Polaramine Syrup
Brand Information
| Brand name | Polaramine Syrup |
| Active ingredient | Dexchlorpheniramine maleate |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Polaramine Syrup.
What is in this leaflet?
This leaflet answers some common questions about Polaramine. It does not contain all of the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor or pharmacist has weighed the risks of you taking Polaramine against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What POLARAMINE is used for
Polaramine relieves symptoms associated with allergic rhinitis (including hayfever), such as sneezing, runny or itchy nose, and burning or itchy eyes.
Polaramine may also be used to relieve symptoms associated with hives (also known as chronic urticaria). These symptoms include itching, redness and lumps on the skin.
Polaramine can also be used to treat drug reactions.
Polaramine belongs to a class of medicines known as antihistamines.
Antihistamines help reduce allergic symptoms by preventing the effects of a substance called histamine. Histamine is produced by the body in response to foreign substances which the body is allergic to.
Your doctor or pharmacist may have prescribed Polaramine for another reason.
Ask your doctor or pharmacist if you have any questions about why Polaramine has been prescribed for you.
There is no evidence that Polaramine is addictive.
Before you take POLARAMINE
When you must not take it
Do not take Polaramine if:
- you are allergic to it or any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include skin rash, difficulty in breathing or faintness. - you are also taking medicines used to treat depression such as monoamine oxidase inhibitor (MAOI).
Taking Polaramine together with a MAOI may exaggerate the effects of Polaramine and cause a severe drop in your blood pressure.
Do not give Polaramine Syrup to children under 2 years of age.
Do not give Polaramine Syrup to children aged between 2-11 years of age except on the advice of a doctor, pharmacist or nurse practitioner.
Do not give Polaramine Tablets to children less than 12 years.
Do not take Polaramine after the expiry date (EXP) printed on the pack.
Do not take Polaramine if the packaging is torn or shows signs of tampering.
If you are not sure whether you should start using Polaramine, talk to your doctor or pharmacist.
Before you start to take it
Tell your doctor or pharmacist if you have allergies to any other medicines, any other substances such as foods, dyes or preservatives
Tell your doctor or pharmacist if you are pregnant or intend to become pregnant. If you are in the third trimester of pregnancy, you must tell your doctor you are taking Polaramine because newborn babies may have severe reactions to antihistamine.
Tell your doctor or pharmacist if you are breast-feeding or plan to breast-feed. Polaramine may be transferred in breast milk to your baby.
Tell your doctor or pharmacist if you have or have had any medical conditions, especially the following:
- seizures
- liver or kidney disease
- an overactive thyroid gland
- heart disease
- high blood pressure
- raised pressure in the eye
- prostate problems
- difficulty passing urine
- a narrowing or blockage between the stomach and small intestine which causes vomiting of undigested food
- peptic ulcer
If you have not told your doctor or pharmacist about any of the above, tell them before you start taking Polaramine.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including medicines that you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines should not be taken with Polaramine. These include: monoamine oxidase inhibitor (MAOI). These medicines may be affected by Polaramine and may cause a decrease in blood pressure.
Polaramine may interfere with other medicines. These include:
- alcohol
- medicines used to treat depression such as tricyclic antidepressants
- strong / narcotic pain killers, such as codeine, morphine and dextropropoxyphene
- some medicines used to help you sleep
- some medicines used to treat anxiety
- some medicines used to stop blood clotting, such as warfarin
These medicines may be affected by Polaramine, or may affect how well it works. You may need different amounts of your medicine, or you may need to take different medicines. Your doctor or pharmacist will advise you.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking Polaramine.
How to take POLARAMINE
How much to take
Polaramine Tablets
Adults and children over 12 years: One Polaramine tablet every 6 hours
After initial relief is obtained, dosage may be reduced to 1 tablet every 6 hours as required.
Do not give Polaramine Tablets to children under 12 years of age.
Polaramine Syrup
Adults and children over 12 years: 5mL every 6 hours
Children 6 to 12 years: 2 to 4mL every 6 to 8 hours
Children 4 to 6 years: 1.75 to 2mL every 6 to 8 hours
Children 2 to 4 years: 1.25 to 1.75mL every 6 to 8 hours
Do not give Polaramine Syrup to children under 2 years of age.
