Proladone
Brand Information
| Brand name | Proladone |
| Active ingredient | Oxycodone |
| Schedule | S8 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Proladone.
Summary CMI
PROLADONE
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
WARNING: Important safety information is provided in a boxed warning in the full CMI. Read before using this medicine.
1. Why am I using Proladone?
Proladone contains the active ingredient oxycodone. Proladone is a suppository containing oxycodone. It belongs to a group of medicines called opioid analgesics and acts to relieve pain. Proladone is used to relieve pain after surgery or for treating cancer pain. For more information, see Section 1. Why am I using Proladone? in the full CMI.
2. What should I know before I use Proladone?
Do not use if you have ever had an allergic reaction to Proladone or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. For more information, see Section 2. What should I know before I use Proladone? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Proladone and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Proladone?
Your doctor will tell you how many suppositories you need to use each day. If possible, go to the toilet and empty your bowels before using your suppository. Suppositories work best if your bowels are empty. Follow the steps under Section 4. How do I use Proladone? in the full CMI.
5. What should I know while using Proladone?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines | Be careful before you drive or use any machines or tools until you know how Proladone affects you. Proladone may cause drowsiness, sleepiness or dizziness in some people and affect alertness. |
| Drinking alcohol | Be careful when drinking alcohol while you are using Proladone. Drinking alcohol while using this medicine could make you feel more dizzy, sleepy or light-headed than usual. Your doctor may suggest you avoid alcohol while you are using Proladone. |
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Proladone? in the full CMI.
6. Are there any side effects?
Speak to your doctor if you have any of these common side effects and they worry you: confusion, drowsiness, constipation, vomiting and nausea. If any of the following happen, stop using Proladone and tell your doctor immediately or go to the Emergency Department at your nearest hospital: inability to breathe properly, severe dizziness, drowsiness and confusion, symptoms of allergy (eg itchy skin, rash, skin blisters or discolouration of skin upon exposure to sunlight). For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
WARNING:
Limitations of use
Proladone suppositories should only be given when your doctor decides that other treatment options are not able to effectively manage your pain or you cannot tolerate them.
Hazardous and harmful use
Proladone suppositories poses risks of abuse, misuse and addiction which can lead to overdose and death. Your doctor will monitor you regularly during treatment.
Life threatening respiratory depression
Proladone suppositories can cause life-threatening or fatal breathing problems (slow, shallow, unusual or no breathing), even when used as recommended. These problems can occur at any time during use, but the risk is higher when first starting Proladone suppositories and after a dose increase, if you are older, or have an existing problem with your lungs. Your doctor will monitor you and change the dose as appropriate.
Use of other medicines while being given Proladone suppositories
Using Proladone suppositories with other medicines that can make you feel drowsy such as sleeping tablets (e.g. benzodiazepines), other pain relievers, antihistamines, antidepressants, antipsychotics, gabapentinoids (e.g. gabapentin and pregabalin), cannabis and alcohol may result in severe drowsiness, decreased awareness, breathing problems, coma and death. Your doctor will minimise the dose and duration of use; and monitor you for signs and symptoms of breathing difficulties and sedation. You must not drink alcohol while using Proladone suppositories.
PROLADONE
Active ingredient: 30 mg oxycodone suppository
Consumer Medicine Information (CMI)
This leaflet provides important information about using Proladone. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Proladone.
Where to find information in this leaflet:
1. Why am I using Proladone?
2. What should I know before I use Proladone?
3. What if I am taking other medicines?
4. How do I use Proladone?
5. What should I know while using Proladone?
6. Are there any side effects?
7. Product details
1. Why am I using Proladone?
Proladone contains the active ingredient oxycodone.
Proladone is a suppository containing oxycodone. It belongs to a group of medicines called opioid analgesics and acts to relieve pain.
Proladone is used to relieve pain after surgery or for treating cancer pain.
Your doctor may have prescribed Proladone for another reason.
Ask your doctor if you have any questions about why Proladone has been prescribed for you.
Addiction
You can become addicted to Proladone even if you take it exactly as prescribed. Proladone may become habit forming causing mental and physical dependence. If abused it may become less able to reduce pain.
