Prolistat
Brand Information
| Brand name | Prolistat |
| Active ingredient | Orlistat |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Prolistat.
Summary CMI
Prolistat®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Prolistat?
Prolistat contains the active ingredient orlistat. It belongs to a group of medicines called lipase inhibitors. These medicines work by targeting the absorption of dietary fat in your body rather than suppressing your appetite. Prolistat is used to treat people who are obese (BMI ≥ 30), or overweight (BMI ≥ 27) with other risk factors (e.g. high blood pressure, high cholesterol, and high blood lipid profile). For more information, see Section 1. Why am I using Prolistat? in the full CMI.
2. What should I know before I use Prolistat?
Do not use if you have ever had an allergic reaction to orlistat or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Prolistat? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Prolistat and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Prolistat?
- The recommended dose of Prolistat for adults over 18 years of age is one 120 mg capsule taken three times a day.
- You will usually take 1 capsule with each of the 3 main meals per day. This means that you will usually take one capsule at breakfast, lunch and dinner. It can be taken during the meal or up to 1 hour after the meal is consumed.
- Prolistat only works in the presence of dietary fat in your body. Therefore, if you miss a main meal or if you know that the meal contains no fat, then Prolistat need not be taken.
More instructions can be found in Section 4. How do I use Prolistat? in the full CMI.
5. What should I know while using Prolistat?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Prolistat? in the full CMI.
6. Are there any side effects?
Mild side effects include Nausea (feeling sick), gum disease (swollen, painful bleeding gums), dyspepsia (indigestion), bloating, increased flatulence (wind) with or without discharge, abdominal pain, urgent need to open the bowels, oily, fatty, or liquid stools, oily discharge from anus, rectal pain, or discomfort, Headache, asthenia (unusual tiredness or weakness).
Tell your pharmacist or doctor if you have type II diabetes and you notice any of the following and they worry you: bloating, hypoglycaemia - symptoms include sweating, weakness, hunger, dizziness, trembling, headache, flushing or paleness, numbness, fast pounding heart.
Serious side effects (Call your doctor straight away, or go straight to the Emergency Department) include: Severe and continuous rectal bleeding, signs of urinary tract infection such as going to the toilet more frequently, burning sensation when passing urine and/or lower back pain, anxiety, signs of respiratory infection such as coughing, runny nose shortness of breath, and/or fever, rash or blistering of the skin, muscle or back pain, hypothyroidism (tiredness, lethargy, muscle weakness, cramps, feeling the cold, a slow heart rate, dry and flaky skin, hair loss, a deep and husky voice, weight gain). Very serious side effects (Call your doctor straight away, or go straight to the Emergency Department) include: Symptoms of liver disease (hepatitis) such as nausea, vomiting, loss of appetite, feeling generally unwell, fever, itching, yellowing of the skin and eyes and dark coloured urine, sudden signs of allergy such as rash, itching or hives on the skin, swelling of face, lips, tongue or other parts of the body, shortness of breath, wheezing or trouble breathing, unexplained bleeding, bruising or blood in your urine, symptoms of inflammation of the pancreas (pancreatitis) such as severe upper stomach pain, often with nausea and vomiting.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Prolistat®
Active ingredient(s): Orlistat
Consumer Medicine Information (CMI)
This leaflet provides important information about using Prolistat. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Prolistat.
Where to find information in this leaflet:
1. Why am I using Prolistat?
2. What should I know before I use Prolistat?
3. What if I am taking other medicines?
4. How do I use Prolistat?
5. What should I know while using Prolistat?
6. Are there any side effects?
7. Product details
1. Why am I using Prolistat?
Prolistat contains the active ingredient Orlistat.
Prolistat is used to treat people who are obese (BMI ≥ 30), or overweight (BMI ≥ 27) with other risk factors (e.g. high blood pressure, high cholesterol, and high blood lipid profile).
Your Body Mass Index (BMI) is calculated by dividing your weight (in kg) by your height (in metres) squared.
Prolistat belongs to a group of medicines called lipase inhibitors.
These medicines work by targeting the absorption of dietary fat in your body rather than suppressing your appetite.
Dietary fats are large molecules that need to be broken down before they can be absorbed into the body. They are broken down by enzymes called lipases. Lipases play an important role in the digestion of dietary fat. When taken with a meal, orlistat interferes with the activity of these enzymes. This allows about 30% of the fat eaten in the meal to pass through the gut undigested. Therefore, your body cannot store these excess calories as fatty tissue or use them as a source of energy. This helps you to reduce your weight by burning up fat that you are already carrying, maintain your lower weight and minimise any weight regain.
Losing even small amounts of weight and keeping it off produces additional health benefits for you, especially when you are at risk for other diseases such as heart disease and diabetes.
Prolistat can also help to improve risk factors, such as high blood pressure, high cholesterol and high blood sugar. If these are not treated, they could lead to other diseases such as hypertension and diabetes.
Prolistat should be taken in conjunction with a well-balanced calorie-controlled diet and other appropriate measures such as exercise.
Ask your pharmacist or doctor if you have any questions or if you are not sure why Prolistat has been recommended for you.
Prolistat is not addictive.