Take Polaramine exactly as your doctor or pharmacist has told you to. If you take the wrong dose, Polaramine may not work as well and you may not get relief from your symptoms.
How to take it
Swallow the tablet whole with a glass of water.
When to take it
It does not matter if you take Polaramine before or after food.
How long to take it
Continue taking Polaramine until your symptoms have resolved.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed
If there is still a long time to go before your next dose, take it as soon as you remember, and then go back to taking your medicine as you would normally
If you are not sure what to do, ask your doctor or pharmacist
If you have trouble remembering to take your medicine, ask your pharmacist for some hints
If you take too much (overdose)
Immediately telephone your doctor, pharmacist or Poisons Information Centre (13 11 26) for advice, or go to casualty at your nearest hospital, if you think that you or anyone else may have taken too much Polaramine
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
If you take too much Polaramine, you may feel dizzy or light-headed and may have difficulty breathing.
While you are taking POLARAMINE
Things you must do
Tell all doctors, dentists and pharmacists who are treating you that you are taking Polaramine.
If you become pregnant while taking Polaramine, tell your doctor
Things you must not do
Do not give Polaramine to anyone else, even if their symptoms seem similar to yours.
Do not take Polaramine to treat any other complaints unless your doctor or pharmacist says to.
Do not drink alcohol while taking Polaramine. The effects of alcohol can be increased by some antihistamine medicines, including Polaramine. If you drink alcohol, the drowsiness, dizziness or light-headedness may become worse.
If you feel tired, drowsy, dizzy or light-headed, do not drive a vehicle or operate machinery. As with some other antihistamine medicines, Polaramine may cause tiredness, drowsiness, dizziness or light-headedness in some people.
Things to be careful of
Protect your skin when you are in the sun, especially between 10am and 3pm.
If you are outdoors, wear protective clothing and use a 30+ sunscreen. Polaramine may cause your skin to be more sensitive than it is normally.
If your mouth continues to feel dry for more than 2 weeks check with your doctor or dentist. Antihistamines may cause dryness of the mouth, nose and throat
Stop taking Polaramine 48 hours before you have any skin tests. Antihistamines may interfere with the results of skin tests
Side effects
All medicines may have some unwanted side effects. Sometimes they are serious, but most of the times they are not. Your doctor or pharmacist has weighed the risk of using this medicine against the benefit they expect it will have for you.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
All medicines have side effects. You may need medical treatment if you get some of the side effects
Ask your doctor or pharmacist any questions you may have.
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Polaramine.
It helps most people with allergies, but it may have unwanted side effects in a few people.
If you are over 60 years of age you may have an increased chance of getting side effects
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- sleepiness
- upset stomach
- loss of appetite
- thickening of mucus
- stuffy nose
- tight chest
- dry mouth, nose or throat
These are mild side effects of medicines, and usually short lived.
Tell your doctor immediately if you notice any of the following:
- wheezing or being short of breath
- fast, pounding or irregular heart beats
- skin rash
- tiredness, headache, dizziness, spinning sensation, with yellowing of the skin and/or eyes
- unable or difficulty passing urine
- passing urine more often than usual
- hallucinations
- tingling or numbness in the hands or feet, also known as 'pins and needles'
These may be serious side effects. You may need urgent medical attention. Serious side effects are rare.
If any of the following happen, stop taking Polaramine and tell your doctor immediately, or go to Accident and Emergency at your nearest hospital:
- chest pain
- sudden signs of allergy such as rash, hives
- swelling of the face, lips, mouth, tongue or throat which may cause difficulty in swallowing or breathing
- convulsions, fits or seizures
- coma
These are very serious side effects. You may need urgent medical attention or hospitalisation.
All of these side effects are very rare.
Other side effects not listed above may also occur in some patients.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
After taking POLARAMINE
Storage
Keep your tablets in the blister pack until it is time to take them. If you take your tablets out, they will not keep well.
Syrup, rinse dispensing syringe with hot water after use. Replace the cap firmly.