Dependence
As with all other opioid containing products, your body may become used to you taking Proladone. Taking it may result in physical dependence. Physical dependence means that you may experience withdrawal symptoms if you stop taking Proladone suddenly, so it is important to take it exactly as directed by your doctor.
Tolerance
Tolerance to Proladone may develop, which means that the effect of the medicine may decrease. If this happens, more may be needed to maintain the same effect.
Withdrawal
Continue taking your medicine for as long as your doctor tells you. If you stop taking this medicine suddenly, your pain may worsen and you may experience some or all of the following withdrawal symptoms:
- nervousness, restlessness, agitation, trouble sleeping or anxiety
- body aches, weakness or stomach cramps
- loss of appetite, nausea, vomiting or diarrhoea
- increased heart rate, breathing rate or pupil size
- watery eyes, runny nose, chills or yawning
- increased sweating.
Proladone given to the mother during labour can cause breathing problems and signs of withdrawal in the newborn.
2. What should I know before I use Proladone?
Warnings
Do not use Proladone if:
- you are allergic to oxycodone, or any of the ingredients listed at the end of this leaflet. Some of the symptoms of an allergic reaction are:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
- discolouration of the skin.
Always check the ingredients to make sure you can use this medicine. - you currently have, or have had, any of the following:
- head injury, or increased pressure in the head
- brain tumour
- epilepsy (also known as fits or convulsions)
- are taking, or have taken in the last 14 days, medicine for depression especially Monoamine Oxidase Inhibitors (MAOIs)
- heart problems such as irregular and/or rapid heartbeat
- asthma
- any difficulty in breathing
- alcoholism - you are a child as there have been no studies of its effects in children
- the expiry date printed on the pack has passed or if the packaging is torn or shows signs of tampering.
If you are not sure whether you should be using Proladone contact your doctor.
Check with your doctor if you:
- have allergies to any other medicines, foods, preservatives or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Your doctor or pharmacist will discuss the possible risks and benefits of using Proladone during pregnancy.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Like most opioid medicines, Proladone is not recommended while you are breast-feeding.
Tell your doctor if you have, or have had, any medical conditions, especially the following:
- myasthenia gravis, a disease of the muscles
- an underactive thyroid gland
- kidney disease
- liver disease
- if male, prostate problems or difficulty passing water
- bowel disorders
- asthma or breathing problems.
If you have not told your doctor about any of the above, tell them before you start using Proladone.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Proladone may interfere with each other. These include:
- medicines used to treat hyperactivity
- medicines used to produce calmness or to help you sleep
- medicines called anaesthetics which are used to put you to sleep during an operation or procedure
- barbiturates, medicines used to treat epilepsy, to produce calmness or to help you sleep
- other opioid medicines used to treat pain
- medicines used to treat depression, psychiatric or mental disorder
- medicines used to treat certain mental and emotional conditions e.g. depression
- medicines used to treat high blood pressure or other heart conditions
- medicines used to prevent blood clots
- medicines used to prevent or relieve the symptoms of allergy (such as hay fever)
- medicines used to treat nausea or vomiting.
These medicines may be affected by Proladone or they may affect how well it works.
Tell your doctor if you regularly drink alcohol. Alcohol may interfere with this medicine. Your doctor may advise you to stop drinking alcohol while using this medicine.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while using Proladone.
4. How do I use Proladone?
How much to use
- Your doctor will tell you how many suppositories you need to use each day
How to use Proladone
If possible, go to the toilet and empty your bowels before using your suppository.
Suppositories work best if your bowels are empty.
Follow these steps to use a suppository:
- Wash your hands thoroughly with soap and water.
- Feel the suppository while it is still in the foil.
- If it feels soft, keep it in the foil and chill it in the fridge or by holding it under cold water for a few minutes. Do not remove the foil wrapper while you are chilling it.
- Put on a disposable glove, if desired. They are available from pharmacies.
- Remove all of the foil wrapper from the suppository.
- Moisten the suppository by dipping it briefly in cool water.
- Lie on your side and raise your knee to your chest.
- Push the suppository gently, pointed end first, into your rectum (back passage).
- Remain lying down for a few minutes while the suppository dissolves.
- Throw away the disposable glove and any used material and wash your hands thoroughly to reduce the risk of infection.
