Plasma-Lyte 148 and 5% Glucose Intravenous Infusion
Brand Information
| Brand name | Plasma-Lyte 148 and 5% Glucose Intravenous Infusion |
| Active ingredient | Sodium chloride + Sodium gluconate + Sodium acetate trihydrate + Potassium chloride + Magnesium chloride hexahydrate + Glucose |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Plasma-Lyte 148 and 5% Glucose Intravenous Infusion.
Summary CMI
Plasma-Lyte 148 and 5% Glucose Intravenous Infusion
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I receiving Plasma-Lyte 148 and Glucose?
Plasma-Lyte 148 and 5% Glucose Intravenous Infusion (Plasma-Lyte 148 and Glucose) contains a mix of fluid, salts (electrolytes) and glucose. Plasma-Lyte 148 and Glucose is used as a source of water, salts and energy.
For more information, see Section 1. Why am I receiving Plasma-Lyte 148 and Glucose? in the full CMI.
2. What should I know before I receive Plasma-Lyte 148 and Glucose?
Do not use if you have ever had an allergic reaction to any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I receive Plasma-Lyte 148 and Glucose? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Plasma-Lyte 148 and Glucose and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is Plasma-Lyte 148 and Glucose given?
- Your doctor or trained nurse will give Plasma-Lyte 148 and Glucose by an infusion (drip) into a vein (intravenously).
- Your doctor will decide how much Plasma-Lyte 148 and Glucose to give. The amount depends on your need and condition.
More instructions can be found in Section 4. How is Plasma-Lyte 148 and Glucose given? in the full CMI.
5. What should I know while receiving Plasma-Lyte 148 and Glucose?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while receiving Plasma-Lyte 148 and Glucose? in the full CMI.
6. Are there any side effects?
For more information on what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Plasma-Lyte 148 and 5% Glucose Intravenous Infusion
Active ingredient(s): sodium chloride, sodium gluconate, sodium acetate trihydrate, potassium chloride, magnesium chloride hexahydrate, glucose
Consumer Medicine Information (CMI)
This leaflet provides important information about using Plasma-Lyte 148 and 5% Glucose Intravenous Infusion (Plasma-Lyte 148 and Glucose). You should also speak to your doctor, nurse or pharmacist if you would like further information or if you have any concerns or questions about using Plasma-Lyte 148 and Glucose.
Where to find information in this leaflet:
1. Why am I receiving Plasma-Lyte 148 and Glucose?
2. What should I know before I receive Plasma-Lyte 148 and Glucose?
3. What if I am taking other medicines?
4. How is Plasma-Lyte 148 and Glucose given?
5. What should I know while receiving Plasma-Lyte 148 and Glucose?
6. Are there any side effects?
7. Product details
1. Why am I receiving Plasma-Lyte 148 and Glucose?
Plasma-Lyte 148 and Glucose contains the active ingredients:
- Sodium chloride
- Sodium gluconate
- Sodium acetate trihydrate
- Potassium chloride
- Magnesium chloride hexahydrate
- Glucose
Plasma-Lyte 148 and Glucose is used as a source of water and electrolytes (salts) and energy (glucose).
2. What should I know before I receive Plasma-Lyte 148 and Glucose?
Warnings
Do not use Plasma-Lyte 148 and Glucose if:
- you are allergic to any of the ingredients listed at the end of this leaflet.
- you have had an allergic reaction to corn or corn products.
- Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have heart disease
- have respiratory lung disease
- have severe kidney disease
- have high blood pressure
- have build-up of fluid under the skin, particularly around the ankles
- have build-up of fluid in the lungs
- have high blood pressure during pregnancy (pre-eclampsia or eclampsia)
- have aldosteronism (a disease that causes high levels of hormone called aldosterone)
- have any condition that means you are more likely to have high blood levels of potassium such as kidney failure, adrenocortical insufficiency, severe dehydration (a loss of water from the body as can occur with vomiting or diarrhoea) or extensive tissue damage (as can occur in severe burns)
- have any other condition associated with retaining sodium or fluid, such as treatment with steroids
- have diabetes (your blood sugar levels will be monitored closely and your insulin treatment may need to be modified)
- have myasthenia gravis (a disease that causes progressive muscle weakness)
- are recovering after an operation
- had a head injury within the past 24 hours
- have high pressure within the skull (intracranial hypertension)
- have had a stroke
- have any other medical conditions
- take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Plasma-Lyte 148 and Glucose may interfere with each other and affect your health.
