Privigen
Brand Information
| Brand name | Privigen |
| Active ingredient | Immunoglobulin, normal (human) |
| Schedule | S4 |
Consumer medicine information (CMI) leaflet
Please read this leaflet carefully before you start using the Privigen
Summary CMI
Privigen®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Privigen®?
Privigen® contains the active ingredient human normal immunoglobulin. Privigen® is used for the replacement of antibodies because your antibody levels are low (referred to as immunodeficiency), or for a condition where there is an imbalance in your immune system requiring treatment with immunoglobulins (referred to as immunomodulation).
For more information, see Section 1. Why am I using Privigen®? in the full CMI.
2. What should I know before I use Privigen®?
Do not use if you have ever had an allergic reaction to Privigen® or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Privigen®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Privigen® and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Privigen®?
- It is usually administered by your doctor or healthcare professional.
- Your doctor will calculate the correct dose for you.
More instructions can be found in Section 4. How do I use Privigen®? in the full CMI.
5. What should I know while using Privigen®?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Privigen®? in the full CMI.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
Side effects tend to be related to the rate of infusion and are likely to occur during the first hour of infusion.
If a side effect occurs, the nurse or doctor may reduce the rate of infusion or the infusion may be stopped.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using Privigen®?
Privigen® contains the active ingredient human normal immunoglobulin.
Immunoglobulins are also known as antibodies and are blood proteins that help your body to fight infections.
Immunoglobulins are produced by your body's immune system to fight infections caused by bacteria and viruses.
Privigen® is used:
- for the replacement of antibodies because your antibody levels are low (referred to as immunodeficiency), or
- for a condition where there is an imbalance in your immune system requiring treatment with immunoglobulins (referred to as immunomodulation).
2. What should I know before I use Privigen®?
Warnings
Do not use Privigen®:
- if you have a history of allergy to human immunoglobulin products (allergic reactions may include skin rash, face swelling, wheezing or breathing difficulties), or previously been told you react to any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - if you have been told you have antibodies to immunoglobulin A (IgA)
Check with your doctor if you:
- are receiving Privigen® or this type of medicine (a human normal immunoglobulin) for the first time or after a long break in treatment (e.g. several months), or if you have been switched from another human normal immunoglobulin medicine
- suffer from hyperprolinaemia type I or II (a genetic disorder causing high levels of the amino acid proline in the blood)
- have diabetes
- have a history of heart, or blood vessel disease, or blood clots, have thick blood, have been immobile for some time. Also tell the doctor what medicine you are using as some medicines, such as those that contain the hormone estrogen (for example, birth control pills), may increase your risk of developing a blood clot.
- have kidney problems or kidney disease
- have high blood pressure
- have low blood volume (hypovolaemia)
- have a condition that causes low antibody levels in your blood (IgA deficiency, hypogammaglobulinaemia or agammaglobulinaemia with or without IgA deficiency)
- have recurrent temporary non-infectious meningitis with intravenous immunoglobulin treatments. If you have a recurrence of aseptic meningitis with intravenous immunoglobulin treatment, your doctor will ask you about the emergence or worsening of your symptoms which may progress to swelling of the brain (brain oedema).
- have frequent headaches or migraine
- have blood group A, B or AB
- are overweight
- are over 65 years of age
- take medicines that may harm your kidneys (nephrotoxic medicines)
- have any allergies to any medicines, foods, preservatives or dyes.
Vaccinations
Please inform your doctor if you are planning to have a vaccination. Privigen® may impair the effect of some virus vaccines such as measles, mumps, rubella and varicella for a period of at least 6 weeks, and up to 3 months. After receiving this medicine, a period of 3 months should be allowed before vaccination with some virus vaccines. In the case of measles vaccine, this effect may last for up to 1 year, so if you are going to receive a measles vaccine you should have your measles antibody status checked.
Tell your vaccinating doctor about your treatment with Privigen® before receiving any vaccination.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Virus safety
Privigen® is made from human plasma (the liquid component of blood). When medicines are made from human blood or plasma, processes are used to prevent infections being passed from the blood/plasma donor to the person receiving the medicine.
These processes include careful selection of the people who donate blood and plasma to make sure that those who might be carrying infections are excluded. In addition, each donation and pools of donations are tested for indicators of virus/virus infection(s).
Manufacturers of these medicines also include steps in the processing of blood or plasma that inactivate or remove viruses. Despite these processes, when medicines are prepared from human blood or plasma, the possibility of passing on an infection cannot be totally ruled out. Unknown or new viruses or other types of infection could also be passed on.
However, the measures taken in the manufacture of this medicine are considered effective for enveloped viruses such as human immunodeficiency virus (HIV), hepatitis B virus, and hepatitis C virus, and for the non-enveloped viruses hepatitis A (HAV) and B19 virus (B19V).
There is reassuring clinical experience regarding the lack of HAV or B19V infections with immunoglobulins. It is assumed that the antibodies which are in the immunoglobulin product make an important contribution to the viral safety.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Privigen® and affect how it works.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Privigen®.
4. How do I use Privigen®?
How much to use
- Privigen® is usually administered by your doctor or healthcare professional.
Privigen® is intended solely for infusion into a vein. Your doctor will calculate the correct dose for you. This will depend on your illness, your present condition and your weight. On the day of your infusion make sure you have had sufficient fluids which will help reduce the risk of side effects. At the beginning of the infusion you will receive Privigen® at a slow infusion rate. If you tolerate this well, your doctor can gradually increase the infusion rate.