Keep the medicines in a cool dry place where the temperature stays below 25°C.
Do not store it or any other medicine in the bathroom, or near a sink, or on a window sill.
Do not leave it in the car on hot days. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines
Disposal
If your doctor or pharmacist tells you to stop taking the tablets or the syrup, or they have passed the expiry date.
Ask your pharmacist what to do with any that are left over.
Product description
Polaramine can be bought without a doctor's prescription.
What it looks like
Polaramine comes in two forms:
Tablets and Syrup
Polaramine Tablets 2mg are off-white, round, bevelled tablets with score on one side, and blank on the other side, in blister packs of 20 and 40 tablets
Polaramine Syrup 2mg/5mL is a clear, red syrup with an orange like flavour in a 100 mL bottle
Ingredients
Polaramine Tablets contain:
Active ingredient: 2mg dexchlorpheniramine maleate
Inactive ingredients:
- lactose
- starch-maize
- starch-pregelatinised maize
- magnesium stearate
Polaramine Syrup contains:
Active ingredient:
2mg/5mL dexchlorpheniramine maleate
Inactive Ingredients:
- sodium citrate
- sodium chloride
- sucrose
- sorbitol (2.8g/20mL)
- methyl hydroxybenzoate
- propyl hydroxybenzoate
- menthol
- ethanol
- water
- brilliant scarlet 4R
- apricot flavour
- blood orange flavour
Polaramine Syrup contains sugars, alcohol, sulfites, hydroxybenzoates, sorbitol (2.8g/20mL).
Products containing sorbitol may have a laxative effect or cause diarrhoea
Sponsor
Bayer Australia Limited
875 Pacific Highway,
Pymble NSW 2073.
Australia
Ph: 1800 008 757
Bayer New Zealand Limited
Auckland 0627.
New Zealand
Ph: 0800 229 376
Australian Registration Numbers
Syrup: AUST R 18197
Tablets: AUST R 154653
Date of Preparation
November 2019
Brand Information
| Brand name | Polaramine Syrup |
| Active ingredient | Dexchlorpheniramine maleate |
| Schedule | S3 |
MIMS Revision Date: 01 July 2022
Name of the medicine
Dexchlorpheniramine maleate.
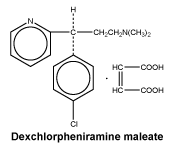
Description
Polaramine (dexchlorpheniramine maleate) is the dextro-isomer of chlorpheniramine maleate. It is an antihistamine with anticholinergic properties.
Dexchlorpheniramine maleate (CAS no. 2438-32-6) is described chemically as (+)-2-[p-chloro-α-[2-(dimethylamino)ethyl]benzyl]pyridine maleate (1:1).
It has the empirical formula of C16H19ClN2.C4H4O4.
Dexchlorpheniramine maleate is a white, odourless, crystalline powder which in aqueous solution has a pH of between 4 and 5. It is freely soluble in water, soluble in alcohol and in chloroform, but only slightly soluble in benzene or ether.
Pharmacology
Pharmacodynamics. Mechanism of action. Dexchlorpheniramine, the d-isomer of the racemic compound chlorpheniramine, is two times more active than chlorpheniramine. Dexchlorpheniramine does not prevent the release of histamine, but rather, competes with free histamine for binding at the H1-receptor sites, and competitively antagonises the effects of histamine on H1-receptors in the GI tract, uterus, large blood vessels and bronchial muscle. Blockade of H1-receptors also suppresses the formation of oedema, flare and pruritus that result from histaminic activity. Since dexchlorpheniramine binds to central and peripheral H1-receptors, sedative effects are likely to occur. H1-antagonists are structurally similar to anticholinergic agents and, therefore, possess the potential to exhibit anticholinergic properties of varying degrees. They also have antipruritic effects. Dexchlorpheniramine has high antihistaminic activity, moderate anticholinergic effects and minimal sedative effects. The drug does not possess antiemetic properties.