Medicines that may lead to more sodium or fluid in the body include:
- corticosteroids (anti-inflammatory medicines)
- corticotrophin (used for diagnosis or to treat swelling)
Medicines that are affected by changes in body pH include:
- aspirin/salicylates (anti-inflammatory medicines)
- barbiturates (used to treat epilepsy)
- lithium (used to treat psychiatric illnesses)
- sympathomimetics such as pseudoephedrine (used in cough and cold preparations), ephedrine, quinidine and dexamphetamine (for treatment of ADHD).
Medicines that may lead to high levels of potassium in your blood include:
- potassium-sparing diuretics (amiloride, spironolactone, triamterene)
- angiotensin converting enzyme inhibitors or angiotensin II receptor antagonist (used in treatment of high blood pressure)
- tacrolimus and cyclosporin (used to prevent rejection of a transplant)
Medicines that may lead to very low sodium levels in your blood include:
- clofibrate, a medicine to lower cholesterol
- antiepileptics (anti-seizure medicines) such as carbamazepine and oxcarbazepine
- chemotherapy medicines for cancer such as chlorpropamide, vincristine, ifosfamide, cyclophosphamide
- medicines to treat depression and anxiety
- antipsychotics, that help manage psychosis
- pain relievers such as opioids and NSAIDS
- medicines to treat diabetes such as desmopressin and vasopressin
- terlipressin, a medicine to treat liver and kidney problems
- oxytocin, a medicine used to induce labour and control postpartum bleeding
- diuretics, medicines used to increase urine output.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Plasma-Lyte 148 and Glucose.
4. How is Plasma-Lyte 148 and Glucose given?
How it is given
- Your doctor or trained nurse will give Plasma-Lyte 148 and Glucose by an infusion (drip) into a vein (intravenously).
- It is usually given in a hospital, clinic or nursing home.
The infusion requires special equipment and medical training. You may receive it at home if your doctor has arranged for it.
How much is given
- Your doctor will decide how much Plasma-Lyte 148 and Glucose to give. The amount depends on your need and condition.
- Your doctor will decide how long you need to be given this medicine.
If you are given too much Plasma-Lyte 148 and Glucose
The doctor or nurse giving you Plasma-Lyte 148 and Glucose is highly trained and qualified, so it is unlikely that you will be given an overdose.
If you think that you have been given too much, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while receiving Plasma-Lyte 148 and Glucose?
Things you should do
Remind any doctor, nurse, dentist or pharmacist you visit that you are using Plasma-Lyte 148 and Glucose.
Things you should not do
- Do not stop using this medicine suddenly.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Plasma-Lyte 148 and Glucose affects you.
The effects of Plasma-Lyte 148 and Glucose on driving and using machines or tools have not been studied.
Drinking alcohol
Tell your doctor if you drink alcohol.
It is unknown how alcohol may affect Plasma-Lyte 148 and Glucose.
Looking after your medicine
You will not have to handle or store Plasma-Lyte 148 and Glucose. It will be stored in the pharmacy or the hospital ward, below 30°C.
If for any reason you need to keep this product at home it should be stored in a locked cupboard.
- Store below 30°C.
- Do not freeze.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
This medicine is for single use, and for one person only. Your doctor or nurse will discard any unused medicine. They will not save unused portions for later, either for you or anyone else.
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Infusion site-related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergic reaction-related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Plasma-Lyte 148 and Glucose contains
| Active ingredients (main ingredients) |
|
| Other ingredients (inactive ingredients) | Hydrochloric acid, Water for Injections |
Do not take this medicine if you are allergic to any of these ingredients.