When to use Privigen®
- Privigen® is given to you under the supervision of your doctor or trained medical professional.
- If you think you should receive Privigen® more or less frequently, please speak to your doctor.
Record of use
- It is recommended that every time you receive a dose of Privigen® the name and batch number of the product are recorded in order to maintain a record of the batches used.
If you forget to use Privigen®
If you think you have missed a dose, speak to your doctor as soon as possible.
If you use too much Privigen®
If you think you have had too much Privigen®, speak to your doctor as soon as possible.
As Privigen® is given to you under the supervision of your doctor or trained medical professional, it is very unlikely that you will receive an overdose. If you experience any side effects, tell your doctor immediately.
5. What should I know while using Privigen®?
Things you should do
If you are going to have surgery, tell the surgeon or anaesthetist that you have been given this medicine. It may affect other medicines used during your surgery.
Call your doctor straight away if you:
- become pregnant while receiving this medicine.
- are about to have any blood test. Privigen® may interfere with some blood tests. After infusion of this medicine you will have high antibody levels in your blood that may change the results of your blood tests.
Remind any doctor, dentist or pharmacist you visit that you are using Privigen®.
Things you should not do
- Do not stop using this medicine suddenly.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Privigen® affects you.
Patients may experience effects, such as dizziness or nausea, during treatment with Privigen® that might affect the ability to drive and use machines. If this happens, you should not drive or use machines until these effects have disappeared.
Looking after your medicine
You will be given this medicine in hospital. You will probably not need to keep any bottles of Privigen® at home. However, if you have to keep this medicine at home:
- Privigen® should be administered as soon as possible after opening the bottle as the solution contains no preservative.
- Store below 25°C. Do not freeze.
- Do not shake.
- Keep the bottle in the outer carton in order to protect from light.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Individuals may react differently to similar doses of the same product. This applies to Privigen®. Side effects tend to be related to the rate of infusion and are likely to occur during the first hour of infusion.
If a side effect occurs, the nurse or doctor may reduce the rate of infusion or the infusion may be stopped.
If you are over 65 years of age you may have an increased chance of getting side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
In rare cases this type of medicine may cause a sudden fall in blood pressure or a condition called anaphylactic shock, which is an allergic reaction that has symptoms such as low blood pressure (feeling faint) and difficulty breathing.
Severe headache, neck stiffness, drowsiness, fever, increased light sensitivity of the eye (photophobia), nausea, and vomiting may occasionally occur after receiving Privigen®.
Rarely, severe breathing problems, light-headedness, drops in blood pressure and fever may occur 1 to 6 hours after receiving this medicine.
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation.
This medicine may affect your blood cell count or liver enzyme levels.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Privigen® contains
| Active ingredient (main ingredient) | Human normal immunoglobulin |
| Other ingredients (inactive ingredients) | Proline Albumin Water |
Do not take this medicine if you are allergic to any of these ingredients.
What Privigen® looks like
Privigen® is a clear, colourless or pale yellow liquid.
5 g in a 50 mL solution: AUST R 143273
10 g in a 100 mL solution: AUST R 143337
20 g in a 200 mL solution: AUST R 143368
40 g in a 400 mL solution: AUST R 219160
Who distributes Privigen®
Privigen® is supplied in Australia by
CSL Behring (Australia) Pty Ltd
ABN 48 160 734 761
189–209 Camp Road
Broadmeadows VIC 3047
Australia
For Medical/Technical Enquiries
TOLL FREE: 1800 642 865
For Customer Service Enquiries
TOLL FREE: 1800 063 892
customerservice@cslbehring.com.au
www.cslbehring.com.au
This leaflet was prepared in March 2025.
® Registered trademark of CSL Limited Group of Companies
Brand Information
| Brand name | Privigen |
| Active ingredient | Immunoglobulin, normal (human) |
| Schedule | S4 |
MIMS Revision Date: 01 December 2024
1 Name of Medicine
Human Normal Immunoglobulin.
2 Qualitative and Quantitative Composition
Privigen is a sterile, clear or slightly opalescent, colourless or pale yellow solution of human normal immunoglobulin for intravenous infusion.
Privigen is a 10% solution containing 100 mg/mL of total human plasma protein with a purity of at least 98% immunoglobulin G (IgG). More than 90% of the IgG consists of monomers and dimers, aggregates (≤ 2% - typically below 0.1%) and albumin (≤ 3%). The distribution of the IgG subclasses is similar to that of normal human plasma (approximate values: 69% IgG1, 26% IgG2, 3% IgG3, 2% IgG4).
Privigen has a nominal osmolality of 320 mOsm/kg and is approximately isotonic. The pH value of the ready-to-use solution is 4.6-5.0. The product contains 250 mmol/L of L-proline as a stabiliser which is a physiological non-essential amino acid. Privigen contains no carbohydrate stabiliser (e.g. sucrose, maltose) and no preservative, and it has a low sodium content of ≤ 1 mmol/L.
The maximum IgA content is 0.025 mg/mL. Prekallikrein activator (PKA) levels are less than 10 IU/mL.
3 Pharmaceutical Form
See Section 2 Qualitative and Quantitative Composition.
4 Clinical Particulars
4.1 Therapeutic Indications
Replacement therapy. Primary immunodeficiency diseases (PID).
Myeloma or chronic lymphocytic leukaemia with severe secondary hypogammaglobulinaemia and recurrent infections.
Symptomatic hypogammaglobulinaemia secondary to underlying disease or treatment.