2. What should I know before I use Prolistat?
Warnings
Do not use Prolistat if:
- you are allergic to Orlistat, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- you have chronic malabsorption syndrome (i.e. your body does not absorb essential nutrients as well as it should).This may occur with conditions such as tropical sprue and idiopathic steatorrhoea.
- You have certain pancreatic problems
- You have had recent major surgery to your stomach or intestines
- You have a blockage of your bile duct (gall stones).
- if the packaging is torn or shows signs of tampering.
- if the expiry date (EXP) printed on the pack has passed.
If you take this medicine after the expiry date has passed, it may not work as well.
If you are not sure whether you should start taking Prolistat, talk to your pharmacist or doctor.
Do not give Prolistat to children or adolescents under 18 years. The safety and effectiveness in children have not been established.
Check with your doctor or pharmacist if you:
- are pregnant or plan to become pregnant.
- are breast-feeding or intend to breast-feed.
- are taking oral contraceptives.
- have vitamin deficiencies.
- have kidney stones.
- have any allergies to any other substances, such as foods, preservatives or dyes.
- have peptic (stomach) ulcer disease.
- receive long term treatment for mental or nerve disorders.
- have symptomatic cholelithiasis (painful gall stones).
- have post surgical adhesions.
- have an eating disorder.
- take large doses of laxatives.
- have epilepsy or suffer from fits.
- have significant heart, kidney, liver, gastrointestinal or endocrine disorders.
- are taking medicines for HIV.
- have a thyroid disorder.
- have any other health problems.
If you have not told your pharmacist or doctor about any of the above, tell them before you take any Prolistat.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
The safety of the use of this medicine in women who are pregnant or may become pregnant has not been established. Prolistat is not recommended for use during pregnancy, unless you and your doctor have discussed the risks and benefits involved.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
It is not known whether orlistat is excreted in human milk. Prolistat should not be taken during breast-feeding.
If you are taking oral contraceptives, commonly known as birth control pills, the use of an additional method of contraception is recommended. This is because you might experience severe diarrhoea whilst taking Prolistat which may decrease the absorption of oral contraceptive into your body. It is recommended that you use an additional method of contraception to prevent pregnancy.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Prolistat and affect how it works.
- ciclosporin, a transplant rejection medicine
- warfarin or other anticoagulants (medicines that prevent blood clots)
- amiodarone, a medicine used to treat irregular heartbeat
- acarbose and other medicines used to treat diabetes
- medicines used to treat epilepsy or fits
- other weight-loss medicines
- some vitamin supplements.
- medicines for HIV
- drugs to control hypothyroidism
- lithium and other medicines used for mental or sleep disorders
The above medicines may be affected by Prolistat or may affect how well it works. You may need different amounts, or take them at different times, or you may need to take different medicines. Because weight loss has many beneficial effects, it may also affect the dose of medicines taken for conditions such as high cholesterol or diabetes.
Be sure to discuss these and other medicines you may be taking with your pharmacist or doctor.
Losing weight may also mean you need adjustments to these medicines.
Your pharmacist and doctor have more information on medicines to be careful with or avoid while taking Prolistat.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Prolistat.
4. How do I use Prolistat?
How much to take
Follow all directions given to you by your pharmacist and doctor carefully.
They may differ from the information contained in this leaflet.
- Take Prolistat exactly as your pharmacist or doctor has prescribed.
- The recommended dose of Prolistat for adults over 18 years of age is one 120 mg capsule taken three times a day.
- Taking more than the recommended dose will not provide an additional benefit.
- It is also recommended that you take a daily multivitamin supplement containing vitamins A, D E and K.
- Use by people under 18 years of age is not recommended except on medical advice.
- If you take the wrong dose, Prolistat may not work as well.
- Take Prolistat exactly as your pharmacist or doctor has prescribed.
- Ask your pharmacist or doctor if you are unsure of the correct dose for you. They will tell you exactly how much to take. This depends on your condition and whether you are taking any other medicines.
When to take Prolistat
- You will usually take 1 capsule with each of the 3 main meals per day. This means that you will usually take one capsule at breakfast, lunch and dinner. It can be taken during the meal or up to 1 hour after the meal is consumed.
- Prolistat only works in the presence of dietary fat in your body. Therefore, if you miss a main meal or if you know that the meal contains no fat, then Prolistat need not be taken.
How long to take it
- Continue taking the medicine for as long as your pharmacist or doctor tells you to, for up to four years.
- Take Prolistat every day for it to be effective.
Weight loss normally starts within 2 weeks and continues for 6 to 12 months on Prolistat treatment. Prolistat will then help you to maintain this new lower weight and help to prevent weight being regained. - The improvement in risk factors (such as reduction in high blood pressure, normalising blood sugar levels and lowering cholesterol levels) is usually observed within 1 month of starting therapy and is maintained during the course of the treatment.
- If you are unsure whether you should stop taking Prolistat, talk to your pharmacist or doctor.
If you forget to use Prolistat
- If it is almost time for your next dose (i.e. close to your next meal), skip the dose you missed and take the next dose when you are meant to.