What Plasma-Lyte 148 and Glucose looks like
Plasma-Lyte 148 and 5% Glucose Intravenous Infusion is a clear solution in VIAFLEX (plastic) bags. It is available as:
- Plasma-Lyte 148 and 5% Glucose 1000mL injection bag (Aust R 19438).
Who distributes Plasma-Lyte 148 and Glucose
Baxter Healthcare Pty Ltd
1 Baxter Drive
Old Toongabbie
NSW 2146
Australia
This leaflet was prepared in September 2025
Baxter, Plasma-Lyte and VIAFLEX are trademarks of Baxter International Inc.
Brand Information
| Brand name | Plasma-Lyte 148 and 5% Glucose Intravenous Infusion |
| Active ingredient | Sodium chloride + Sodium gluconate + Sodium acetate trihydrate + Potassium chloride + Magnesium chloride hexahydrate + Glucose |
| Schedule | Unscheduled |
MIMS Revision Date: 01 July 2019
1 Name of Medicine
Sodium chloride, sodium gluconate, sodium acetate trihydrate, potassium chloride, magnesium chloride hexahydrate and anhydrous glucose.
2 Qualitative and Quantitative Composition
Plasma-Lyte 148 and 5% Glucose Intravenous (IV) Infusion is a sterile, clear, non-pyrogenic hypertonic solution in a single dose container for intravenous administration. It contains no antimicrobial agents. The approximate osmolality is 584 mOsm/kg. An injection with an osmolality within the range of 250 to 350 mOsm/kg is considered to be isotonic. Administration of substantially hypertonic solutions (≥ 600 mOsm/kg) may cause vein damage.
Each 1000 mL of Plasma-Lyte 148 and 5% Glucose IV Infusion contains (see Table 1):

For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for intravenous infusion.
4 Clinical Particulars
4.1 Therapeutic Indications
Plasma-Lyte 148 and 5% Glucose IV Infusion is indicated as a source of water, electrolytes and calories or as an alkalinising agent.
4.2 Dose and Method of Administration
Dosage. As directed by the physician. Dosage, rate and duration of administration are to be individualised and depend upon the indication for use, the patient's age, weight, clinical condition and concomitant treatment, and on the patient's clinical and laboratory response to treatment.
Each Viaflex container is for single patient use only. All injections in Viaflex plastic containers are intended for intravenous administration using sterile equipment.
As reported in the literature, the dosage, volume and infusion rate of intravenous glucose must be selected with caution in paediatric patients, particularly neonates and low birth weight infants, because of the increased risk of hyperglycaemia/hypoglycaemia (see Section 4.4 Special Warnings and Precautions for Use, Paediatric use).
Directions for use of Viaflex plastic container. Warning. Do not use plastic containers in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed (see Section 4.4 Special Warnings and Precautions for Use, Risk of air embolism).
Parenteral drug products should be inspected visually for particular matter and discolouration prior to the administration whenever solution and container permit. Do not administer unless solution is clear and seal is intact.
To open. Tear overwrap down side at slit and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilisation process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Check for minute leaks by squeezing the inner bag firmly. If leaks are found, discard solution, as sterility may be impaired. If supplemental medication is desired, follow the directions below.
Preparation for administration. Plasma-Lyte 148 and 5% Glucose IV Infusion is a sterile preparation. Thus, aseptic technique must be applied throughout the administration.
(1) Suspend container from eyelet support.
(2) Remove plastic protector from outlet port at the bottom of container.
(3) Attach administration set.
To add medications. Warning. Additives may be incompatible. Those additives known to be incompatible should not be used. Consult with a pharmacist if available. As with all parenteral solutions, compatibility of the additives with the solution must be assessed before addition. Before adding a substance or medication, verify that it is soluble and/or stable in water and that the pH range of Plasma-Lyte 148 and 5% Glucose IV Infusion is appropriate. After addition, check for possible colour change and/or the appearance of precipitates, insoluble complexes or crystals. The instructions for use of the medication to be added and other relevant literature must be consulted (see Section 6.2 Incompatibilities).