Immunomodulatory therapy. Idiopathic thrombocytopenic purpura (ITP) in patients at high risk of bleeding or prior to surgery to correct the platelet count.
Guillain-Barré syndrome (GBS).
Kawasaki disease.
Chronic inflammatory demyelinating polyneuropathy (CIDP).
Multifocal motor neuropathy (MMN).
Myasthenia gravis (MG) exacerbations.
Lambert-Eaton myasthenic syndrome (LEMS).
Stiff person syndrome (SPS).
4.2 Dose and Method of Administration
Dosage. The dose and dose regimen are dependent on the indication. The recommended dosage and dosage regimen are summarised in Table 1 and are given as a guideline.
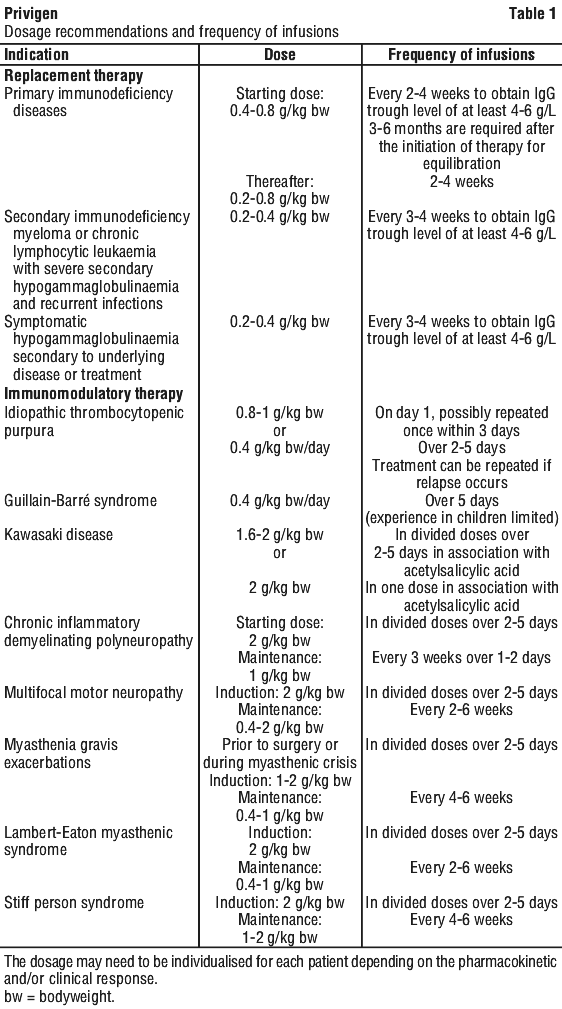
Privigen should be infused intravenously. Patients naive to human normal immunoglobulin, patients switched from an alternative IVIg product or patients who have not received IVIg for a long time should have vital signs and general status monitored regularly during and for the first hour after the first infusion. In such patients the initial infusion rate is 0.3 mL/kg bodyweight/hr. If well tolerated, the rate of administration may gradually be increased to 4.8 mL/kg bodyweight/hr. In a clinical study in PID patients, the maximum infusion rate was 7.2 mL/kg bodyweight/hr (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
In patients at risk for acute renal failure or thromboembolic adverse reactions, IVIg products should be administered at the minimum rate of infusion and dose practicable.
In case of an adverse reaction, the rate of administration must be reduced or the infusion stopped. The treatment required depends on the nature and severity of the side effect.
In case of shock, the current medical standards for shock treatment should be implemented.
Privigen is packaged as a ready to use solution in single use bottles. The product should be at room or body temperature before use. The solution should be clear or slightly opalescent. Do not use solutions that are cloudy or have particulate matter. Do not shake. Slight yellow colouration is of no concern and the product can still be used. Do not use if turbid.
Privigen should always be administered by intravenous (IV) infusion using appropriate administration equipment. Privigen is packaged in a glass bottle that must be vented during use.
Do not mix other medicinal products in the same infusion line. Privigen must not be mixed with physiological saline. The infusion line may, however, be primed or flushed with physiological saline (0.9% sodium chloride).
Always pierce the stopper at its centre, within the marked area. If desired, Privigen can be diluted with glucose 5% solution, using aseptic technique.
4.3 Contraindications
Hypersensitivity to the active substance or the excipient.
Hypersensitivity to human immunoglobulins, especially in patients with IgA deficiency where the patient has anti-IgA antibodies.
For patients with hyperprolinaemia type I and type II, see Section 4.4 Special Warnings and Precautions for Use.
4.4 Special Warnings and Precautions for Use
Privigen contains the excipient L-proline. Physicians should weigh the risk/benefit of Privigen in patients with hyperprolinaemia type I and type II on an individual basis.
Certain severe adverse reactions may be related to the rate of infusion. Reactions to IVIg tend to be related to the infusion rate and are most likely to occur during the first hour of the infusion. The recommended infusion rate given in Section 4.2 Dose and Method of Administration must be closely followed. Patients must be closely monitored and carefully observed for any symptoms throughout the infusion period (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)).
Certain adverse reactions may occur more frequently:
in case of high rate of infusion;
in patients with hypogammaglobulinaemia or agammaglobulinaemia, with or without IgA deficiency;
in patients who receive human immunoglobulin for the first time or, in rare cases, when the human normal immunoglobulin product is switched or when there has been a long interval since the previous infusion.