Pharmacokinetics. The absorption, distribution, metabolism and elimination of dexchlorpheniramine have not been specifically described. However, since dexchlorpheniramine is the primary active isomer of the racemic compound chlorpheniramine, the pharmacokinetics of dexchlorpheniramine are likely to be similar to that of chlorpheniramine.
Dexchlorpheniramine is administered orally. H1-antagonists are generally well absorbed from the GI tract. The onset of action of immediate release formulations of chlorpheniramine is about 30-60 minutes. The Cmax of chlorpheniramine occurs in about 2 hours, the maximum therapeutic effect in about 6 hours and the duration of action lasts between 4-8 hours. Protein binding is approximately 72%. Chlorpheniramine is widely distributed in body tissues and fluids, and it crosses the placenta and is excreted into breast milk.
The metabolism of chlorpheniramine is extensive and rapid, first occurring in the gastric mucosa and then on first-pass through the liver, which may be saturable. N-dealkylation produces several metabolites, which are excreted in the urine along with the parent compound. The half-life in healthy adults and children is 20-24 hours and 10-13 hours, respectively. Excretion rates are dependent on the pH of urine and urinary flow, with the rate decreasing as the pH rises and urinary flow decreases.
Indications
Polaramine is indicated for symptomatic treatment of perennial and seasonal allergic rhinitis, vasomotor rhinitis, allergic conjunctivitis, mild uncomplicated allergic skin manifestations of urticaria and angioedema. Polaramine may relieve itching due to skin conditions such as allergic eczema, pruritus ani, pruritus vulvae, atopic dermatitis, contact dermatitis, insect bites, dermographism and drug reactions, including serum sickness.
Contraindications
Polaramine is contraindicated for use in: children under 2 years of age due to the risk of respiratory depression; patients taking monoamine oxidase inhibitors (MAOIs) (see Interactions with Other Medicines); patients with a history of hypersensitivity to dexchlorpheniramine, to other drugs of similar chemical structure, or to any other excipients.
Precautions
Polaramine may cause drowsiness and may add to the effects of alcohol. Drowsiness may continue the following day. Those affected should not drive or operate machinery; alcohol should be avoided.
Polaramine should be used with caution in patients with: narrow angle glaucoma; stenosing peptic ulcer; prostatic hypertrophy; bladder neck obstruction; pyloroduodenal obstruction; cardiovascular disease including hypertension; increased intraocular pressure; hyperthyroidism; renal or hepatic impairment; seizures.
Polaramine may cause photosensitivity in some patients.
Use in pregnancy. (Category A)
Safety during pregnancy has not been established. Polaramine should be used during the first two trimesters of pregnancy only if clearly needed.
Dexchlorpheniramine maleate should not be used in the third trimester of pregnancy because newborn and premature infants may have severe reactions to antihistamines.
Polaramine has been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the foetus having been observed.
Use in lactation. Polaramine is excreted in breast milk. Therefore, caution should be exercised when administered to nursing mothers.
Paediatric use. Polaramine is contraindicated in children under 2 years of age due to the risk of respiratory depression.
Caution should be exercised when administering Polaramine to paediatric patients 2 years of age and older. It is recommended that the lowest effective dose of Polaramine be used in paediatric patients 2 years of age and older and concomitant administration of other drugs with respiratory depressant effects be avoided.
Children may experience paradoxical excitation with dexchlorpheniramine maleate. In children this may cause excitability.
Polaramine syrup contains sorbitol which may have a laxative effect or cause diarrhoea.
Use in the elderly. The elderly may experience paradoxical excitation with dexchlorpheniramine maleate. In patients over 60 years of age, antihistamines may cause dizziness, sedation and hypotension. Also, they are more likely to have central nervous system (CNS) depressive side effects, including confusion.
Effect on laboratory tests. Antihistamines should be discontinued approximately 48 hours prior to skin testing procedures since these drugs may prevent or diminish otherwise positive reactions to dermal reactivity indicators.
Interactions
The following interactions with Polaramine have been noted:
Central nervous system (CNS) depressants (alcohol, sedatives, opioid analgesics, hypnotics) may cause an increase in sedative effects of Polaramine.
Concomitant administration with tricyclic antidepressants (TCAs) may result in additive antimuscarinic activity.