If, in the informed judgement of the physician, it is deemed advisable to introduce additives, use aseptic technique. Mix thoroughly when additives have been introduced. Do not store solutions containing additives.
To add medication before solution administration. Prepare medication site. Using syringe with 0.63 to 0.80 mm needle, puncture resealable medication port and inject. Mix solution and medication thoroughly. For high-density medication such as potassium chloride, squeeze ports while ports are upright and mix thoroughly.
To add medication during solution administration. Close clamp on the set. Prepare medication site. Using syringe with 0.63 to 0.80 mm needle, puncture resealable medication port and inject. Remove container from IV pole and/or turn to upright position. Evaluate both ports by squeezing them while container is in the upright position. Mix solution and medication thoroughly. Return container to in use position and continue administration.
After opening the container, the contents should be used immediately and should not be stored for a subsequent infusion. Do not reconnect any partially used containers.
4.3 Contraindications
Plasma-Lyte 148 and 5% Glucose IV Infusion is contraindicated in patients with a known hypersensitivity to the product. Solutions containing glucose may be contraindicated in patients with known allergy to corn or corn products.
Plasma-Lyte 148 and 5% Glucose IV Infusion must not be used in patients with clinically significant hyperglycemia.
4.4 Special Warnings and Precautions for Use
Plasma-Lyte 148 and 5% Glucose IV Infusion is not indicated for:
the treatment of hypochloremic hypokalaemic alkalosis;
the primary treatment of severe metabolic acidosis;
the treatment of hypomagnesaemia.
Although Plasma-Lyte 148 and 5% Glucose IV Infusion has a potassium concentration similar to the concentration in plasma, it is insufficient to produce a useful effect in case of severe potassium deficiency; therefore, it should not be used for correction of severe potassium deficiency.
Plasma-Lyte 148 and 5% Glucose IV Infusion should not be administered simultaneously with blood through the same administration set because of the possibility of pseudo-agglutination or haemolysis.
Hypersensitivity reactions. Hypersensitivity/infusion reactions, including anaphylactoid reactions, have been reported with Plasma-Lyte 148 and 5% Glucose IV Infusion. The infusion must be stopped immediately if any signs or symptoms of a suspected hypersensitivity reaction develop. Appropriate therapeutic countermeasures must be instituted as clinically indicated.
Hypertonic solution. Plasma-Lyte 148 and 5% Glucose IV Infusion is a hypertonic solution, having an osmolality of 584 mOsm/kg. Administration of hypertonic solutions may cause venous irritation including phlebitis. Hyperosmolar solution should be administered with caution, if at all, to patients with hyperosmolar states e.g. hypochloraemic hypokalaemic alkalosis due to prolonged vomiting, pyloric stenosis, prolonged nasogastric suctioning.
Fluid and/or solute overload and electrolyte disturbances. Depending on the volume and rate of infusion, intravenous administration of Plasma-Lyte 148 and 5% Glucose IV Infusion can cause:
fluid and/or solute overload resulting in overhydration/hypervolaemia and for example congested states, including pulmonary congestion and oedema;
clinically relevant electrolyte disturbances and acid-base imbalance.
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid base balance during prolonged parenteral therapy or whenever the condition of the patient or the rate of administration warrants such evaluation.
Hyponatraemia. Glucose intravenous infusions are usually isotonic solutions. Plasma-Lyte 148 and 5% Glucose IV Infusion has an osmolarity of 584 mOsm/kg. In the body, however, glucose containing fluids can become extremely physiologically hypotonic due to rapid glucose metabolisation. Monitoring of serum sodium is particularly important for hypotonic fluids.
Depending on the tonicity of the solution, the volume and rate of infusion, and depending on a patient's underlying clinical condition and capability to metabolise glucose, intravenous administration of glucose can cause electrolyte disturbances, most importantly hypo- or hyperosmotic hyponatraemia.
