Primoteston Depot
Brand Information
| Brand name | Primoteston Depot |
| Active ingredient | Testosterone enantate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Primoteston Depot.
Summary CMI
PRIMOTESTON DEPOT
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using PRIMOTESTON DEPOT?
PRIMOTESTON DEPOT contains the active ingredient testosterone enantate. PRIMOTESTON DEPOT is used to replace the body's natural testosterone when not enough is made by the body.
For more information, see Section 1. Why am I using PRIMOTESTON DEPOT? in the full CMI.
2. What should I know before I use PRIMOTESTON DEPOT?
Do not use if you have ever had an allergic reaction to PRIMOTESTON DEPOT or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use PRIMOTESTON DEPOT? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with PRIMOTESTON DEPOT and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use PRIMOTESTON DEPOT?
- It is injected slowly into the muscle by your doctor.
- Your doctor will determine the time period between injections.
More instructions can be found in Section 4. How do I use PRIMOTESTON DEPOT? in the full CMI.
5. What should I know while using PRIMOTESTON DEPOT?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using PRIMOTESTON DEPOT? in the full CMI.
6. Are there any side effects?
Common side effects include pain, redness, swelling, heat and itching at the injection site, cough, shortness of breath, difficulty in breathing, rash, itching or hives, acne, hair loss or growth, changes in libido, breast development, fluid retention, weight gain and hostility/aggression. Serious side effects include severe stomach pain or tenderness, weakness, tiredness, headache, light-headedness, fainting, signs of allergy such as rash, swelling of the face, lips, mouth, throat, wheezing or trouble breathing, chest pain, yellowing of the skin and eyes and unwanted, frequent or prolonged and painful erections.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
PRIMOTESTON DEPOT (PREE mo TEST on)
Active ingredient(s): testosterone enantate
Consumer Medicine Information (CMI)
This leaflet provides important information about using PRIMOTESTON DEPOT. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using PRIMOTESTON DEPOT.
Where to find information in this leaflet:
1. Why am I using PRIMOTESTON DEPOT?
2. What should I know before I use PRIMOTESTON DEPOT?
3. What if I am taking other medicines?
4. How do I use PRIMOTESTON DEPOT?
5. What should I know while using PRIMOTESTON DEPOT?
6. Are there any side effects?
7. Product details
1. Why am I using PRIMOTESTON DEPOT?
PRIMOTESTON DEPOT contains the active ingredient testosterone enantate. PRIMOTESTON DEPOT provides testosterone which is a natural hormone known as an androgen, which controls normal sexual development and function in men.
PRIMOTESTON DEPOT is used to replace the body's natural hormone testosterone when not enough is made by the body.
2. What should I know before I use PRIMOTESTON DEPOT?
Warnings
Do not use PRIMOTESTON DEPOT if:
- you are allergic to testosterone enantate, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
- you have prostate cancer.
- you have breast cancer (males).
- you have high blood calcium levels associated with malignant tumours.
- you have previous or existing liver tumours.
Check with your doctor if you:
- have tendency to retain fluid (signs may include swollen feet or ankles).
- have high blood pressure or if you are being treated for high blood pressure as testosterone may cause a rise in blood pressure.
- have sleep apnoea (abnormal pauses in breathing during sleep).
- have a bleeding disorder.
- have thrombophilia (an abnormality of blood coagulation that increases the risk of thrombosis – blood clots in blood vessels).
- you are suffering from severe heart, liver or kidney disease (as testosterone may cause severe complications in the form of water retention in your body and sometimes accompanied by congestive heart failure.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
PRIMOTESTON DEPOT is intended for use in men only.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with PRIMOTESTON DEPOT and affect how it works. These include:
- barbiturates, medicines used to treat nervousness or sleeping problems.
- oxyphenbutazone, a medicine used to treat pain and inflammation.
- oral anticoagulants, ‘blood thinning’ medications to treat or prevent blood clots such as warfarin.