- If you remember your missed dose within 1 hour of your last meal, take it and then take the next dose when you are meant to (i.e. after your next meal). Then go back to taking it as you would normally.
- If you are not sure what to do, ask your pharmacist or doctor.
- Do not try to make up for missed doses by taking more than one dose at a time. This may increase the chance of getting an unwanted side effect.
- If you have trouble remembering to take your medicine, ask your pharmacist for hints.
How to take it
- Swallow the capsule whole with a full glass of water.
- Prolistat should be taken with a well-balanced calorie-controlled diet that is rich in fruit and vegetables and contains an average of 30% calories from fat. Your daily intake of fat, carbohydrate and protein should be distributed over 3 main meals. To gain the most benefit, avoid eating food containing fat between meals, such as biscuits, chocolate or savoury snacks.
- Look out for fats in foods by reading all food labels.
Your fat intake may also be reduced by removing all fat from meat, skin from chicken and using non-fat or low-fat milk. - Be careful not to replace the fat in your diet with large quantities of other foods, such as sugar and alcohol.
- For further dietary advice, refer to the NHMRC website at www.nhmrc.gov.au.
If you use too much Prolistat
If you think that you have used too much Prolistat, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Prolistat?
Things you should do
- If you become pregnant while taking this medicine, tell your doctor immediately.
- Tell your pharmacist or doctor if, for any reason, you have not taken your medicine exactly as prescribed.
Otherwise, your pharmacist or doctor may think that it was not effective and change your treatment unnecessarily. - Tell your pharmacist or doctor if you feel the capsules are not helping your condition.
- If you are about to be started on any new medicine, tell your pharmacist and doctor that you are taking Prolistat. Likewise, tell any other doctors, dentists and pharmacists who are treating you that you are taking this medicine.
- If you are being treated for epilepsy or fits, contact your pharmacist or doctor if you think that the frequency or severity of convulsions have changed while taking Prolistat.
- Your doctor may need you to have regular blood tests to monitor your blood sugar or blood cholesterol level. To gain the most benefit from Prolistat you should follow the nutrition program recommended to you by your pharmacist or doctor. As with any weight-control program, over-consumption of fat and calories may
Remind any doctor, dentist or pharmacist you visit that you are using Prolistat.
Things you should not do
- Do not give your medicine to anyone else, even if they have the same condition as you. This medicine is only intended for the person it has been recommended for.
- Do not take Prolistat to treat any other complaints unless your pharmacist or doctor tells you to.
Weight reduction diet
People on a weight reduction diet may become deficient in some vitamins and minerals. To avoid this, you should follow your doctor's or pharmacist's advice in taking a well-balanced diet rich in fruit and vegetables. Because Prolistat works by preventing the absorption of some of the fat from the diet, it may affect absorption of some fat-soluble nutrients. Although most people taking Prolistat will still have normal vitamin levels, you should take a multivitamin supplement containing fat-soluble vitamins. Take this supplement at least 2 hours before or after Prolistat, or at bedtime.
Distribute your daily intake of fat over 3 main meals.
This medicine can cause harmless changes in your bowel habits, such as fatty or oily stools. This is due to the removal of undigested fat in your faeces. The possibility of this happening will increase if Prolistat is taken with a diet high in fat. In addition, your daily intake of fat should be distributed over 3 main meals. If Prolistat is taken with any one meal very high in fat, the possibility of unwanted effects on the digestive system will increase.
Oral contraceptive use
If you are taking oral contraceptives, commonly known as birth control pills, the use of an additional method of contraception is recommended. This is because you might experience severe diarrhoea whilst taking Prolistat which may decrease the absorption of oral contraceptive into your body. It is recommended that you use an additional method of contraception to prevent pregnancy.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Prolistat affects you.
Prolistat may cause dizziness in some people
Drinking alcohol
Tell your doctor if you drink alcohol.
Alcohol is not expected to interact with Prolistat.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
Keep Prolistat in a cool dry place where the temperature stays below 25°C. Store in original container in order to protect from light and moisture.;
for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on windowsills.
Heat and damp can destroy some medicines.
Keep it where young children cannot reach it.
- A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Do not keep Prolistat past its expiry date.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
The majority of unwanted effects related to Prolistat use result from its local action in your digestive system.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
| Tell your pharmacist or doctor if you have type II diabetes and you notice any of the following and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. You may require medical attention. |
Very Serious side effects
| Very Serious side effects | What to do |
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. You may need urgent medical attention or hospitalisation. |
These very serious side effects are very rare.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available without a doctor's prescription.
What Prolistat contains
| Active ingredient (main ingredient) | Orlistat 120 mg |
| Other ingredients (inactive ingredients) |
|
Prolistat does NOT contain lactose, sucrose, tartrazine or any other azo dyes.
Do not take this medicine if you are allergic to any of these ingredients.
What Prolistat looks like
Prolistat 120 mg capsules is available in blister packs of 42 or 84 capsules. The capsules are blue in colour and contain a white powder or slightly compacted agglomerates.
Australian Registration Number: AUST R 268252
Who distributes Prolistat
Boucher & Muir Pty Ltd
Level 9, 76 Berry Street
North Sydney NSW 2060
This leaflet was prepared in September 2025.