High volume infusion must be used under specific monitoring in patients with cardiac or pulmonary failure, and in patients with non-osmotic vasopressin release (including SIADH), due to the risk of hospital-acquired hyponatraemia. Acute hyponatraemia can lead to acute hyponatraemic encephalopathy (cerebral oedema) characterized by headache, nausea, seizures, lethargy and vomiting. Patients with cerebral oedema are at particular risk of severe, irreversible and life-threatening cerebral injury.
Use in patients with or at risk of hypermagnesaemia. Solutions containing magnesium should be used with caution, if at all, in patients with:
hypermagnesaemia or conditions predisposing to hypermagnesaemia including, but not limited to, severe renal impairment or magnesium therapy such as eclampsia;
myasthenia gravis.
Use in patients with or at risk of alkalosis. Plasma-Lyte 148 and 5% Glucose IV Infusion should be used with particular caution, if at all, in patients with alkalosis or at risk for alkalosis. Excess administration may result in metabolic alkalosis.
The administration of acetate or gluconate ions should be done with great care in those conditions in which there is an increased level or an impaired utilisation of these ions, such as severe hepatic insufficiency.
Use in patients with hypervolaemia or overhydration, or conditions that cause sodium retention and oedema. The risk of dilutional states is inversely proportional to the electrolyte concentrations of the infusion. The risk of solute overload causing congested states with peripheral and pulmonary oedema is directly proportional to the electrolyte concentrations of the infusion.
Plasma-Lyte 148 and 5% Glucose IV Infusion should be administered with particular caution, to hypervolaemia or overhydrated patients.
Plasma-Lyte 148 and 5% Glucose IV Infusion should be used with great care, if at all, in patients with congestive heart failure, severe renal insufficiency, and in clinical states in which there exists oedema with sodium retention and fluid overload, such as patients with primary hyperaldosteronism, secondary hyperaldosteronsim (associated with, for example, hypertension, congestive heart failure, renal artery stenosis or nephrosclerosis) or preeclampsia.
Use in patients with hypocalcaemia. Plasma-Lyte 148 and 5% Glucose IV Infusion contains no calcium and an increase in plasma pH due to its alkalinising effect may lower the concentration of ionised (not protein-bound) calcium. Plasma-Lyte 148 and 5% Glucose IV Infusion should be administered with particular caution, if at all, to patients with hypocalcaemia.
Use in patients with or at risk of hyperkalaemia. Plasma-Lyte 148 and 5% Glucose IV Infusion should be used with caution, if at all, in patients with hyperkalaemia or conditions predisposing to hyperkalaemia (such as severe renal impairment or adrenocortical insufficiency, acute dehydration or extensive tissue injury or burns) and in patients with cardiac disease and in conditions where potassium retention is present.
Use in patients with severe renal impairment. In patients with diminished renal function, administration of Plasma-Lyte 148 and 5% Glucose IV Infusion may result in sodium and/or potassium or magnesium retention.
Use in patients with or at risk of hyperglycaemia. Plasma-Lyte 148 and 5% Glucose IV Infusion should be used with caution in patients with impaired glucose tolerance or diabetes mellitus.
Because Plasma-Lyte 148 and 5% Glucose IV Infusion contains glucose as well as gluconate (a portion of which may be metabolised into glucose), administration of Plasma-Lyte 148 and 5% Glucose IV Infusion that exceeds the metabolic capacity for glucose may lead to hyperglycaemia (see Section 4.4 Special Warnings and Precautions for Use, Paediatric use).
In order to avoid hyperglycaemia, the infusion rate should not exceed the patient's ability to utilise glucose. Hyperglycaemia has been implicated in increasing cerebral ischemic brain damage and impairing recovery after acute ischemic strokes. Caution is recommended in using glucose-containing solutions in such patients.
Early hyperglycaemia has been associated with poor outcomes in patients with severe traumatic brain injury. Glucose-containing solutions should, therefore, be used with caution in patients with head injury, in particular during the first 24 hours following the trauma.