Potential complications can often be avoided by ensuring that patients:
are not sensitive to human normal immunoglobulin by initially infusing the product slowly (0.3 mL/kg bodyweight/hr);
are carefully monitored for any symptoms throughout the infusion period. In particular, patients naive to human normal immunoglobulin, patients switched from an alternative IVIg product or when there has been a long interval since the previous infusion, should be monitored during the first infusion and for the first hour after the first infusion, in order to detect potential adverse signs. All other patients should be observed for at least 20 minutes after administration.
In case of adverse reaction, either the rate of administration must be reduced or the infusion stopped (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)). The treatment required depends on the nature and severity of the adverse reaction. In case of shock, standard medical treatment for shock should be implemented.
In all patients, IVIg administration requires:
adequate hydration prior to the initiation of the infusion;
avoidance of concomitant use of loop diuretics.
Hypersensitivity. True hypersensitivity reactions are rare. They can occur in patients with anti-IgA antibodies. IVIg is not indicated in patients with selective IgA deficiency where the IgA deficiency is the only abnormality of concern.
Rarely, human normal immunoglobulin can induce a fall in blood pressure with anaphylactic reaction, even in patients who have tolerated previous treatment with human normal immunoglobulin.
Haemolytic anaemia. IVIg products can contain blood group antibodies (e.g. anti-A and anti-B) which may act as haemolysins and induce in vivo coating of red blood cells (RBC) with immunoglobulin, causing a positive direct antiglobulin reaction (Coombs' test) and, rarely, haemolysis. Haemolytic anaemia can develop subsequent to IVIg therapy due to enhanced RBC sequestration. The Privigen manufacturing process includes an immunoaffinity chromatography (IAC) step that specifically reduces blood group A and B antibodies (isoagglutinins A and B). Clinical data with Privigen manufactured with the IAC step show statistically significant reductions of haemolytic anaemia (see Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties, Clinical trials).
Isolated cases of haemolysis related renal dysfunction/renal failure or disseminated intravascular coagulation and death have occurred.
The following risk factors are associated with the development of haemolysis: high doses, whether given as a single administration or divided over several days; blood group A, B and AB (non-O blood group), and underlying inflammatory state. As this event was commonly reported in blood group A, B or AB (non-O blood group) patients receiving high doses for non-PID indications, increased vigilance is recommended.
Haemolysis has rarely been reported in patients given replacement therapy for PID.
IVIg recipients should be monitored for clinical signs and symptoms of haemolysis. If signs and/or symptoms of haemolysis develop during or after an IVIg infusion, discontinuation of the IVIg treatment should be considered by the treating physician (also see Section 4.8 Adverse Effects (Undesirable Effects)).
Aseptic meningitis syndrome (AMS). Aseptic meningitis syndrome has been reported to occur in association with IVIg treatment (see Section 4.8 Adverse Effects (Undesirable Effects).
Discontinuation of IVIg treatment has resulted in remission of AMS within several days without sequelae. The syndrome usually begins within several hours to 2 days following IVIg treatment. Symptoms may include severe headache, nuchal rigidity, drowsiness, fever, photophobia, nausea, and vomiting. Patients exhibiting such symptoms should receive a thorough neurological examination, including cerebrospinal fluid studies, to rule out other causes of meningitis. Cerebrospinal fluid studies are frequently positive with pleocytosis up to several thousand cells per mm3, predominantly from the granulocytic series, and elevated protein levels up to several g/L, but negative culture results.
AMS may occur more frequently in association with high dose (≥ 2 g/kg) IVIg treatment and/or rapid infusion (see Section 4.2 Dose and Method of Administration; Section 4.4 Special Warnings and Precautions for Use).
Patients with recurrence of AMS in association with IVIg treatment should be monitored for the emergence or worsening of symptoms potentially progressing to brain oedema (cerebral oedema).
Thromboembolism. There is clinical evidence of an association between IVIg administration and thromboembolic events such as myocardial infarction, cerebral vascular accident (including stroke), pulmonary embolism and deep vein thromboses, which is assumed to be related to a relative increase in blood viscosity through the high influx of immunoglobulin in at risk patients. Caution should be exercised in prescribing and infusing IVIg in obese patients and in patients with pre-existing risk factors for thrombotic events (such as advanced age, hypertension, diabetes mellitus and a history of vascular disease or thrombotic episodes, patients with acquired or inherited thrombophillic disorders, patients with prolonged periods of immobilisation, severely hypovolaemic patients, patients with diseases which increase blood viscosity).
In patients at risk for thromboembolic adverse reactions, IVIg products should be administered at the minimum rate of infusion and dose practicable based on clinical judgement.
Acute renal failure. Cases of acute renal failure have been reported in patients receiving IVIg therapy. In most cases, risk factors have been identified, such as pre-existing renal impairment, diabetes mellitus, hypovolaemia, overweight, concomitant nephrotoxic medicinal products or age over 65 years.
In cases of renal impairment, IVIg discontinuation should be considered. While these reports of renal dysfunction and acute renal failure have been associated with the use of many of the licensed IVIg products containing various excipients such as sucrose, glucose and maltose, those containing sucrose as a stabiliser accounted for a disproportionate share of the total number. In patients at risk, the use of IVIg products that do not contain sucrose should therefore be considered. Privigen does not contain sucrose, maltose or glucose.
In patients at risk of acute renal failure, IVIg products should be administered at the minimum rate of infusion and dose practicable based on clinical judgement.
For patients suffering from diabetes mellitus and requiring dilution of Privigen to lower concentrations, the presence of glucose in the recommended diluent should be taken into account.