Brand Information
| Brand name | Prolistat |
| Active ingredient | Orlistat |
| Schedule | S3 |
MIMS Revision Date: 01 August 2022
1 Name of Medicine
Orlistat.
2 Qualitative and Quantitative Composition
Prolistat capsules contain orlistat 120 mg.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
The capsules are hard gelatin capsules with blue cap and body (size no. 1).
4 Clinical Particulars
4.1 Therapeutic Indications
Prolistat is indicated for the treatment of obese patients with a body mass index (BMI) ≥ 30, and overweight patients with a BMI ≥ 27 in the presence of other risk factors, in conjunction with a mildly hypocaloric diet.
4.2 Dose and Method of Administration
The recommended oral dose of Prolistat is one 120 mg capsule with each main meal (during or up to one hour after the meal). If a meal is missed or contains no fat, the dose of Prolistat may be omitted. The therapeutic benefits of Prolistat (including weight control and improvement of risk factors) are continued with long-term administration.
At the recommended therapeutic dose, the inhibition of the absorption of approximately one-third of dietary fat produces meaningful weight loss and reduces risk factors. At the same time, this effect allows absorption of adequate amounts of dietary fat and other nutrients that are essential for the maintenance of good health.
The patient should be on a nutritionally balanced, mildly hypocaloric diet that contains approximately 30% of calories from fat. It is recommended that the diet should be rich in fruit and vegetables. The daily intake of fat, carbohydrate and protein should be distributed over three main meals.
Because orlistat has been shown to reduce the absorption of some fat soluble vitamins and beta-carotene, patients should be counselled to take a multivitamin containing fat soluble vitamins to ensure adequate nutrition. The supplement should be taken at least 2 hours before or after the administration of Prolistat, such as bedtime.
In the 4 year clinical trial, patients took vitamin supplements containing vitamin A 800 microgram, vitamin D 5 microgram, vitamin E 10 mg, vitamin K 30 microgram and beta-carotene 400 microgram.
Doses above 120 mg three times daily have not been shown to provide additional benefit.
Special patient groups. There is no dose adjustment required for the elderly, nor in hepatic and/or renal impairment.
Children. The use of Prolistat in patients under 18 years of age is not recommended (see Section 4.4 Special Warnings and Precautions for Use, Paediatric use).
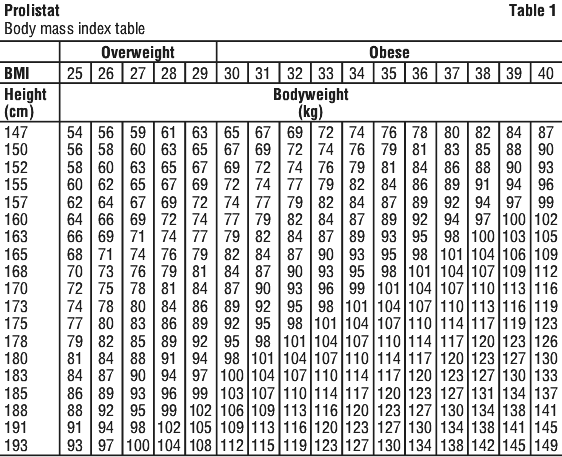
Body mass index (BMI) calculation. BMI (kg/m2) = weight (kilograms)/height2 (metres). See Table 1.
4.3 Contraindications
Prolistat is contraindicated in patients with:
hypersensitivity to orlistat or to any of the excipients in the capsule;
pancreatic enzyme deficiency or chronic pancreatitis and major gastrointestinal surgery;
chronic malabsorption syndrome;
cholestasis.
during pregnancy or breastfeeding.
4.4 Special Warnings and Precautions for Use
General. No serious adverse reactions or safety hazards related to the use of orlistat have been reported to date during large, long-term clinical trials (see Section 4.8 Adverse Effects (Undesirable Effects)).
In addition to clinical trial data, post-marketing information is now available (see Section 4.8 Adverse Effects (Undesirable Effects), Post-marketing experience).
Information for patients. Patients should be advised to adhere to dietary guidelines (see Section 4.2 Dose and Method of Administration). The possibility of experiencing gastrointestinal events (see Section 4.8 Adverse Effects (Undesirable Effects)) may increase when Prolistat is taken with a diet high in fat (in a 8.4 kilojoule/day diet (2000 calorie/day), a diet high in fat would contain > 30% calories from fat, which equates to > 67 g fat). The daily intake of fat should be distributed over three main meals. If Prolistat is taken with any one meal very high in fat, the possibility of gastrointestinal effects may increase.
Consumption of a diet low in fat will decrease the likelihood of experiencing adverse gastrointestinal events and this may help patients to monitor and regulate their fat intake. Patients should be advised of the merits of reducing the saturated fats in their diet whilst preserving an adequate intake of unsaturated fats including omega fatty acids.
Patients should be on a nutritionally balanced hypocaloric diet containing no more than 30% of calories as fat (≤ 67 g of fat/day) and should also have an adequate intake of fat soluble vitamins.