Try not to go to the toilet and open your bowels for at least an hour after using the suppository.
The suppository takes about one hour to be completely absorbed and do its work.
If you are not sure how to use a suppository, ask your doctor or pharmacist.
If you forget to use Proladone
If it is almost time for your next suppository (within 2 or 3 hours), skip the suppository you missed and use your next suppository when you are meant to.
Otherwise, use it as soon as you remember, and then go back to using the suppositories as you would normally.
Do not use two suppositories to make up for the one that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to use your medicine, ask your pharmacist for some hints.
How long to use Proladone
The length of treatment will depend on your condition. Your doctor will advise you when to stop using Proladone.
Do not suddenly stop using Proladone. Your doctor may want you to gradually reduce the amount of Proladone you are using before stopping completely. Stopping gradually will lessen the risk of unwanted side effects.
If you use too much Proladone
If you or someone else receive too much (overdose), and experience one or more of the symptoms below, immediately call triple zero (000) for an ambulance.
Keep the person awake by talking to them or gently shaking them every now and then. You should follow the above steps even if someone other than you have accidentally used Proladone that was prescribed for you.
If someone takes an overdose they may experience one or more of the following symptoms:
- Slow, unusual or difficult breathing
- Drowsiness, dizziness or unconsciousness
- Slow or weak heartbeat
- Nausea or vomiting
- Convulsions or fits
- Shakiness, sweating or hunger
- Memory loss, difficulty concentrating, changes in personality or behaviour, weakness or paralysis in the limbs (arms and legs), loss of coordination
If you think you or someone else may have used too much Proladone, you should immediately:
- phone the Poisons Information Centre by calling 13 11 26 (Australia), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
When seeking medical attention, take this leaflet and remaining medicine with you to show the doctor. Also tell them about any other medicines or alcohol which have been taken.
5. What should I know while using Proladone?
Things you should do
Tell any other doctors, dentists, and pharmacists who are treating you that you are using Proladone.
If you are about to be started on any new medicine, tell your doctor, dentist or pharmacist that you are using Proladone.
If you plan to have surgery that needs a general anaesthetic, tell your doctor or dentist that you are using Proladone.
If you become pregnant while using Proladone, tell your doctor.
Things you should not do
Do not give Proladone to anyone else, even if they have the same condition as you.
Do not use Proladone to treat any other complaints unless your doctor tells you to.
Do not stop using Proladone or lower or increase the dose without checking with your doctor.
Do not run out of this medicine over the weekend or on holidays. Your doctor may want you to gradually reduce the amount of Proladone you are using before stopping completely. This may help reduce the possibility of withdrawal symptoms such as nausea, diarrhoea, body aches (see Side effects).
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Proladone affects you.
Proladone may cause drowsiness, sleepiness or dizziness in some people and affect alertness. Make sure you know how you react to Proladone before you drive or operate machinery or do anything else that could be dangerous if you are sleepy or dizzy. If this occurs do not drive.
Drinking alcohol
Be careful when drinking alcohol while you are using Proladone. Drinking alcohol while using this medicine could make you feel more dizzy, sleepy or light-headed than usual. Your doctor may suggest you avoid alcohol while you are using Proladone.
Be careful using the suppositories for people over 65 years
Families and carers should be aware that these people may become confused while using Proladone. Special care may be needed.
Looking after your medicine
Keep your suppositories in the foil pack until it is time to use them. If you take the suppositories out of the pack they will not keep well.
Keep your suppositories in a cool dry place where the temperature stays below 25°C.
Do not store Proladone or any other medicines in the bathroom or near a sink.
Do not leave it in the car on hot days or on window sills.
Heat and dampness can destroy some medicines.
Keep it where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If your doctor tells you to stop using the suppositories, if the medicine is damaged, you no longer need to use this medicine or you find that they have passed their expiry date, ask your pharmacist what to do with any that are left over.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
Do not be alarmed by this list of possible side effects.
You may not experience any of them.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Common side effects
| Common side effects | What to do |
| Speak to your doctor if you have any of these common side effects and they worry you |
Other side effects
| Other side effects | What to do |
| Speak to your doctor if you have any of these side effects and they worry you |
Serious side effects
| Serious side effects | What to do |
| If any of the following happen, stop using Proladone and tell your doctor immediately or go to the Emergency Department at your nearest hospital |
High doses of Proladone can cause:
- heart failure
- low blood pressure
- inability to breathe properly.
