Prednefrin Forte
Brand Information
| Brand name | Prednefrin Forte |
| Active ingredient | Prednisolone acetate + Phenylephrine hydrochloride |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Prednefrin Forte.
Summary CMI
PREDNEFRIN® FORTE
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using PREDNEFRIN® FORTE?
PREDNEFRIN® FORTE eye drops contain the active ingredients phenylephrine hydrochloride and prednisolone acetate. PREDNEFRIN® FORTE is used to treat severe inflammation (pain, swelling and redness) of the eye that is NOT caused by an infection and persistent allergy of the eye.
For more information, see Section 1. Why am I using PREDNEFRIN® FORTE? in the full CMI.
2. What should I know before I use PREDNEFRIN® FORTE?
Check the list of ingredients at the end of the CMI. Do not use PREDNEFRIN® FORTE if you have ever had an allergic reaction to any of them.
Talk to your doctor before you use this medicine if he/she is not aware that you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use PREDNEFRIN® FORTE? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with PREDNEFRIN® FORTE and affect how it works, or PREDNEFRIN® FORTE may interfere with other medicines and affect how they work.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use PREDNEFRIN® FORTE?
- Your doctor will tell you the number of drops to use, when to use PREDNEFRIN® FORTE and how long to use it.
More instructions can be found in Section 4. How do I use PREDNEFRIN® FORTE? in the full CMI.
5. What should I know while using PREDNEFRIN® FORTE
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using PREDNEFRIN® FORTE? in the full CMI.
6. Are there any side effects?
Increased pressure in the eye, eye irritation, dilation (widening) of the pupil, dry eye, eye discharge, eye infection, eye pain, foreign body sensation, itchy eye, watery eye(s), eye redness, blurry vision or changes in vision, headache, changes to taste, itchy, red skin rash, severe allergic reaction.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
PREDNEFRIN® FORTE (pred-nef-frin forte)
Active ingredients: [phenylephrine hydrochloride & prednisolone acetate] (fen-nil-ef-rin hi-dro-chlor-ride & pred-nis-so-lone as-see-tate)
Consumer Medicine Information (CMI)
This leaflet provides important information about using PREDNEFRIN® FORTE eye drops. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using PREDNEFRIN® FORTE.
Where to find information in this leaflet:
1. Why am I using PREDNEFRIN® FORTE?
2. What should I know before I use PREDNEFRIN® FORTE?
3. What if I am taking other medicines?
4. How do I use PREDNEFRIN® FORTE?
5. What should I know while using PREDNEFRIN® FORTE?
6. Are there any side effects?
7. Product details
1. Why am I using PREDNEFRIN® FORTE?
PREDNEFRIN® FORTE contains the active ingredients phenylephrine hydrochloride and prednisolone acetate.
PREDNEFRIN® FORTE is used to treat severe inflammation (pain, swelling and redness) of the eye that is NOT caused by an infection.
The first active ingredient in PREDNEFRIN® FORTE eye drops, prednisolone acetate, belongs to a chemical group called the glucocorticoids, which reduce pain, swelling and redness. The second active ingredient, phenylephrine hydrochloride, is responsible for reducing swollen blood vessels in the eye and lid.
2. What should I know before I use PREDNEFRIN® FORTE?
Warnings
Do not use PREDNEFRIN® FORTE if:
- you are allergic to phenylephrine hydrochloride, prednisolone acetate, or any of the ingredients listed at the end of this leaflet.
- you have any infection in or around the eye (bacterial, viral or fungal)
- you have an open sore on the front layer of the eyeball
- you have glaucoma (increased pressure in the eye).
Check with your doctor if you:
- have any condition which might cause thinning of the front layer of the eyeball
- have thickening of the walls of blood vessels (arteriosclerosis)
- have or have had high blood pressure
- have an overactive thyroid
- have an enlarged prostate
- have diabetes
- are susceptible to allergies
- have or have had adrenal suppression (body doesn't make enough hormones, which can make you feel tired and weak)
- have or have had asthma
- take any medicines for any other condition.
The use of steroids after cataract surgery may delay healing.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Make sure your doctor is aware that you are pregnant or plan to become pregnant or are breastfeeding or intend to breastfeed.
Use in children
Use of PREDNEFRIN® FORTE is not recommended in children or adolescents younger than 18 years of age.