Transfusion related acute lung injury (TRALI). Noncardiogenic pulmonary oedema may very rarely occur following treatment with IVIg products, including Privigen. TRALI is characterised by severe respiratory distress, pulmonary oedema, hypoxaemia, normal left ventricular function, and fever. Symptoms typically appear within 1 to 6 hours following treatment.
Monitor patients for pulmonary adverse reactions. TRALI may be managed using oxygen therapy with adequate ventilatory support.
Pathogen safety. Privigen manufacture includes standard measures to prevent infections resulting from the use of medicinal products prepared from human blood or plasma. These include: donor selection, screening of individual donations and plasma pools for specific markers of infection and the inclusion of effective manufacturing steps for the inactivation/removal of viruses. These multiple, complementary manufacturing processes include two dedicated steps to reduce the possibility of pathogen transmission: virus filtration and incubation at pH 4.
Despite this, when medicinal products prepared from human blood or plasma are administered, the possibility of transmitting infective agents cannot be totally excluded. This also applies to unknown or emerging viruses and other pathogens.
The measures taken are considered effective for enveloped viruses such as human immunodeficiency virus (HIV), hepatitis B (HBV), and hepatitis C (HCV), and for the nonenveloped viruses such as hepatitis A (HAV) and parvovirus B19.
There is reassuring clinical experience regarding the lack of hepatitis A or parvovirus B19 transmission with immunoglobulins and it is also assumed that the antibody content makes an important contribution to the viral safety.
Vaccination for patients in receipt of medicinal products from human plasma should be considered where appropriate.
Use in the elderly. Clinical studies of Privigen did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently than younger patients.
Paediatric use. Although limited data is available, it is expected that the same warnings, precautions and risk factors apply to the paediatric population.
Treatment of PID. Privigen was evaluated in 19 children and 12 adolescents with PID. There were no apparent differences in the safety and efficacy profiles when compared to these profiles in adult patients. No paediatric specific dose was necessary to achieve the desired serum IgG levels. The use of Privigen has not been established in paediatric patients with PID under the age of three years.
Treatment of ITP. The use of Privigen has not been established in patients with ITP under the age of 15 years.
Treatment of CIDP, MMN, MG, LEMS and SPS. The use of Privigen has not been established in patients with neurological indications under the age of 18 years.
For further information regarding the safety profile for use in paediatric patients with CIDP, see Section 5.1 Pharmacodynamic Properties, Clinical trials, Post-authorisation safety study (PASS) in patients with various immunological conditions.
Effects on laboratory tests. Interference with serological testing. After infusion of immunoglobulin the transitory rise of the various passively transferred antibodies in the patient's blood may result in misleading positive serological tests.
Passive transmission of antibodies to erythrocyte antigens, e.g. A, B, D may interfere with some serological tests for red cell allo-antibodies (e.g. direct agglutination test (DAT), direct Coombs' test).
4.5 Interactions with Other Medicines and Other Forms of Interactions
Live attenuated virus vaccines. Immunoglobulin administration may impair the efficacy of live attenuated virus vaccines such as measles, mumps, rubella and varicella for a period of at least six weeks and up to three months. After administration of Privigen, an interval of three months should elapse before vaccination with live attenuated virus vaccines. In the case of measles, this impairment may persist for up to one year. Therefore, patients receiving measles vaccine should have their antibody status checked.
Medicine interactions. The interaction of Privigen with other medicines has not been established.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No fertility studies have been conducted in animals using Privigen or the excipient present in the formulation (L-proline). Clinical experience with immunoglobulins suggests that no harmful effects on fertility are to be expected.
Use in pregnancy. The safety of this medicinal product for use in human pregnancy has not been established in controlled clinical trials and therefore should only be given with caution to pregnant women and breastfeeding mothers. IVIg products have been shown to cross the placenta, increasingly during the third trimester. Clinical experience with immunoglobulins suggests that no harmful effects on the course of pregnancy, or on the foetus and the neonate are to be expected.
Use in lactation. Immunoglobulins are excreted into breast milk and may contribute to protecting the neonate from pathogens which have a mucosal portal of entry.
4.7 Effects on Ability to Drive and Use Machines
The ability to drive and operate machines may be impaired by some adverse reactions associated with Privigen. Patients who experience adverse reactions during treatment should wait for these to resolve before driving or operating machines.
4.8 Adverse Effects (Undesirable Effects)
Intravenously administered human normal immunoglobulins have a well established history of safety and efficacy in humans.
Adverse reactions such as chills, headache, dizziness, fever, vomiting, allergic reactions, nausea, arthralgia, low blood pressure and moderate back pain may occur occasionally in connection with intravenous administration of human immunoglobulin.
Rarely human immunoglobulin may cause a sudden fall in blood pressure and, in isolated cases, anaphylactic shock, even when the patient has shown no hypersensitivity to previous administration (see Section 4.4 Special Warnings and Precautions for Use, Hypersensitivity).
Cases of reversible aseptic meningitis and rare cases of transient cutaneous reactions have been observed with human normal immunoglobulin (see Section 4.4 Special Warnings and Precautions for Use, Aseptic meningitis syndrome).
Reversible haemolytic reactions have been observed in patients, especially those with blood groups A, B, and AB (non-O blood groups) in immunomodulatory treatment.
Haemolysis has been reported rarely in patients given replacement therapy for PID. Rarely, haemolytic anaemia requiring transfusion may develop after high dose IVIg treatment (see Section 4.4 Special Warnings and Precautions for Use, Haemolytic anaemia).