- medicines used to control blood sugar levels in diabetes, including oral antidiabetic drugs and insulin.
- ciclosporin, a medicine used to suppress the immune system.
- corticosteroids such as prednisolone or hydrocortisone, or adrenocorticotropic hormone (ACTH), used to control inflammation.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect PRIMOTESTON DEPOT.
4. How do I use PRIMOTESTON DEPOT?
How much to take / use
- PRIMOTESTON DEPOT is injected slowly into the muscle by your doctor.
- Experience shows that the short-lasting reactions (urge to cough, coughing fits, difficulty in breathing) which occur in very rare cases during or immediately after the injection of oily solutions can be avoided by injecting the solution slowly.
When to take / use PRIMOTESTON DEPOT
- Your doctor will determine how often to inject PRIMOTESTON DEPOT after measuring your testosterone levels.
- Initially, one prefilled syringe is injected into the muscle every 2-3 weeks by your doctor. The injection interval will depend on your testosterone levels.
- To maintain the effect of PRIMOTESTON DEPOT, 1 prefilled syringe is injected into the muscle every 3 – 4 weeks.
- PRIMOTESTON DEPOT helps control the symptoms of your condition but does not cure it.
If you forget to use PRIMOTESTON DEPOT
PRIMOTESTON DEPOT will be injected by your doctor at regular interval. Keep your regular appointments to see the doctor.
If you use too much PRIMOTESTON DEPOT
As PRIMOTESTON DEPOT is given to you under the supervision of your doctor, it is very unlikely that you will receive too much. However, if you experience any side effects after being given it, tell your doctor immediately.
If you think that you have used too much PRIMOTESTON DEPOT, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using PRIMOTESTON DEPOT?
Things you should do
Keep all of your doctor appointments.
Call your doctor straight away if you:
- experience prolonged stomach pain, blood in the stools or vomit, or yellowing of the skin and eyes during treatment with PRIMOTESTON DEPOT. There have been cases reported of patients developing liver tumours while using PRIMOTESTON DEPOT. Your doctor may tell you to stop using PRIMOTESTON DEPOT.
Remind any doctor, dentist or pharmacist you visit that you are using PRIMOTESTON DEPOT.
Your doctor may examine your prostate and conduct other tests (including blood tests) from time to time, particularly if you are elderly. This is to make sure the medicine is working and to check for unwanted side effects.
Things you should not do
- Do not abuse the use of testosterone, especially if you take too much of this medicine alone or with other anabolic androgenic steroids, as this can cause serious health problems to your heart and blood vessels (that can lead to death), mental health and/or the liver. Individuals who have abused testosterone may experience withdrawal symptoms when the dosage changes significantly or is stopped immediately. You should not abuse this medicine alone or with other anabolic androgenic steroids because it carries serious health risks. See “Side effects”.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how PRIMOTESTON DEPOT affects you.
Looking after your medicine
- Store below 25°C.
- Keep in the pack until it is time to use it.
Follow the instructions on the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
The doctor will discard the syringe after administration.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
These side effects may occur during or immediately after the injection.
Long term treatment or treatment with high doses of PRIMOTESTON DEPOT can affect sperm production.
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What PRIMOTESTON DEPOT contains
| Active ingredient (main ingredient) | Testosterone enantate |
| Other ingredients (inactive ingredients) | Benzyl benzoate, castor oil |
| Potential allergens | n/a |
Do not take this medicine if you are allergic to any of these ingredients.
What PRIMOTESTON DEPOT looks like
PRIMOTESTON DEPOT is a clear, yellowish oily solution and comes in a 1 mL prefilled syringe. Each pack contains 1 prefilled syringe (AUST R 10707).
Who distributes PRIMOTESON DEPOT
Bayer Australia Ltd
ABN 22 000 138 714
875 Pacific Highway
Pymble NSW 2073
This leaflet was prepared in December 2025.