The following side effects may occur when you stop using Proladone:
- body aches
- diarrhoea
- gooseflesh or goose bumps
- loss of appetite
- nervousness
- restlessness
- runny nose
- sneezing
- shivering
- stomach cramps
- feeling sick, also called nausea
- trouble sleeping
- an increase in sweating and yawning
- weakness
- fast heart beat
- unexplained fever
These symptoms are usually mild if Proladone is stopped gradually.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Proladone contains
| Active ingredient (main ingredient) | oxycodone |
| Other ingredients (inactive ingredients) | lactose monohydrate maize starch pectin povidone magnesium stearate hard fat |
Do not take this medicine if you are allergic to any of these ingredients.
Proladone does not contain sucrose, gluten, tartrazine, alcohol, dyes or preservatives.
What Proladone looks like
Proladone is a smooth, off-white, oval/cone-shaped suppository for rectal use which will dissolve in the rectum (back passage). It is covered by a wax coating for easy insertion into the rectum.
Proladone 30 mg
12 suppositories per foil strip
AUST R 14965
Who distributes Proladone
Proladone is supplied in Australia by:
Phebra Pty Ltd,
19 Orion Road,
Lane Cove West, NSW 2066,
Australia
This leaflet was prepared in November 2025.
Phebra and the Phi symbol are trademarks of Phebra Pty Ltd, 19 Orion Road, Lane Cove West, NSW 2066, Australia.

Brand Information
| Brand name | Proladone |
| Active ingredient | Oxycodone |
| Schedule | S8 |
MIMS Revision Date: 01 December 2025
Hazardous and harmful use. Proladone suppositories poses risks of hazardous and harmful use which can lead to overdose and death. Assess the patient's risk of hazardous and harmful use before prescribing and monitor the patient regularly during treatment (see Section 4.4 Special Warnings and Precautions for Use).
Life threatening respiratory depression. Serious, life-threatening or fatal respiratory depression may occur with the use of Proladone suppositories. Be aware of situations which increase the risk of respiratory depression, modify dosing in patients at risk and monitor patients closely, especially on initiation or following a dose increase (see Section 4.4 Special Warnings and Precautions for Use).
Concomitant use of benzodiazepines and other central nervous system (CNS) depressants, including alcohol. Concomitant use of opioids with benzodiazepines, gabapentinoids, antihistamines, tricyclic antidepressants, antipsychotics, cannabis or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Limit dosages and durations to the minimum required; and monitor patients for signs and symptoms of respiratory depression and sedation. Caution patients not to drink alcohol while taking Proladone suppositories.
1 Name of Medicine
Oxycodone.
2 Qualitative and Quantitative Composition
Proladone suppositories contain oxycodone 30 mg per suppository as the active ingredient.
Proladone suppositories also contain lactose monohydrate.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Proladone suppositories consist of an oval, 22 mm x 10 mm compressed cone, which will dissolve in use and which is covered by a wax coating to aid insertion, presented as a smooth, mottled, off-white product.
4 Clinical Particulars
4.1 Therapeutic Indications
Semisynthetic narcotic analgesic. Relief of post-operative pain following a wide range of major operative procedures such as major orthopaedic, abdominal, gynaecological and thoracic surgery, and for the relief of pain in malignant disease.
4.2 Dose and Method of Administration
One suppository every six to eight hours; in case of terminal disease, one suppository as required to control pain.
4.3 Contraindications
Hypersensitivity to opiate narcotics, severe respiratory disease, acute respiratory disease, acute respiratory depression, cor pulmonale, cardiac arrhythmias, bronchial asthma, acute alcoholism, brain tumour, head injuries, increased cerebrospinal or intracranial pressure, severe central nervous system (CNS) depression, convulsive disorders, delirium tremens, suspected surgical abdomen, and concomitant monoamine oxidase inhibitors (MAOIs) or within 14 days of such therapy.
The use of Proladone suppositories is contraindicated in chronic (long-term) non-cancer pain.