3. What if I am taking other medicines?
Tell your doctor, pharmacist or optical practitioner if you are using any other medicines, including any medicines, vitamins, or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with PREDNEFRIN® FORTE and affect how it works; while PREDNEFRIN® FORTE may affect how other medicines work.
Do not use PREDNEFRIN® FORTE if:
- you are taking specific medicines to treat depression called "monoamine oxidase (MAO) inhibitors," tricyclic antidepressants, or you have taken them in the last 14 days.
There is a small chance that taking certain CYP3A inhibitor medicines, like some antibiotics, antifungal, and antiretroviral drugs, could increase your risk of having side effects.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are using and if these affect PREDNEFRIN® FORTE.
4. How do I use PREDNEFRIN® FORTE?
How much to use
- use one to two drops in the affected eye(s), according to your doctor's instructions.
When to use PREDNEFRIN® FORTE
- Use PREDNEFRIN® FORTE as instructed by your doctor.
- Make sure you understand how frequently you should use it and when to start using it and when to finish using it.
In some instances, PREDNEFRIN® FORTE needs to be used with other eye drops. Your doctor will let you know which drops, how to use them and how long to use them.
Wait at least 5 to 10 minutes after using PREDNEFRIN® FORTE before using other eye drops.
How to use PREDNEFRIN® FORTE
- If you wear contact lenses, remove them before using PREDNEFRIN® FORTE.
- Wash your hands well with soap and water.
- Shake the bottle well.
- Tear off the protective seal on any new bottle and throw the seal away. Remove the lid/cap.
- Hold the bottle upside down in one hand between your thumb and index finger.
- Using your other hand, gently pull down your lower eyelid to form a pouch or pocket.
- Tilt your head back and look up. This might be easier if you are sitting or lying down.
- Put the tip of the bottle close to your lower eyelid. Do not let the tip touch your eye or eyelid.
- Release one drop into the pouch or pocket formed between your eye and eyelid by gently squeezing the bottle.
- If a drop misses your eye, try again, and wipe off any excess that runs down your cheek.
- Close your eye. Do not blink or rub your eye or eyelid.
- While your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This will help to stop the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body.
- Replace the lid/cap, sealing it tightly.
- Wash your hands again with soap and water.
- If you wear contacts, wait 15 minutes before replacing your contact lenses.
If you forget to use PREDNEFRIN® FORTE
It is important that you use PREDNEFRIN® FORTE according to your doctor's instructions.
If you forget to use PREDNEFRIN® FORTE and it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
Otherwise, use the drops as soon as you remember, and then go back to using/taking them as you would normally.
Do not use double the number of drops to make up for the dose you missed.
If you use too much PREDNEFRIN® FORTE
Immediately rinse your eyes with warm water.
If PREDNEFRIN® FORTE is accidentally swallowed, you should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there appears to be no signs of discomfort or poisoning.
5. What should I know while using PREDNEFRIN® FORTE?
Things you should do
- Follow all of your doctor's instructions carefully.
- If you wear soft contact lenses, remove them before using PREDNEFRIN® FORTE, and wait at least 15 minutes before inserting them again. The preservative in PREDNEFRIN® FORTE eye drops (benzalkonium chloride) may be absorbed and cause discolouration of soft contact lenses.
- Keep all your follow-up appointments with your doctor so your eye(s) can be checked.
- Use all the medication prescribed to treat your eye condition.
- Keep the bottle tightly sealed to avoid contaminating the eye drops.
Call your doctor straight away if:
- you develop an eye infection, receive an eye injury, or have eye surgery
- you develop blurred vision or you notice any changes in your vision
- you become pregnant while using PREDNEFRIN® FORTE
- your eye(s) seem(s) to be getting worse or is/are not improving.
Remind any doctor, dentist, pharmacist, or optometrist you visit that you are using PREDNEFRIN® FORTE.
Things you should not do
- Do not touch the dropper tip against your eye, eyelid, or anything else, to avoid contaminating the eye drops. PREDNEFRIN® FORTE eye drops contain a corticosteroid. Corticosteroid eye drops should not be used for more than 10 days, except under strict supervision by your doctor.
- Do not stop using this medicine suddenly without checking with your doctor.
Driving or using machines
Wait for your vision to clear after using PREDNEFRIN® FORTE before you drive or use machines.