A reduction in ciclosporin plasma levels has been observed when Orlistat is coadministered. Therefore, it is recommended to monitor more frequently than usual the ciclosporin plasma levels when Prolistat is coadministered (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Weight loss induced by Prolistat is accompanied by improved metabolic control in type 2 diabetics which might allow or require reduction in the dose of hypoglycaemic medication (e.g. sulfonylureas) (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Cases of rectal bleeding have been reported with Orlistat. Prescribers should investigate further in case of severe and/or persistent symptoms.
The use of an additional contraceptive method is recommended to prevent possible failure of oral contraception that could occur in case of severe diarrhoea (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Coagulation parameters should be monitored in patients treated with concomitant oral anticoagulants (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions; Section 4.8 Adverse Effects (Undesirable Effects)).
Rare occurrence of hypothyroidism and/or reduced control of hypothyroidism may occur. The mechanism, although not proven, may involve a decreased absorption of iodine salts and/or levothyroxine (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Antiepileptics patient. Orlistat may unbalance anticonvulsant treatment by decreasing the absorption of antiepileptic drugs, leading to convulsions (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Antiretrovirals for HIV. Orlistat may potentially reduce the absorption of antiretroviral medicines for HIV and could negatively affect the efficacy of antiretroviral medications for HIV (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Use in patients with other disorders. Prolistat should be administered with due caution in patients with active peptic ulcer disease, chronically treated psychiatric/ neurologic disorders, symptomatic cholelithiasis, postsurgical adhesions, bulimia or laxative abuse and patients with significant cardiac, renal, hepatic, GI or endocrine disorders.
Use in patients with vitamin deficiency. There are no clinical data on the use of orlistat in patients with vitamin deficiency.
Treatment with prolistat may potentially impair the absorption of fat-soluble vitamins (A, D, E and K). Prolistat should be administered with due caution in those patients who have a deficiency of fat soluble vitamins (A, D, E and K). Patients taking orlistat showed a greater reduction in vitamins D, E and beta-carotene levels compared to placebo during two or more consecutive visits in studies of 1-2 years duration. These patients did not receive prior vitamin supplementation. In order to ensure adequate nutrition, the use of a multivitamin supplement should be considered.
In a 4 year study, vitamin supplements were administered to patients who had low baseline vitamin values, or who experienced a decrease in their vitamin values during treatment. Over the course of the study treatment with orlistat resulted in a decrease in the levels of fat soluble vitamins A, D, E and K. More orlistat than placebo treated patients had falls in vitamins A and E. However in both orlistat and placebo treatment groups the mean plasma levels of fat soluble vitamins remained within the normal reference ranges at all times during the study.
Use in hepatic impairment. Clinical investigations in patients with hepatic impairment have not been undertaken.
Use in renal impairment. The use of orlistat may be associated with hyperoxaluria and oxalate nephropathy leading sometimes to renal failure. This risk is increased in patients with underlying chronic kidney disease and/or volume depletion (see Section 4.8 Adverse Effects (Undesirable Effects)).
Prolistat should be administered with due caution in obese patients with nephrolithiasis (renal stones) due to trends showing increased oxalate excretion in both healthy and obese subjects.
Use in the elderly. Clinical investigations in geriatric patients have not been undertaken.
Paediatric use. The safety and efficacy of orlistat in children have not been established.
Effects on laboratory tests. Coagulation parameters, such as international normalised ratio (INR) values, should be monitored in patients treated with concomitant oral anticoagulants.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Alcohol. In a multiple dose study in 30 normal weight subjects, coadministration of orlistat and 40 grams of alcohol (e.g. approximately 3 glasses of wine) did not result in alteration of alcohol pharmacokinetics, orlistat pharmacodynamics (faecal fat excretion) and systemic exposure to orlistat.
Oral contraceptives (various ethinyloestradiol and gestagen combinations commercially available). In 20 normal weight female subjects, the treatment of orlistat 120 mg three times daily for 23 days resulted in no changes in the ovulation suppressing action of oral contraceptives.
However, it should be borne in mind that orlistat-induced bowel irregularities may impair the efficacy of oral contraceptives. An additional contraceptive method should therefore be used, particularly in the event of diarrhoea.
Nifedipine (extended release tablets). No interactions based on specific drug-drug interaction studies with nifedipine gastrointestinal therapeutic system (GITS) and nifedipine slow release have been observed. In 17 normal weight subjects receiving orlistat 120 mg tid for 6 days, orlistat did not alter the bioavailability of nifedipine extended release tablets.
Ciclosporin. A reduction in ciclosporin plasma levels has been observed when orlistat is coadministered (see Section 4.4 Special Warnings and Precautions for Use). Therefore, it is recommended to monitor more frequently than usual the ciclosporin plasma levels when orlistat is coadministered (see Section 4.4 Special Warnings and Precautions for Use).
Anticoagulants. Coagulation parameters, including international normalised ratio (INR), should be monitored in patients treated with oral anticoagulants given concomitantly with orlistat.
Amiodarone. In a pharmacokinetic study, oral administration of amiodarone during orlistat treatment demonstrated a 25-30% reduction in the systemic exposure to amiodarone and desethylamiodarone. The pharmacokinetics of amiodarone are complex. The effect of commencing orlistat treatment in patients on stable amiodarone therapy has not been studied, but reduced systemic exposure to amiodarone and desethylamiodarone is possible, especially after chronic dosage with orlistat. A reduced therapeutic effect of amiodarone is possible.