4.4 Special Warnings and Precautions for Use
Oxycodone can produce drug dependence and, therefore, has the potential of being abused. Psychological dependence, physical dependence and tolerance may develop upon repeated administration. Abrupt withdrawal of oxycodone in those physically dependent may precipitate withdrawal symptoms. Therefore, patients on prolonged therapy should be withdrawn gradually from the drug if it is no longer required for pain control.
Oxycodone should be used with extreme caution in patients with head injuries and raised intracranial pressure, as respiratory depression and ability to increase cerebrospinal fluid (CSF) pressure may be exaggerated, thereby complicating the clinical course.
Therapeutic doses of oxycodone may decrease respiratory drive and increase airways resistance in patients with acute asthma, chronic obstructive airways disease or those with substantially decreased pulmonary reserve or respiratory depression.
Oxycodone should be used only with caution and in reduced dosage during concomitant administration of other narcotic analgesics, general anaesthetics, phenothiazines and other tranquillisers, sedative/ hypnotics, some tricyclic antidepressants and other CNS depressants (including alcohol). Respiratory depression, hypotension and profound sedation or coma may result.
Administration of oxycodone may result in severe hypotension in patients whose ability to maintain adequate blood pressure is compromised by reduced blood volume, or concurrent administration of such drugs as phenothiazines or certain anaesthetics. Oxycodone may obscure the diagnosis or clinical course of patients with acute abdominal conditions.
Opioid analgesics should be used with caution in patients with myasthenia gravis.
The euphoric activity of opioid compounds has led to their abuse. It should be given with caution or in reduced doses to patients with hypothyroidism, adrenocortical insufficiency, impaired kidney or liver function, prostatic hypertrophy or shock. It should be used with caution in patients with obstructive bowel disorders.
Hazardous and harmful use. Proladone suppositories contains the opioid oxycodone and is a potential drug of abuse, misuse and addiction. Addiction can occur in patients appropriately prescribed Proladone suppositories at recommended doses.
The risk of addiction is increased in patients with a personal or family history of substance abuse (including alcohol and prescription and illicit drugs) or mental illness. The risk also increases the longer the drug is used and with higher doses. Patients should be assessed for their risks for opioid abuse or addiction prior to being prescribed Proladone suppositories.
All patients receiving opioids should be routinely monitored for signs of misuse and abuse. Opioids are sought by people with addiction and may be subject to diversion. Strategies to reduce these risks include prescribing the drug in the smallest appropriate quantity and advising the patient on the safe storage and proper disposal of any unused drug (see Section 6.4 Special Precautions for Storage; Section 6.6 Special Precautions for Disposal). Caution patients that abuse of oral or transdermal forms of opioids by parenteral administration can result in serious adverse events, which may be fatal.
Patients should be advised not to share Proladone suppositories with anyone else.
Respiratory depression. Serious, life-threatening or fatal respiratory depression can occur with the use of opioids even when used as recommended. It can occur at any time during the use of Proladone suppositories but the risk is greatest during initiation of therapy or following an increase in dose. Patients should be monitored closely for respiratory depression at these times.
The risk of life-threatening respiratory depression is also higher in elderly, frail, or debilitated patients and in patients with existing impairment of respiratory function (e.g. chronic obstructive pulmonary disease; asthma) and in patients with hepatic or renal impairment (see Use in hepatic impairment, Use in renal impairment). Opioids should be used with caution and with close monitoring in these patients (see Section 4.2 Dose and Method of Administration). The use of opioids is contraindicated in patients with severe respiratory disease, acute respiratory disease and respiratory depression (see Section 4.3 Contraindications).
The risk of respiratory depression is greater with the use of high doses of opioids, especially high potency and modified release formulations, and in opioid naïve patients. Initiation of opioid treatment should be at the lower end of the dosage recommendations with careful titration of doses to achieve effective pain relief. Careful calculation of equianalgesic doses is required when changing opioids or switching from immediate release to modified release formulations, (see Section 4.2 Dose and Method of Administration), together with consideration of pharmacological differences between opioids. Consider starting the new opioid at a reduced dose to account for individual variation in response.
Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol. Concomitant use of opioids and benzodiazepines or other CNS depressants, including alcohol, may result in sedation, respiratory depression, coma and death. Because of these risks, concomitant prescribing of Proladone suppositories with CNS depressant medicines, such as other opioid analgesics, benzodiazepines, gabapentinoids, cannabis, sedatives, hypnotics, tricyclic antidepressants, antipsychotics, antihistamines, centrally-active anti-emetics and other CNS depressants, should be reserved for patients for whom other treatment options are not possible. If a decision is made to prescribe Proladone suppositories concomitantly with any of the medicines, the lowest effective dose should be used, and the duration of treatment should be as short as possible. Patients should be followed closely for signs and symptoms of respiratory depression and sedation. Patients and their caregivers should be made aware of these symptoms. Patients and their caregivers should also be informed of the potential harms of consuming alcohol while taking Proladone suppositories.
Tolerance, dependence and withdrawal. Neuroadaptation of the opioid receptors to repeated administration of opioids can produce tolerance and physical dependence. Tolerance is the need for increasing doses to maintain analgesia. Tolerance may occur to both the desired and undesired effects of the opioid.
Physical dependence, which can occur after several days to weeks of continued opioid usage, results in withdrawal symptoms if the opioid is ceased abruptly or the dose is significantly reduced. Withdrawal symptoms can also occur following the administration of an opioid antagonist (e.g. naloxone) or partial agonist (e.g. buprenorphine). Withdrawal can result in some or all of the following symptoms: dysphoria, restlessness/agitation, lacrimation, rhinorrhoea, yawning, sweating, chills, myalgia, mydriasis, irritability, anxiety, increasing pain, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhoea, increased blood pressure, increased respiratory rate and increased heart rate.
When discontinuing Proladone suppositories in a person who may be physically-dependent, the drug should not be ceased abruptly but withdrawn by tapering the dose gradually (see Ceasing opioids and see Section 4.2 Dose and Method of Administration).
Accidental ingestion/exposure. Accidental ingestion or exposure of Proladone suppositories, especially by children, can result in a fatal overdose of oxycodone. Patients and their caregivers should be given information on safe storage and disposal of unused Proladone suppositories (see Section 6.4 Special Precautions for Storage; Section 6.6 Special Precautions for Disposal).
Hyperalgesia. Hyperalgesia may occur with the use of opioids, particularly at high doses. Hyperalgesia may manifest as an unexplained increase in pain, increased levels of pain with increasing opioid dosages or diffuse sensitivity not associated with the original pain. Hyperalgesia should not be confused with tolerance (see Tolerance, dependence and withdrawal). If opioid induced hyperalgesia is suspected, the dose should be reduced and tapered off if possible. A change to a different opioid may be required.
Sleep-related breathing disorders. Opioids can cause sleep-related breathing disorders including central sleep apnoea (CSA) and sleep-related hypoxemia. Opioid use increases the risk of CSA in a dose-dependent fashion. In patients who present with CSA, consider decreasing the total opioid dosage.
Adrenal insufficiency. Adrenal insufficiency has been reported with opioid use, more often following long-term use. Symptoms may include nausea, vomiting, anorexia, fatigue, weakness, dizziness, or low blood pressure. If adrenal insufficiency is suspected, appropriate laboratory testing is recommended and discontinuation of treatment with Proladone should be considered.
Endocrine effects. Opioids, such as Proladone, may influence the hypothalamic-pituitary-adrenal or –gonadal axes. Hormonal disturbances that have been observed include an increase in serum prolactin and decreases in plasma cortisol and testosterone. Clinical symptoms may manifest from these hormonal changes.
Androgen deficiency may manifest as low libido, impotence, erectile dysfunction, amenorrhea, or infertility.
Neonatal withdrawal syndrome. Chronic use of oxycodone by the mother at the end of pregnancy may result in a withdrawal syndrome (e.g. hypertonia, neonatal tremor, neonatal agitation, myoclonus, convulsions, apnoea or bradycardia) in the neonate. In many reported cases the withdrawal was serious and required treatment. The syndrome is generally delayed for several hours to several days after birth. (See Section 4.6, Use in pregnancy).
Gastrointestinal toxicity. Reports of significant oesophageal dysfunction have been observed via high-resolution manometry in patients taking opioid medicines on a long-term basis. Discontinuation or weaning of opioids should be considered in patients presenting with oesophageal complaints including but not limited to dysphagia, regurgitation, or noncardiac chest pain.