Looking after your medicine
Follow the instructions on the carton and bottle label on how to take care of your medicine properly.
Store PREDNEFRIN® FORTE upright.
Store it in a cool dry place away from moisture, heat or sunlight; for example:
- do not store it in the bathroom or near a sink, or
- do not store it in the car or on window sills.
Do not freeze the eye drops.
Keep it where young children cannot reach it.
When to discard your medicine
Discard the bottle 4 weeks after first opening it.
Getting rid of any unwanted medicine
If your doctor advises that you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not open a new bottle to use it if the expiry date has passed.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Effects on the eye:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Effects on the eye:
| Call your doctor straight away, if you notice any of these serious side effects. |
Effect on the body as a whole:
| Go straight to the Emergency Department at your nearest hospital if you notice any of these very serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop using any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What PREDNEFRIN® FORTE contains
| Active ingredients (main ingredients) |
|
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not use this medicine if you are allergic to any of these ingredients.
What PREDNEFRIN® FORTE looks like
PREDNEFRIN® FORTE is a milky suspension. It is supplied in a plastic dropper bottle containing 10 mL of liquid. (Aust R 23235).
Who distributes PREDNEFRIN® FORTE?
PREDNEFRIN® FORTE is distributed in Australia by:
AbbVie Pty Ltd
Mascot NSW 2020
Australia
Toll-free:1800 252 224
This leaflet was prepared in July 2025
Version 1
© 2025 AbbVie. All rights reserved.
PREDNEFRIN® FORTE and its design are trademarks of Allergan Inc., an AbbVie company.
Brand Information
| Brand name | Prednefrin Forte |
| Active ingredient | Prednisolone acetate + Phenylephrine hydrochloride |
| Schedule | S4 |
MIMS Revision Date: 01 October 2024
1 Name of Medicine
Phenylephrine hydrochloride and prednisolone acetate.
2 Qualitative and Quantitative Composition
Prednefrin Forte eye drops contains prednisolone acetate (microfine suspension) 10 mg (1%) and phenylephrine hydrochloride 1.2 mg (0.12%).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Prednefrin Forte eye drops is a sterile ophthalmic suspension.
4 Clinical Particulars
4.1 Therapeutic Indications
Severe inflammation (non-infectious) of the eye, such as acute iritis, iridocyclitis, scleritis, episcleritis, uveitis, resistant ocular allergy and inflammation following surgery (where no infectious aetiology is suspected), particularly where unusually rapid control of the inflammation is desired.
4.2 Dose and Method of Administration
1 to 2 drops instilled into the conjunctival sac two to four times daily. During the initial 24 to 48 hours the dosage may be safely increased to 2 drops every hour. The physician may choose the dosage which affords optimal therapeutic effect in each case.
In order to minimise systemic absorption of Prednefrin Forte eye drops, apply pressure to the tear duct immediately following administration of the drug.
Care should be taken not to discontinue therapy prematurely.
4.3 Contraindications
Prednefrin Forte eye drops is contraindicated in:
Acute untreated purulent infections, such as superficial (or epithelial) Herpes simplex keratitis (dendritic keratitis), vaccinia, varicella and most other viral diseases of the cornea and conjunctiva.
Ocular tuberculosis and fungal infections of the ocular structures of the eye.
Mycobacterial infection of the eye.
Narrow angle glaucoma.
Hypersensitivity to prednisolone acetate, phenylephrine hydrochloride, benzalkonium chloride or any of the other constituents.
4.4 Special Warnings and Precautions for Use
In diseases due to microorganisms, acute untreated infection may be masked, enhanced or activated by the steroid. Since Prednefrin Forte eye drops is not an anti-infective, if infection is present, appropriate measures must be taken to counteract the organisms involved. Prolonged use may also suppress the host immune response and thus increase the hazard of secondary ocular infections. As fungal infections of the cornea are particularly prone to develop with long-term local steroid application, fungal invasion must be suspected in any persistent corneal ulceration where a steroid has been used, or is in use. Fungal cultures should be taken when appropriate. Use of intraocular steroid medication in the presence of stromal Herpes simplex may prolong the course and may exacerbate the severity of many viral infections of the eye. Use of corticosteroid medication in the treatment of patients with a history of Herpes simplex requires caution and should be followed by frequent mandatory slit lamp microscopy (see Section 4.3 Contraindications). Prednefrin Forte eye drops contains benzalkonium chloride as a preservative.