Brand Information
| Brand name | Primoteston Depot |
| Active ingredient | Testosterone enantate |
| Schedule | S4 |
MIMS Revision Date: 01 March 2026
1 Name of Medicine
Testosterone enantate.
2 Qualitative and Quantitative Composition
1 mL Primoteston Depot contains 250 mg testosterone enantate (equivalent to approximately 180 mg testosterone).
Primoteston Depot contains: benzyl benzoate.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection.
Clear, yellowish oily solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Androgen replacement therapy for confirmed testosterone deficiency in males.
4.2 Dose and Method of Administration
Like all oily solutions, Primoteston Depot must be injected intramuscularly, and extremely slowly (see Section 4.4 Special Warnings and Precautions for Use).
For the development and stimulation of still underdeveloped androgen-dependent target organs and for the initial treatment of deficiency symptoms: 1 prefilled syringe i.m. every 2-3 weeks.
To maintain an adequate androgenic effect, 1 prefilled syringe i.m. every 3-4 weeks. Shorter injection intervals may be necessary depending on the individual requirement for hormone, but longer intervals of up to 6 weeks are also sufficient in many cases.
Serum testosterone levels should be measured before the start of treatment and periodically during treatment as recommended by current treatment guidelines.
The product should be inspected visually for particles prior to administration. Only clear solution free from particles should be used.
4.3 Contraindications
Prostatic carcinoma, mammary carcinoma in males.
Hypercalcaemia accompanying malignant tumours.
Previous or existing liver tumours.
Hypersensitivity to any of the ingredients.
4.4 Special Warnings and Precautions for Use
The general aim of androgen replacement therapy for confirmed testosterone deficiency in males is to keep serum testosterone levels within the reference range for the age group concerned. Over-replacement should be avoided.
Androgens are not indicated for enhancing muscular development in healthy individuals or for increasing physical ability.
Older patients treated with androgens may be at increased risk for the development of prostatic hyperplasia. Androgens can enhance the growth of an existing prostatic carcinoma. Therefore, carcinoma of the prostate has to be excluded before starting therapy with testosterone preparations.
As a precaution, regular examinations of the prostate are recommended. Haemoglobin and haematocrit should be checked periodically in patients on long-term androgen therapy to detect cases of polycythaemia (see Section 4.8 Adverse Effects (Undesirable Effects)).
In general, the risk of bleeding from using intramuscular injections in patients with acquired or inherited bleeding disorders always has to be taken into account. Testosterone and its derivatives have been reported to increase the activity of coumarin-derived oral anticoagulants (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Testosterone should be used with caution in patients with thrombophilia, as there have been post-marketing studies and reports of thrombotic events in these patients during testosterone therapy.
Cases of benign and malignant liver tumours, which may lead to life-threatening intra-abdominal haemorrhage, have been observed after the use of hormonal substances such as the one contained in Primoteston Depot. The doctor must therefore be informed of the occurrence of unusual upper abdominal complaints which do not disappear spontaneously within a short time as it may then be necessary to withdraw the preparation. A hepatic tumour should be considered in the differential diagnosis when severe upper abdominal pain, liver enlargement or signs of intra-abdominal haemorrhage occur in men using Primoteston Depot.
Caution should be exercised in patients predisposed to oedema. e.g. in case of severe cardiac, hepatic, or renal insufficiency or ischaemic heart disease, as treatment with androgens may result in increased retention of sodium and water. In case of severe complications characterised by oedema with or without congestive heart failure, treatment must be stopped immediately (see Section 4.8 Adverse Effects (Undesirable Effects)).
Testosterone may cause a rise in blood pressure and Primoteston Depot should be used with caution in men with hypertension.
Primoteston Depot must not be used in women, due to possible virilising effects.
Testosterone has been subject to abuse, typically at doses higher than recommended for the approved indication(s) and in combination with other anabolic androgenic steroids.
Testosterone abuse may result in dependence and withdrawal symptoms upon significant dose reduction or abrupt discontinuation of use.