Ceasing opioids. Abrupt discontinuation or rapid decreasing of the dose in a person physically dependent on an opioid may result in serious withdrawal symptoms and uncontrolled pain (see Tolerance, dependence and withdrawal). Such symptoms may lead the patient to seek other sources of licit or illicit opioids. Opioids should not be ceased abruptly in a patient who is physically dependent but withdrawn by tapering the dose slowly. Factors to take into account when deciding how to discontinue or decrease therapy include the dose and duration of the opioid the patient has been taking, the type of pain being treated and the physical and psychological attributes of the patient. A multimodal approach to pain management should be in place before initiating an opioid analgesic taper. During tapering, patients require regular review and support to manage any increase in pain, psychological distress and withdrawal symptoms.
There are no standard tapering schedules suitable for all patients and an individualised plan is necessary. In general, tapering should involve a dose reduction of no more than 10 percent to 25 percent every 2 to 4 weeks (see Section 4.2 Dose and Method of Administration). If the patient is experiencing increased pain or serious withdrawal symptoms, it may be necessary to go back to the previous dose until stable before proceeding with a more gradual taper.
When ceasing opioids in a patient who has a suspected opioid use disorder, the need for medication assisted treatment and/or referral to a specialist should be considered.
Use in hepatic impairment. Oxycodone should be given with caution or in reduced doses to patients with impaired liver function.
Use in renal impairment. Oxycodone should be given with caution or in reduced doses to patients with impaired kidney function.
Use in the elderly. Oxycodone should be administered with caution and in reduced dosages to elderly patients.
Paediatric use. Oxycodone should not be administered to children.
Effects on laboratory tests. No data available.
Hepatobillary disorders. Opioids may cause dysfunction and spasm of the sphincter of Oddi, thus raising intrabiliary pressure and increasing the risk of biliary tract symptoms and pancreatitis. Therefore, Proladone has to be administered with caution in patients with pancreatitis and diseases of the biliary tract.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Generally, the effects of oxycodone may be antagonised by acidifying agents and potentiated by alkalising agents.
The analgesic effect of oxycodone is potentiated by amphetamines, chlorpromazine and methocarbol. CNS depressants, such as other opioids, alcohol, anaesthetics, sedatives, benzodiazepines, hypnotics, barbiturates, phenothiazines, chloral hydrate, glutethimide, gabapentinoids, cannabis, tricyclic antidepressants, antipsychotics, antihistamines, centrally-active anti-emetics and other CNS depressants may enhance the depressant effects of oxycodone (see Section 4.4 Special Warnings and Precautions for Use, Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol). MAOIs (including procarbazine hydrochloride), pyrazolidone antihistamines, β-blockers and alcohol may also enhance the depressant effect of oxycodone.
Oxycodone may increase the anticoagulant activity of coumarin derivatives.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category C)
Oxycodone is Pregnancy Category C - Drugs which, owing to their pharmacological effects, have caused or may be suspected of causing, harmful effects on the human fetus or neonate without causing malformations. These effects may be reversible. Accompanying texts should be consulted for further details.
During labour, narcotic analgesics may cause respiratory depression in the newborn infant.
Neonatal withdrawal syndrome has been reported in newborn infants with chronic maternal use of Proladone during pregnancy.
Use in lactation. It is not known whether oxycodone is excreted in breast milk nor whether it has a harmful effect on the newborn. Therefore, oxycodone is not recommended for nursing mothers unless the expected benefits outweigh the potential risk.
4.7 Effects on Ability to Drive and Use Machines
Oxycodone may impair the mental and/or physical abilities needed for certain potentially hazardous activities, such as driving a car or operating machinery. Patients should be cautioned accordingly.