Eye drops containing corticosteroids should not be used for more than 10 days except under strict ophthalmic supervision with regular checks for intraocular pressure. Extended use of topical corticosteroids may cause increased intraocular pressure in susceptible individuals resulting in glaucoma, with damage to the optic nerve, defects in visual acuity and fields of vision. Prolonged use may result in posterior subcapsular cataract formation.
In diseases causing thinning of the cornea, perforation has been known to have occurred with the use of topical steroids. Reports in the literature indicate that posterior subcapsular lenticular opacities have been reported to occur after heavy or protracted use of topical ophthalmic corticosteroids.
Prednefrin Forte eye drops contains sodium metabisulfite, a sulfite that may cause allergic-type reactions, including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. Sulfite sensitivity is seen more frequently in asthmatic patients.
The possibility of adrenal suppression should be considered with prolonged, frequent use of high dose topical steroids, particularly in infants and children.
The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation.
If patient experiences eye pain, changes in vision, ocular irritation or if eye irritation and redness persist over 72 hours, discontinue use and consult a doctor.
Potential systemic effects. Prednefrin Forte eye drops should be used with caution in patients with arteriosclerosis, hypertension, hyperthyroidism, prostatic enlargement, or diabetes.
Treatment should be used with caution in patients receiving monoamine oxidase (MAO) inhibitor therapy or within 14 days of stopping such treatment as patients may experience hypertensive crisis.
To minimise risk of potential systemic effects, the puncta should be depressed after instillation of drops to reduce drainage through the nasolacrimal duct to the oral and nasal mucosa.
Eye inflammation. Use Prednefrin Forte eye drops with caution on an inflamed eye, as hyperaemia greatly increases the rate of systemic absorption through the conjunctiva.
Use with contact lenses. Prednefrin Forte eye drops contain the preservative benzalkonium chloride, which may be absorbed by and cause discoloration of soft contact lenses. Patients wearing soft (hydrophilic) contact lenses should be instructed to remove contact lenses prior to administration of Prednefrin Forte eye drops and wait at least 15 minutes following administration before reinserting soft contact lenses.
Potential for eye injury or contamination. To prevent eye injury or contamination, care should be taken to avoid touching the bottle tip to the eye or to any other surface.
Visual disturbance. Visual disturbance may be reported with systemic and topical corticosteroid use. If a patient presents with symptoms such as blurred vision or other visual disturbances, consider evaluating for possible causes which may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR) which have been reported after use of systemic and topical corticosteroids.
Information for patients. Upon instillation, patients may experience transient blurred vision which may impair the ability to drive or use machinery. If affected, patients should not drive or use machinery until their vision has cleared.
Use in the elderly. No data available.
Paediatric use. Safety and effectiveness have not been demonstrated with Prednefrin Forte eye drops in paediatric patients. Prednefrin Forte eye drops is not recommended to be used in pediatric patients.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No interaction studies have been performed. Concurrent use of MAOI, tricyclic antidepressants, guanethidine or systemic adrenergic blockers may alter the effects of Prednefrin Forte eye drops.
Although the systemic exposure is expected to be low with topical ophthalmic corticosteroid administration, co-treatment with CYP3A inhibitors may increase the risk of systemic corticosteroid-related adverse effects.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category C)
In animal experiments, corticosteroids have been found to cause malformations of various kinds (cleft palate, skeletal malformations) and abortion. These findings do not seem to be relevant to humans. Reduced intrauterine growth and lower birth weight have been recorded in animals and humans after long-term or high dose treatment. Suppression of the adrenal cortex in the newborn baby may occur after long-term treatment. The short-term use of corticosteroids prior to delivery for the prevention of respiratory distress syndrome does not seem to pose a risk to the fetus or the newborn infant.
There are no adequate and well controlled studies in pregnant women. Prednefrin Forte should be used during pregnancy only if the potential benefit justifies the potential risk to the foetus.
Maternal pulmonary oedema has been reported with tocolysis and fluid overload.
Use in lactation. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in breast milk. Therefore, use is not recommended in women breast feeding infants.
4.7 Effects on Ability to Drive and Use Machines
Prednefrin Forte eye drops may cause pupillary dilation, transient blurring of vision, which may impair the ability to drive or operate machines. The patient should wait until their vision has cleared before driving or using machinery.