If hyperglycaemia occurs, the rate of glucose administration should be reduced and/or insulin administered, or the insulin dose adjusted.
Risk of air embolism. Do not connect flexible plastic containers in series in order to avoid air embolism due to possible residual air contained in the primary container. Pressurising intravenous solutions contained in flexible plastic containers to increase flow rates can result in air embolism if the residual air in the container is not fully evacuated prior to administration.
Use of a vented intravenous administration set with the vent in the open position could result in air embolism. Vented intravenous administration sets with the vent in the open position should not be used with flexible plastic containers.
Other. The Viaflex plastic container is fabricated from a specially formulated polyvinyl chloride (PL 146 Plastic). The amount of water that can permeate from inside the container into the overwrap is insufficient to affect the solution significantly. Solution in contact with the plastic container can leach out certain chemical components from the plastic in very small amounts: however, biological testing was supportive of the safety of the plastic container material.
Use in the elderly. Clinical studies of Plasma-Lyte 148 and 5% Glucose IV Infusion did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or drug therapy.
When selecting the type of infusion solution and the volume/rate of infusion for an elder patient, consider that elderly patients are generally more likely to have cardiac, renal, hepatic and other diseases or concomitant drug therapy.
Paediatric use. Safety and effectiveness of Plasma-Lyte 148 and 5% Glucose IV Infusion in paediatric patients have not been established by adequate or well controlled trials, however, the use of electrolyte solutions in the paediatric population is referenced in the medical literature. The precautions and adverse reactions identified in this document should be observed in the paediatric population.
The infusion rate and volume depends on the age, weight, clinical and metabolic conditions of the patient, concomitant therapy and should be determined by the consulting physician experienced in paediatric intravenous fluid therapy.
In very low birth weight infants, excessive or rapid administration of glucose infusion may result in increased serum osmolarity and possible haemorrhage.
Newborns - especially those born premature and with low birth weight - are at increased risk of developing hypo- or hyperglycaemia and therefore need close monitoring during treatment with intravenous glucose solutions to ensure adequate glycaemic control in order to avoid potential long term adverse effects. Hypoglycaemia in the newborn can cause prolonged seizures, coma and brain damage. Hyperglycaemia has been associated with intraventricular haemorrhage, late onset bacterial and fungal infection, retinopathy of prematurity, necrotising enterocolitis, bronchopulmonary dysplasia, prolonged length of hospital stay, and death.
Effects on laboratory tests. There have been reports of false-positive test results using the Bio Rad Laboratories Platelia Aspergillus EIA test in patients receiving Baxter gluconate containing Plasma-Lyte solutions. These patients were subsequently found to be free of Aspergillus infection. Therefore, positive test results for this test in patients receiving Baxter gluconate containing Plasma-Lyte solutions should be interpreted cautiously by other diagnostic methods.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Plasma-Lyte 148 and 5% Glucose IV Infusion should not be administered simultaneously with blood through the same administration set because of the possibility of pseudo-agglutination or haemolysis (see Section 4.4 Special Warnings and Precautions for Use).
Caution must be exercised in the administration of Plasma-Lyte 148 and 5% Glucose IV Infusion to patients treated with drugs that may increase the risk of sodium and fluid retention such as corticosteroids or corticotropin.
Caution is advised when administering Plasma-Lyte 148 and 5% Glucose IV Infusion to patients treated with drugs for which renal elimination is pH dependent. Due to its alkalinising effect (formation of bicarbonate), Plasma-Lyte 148 and 5% Glucose IV Infusion may interfere with the elimination of such drugs:
renal clearance of acidic drugs such as salicylates, barbiturates and lithium may be increased;
renal clearance of alkaline drugs such as sympathomimetics (e.g. ephedrine, pseudoephedrine, quinidine or dextroamphetamine (dexamphetamine) sulfate may be decreased.
Because of its potassium content, Plasma-Lyte 148 and 5% Glucose IV Infusion should be administered with caution in patients treated with agents or products that can cause hyperkalaemia or increase the risk of hyperkalaemia, such as potassium sparing diuretics (amiloride, spironolactone, triamterene) with ACE inhibitors, angiotensin II receptor antagonists or the immunosuppressants tacrolimus and cyclosporin.