Abuse of testosterone along with other anabolic androgenic steroids can lead to serious adverse reactions including: cardiovascular (with fatal outcomes in some cases), hepatic and/or psychiatric events.
Pre-existing sleep apnoea may be potentiated.
As with all oily solutions, Primoteston Depot must be injected intramuscularly and extremely slowly. Pulmonary microembolism of oily solutions can lead to signs and symptoms such as cough, dyspnoea and chest pain. There may be other signs and symptoms including vasovagal reactions such as malaise, hyperhidrosis, dizziness, paraesthesia, or syncope. These reactions may occur during or immediately after the injection and are reversible.
Treatment is usually supportive, e.g. by administration of oxygen.
Clotting disorders. Testosterone should be used with caution in patients with thrombophilia or risk factors for venous thromboembolism (VTE), as there have been post-marketing studies and reports of thrombotic events (e.g. deep-vein thrombosis, pulmonary embolism, ocular thrombosis) in these patients during testosterone therapy. In thrombophilic patients, VTE cases have been reported even under anticoagulation treatment, therefore continuing testosterone treatment after first thrombotic event should be carefully evaluated. In case of treatment continuation, further measures should be taken to minimise the individual VTE risk.
Use in hepatic impairment. No formal studies have been performed in patients with liver impairment. The use of Primoteston Depot is contraindicated in men with past or present liver tumours.
Use in renal impairment. See Section 4.4 Special Warnings and Precautions for Use.
Use in the elderly. Limited data do not suggest the need for a dosage adjustment in elderly patients.
Paediatric use. Primoteston Depot is not indicated for use in children and adolescents.
In addition to causing masculinisation in children, testosterone can cause accelerated growth, bone maturation and premature epiphyseal closure, thereby reducing adult height.
Effects on laboratory tests. See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Drugs that affect testosterone. Barbiturates and other enzyme inducers. Interactions can occur with drugs that induce microsomal enzymes which can result in increased clearance of testosterone.
Effects of androgens on other drugs. Oxyphenbutazone. Increased oxyphenbutazone serum levels have been reported.
Oral anticoagulants. Testosterone and its derivatives have been reported to increase the activity of coumarin-derived oral anticoagulants, possibly requiring dose adjustment. Independently of this finding, the risk of bleeding from using intramuscular injections in patients with acquired or inherited bleeding disorders always has to be taken into account as a general rule.
Hypoglycaemic agents. Androgens may enhance the blood sugar reducing effects of hypoglycaemic agents including oral antidiabetic drugs and insulin. Therefore, the dosage of the hypoglycaemic agent may need to be lowered.
Other interactions. Androgens may potentiate the effects of ciclosporin and increase risk of nephrotoxicity.
The concurrent administration of testosterone with ACTH or corticosteroids may enhance oedema formation; thus these active substances should be administered cautiously, particularly in patients with cardiac or hepatic disease or in patients predisposed to oedema.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. See Section 4.8 Adverse Effects (Undesirable Effects).
Use in pregnancy. Primoteston Depot is intended for use in men only. Primoteston Depot is not indicated for use in pregnant women.
Use in lactation. Primoteston Depot is intended for use in men only. Primoteston Depot is not indicated for use in breast feeding women.
4.7 Effects on Ability to Drive and Use Machines
Not known.
4.8 Adverse Effects (Undesirable Effects)
The most commonly reported adverse reactions with Primoteston Depot are injection site pain, injection site erythema, and cough and/or dyspnoea during or immediately after the injection.
Pulmonary microembolism of oily solutions can in rare cases lead to signs and symptoms such as cough, dyspnoea, malaise, hyperhidrosis, chest pain, dizziness, paraesthesia or syncope. These reactions may occur during or immediately after the injections and are reversible. Cases suspected by the company or the reporter representing oily pulmonary microembolism have been reported from postmarketing experience (see Section 4.4 Special Warnings and Precautions for Use).
High-dosed or long-term administration of testosterone, including Primoteston Depot, increases the tendency to water retention and oedema.