In patients receiving concomitant amiodarone treatment, reinforcement of clinical and ECG monitoring is warranted.
Antiepileptic medicines. Convulsions have been reported in patients treated concomitantly with orlistat and antiepileptic medicines. A causal relationship has not been established; however, patients should be monitored for possible changes in the frequency and/or severity of convulsions.
No interactions based on specific drug-drug-interaction studies with amitriptyline, atorvastatin, biguanides, fibrates, fluoxetine, losartan, phentermine, pravastatin, or sibutramine have been observed.
Rare occurrence of hypothyroidism and/or reduced control of hypothyroidism may occur. The mechanism, although not proven, may involve a decreased absorption of iodine salts and/or levothyroxine.
There are some case reports of reduced efficacy of antiretroviral HIV medicines, antidepressants, antipsychotics (including lithium) and benzodiazepines coincidental to the initiation of orlistat treatment in previously well-controlled patients. Therefore, orlistat treatment should only be initiated after careful consideration of the possible impact in these patients.
Metabolic agents. Weight loss induced by orlistat is accompanied by improved metabolic control in type 2 diabetes, which might allow or require reduction in the dose of hypoglycaemic medication.
Glibenclamide. In 12 normal weight subjects receiving orlistat 80 mg three times daily for 4 1/3 days, orlistat did not alter the pharmacokinetics or pharmacodynamics (blood glucose lowering) of glibenclamide.
Acarbose. No studies evaluating any possible pharmacokinetic/ pharmacodynamic interactions between orlistat and the α-glucosidase inhibitor acarbose have been conducted, therefore concomitant administration with Prolistat is not recommended.
In the absence of interaction studies, the concomitant administration of Prolistat with other antiobesity agents is not recommended.
Narrow therapeutic index medicines. Drug interaction studies were performed with orlistat and a number of medicines with a narrow therapeutic index. Orlistat had no inhibitory effects on pharmacokinetic or pharmacodynamic parameters of the following medicines.
Phenytoin. In 12 healthy normal weight subjects receiving orlistat 120 mg three times daily for 6 days, orlistat did not alter the pharmacokinetics of a 300 mg dose of phenytoin.
Warfarin. In 12 normal weight subjects, administration of orlistat (120 mg three times daily for 16 days) did not result in any change in either warfarin pharmacokinetics (both R and S-enantiomers) or pharmacodynamics (prothrombin time and serum factor VII). Although undercarboxylated osteocalcin, a marker of vitamin K nutritional status, was unaltered with Prolistat administration, vitamin K levels tended to decline in subjects taking orlistat. Therefore, as vitamin K absorption may be decreased with orlistat, patients on chronic stable doses of warfarin who are prescribed Prolistat should be monitored closely for changes in coagulation parameters.
Digoxin. In 12 normal weight subjects receiving orlistat 120 mg three times daily for 6 days, orlistat did not alter the pharmacokinetics of a single dose of digoxin.
Fat soluble vitamin supplements and analogues. Treatment with orlistat may potentially impair the absorption of fat-soluble vitamins (A, D, E and K). During clinical studies of orlistat there were decreases in levels of some fat soluble vitamins and analogues.
Furthermore, the absorption of supplementary doses of vitamins A, D and E and beta-carotene is significantly reduced with concomitant orlistat administration.
If a multivitamin supplement is required, this should be taken at least 2 hours before or after the dose of orlistat or at bedtime.
Other concomitant medications. During clinical studies, orlistat was administered with a wide range of commonly prescribed medicines without evidence of clinically significant interactions.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Fertility and reproductive performance were not affected in a study in rats given oral doses of up to 400 mg/kg/day which corresponds to a systemic exposure, in terms of plasma Cmax for parent drug, at least 95 times higher than that in women at the recommended dose.
Use in pregnancy. (Category B1)
Orlistat was not embryotoxic or teratogenic in rats or rabbits at oral doses of up to 800 mg/kg/day, associated with systemic exposures (in terms of plasma Cmax for parent drug) of 100 times (rat) and 3 times (rabbit) than in humans at the recommended dose. Administration of orlistat to rats from late gestation to weaning at oral doses up to 400 mg/kg/day, associated with systemic exposures (plasma Cmax for parent drug) of 50 times that in humans at the recommended dose, did not affect pup survival or development. However, the safety of orlistat has not been established in pregnant women, and because animal reproduction studies are not always predictive of human response, orlistat is contraindicated during pregnancy.
Use in lactation. The secretion of orlistat in human breast milk has not been investigated. Prolistat is contraindicated during breastfeeding.
4.7 Effects on Ability to Drive and Use Machines
No effects on the ability to drive and to use machines have been reported by patients taking orlistat.
4.8 Adverse Effects (Undesirable Effects)
Gastrointestinal symptoms were the most commonly observed adverse events associated with orlistat, and were related to its mechanism of action. There was no difference to placebo in the occurrence and intensity of adverse reactions in other body systems.