Increase in serum creatinine level and/or acute renal failure have been observed (see Section 4.4 Special Warnings and Precautions for Use, Acute renal failure).
Very rarely: transfusion related acute lung injury and thromboembolic reactions such as myocardial infarction, stroke, pulmonary embolism and deep vein thromboses events have been reported (see Section 4.4 Special Warnings and Precautions for Use).
Tabulated list of adverse reactions. Seven clinical studies were performed with Privigen, which included patients with PID, ITP and CIDP. In the pivotal PID study, 80 patients were enrolled and treated with Privigen. Of these, 72 completed the 12 months of treatment. In the PID extension study, 55 patients were enrolled and treated with Privigen. Another study in 11 PID patients was conducted in Japan. Two ITP studies were performed with 57 patients each. Two CIDP studies were performed with 28 and 207 patients, respectively.
Most adverse drug reactions (ADRs) observed in the 7 clinical studies were mild to moderate in nature.
Table 2 shows an overview of the ADRs observed in the 7 clinical studies categorised according to MedDRA System organ class, preferred term level and frequency.
Within each frequency grouping, ADRs are presented in order of decreasing frequency.
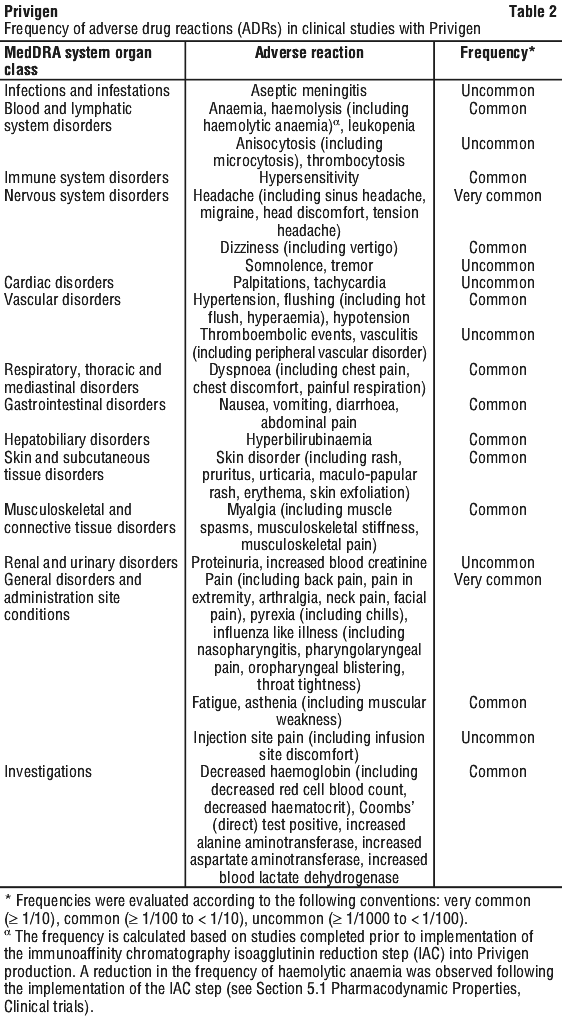
Blood and lymphatic system disorders. Decreased neutrophil count.
Immune system disorders. Anaphylactic shock.
Nervous system disorders. Brain oedema (cerebral oedema).
Vascular disorders. Transfusion-related acute lung injury.
Skin and subcutaneous tissue disorders. Exfoliative dermatitis.
Renal and urinary disorders. Acute renal failure.
Paediatric population. In Privigen clinical studies with paediatric patients, the frequency, nature and severity of adverse reactions did not differ between children and adults.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdose may lead to fluid overload and hyperviscosity, particularly in patients at risk, including elderly patients or patients with cardiac, or renal impairment.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
The manufacturing process for Privigen includes the following steps: ethanol precipitation of the IgG plasma fraction, followed by octanoic acid fractionation and incubation at pH 4. Subsequent purification steps comprise depth filtration, anion exchange chromatography, immunoaffinity chromatography to specifically reduce blood group A and B antibodies (isoagglutinins A and B, see Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties, Clinical trials) and a virus filtration step that can remove particles to a size of 20 nanometre.
Mechanism of action. Privigen contains mainly IgG that are present in the normal human population and that show a broad spectrum of functionally intact antibodies against infectious agents. Privigen is prepared from plasma from 1000 or more human donors. The IgG subclass distribution in Privigen corresponds approximately to that of native human plasma. The Fc and Fab functions of the IgG molecule are retained.
The formulation of Privigen minimises the formation of IgG dimers. The minimisation of IgG dimers is important for the tolerability of the product.
In replacement therapy adequate doses of Privigen may restore abnormally low IgG levels to within the normal range.
The mechanism of action in indications other than replacement therapy is not fully elucidated, but includes immunomodulatory effects.
Clinical trials. The safety and efficacy of Privigen was evaluated in 5 prospective, open-label, single-arm, multicentre studies carried out: in Europe (ITP, PID and CIDP studies), Japan (PID and CIDP studies) and in the US (PID and CIDP studies).
Additional safety data were collected in a Post-Authorisation Safety Study (PASS), an observational retrospective cohort study in patients with various immunological conditions performed in the US.
Treatment of primary immunodeficiency diseases (PID). In the PID study, a total of 80 patients between 3 and 69 years of age were treated with a median dose of 200 to 888 mg/kg bodyweight (bw) for a maximum of 12 months. The administration of Privigen every 3 or 4 weeks resulted in stable serum IgG trough levels throughout the treatment period with mean IgG trough levels ranging from 8.84 g/L to 10.27 g/L.