Spermatogenesis is reversibly interrupted or reduced by long-term and high-dosed treatment with Primoteston Depot.
If, in individual cases, frequent or persistent, painful erections (priapism) occur, the dose should be reduced or the treatment discontinued in order to avoid injury to the penis.
Various skin reactions including injection site reactions (injection site pain, injection site erythema, injection site induration, injection site swelling, injection site inflammation) may occur.
Other events reported with Primoteston Depot include benign and malignant liver tumours, polycythaemia, hypersensitivity reactions, libido increased, libido decreased, depression, insomnia, restlessness, aggression, irritability, headache, dizziness, hot flush, hypertension, cough, dyspnoea, dysphonia, diarrhoea, nausea, jaundice, acne, alopecia, rash, urticaria, pruritus, erythema, hyperhidrosis, arthralgia, pain in extremity, myalgia, various kinds of injection site reactions, fatigue, asthenia, prostatic specific antigen (PSA) increased, weight increased, liver function test abnormal, haematocrit increased, red cell blood count increased, haemoglobin increased, blood pressure increased, blood testosterone increased, gynaecomastia, testicular pain and post marketing reports of venous thromboembolism.
Hostility, nervousness and increased hair growth have been reported under treatment with testosterone-containing preparations.
Regarding adverse effects associated with the use of androgens, please also see Section 4.4 Special Warnings and Precautions for Use.
The following adverse effects have been reported with testosterone containing products. See Table 1.
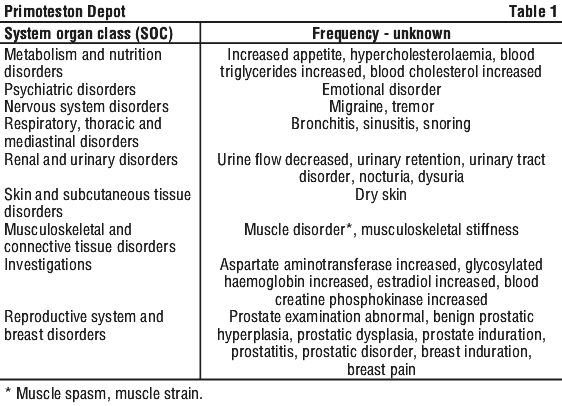
4.9 Overdose
No special therapeutic measure apart from termination of therapy with the drug or dose reduction is necessary after overdosage.
Acute toxicity data show that testosterone enantate, the ester contained in Primoteston Depot, can be classified as non-toxic following single intake. Even following single administration of a multiple of the dose required for therapy, no toxicity risk is to be expected.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Primoteston Depot contains a derivative of the natural male sex hormone testosterone as its active ingredient. The general aim of androgen replacement therapy for confirmed testosterone deficiency in males is to keep serum testosterone levels within the reference range for the age group concerned.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
The depot effect of testosterone enantate permits long intervals between injections. This ester not only has a long-lasting, but also a very intensive androgenic effect. The duration of action of 1 mL Primoteston Depot is approximately 2-4 weeks depending on the initial hormonal status.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Benzyl benzoate, castor oil.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
Please see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Primoteston Depot should be stored below 25°C. Keep out of reach of children. Protect from light.
6.5 Nature and Contents of Container
1 mL prefilled syringe containing 250 mg testosterone enantate.
Pack size of 1 x 1 mL.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
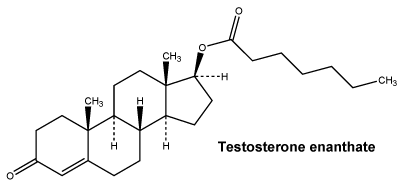
Testosterone enantate is designated chemically as 17 beta-heptanoyloxy-4-androstene-3-one.
The empirical formula of testosterone enantate is C26H40O3 and its molecular weight is 400.66 g/mol.
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine.
Date of First Approval
19 August 1991
Date of Revision
13 January 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.