Caution is advised when administering Plasma-Lyte 148 and 5% Glucose IV Infusion to patients treated with drugs leading to an increased vasopressin effect. The below listed drugs increase the vasopressin effect, leading to reduced renal electrolyte free water excretion and may increase the risk of hyponatraemia following treatment with IV fluids (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)):
Drugs stimulating vasopressin release such as chlorpropamide, clofibrate, carbamazepine, vincristine, selective serotonin reuptake inhibitors (SSRIs), 3,4-methylenedioxy-N-methamphetamine, ifosfamide, antipsychotics, opioids.
Drugs potentiating vasopressin action such as chlorpropamide, non-steroidal anti-inflammatories (NSAIDS), cyclophosphamide.
Vasopressin analogues such as desmopressin, oxytocin, vasopressin, terlipressin.
Caution is advised when administering Plasma-Lyte 148 and 5% Glucose IV Infusion to patients treated with drugs that may increase the risk of hyponatraemia, such as diuretics and antiepileptics (e.g. oxcarbazepine).
See Section 6.2 Incompatibilities.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Studies with Plasma-Lyte 148 and 5% Glucose IV Infusion have not been performed to evaluate effect on fertility.
Use in pregnancy. (No category)
Intrapartum maternal intravenous infusion of glucose containing solutions may result in foetal insulin production, with an associated risk of foetal hyperglycaemia and metabolic acidosis as well as rebound hypoglycaemia in the neonate. Physicians should carefully consider the potential risks and benefits for each specific patient before administering Plasma-Lyte 148 and 5% Glucose IV Infusion.
Use in lactation. There are no adequate data from the use of Plasma-Lyte 148 and 5% Glucose IV Infusion in lactating women. The potential risks and benefits for each specific patient should be carefully considered before using Plasma-Lyte 148 and 5% Glucose IV Infusion in lactating women.
4.7 Effects on Ability to Drive and Use Machines
There is no information of the effects of Plasma-Lyte 148 and 5% Glucose IV Infusion on the ability to operate an automobile or other heavy machinery.
4.8 Adverse Effects (Undesirable Effects)
Reactions that may occur because of the solution or the technique of administration include febrile response or infection at the site of infusion. Other reactions that may occur include:
Circulatory effects. Extravasation, hypervolaemia, venous thrombosis, phlebitis extending from the site of injection.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
The following adverse reactions have been reported in the post-marketing experience with unspecified Plasma-Lyte products (listed by MedDRA System Organ Class (SOC), then by Preferred Term in order of severity, where feasible):
Immune system disorders. Hypersensitivity/infusion reactions including anaphylactoid reaction and the following manifestation: hypotension, chest discomfort, dyspnoea, wheezing, flushing, hyperaemia, asthenia, urticaria, cold sweat, pyrexia and chills.
Metabolism and nutrition disorders. Hyperkalaemia and hyperglycaemia.
General disorders and administration site conditions. Infusion site reaction (e.g. burning sensation).
Other adverse reactions reported with Plasma-Lyte or other similar products are other manifestations of hypersensitivity/ infusion reactions including tachycardia, palpitations, chest pain, respiratory rate increased, feeling abnormal, piloerection, oedema peripheral and infusion site pain.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
If overdosage is suspected (through the monitoring of electrolytes, especially sodium and potassium), administration of the medicine should be discontinued and the patient observed closely.
Excessive administration of Plasma-Lyte 148 and 5% Glucose IV Infusion may lead to metabolic alkalosis. Metabolic alkalosis may be accompanied by hypokalaemia as well as a decrease in ionised serum calcium and magnesium (see Section 4.4 Special Warnings and Precautions for Use).
An excessive volume of Plasma-Lyte 148 and 5% Glucose IV Infusion may lead to fluid and sodium overload with a risk of oedema (peripheral and/or pulmonary) particularly when renal sodium excretion is impaired (see Section 4.4 Special Warnings and Precautions for Use).