Monoamine oxidase inhibitors (MAOIs) may prolong and intensify the anticholinergic and CNS depressive effects of some antihistamines and may cause a decrease in blood pressure.
Oral anticoagulants may have their actions decreased by antihistamines.
Adverse effects
Slight to moderate drowsiness is the most frequent side effect of dexchlorpheniramine maleate.
Other reported reactions associated with antihistamine therapy in general include:
General. Urticaria, drug rash, anaphylactic shock, photosensitivity, excessive perspiration, chills, dryness of mouth, nose and throat.
Cardiovascular. Hypotension, hypertension, headache, palpitations, tachycardia, extrasystoles.
Haematological. Haemolytic anaemia, hypoplastic anaemia, thrombocytopenia, agranulocytosis.
Gastrointestinal. Epigastric distress, anorexia, nausea, vomiting, diarrhoea, constipation.
Genitourinary. Urinary frequency, difficult urination, urinary hesitation and retention, early menses.
Nervous system. Sedation, dizziness, disturbed coordination, fatigue, confusion, restlessness, excitation, nervousness, tremor, irritability, insomnia, euphoria, paraesthesia, blurred vision, diplopia, vertigo, tinnitus, acute labyrinthitis, hysteria, neuritis, convulsions, lassitude, depression, inability to concentrate, dilated pupils, hypereflexia, hyporeflexia, xerostomia, hallucinations, appetite stimulation, anxiety, facial dyskinesias and seizures.
Respiratory. Thickening of bronchial secretions, tightness of chest, wheezing, nasal stuffiness.
Dosage and Administration
Polaramine tablets. Adults and children over 12 years. 1 tablet every 6 hours.
Polaramine syrup. Adults and children over 12 years. 5 mL every 6 hours.
Children 6 to 12 years. 2 to 4 mL every 6 to 8 hours.
Children 4 to 6 years. 1.75 to 2 mL every 6 to 8 hours.
Children 2 to 4 years. 1.25 to 1.75 mL every 6 to 8 hours.
Polaramine syrup is not to be used in children under 2 years of age.
Overdosage
In case of overdose, immediately contact the Poisons Information Centre (in Australia, call 13 11 26; in New Zealand, call 0800 764 766) for advice.
Manifestations. Antihistamine overdosage effects may vary from central nervous system depression (apnoea, arrhythmias, cardiovascular collapse, cyanosis, diminished mental alertness, sedation) to stimulation (convulsions, hallucinations, insomnia or tremors) to death. Other signs and symptoms may be ataxia, blurred vision, dizziness, hypotension and tinnitus. Stimulation is particularly likely in children, as are atropine-like signs and symptoms (dry mouth; fixed, dilated pupils; flushing; gastrointestinal symptoms and hyperthermia).
Treatment. Dialysis is of little value in antihistamine poisoning. Treatment of the signs and symptoms of an overdosage are symptomatic and supportive. Consider standard measures to remove any unabsorbed drug. There is no specific antidote. Measures to enhance excretion (urinary acidification, haemodialysis) are not recommended.
Presentation
Polaramine is available for oral use as either as: film coated immediate release tablet (Polaramine tablets) or as a syrup (Polaramine syrup).
Polaramine tablets. Off-white, round, bevelled tablet with score on the one side, blank on the other side.
Available in blister packs of 20 and 40 tablets.
Active: dexchlorpheniramine maleate 2 mg.
Inactive: lactose, starch-maize, starch-pregelatinised maize, magnesium stearate.
Polaramine syrup. A clear, red syrup with an orange like odour.
Available in a bottle of 100 mL.
Active: dexchlorpheniramine maleate 2 mg per 5 mL.
Inactive: sodium citrate, sodium chloride, sucrose, sorbitol solution (70 per cent), methyl hydroxybenzoate, propyl hydroxybenzoate, menthol, ethanol, water, brilliant scarlet 4R, apricot flavour, blood orange flavour.
Storage
Store below 25°C.
Poison schedule
Pharmacist-Only Medicine (S3).
Date of First Approval
01 August 2008
Date of Revision
01 November 2019
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.