the 15 and 50 mg/kg doses in males and none were detected in controls. No metastases of these carcinoids were detected. There was no increase in incidence of liver tumours. The dose of 15 mg/kg is seen to be the no-effect level for liver tumours in rodents.
Consideration of the possible mechanisms involved in the development of the above drug-related tumour types suggests that it is unlikely that there is any carcinogenic risk in humans at therapeutic dose levels of pantoprazole for short term treatment.
6 Pharmaceutical Particulars
6.1 List of Excipients
Pharmacy Action Heartburn Relief tablets contain mannitol, sodium carbonate, sodium starch glycollate, crospovidone, colloidal anhydrous silica, calcium stearate, hypromellose, macrogol 6000, sodium hydroxide, EUDRAGIT L30-D55 and OPADRY AMB aqueous moisture barrier coating system 80W52172 yellow.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Pharmacy Action Heartburn Relief 20 mg tablets are available in Al/Al blister packs of 7 and 14 tablets.
Not all pack sizes are available.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
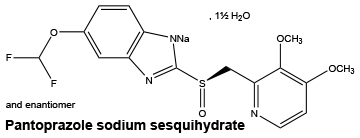
Pantoprazole is a substituted benzimidazole which inhibits basal and stimulated gastric secretion. It is a white to off white crystalline powder. Freely soluble in water and in ethanol (96%), practically insoluble in hexane. Solubility is low at neutral pH and increases with increasing pH.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
(S3) Pharmacist Only Medicine (14 tablets).
(S2) Pharmacy Medicine (7 tablets).
Date of First Approval
04 June 2018
Date of Revision
03 February 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.