The primary endpoint was the annual rate of acute serious bacterial infections (SBI), defined as pneumonia, bacteraemia/septicaemia, osteomyelitis/septic arthritis, bacterial meningitis, and visceral abscess, per subject, per year. The observed annual rate of acute SBI was 0.08 infections per subject per year (upper 1-sided 97% CI 0.182), which met the predefined success rate of less than one acute SBI per subject, per year.
The secondary endpoints in the PID study included: rate of any infection (3.55 per subject year), days out of work/school/daycare/unable to perform normal activities due to illness (7.94 days, per subject year), days of hospitalisation (2.31 days, per subject year), and use of antibiotics (87.4 days, per subject year).
An open-label, single-arm phase 3 study conducted in Japan assessed the PK and safety of Privigen in 11 subjects with PID.
Treatment of idiopathic thrombocytopenic purpura (ITP). A total of 57 patients aged 15 to 69 years with chronic ITP and a platelet count of ≤ 20 x 109/L were treated with 1 g/kg bw of Privigen on each of the two consecutive days. A rise in platelet count to at least 50 x 109/L within 7 days after the first infusion was observed in 46 of the 57 patients studied. The median time to achieve this platelet response was 2.5 days after the first infusion (primary endpoint). After day one (i.e. on day two prior to the second dosing) 43% of the patients reached this response. For those patients who responded, the median duration of platelet count ≥ 50 x 109/L was 15.4 days (range: 1 to > 82 days).
Treatment of neurological disorders. Chronic inflammatory demyelinating polyneuropathy (CIDP). In the first CIDP study, a prospective multicentre open label trial (PRIMA Privigen Impact on Mobility and Autonomy study), 28 patients with CIDP (13 patients were and 15 patients were not on IVIg treatment at study screening) received a loading dose of 2 g/kg bw given over 2-5 days followed by up to 7 maintenance doses of 1 g/kg bw over 1-2 days every 3 weeks. Patients on IVIg treatment at study screening were withdrawn from IVIg treatment until confirmed deterioration had occurred before the start of Privigen treatment. On the adjusted 10 point INCAT (Inflammatory Neuropathy Cause and Treatment) scale a clinically meaningful improvement of at least 1 point from baseline to treatment week 25 was observed in 17 out of 28 patients. The INCAT responder rate, therefore, was 60.7% (95% confidence interval [42.41, 76.43]). 53% (9/17) of the responding patients responded after receiving the initial induction dose at week 4 and 94% (16/17) of patients responded by week 10.
In the second CIDP clinical study, a prospective, multicentre, randomised, placebo-controlled study (PATH Polyneuropathy and Treatment with Hizentra), 207 subjects with CIDP received Privigen in the pre-randomisation phase of the study. All subjects had previously received IVIg treatment for at least 8 weeks prior to entering the study. IVIg-dependence was confirmed by clinically evident deterioration during an IVIg withdrawal phase of up to 12 weeks. In subjects who were IVIg-dependent 207 received a Privigen loading dose of 2 g/kg bw, followed by up to 4 Privigen maintenance doses of 1 g/kg bw every 3 weeks for up to 13 weeks.
Following clinical deterioration during IVIg withdrawal, clinical improvement of CIDP was primarily defined by a decrease of ≥ 1 point in the adjusted INCAT score. Additional measures of CIDP improvement were a Rasch-built Overall Disability Scale (R-ODS) increase of ≥ 4 points, a mean grip strength increase of ≥ 8 kPa, or a Medical Research Council (MRC) sum score increase of ≥ 3 points. Overall, 91% of subjects (188 patients) showed improvement in at least one of the criteria above by week 13.
By adjusted INCAT score, the responder rate by week 13 was 72.9% (151/207 patients), with 149 patients responding already by week 10. A total of 43 of the 207 patients achieved a better CIDP status as assessed by adjusted INCAT score compared to their CIDP status at study entry.
The comparability of the response rates by adjusted INCAT score between the PRIMA and PATH studies are shown in Figure 1.
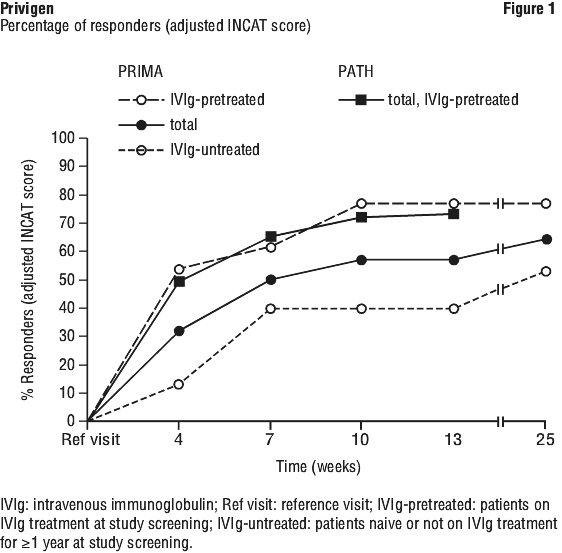
In PRIMA, the percentage of responders based on MRC score (defined as an increase by ≥ 3 points) was 85% (87% in the IVIg-untreated and 82% in IVIg-pretreated) and 57% in PATH. The overall median time to first MRC sum score response in PRIMA was 6 weeks (6 weeks in the IVIg-untreated and 3 weeks in the IVIg-pretreated) and 9.3 weeks in PATH. MRC sum score in PRIMA improved by 6.9 points (7.7 points for IVIg-untreated and 6.1 points for IVIg-pretreated) and by 3.6 points in PATH. The grip strength of the dominant hand improved by 14.1 kPa (17.0 kPa in IVIg-untreated and 10.8 kPa in IVIg-pretreated subjects) in the PRIMA study, while in PATH the grip strength of the dominant hand improved by 12.2 kPa. For the non dominant hand similar results were observed in both studies, PRIMA and PATH.