The majority of GI symptoms were mild and transient. Commonly or very commonly observed adverse events that were considered by the investigator to be possibly or probably related to study medication were oily spotting, flatus with discharge, faecal urgency, fatty/ oily stool, flatulence, abdominal pain, oily evacuation, liquid stools, increased defecation and faecal incontinence.
The incidence of these increases the higher the fat content of the diet. Patients should be counselled as to the possibility of gastrointestinal effects occurring and how best to handle them such as reinforcing the diet, particularly the percentage of fat it contains. Consumption of a diet low in fat will decrease the likelihood of experiencing adverse gastrointestinal events and this may help patients to monitor and regulate their fat intake.
Very commonly observed adverse reactions were generally mild and transient. Events occurred early in treatment (within 3 months) and most patients experienced only one episode. Only 3% of patients experienced more than two episodes of any one adverse event.
During the first year of controlled clinical trials, 8.8% of patients treated with orlistat discontinued treatment due to adverse events, compared to 4.9% of placebo treated patients. During the second year of controlled clinical trials, 3.6% of patients treated with 120 mg orlistat for both years (n = 613) discontinued treatment due to adverse events, compared to 2.5% of patients treated with placebo for both years (n = 524). In both periods, the most common reasons for discontinuation from the orlistat groups were gastrointestinal adverse events.
In a 4 year clinical trial, the general pattern of adverse event distribution was similar to that reported for the 1 and 2 year studies.
Other adverse events observed in 1 and 2 year placebo controlled clinical trials. Events listed below are classified within body system categories possibly or probably related to study medication according to the investigator, and enumerated in order of decreasing frequency using the following definitions. All events included occur at an equal or greater frequency in the patients treated with orlistat (n = 2847) compared to placebo (n = 1741).
Common: ≥ 1/100 and < 1/10 patients (1 to < 10%), uncommon: ≥ 1/1000 and < 1/100 patients (0.1 to < 1%), rare: ≥ 1/10,000 and < 1/1000 patients (0.01 to < 0.1%), very rare: 1/100,000 and < 1/10,000 patients (0.001 to < 0.01%).
Gastrointestinal disorders. Common: soft stools, nausea, dyspepsia.
Uncommon: haemorrhoids, abdominal discomfort, discoloured faeces, vomiting, infectious diarrhoea, borborygmus, eructation, bloodstained faeces, stomach upset, anal irritation, abdominal distention, rectal haemorrhage, anal burning, abdominal fullness, dry mouth, diverticulitis, unpleasant smelling faeces, enteritis, rectal pain, gastritis, colic, painful defecation, tooth disorder, gingival disorder.
Skin and subcutaneous tissue disorders. Uncommon: xeroderma, rash, pruritus ani, nail disorder, abnormal hair texture, pruritus, eczema, acne, increased sweating.
Nervous system disorders. Common: headache.
Uncommon: dizziness, vertigo, paraesthesia.
General disorders and administration site conditions. Common: asthenia.
Uncommon: oedema, insomnia, influenza syndrome.
Musculoskeletal and connective tissue disorders. Uncommon: leg cramps, back pain, myalgia, pain in ribs.
Metabolism and nutrition disorders. Uncommon: hypoglycaemia, increased appetite, thirst.
Hepatobiliary disorders. Uncommon: cholelithiasis, biliary colic, cholecystitis.
Infections and infestations. Uncommon: upper respiratory tract infection, lower respiratory tract infection, influenza, urinary tract infection.
Psychiatric disorders. Uncommon: depression, anxiety.
Cardiac disorders. Uncommon: chest pain.
Two cases of each of the following adverse events have been reported during clinical trials of orlistat considered by the investigator to be possibly or probably related to study medication: gastrointestinal disorder, solid stools, lower abdominal pain, epigastric pain not food related, hard stools, diverticulum caecum, papular rash, brittle nails, loss of appetite, fever, fatigue, malaise, muscle cramps, decreased appetite, hypokalaemia, avitaminosis, sinusitis, pharyngitis, unpleasant smelling urine, polyuria, dysmenorrhoea, amenorrhoea, menstrual irregularity, palpitation, blurred vision, dermal bleeding, bitter taste, tumour.
Post-marketing experience. Gastrointestinal disorders. Very rare: pancreatitis.
Immune system disorders. Rare: hypersensitivity reactions including urticaria, pruritus, rash, angioedema, bronchospasm and anaphylaxis.
Skin and subcutaneous tissue disorders. Very rare: cases of bullous eruptions have been reported during post-marketing experience.
Investigations. Reports of decreased prothrombin, increased INR and unbalanced anticoagulant treatment resulting in change to haemostatic parameters have been reported in patients treated concomitantly with orlistat and anticoagulants during post-marketing experience (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Very rare: increases in transaminases and in alkaline phosphatase.
Hepatobiliary disorders. Very rare: hepatitis that may be serious, exceptional cases of severe liver injury, some fatal cases or cases requiring liver transplantation have been reported. No causal relationship or physiopathological mechanism between liver injury and orlistat therapy has been established.