4.8 Adverse Effects (Undesirable Effects)
Immune system disorders. Hypersensitivity, urticaria.
Nervous system disorders. Headache.
Eye disorders. Adverse reactions include increased intraocular pressure, which may be associated with optic nerve damage and defects in the visual fields; posterior subcapsular cataract formation; eye penetration (sclera or corneal perforation), ocular infections from bacteria, fungi or viruses liberated from ocular tissues and perforation of the globe when used in conditions where there is thinning of the cornea or sclera, eye irritation, vision blurred/visual disturbances and mydriasis.
Gastrointestinal disorders. Dysgeusia.
Skin and subcutaneous tissue disorders. Pruritus and rash.
General disorders and administration site conditions. Systemic side effects may occur rarely with extensive use of topical steroids.
Post-marketing experiences. The following adverse reactions have been identified during post-marketing use of Prednefrin Forte eye drops. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made.
Eye disorders. Dry eye, eye discharge, cataract subcapsular, eye irritation, eye pain, eye penetration (scleral or corneal perforation), foreign body sensation, intraocular pressure increased, ocular infection (including bacterial, fungal, and viral infections), eye pruritus, lacrimation increased, mydriasis, ocular hyperemia, vision blurred, eyelid ptosis.
Immune system disorders. Hypersensitivity reaction including symptoms or signs of eye allergy and skin allergy.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Although overdose by the topical ophthalmic route will not ordinarily cause acute problems, it may cause systemic sympathomimetic effects (e.g. palpitation, headache and hypertension). In case of overdose, immediately flush eyes with water or normal saline. If accidentally ingested, drink fluids to dilute.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Prednisolone acetate is a glucocorticoid that, on the basis of weight, has 3 to 5 times the anti-inflammatory potency of hydrocortisone. Glucocorticoids inhibit the oedema, fibrin deposition, capillary dilation and phagocytic migration of the acute inflammatory response as well as capillary proliferation, deposition of collagen and scar formation. The phenylephrine component of Prednefrin Forte eye drops constricts the engorged vessels of the eye and lid.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Preservative. Benzalkonium chloride 0.04 mg (0.004%) per 1 mL.
Inactives. Hypromellose, phenazone, polysorbate 80, boric acid, sodium citrate, sodium metabisulfite, sodium chloride, disodium edetate and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
18 months.
Contents are sterile if seal is intact.
AUST R23235.
6.4 Special Precautions for Storage
Store below 25°C. Protect from freezing. Store upright.
Shake well before using.
6.5 Nature and Contents of Container
A sterile suspension in 10 mL dropper bottles.
To avoid contamination of the suspension, keep container tightly closed.
Do not touch dropper tip to any surface.
6.6 Special Precautions for Disposal
Discard unused contents 4 weeks after opening.
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
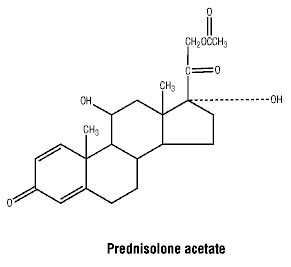
Chemical structure. Structure of prednisolone acetate.
Structure of phenylephrine hydrochloride.
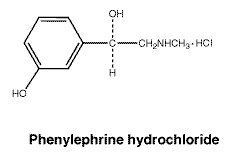
CAS registry number. Prednisolone acetate: 52-21-1.
Phenylephrine hydrochloride: 61-76-7.
Prednisolone acetate is an odourless, white or almost white, crystalline powder. Practically insoluble in water, soluble 1 in 120 of alcohol; slightly soluble in acetone and chloroform.
Molecular weight: 402.5.
Chemical name: 11β,17α,21-trihydroxypregna-1,4-diene-3,20-dione.
Empirical formula: C21H28O5.
Phenylephrine hydrochloride is white or almost white, odourless crystals or crystalline powder. Soluble 1 in 2 of water, and 1 in 4 of alcohol; practically insoluble in chloroform.
Molecular weight: 203.7.
Chemical name: (R)-3-hydroxy- α-[(methylamino)methyl]benzenemethanol hydrochloride.
Empirical formula: C9H14ClNO2.
7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Date of First Approval
14 October 1991
Date of Revision
29 August 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.