Overall, the efficacy and safety profiles of Privigen in the PRIMA and the PATH studies in CIDP patients were comparable.
Privigen has comparable characteristics to other IVIg products that have been used in the management of Chronic Inflammatory Demyelinating Polyneuropathy (CIDP), Multifocal Motor Neuropathy (MMN), Myasthenia Gravis (MG), Lambert Eaton Myasthenic Syndrome (LEMS) and Stiff Person Syndrome (SPS). In addition, the adverse reactions reported in the literature for IVIg when used in these indications were comparable with those reported for other indications. It is therefore expected that Privigen will have a comparable efficacy and safety profile to other IVIg products in the management of these indications.
There are several randomised controlled clinical trials in the literature demonstrating the efficacy and safety of the use of IVIg in the treatment of patients with CIDP, MMN and MG. Whilst the evidence for the efficacy of IVIg in the management of CIDP and acute exacerbations of MG is clear, data for the treatment of chronic MG and MMN is not as definitive. Clinical trials for the use of IVIg for MMN showed an increase in muscle strength but no impact on the disability scale.
The efficacy and safety of IVIg in the treatment of patients with stiff person syndrome and LEMS has only been demonstrated in a single randomised controlled clinical trial for each condition.
Post-authorisation safety study (PASS) in patients with various immunological conditions. In an observational hospital-based retrospective cohort PASS, the risk of haemolytic anaemia following Privigen therapy was evaluated in patients with various immunological conditions across 2 time periods. The risk of haemolytic anaemia was assessed prior (baseline) and after the implementation of a risk minimisation measure, the introduction of the IAC step in the Privigen manufacturing process. The IAC step was shown to produce a 2-titre step reduction of isoagglutinins, from a median anti-A of 32 to 8 and from a median anti-B of 16 to 4.
Of the 9439 patients who received Privigen prior to the introduction of the IAC step, 47 cases of haemolytic anaemia were identified. Following the introduction of IAC, 7759 patients received Privigen, with 4 cases of haemolytic anaemia. A statistically significant reduction of 89% in the rate of haemolytic anaemia (based on an incidence rate ratio of 0.11; adjusted for in-/outpatient setting, age, sex, Privigen dose and indication for Privigen use; one-sided p-value < 0.01) was observed after implementation of the IAC step compared to baseline.
The reduction in probable haemolytic anaemia incidence rate after IAC implementation versus baseline was especially pronounced in patients treated with Privigen doses ≥ 0.75 g/kg bw. These calculations are estimated from the demographic characteristics.
Additionally, 28 paediatric patients with CIDP < 18 years of age were identified throughout the entire study period from 1 January 2008 to 30 April 2019. None of the paediatric patients given a total of 486 Privigen administrations experienced haemolytic anaemia, AMS, acute renal failure, severe anaphylactic reaction or a thromboembolic event. Two out of 28 patients experienced a moderate anaphylactic reaction equating to 7.1% of the patients, and 0.4% of all 486 Privigen administrations.
5.2 Pharmacokinetic Properties
Human normal immunoglobulin is immediately and completely bioavailable in the recipient's circulation after intravenous administration. It is distributed relatively rapidly between plasma and extravascular fluid. After approximately 3-5 days, equilibrium is reached between the intra- and extravascular compartments.
The pharmacokinetic parameters for Privigen were determined in a clinical study in Primary Immunodeficiency Diseases (PID) patients (see Section 5.1 Pharmacodynamic Properties, Clinical trials). Twenty five patients (aged 13 to 69 years) participated in the pharmacokinetic assessment (see Table 3). The median half life of Privigen in PID patients was 36.6 days.
This half life may vary from patient to patient, particularly in patients with primary immunodeficiencies.

5.3 Preclinical Safety Data
Genotoxicity. No genotoxicity studies have been conducted with Privigen. The excipient L-proline was not genotoxic in a standard array of genotoxicity tests.
Carcinogenicity. No carcinogenicity studies have been conducted with Privigen.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
See Section 4.2 Dose and Method of Administration.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C (Do not freeze).
Do not shake.
Keep the bottle in the outer carton in order to protect from light.
Do not use after the expiry date printed on the carton and the label.
6.5 Nature and Contents of Container
Privigen is presented as a 10% (100 g/L) solution for intravenous infusion. The solution is dispensed into a clear glass bottle and closed with a latex free rubber stopper and aluminium crimp cap, with a plastic flip off disc providing a tamper evident seal.
The product is supplied in the following pack sizes:
One bottle of 50 mL solution containing 5 g human normal immunoglobulin.
One bottle of 100 mL solution containing 10 g human normal immunoglobulin.
One bottle of 200 mL solution containing 20 g human normal immunoglobulin.
One bottle of 400 mL solution containing 40 g human normal immunoglobulin.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. No data available.
CAS number. 9007-83-4.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
12 March 2009
Date of Revision
16 October 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. NPS MedicineWise disclaims all liability (including for negligence) for any loss, damage or injury resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.