Excessive administration of potassium may lead to the development of hyperkalaemia, especially in patients with severe renal impairment (see Section 4.4 Special Warnings and Precautions for Use).
Excessive administration of magnesium many lead to hypermagnesaemia (see Section 4.4 Special Warnings and Precautions for Use).
Excessive administration of glucose-containing solution may lead to hyperglycaemia, hyperosmolarity, osmotic diuresis and dehydration.
When assessing an overdose, any additives in the solution must also be considered. The effect of overdose may require immediate medical attention and treatment.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Plasma-Lyte 148 and 5% Glucose IV Infusion is a source of water, electrolytes and calories. It is capable of inducing diuresis depending on the clinical condition of the patient.
Plasma-Lyte 148 and 5% Glucose IV Infusion produces a metabolic alkalinising effect. Acetate and gluconate ions are metabolised ultimately to carbon dioxide and water, which requires the consumption of hydrogen cations.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. Studies with Plasma-Lyte 148 and 5% Glucose IV Infusion have not been performed to evaluate mutagenic potential.
Carcinogenicity. Studies with Plasma-Lyte 148 and 5% Glucose IV Infusion have not been performed to evaluate carcinogenic potential.
6 Pharmaceutical Particulars
6.1 List of Excipients
Water for injections; hydrochloric acid (for pH adjustment).
6.2 Incompatibilities
Additives may be incompatible. Those additives known to be incompatible should not be used. Consult with a pharmacist if available.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Do not freeze.
6.5 Nature and Contents of Container
Plasma-Lyte 148 and 5% Glucose IV Infusion in Viaflex plastic containers is available as shown in Table 2.
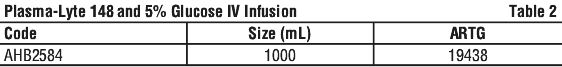
6.6 Special Precautions for Disposal
Any unused product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Sodium chloride. Molecular formula: NaCl.
Molecular Weight: 58.44.
Appearance: colourless or white crystal.
Solubility: freely soluble in water.
Sodium gluconate (sodium D-gluconate). Molecular formula: C6H11NaO7.
Molecular Weight: 218.14.
Appearance: white to off white crystalline powder.
Solubility: very soluble in water; sparingly soluble in alcohol; insoluble in ether.
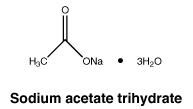
Sodium acetate trihydrate. Molecular formula: C2H3NaO2,3H2O.
Molecular Weight: 136.1.
Appearance: white crystalline solid.
Solubility: hygroscopic, very soluble in water; moderately soluble in ethanol.
Potassium chloride. Molecular formula: KCl.
Molecular Weight: 74.55.
Appearance: colourless or white crystal.
Solubility: freely soluble in water.
Magnesium chloride hexahydrate. Molecular formula: MgCl2.6H2O.
Molecular Weight: 203.31.
Appearance: white or colourless crystalline solid.
Solubility: very soluble in water.
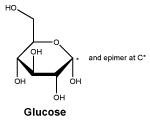
Glucose (D-(+)glucopyranose). Molecular formula: C6H12O6.
Molecular Weight: 180.2.
Appearance: a white or almost white, crystalline powder.
Solubility: freely soluble in water, sparingly soluble in ethanol (96%).
Chemical structure.
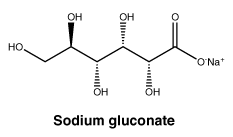
Sodium gluconate (sodium D-gluconate). CAS No.: 527-07-1.
Sodium acetate trihydrate. CAS No.: 6131-90-4.
Potassium chloride. CAS No.: 7447-40-7.
Magnesium chloride hexahydrate. CAS No.: 7791-18-6.
Glucose (D-(+)glucopyranose). CAS No.: 50-99-7.
7 Medicine Schedule (Poisons Standard)
Unscheduled.
Date of First Approval
30 September 1991
Date of Revision
30 May 2019
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.