4.8 Adverse Effects (Undesirable Effects)
In normal doses, the most common side effects of opioid analgesics are nausea, vomiting, constipation, drowsiness and confusion. Micturition may be difficult and there may be ureteric or biliary spasm; there is also an antidiuretic effect. Dry mouth, sweating, facial flushing, anorexia, faintness, vertigo, bradycardia, supraventricular tachycardia, syncope, palpitations, orthostatic hypotension, hypothermia, restlessness, changes of mood and miosis also occur. Raised intracranial pressure occurs in some patients. Due to the histamine-releasing effect, reactions such as urticaria and pruritus occur in some individuals. Muscle rigidity has been reported following the administration of opioids. Larger doses produce respiratory depression and hypotension, with circulatory failure and deepening coma. Convulsions may occur in infants and children. Death may occur from respiratory failure. Toxic doses vary considerably with the individual and regular users may tolerate large doses. Side effects with an unknown frequency include sphincter of Oddi dysfunction.
In long term use, physical dependence and tolerance may develop.
The following withdrawal symptoms may be observed after narcotics are discontinued: body aches, diarrhoea, gooseflesh, loss of appetite, nervousness, restlessness, runny nose, sneezing, tremors or shivering, stomach cramps, nausea, trouble with sleeping, unusual increase in sweating and yawning, weakness, tachycardia and unexplained fever. With appropriate medical use of narcotics and gradual withdrawal from the drug, these symptoms are usually mild.
Post-marketing experience. Respiratory disorders with a frequency not known. Central sleep apnoea syndrome.
Gastrointestinal disorders with a frequency not known. Pancreatitis.
Endocrine disorders with a frequency not known. Adrenal insufficiency and androgen deficiency.
Reporting suspected adverse reactions. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Serious overdosage with oxycodone is characterised by respiratory depression and somnolence progressing to coma and skeletal muscle flaccidity. Hypoglycaemia, cardiac arrest and death may occur.
Toxic leukoencephalopathy has been observed with opioid overdose.
Treatment. Primary attention should be given to the re-establishment of adequate respiratory exchange through provision of a patent airway and the institution of assisted or controlled ventilation. The narcotic antagonist naloxone is a specific antidote against respiratory depression which may result from overdosage or unusual sensitivity to narcotics, including oxycodone. Therefore, an appropriate dose of naloxone (usual adult dose: 0.4 mg) should be administered, preferably by the intravenous route, simultaneously with efforts at respiratory resuscitation. Since the duration of action of oxycodone may exceed that of the antagonist, the patient should be kept under continued surveillance and repeated doses of the antagonist should be administered as needed to maintain adequate respiration.
An antagonist should not be administered in the absence of clinically significant respiratory or cardiovascular depression.
Oxygen, intravenous fluids, vasopressors and other supportive measures should be used as indicated.
Gastric emptying may be useful in removing unabsorbed drug.
In an individual physically dependent on narcotics, the administration of the usual dose of a narcotic antagonist will precipitate an acute withdrawal syndrome. The severity of this syndrome will depend on the degree of physical dependence and the dose of antagonist administered. The use of narcotic antagonists in such individuals should be avoided if possible. If a narcotic antagonist must be used to treat serious respiratory depression in the physically dependent patient, only 10 to 20% of the usual initial dose of the antagonist should be administered.
In severe toxicity, the cardiovascular system is usually depressed and requires supportive treatment. If hypotension is due to vasodilatation, plasma expansion or even vasopressors may be required. Additional measures include support of electrolyte balance, maintenance of normal temperature, catheterisation of the bladder to avoid distension and symptomatic treatment of itching, nausea, vomiting, headache and confusion during the recovery period.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The effects of oxycodone when combined with pectin are of longer duration than those of oxycodone hydrochloride.
The suppository has been found to reduce the severity of intractable pain due to carcinomatosis for up to 8 hours.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. No data available.
Distribution. No data available.
Metabolism. No data available.
Excretion. No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Proladone suppositories also contain lactose monohydrate, maize starch, pectin, povidone, magnesium stearate and hard fat.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG)1. The expiry date can be found on the packaging.
1 AUST R 14965.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Proladone is presented as a strip pack of 12 suppositories.
Phebra product code - TAB007.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
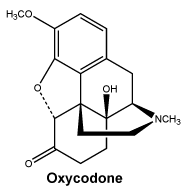
Chemical name: 6-deoxy-7, 8-dihydro-14-hydroxy-3-O-methyl-6-oxomorphine.
The molecular weight of oxycodone is 315.4. The molecular formula is C18H21NO4.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S8 - Controlled drug.
Date of First Approval
09 September 1991
Date of Revision
23 October 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.