Nervous system disorders. Convulsions have been reported in patients treated concomitantly with orlistat and anti-epileptic medicines (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Renal and urinary disorders. Cases of hyperoxaluria and oxalate nephropathy that may lead to renal failure have been reported.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdose of orlistat has not been reported. Single doses of 800 mg orlistat and multiple doses of up to 400 mg three times daily for 15 days have been studied in normal weight and obese subjects without significant adverse findings. In addition, doses of 240 mg three times daily have been administered to obese patients for 6 months without significant increase of adverse findings. Doses above the recommended dose of 120 mg three times daily have not been found to appreciably improve efficacy and may increase gastrointestinal events.
Orlistat overdose cases received during post-marketing reported either no adverse events or adverse events that are similar to those reported with recommended dose.
Should a significant overdose of Prolistat occur, it is recommended that the patient be observed for 24 hours. Based on human and animal studies, any systemic effects attributable to the lipase inhibiting properties of orlistat should be rapidly reversible.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Therapeutic/ pharmacological class of drug. Peripherally acting anti-obesity agent, ATC code A08AB01.
Orlistat is the first of a new class of antiobesity agents with a unique mode of action. Orlistat is a potent, specific and reversible long acting inhibitor of gastrointestinal lipases, which are required for the systemic absorption of dietary triglycerides.
Orlistat selectively inhibits gastrointestinal (GI) lipase activity within the GI tract and prevents the absorption of 30% of dietary fat, thus producing a weight loss effect. Orlistat acts by forming a covalent bond with the active serine site of gastric and pancreatic lipases in the lumen of the stomach and small intestine. The inactivated enzyme is thus unable to hydrolyse dietary fat, in the form of triglycerides, into absorbable free fatty acids and monoglycerides.
As undigested triglycerides are not absorbed, the resulting caloric deficit has a positive effect on weight control. No systemic absorption is required to exert its therapeutic effect.
Orlistat had no significant effects on the physiological function of the GI tract, nor did it significantly influence systemic lipase activity.
Based on faecal fat measurements, the effect of orlistat is seen as soon as 24 to 48 hours after dosing. Upon discontinuation of therapy, faecal fat content usually returns to pretreatment levels, within 48 to 72 hours.
Clinical trials. Observational epidemiologic studies have established a relationship between obesity, visceral fat and the risks for cardiovascular disease, type II diabetes, certain forms of cancer, gallstones, certain respiratory disorders and an increase in overall mortality. These studies suggest that weight loss, if maintained, may produce health benefits for obese patients who have or are at risk of developing weight related comorbidities. Since fat provides twice the number of calories per gram as carbohydrates and protein, a selective reduction of calories from the dietary fat intake is a more efficient way of producing weight loss. The long-term effects of orlistat on morbidity and mortality have not been established.
The effects of orlistat on weight loss, weight maintenance, weight regain and on a number of comorbidities were assessed in the 4 year XENDOS study and in seven multicentre phase III double blind, placebo controlled, parallel group studies of 1 to 2 years duration. During the first year of therapy, the studies of 2 year duration assessed weight loss and weight maintenance. During the second year of therapy, some studies assessed continued weight loss and weight maintenance and others assessed the prevention of weight regain. These studies included over 2800 patients treated with orlistat and 1400 patients treated with placebo. The majority of studies had inclusion criteria of a body mass index (BMI) of 28-43 and age greater than 18 years. Orlistat also demonstrated beneficial effects in those patients who were diet resistant. In the XENDOS study, which included 3304 patients, the time to onset of noninsulin dependent diabetes mellitus was assessed in addition to long-term weight management.
During the weight loss and weight maintenance period, a well balanced, mildly hypocaloric diet that provided 30% of calories from fat was recommended to all patients.
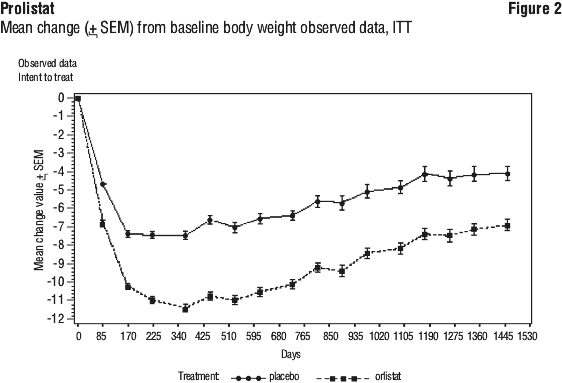
Orlistat has been shown to be safe and efficacious for long-term treatment for up to 4 years. Possible weight regain and/or attenuation of weight loss may occur after 1 year (see Figure 2).
Patients who participated in the 4 year XENDOS trial were aged between 30-60 years at enrolment.
The weight loss achieved with orlistat was associated with significant improvement of risk factors i.e. improved lipid profile (LDL/HDL ratio), reduced total cholesterol, reduced LDL cholesterol, reduced systolic and diastolic blood pressure, reduced fasting glucose and insulin levels and a reduction in waist circumference, which is associated with reduced visceral adiposity. Improved cardiovascular risk status and improvements in glycaemic control, as shown by reductions in the number of diabetic or impaired patients upon glucose tolerance testing, were determined and are in accordance with the reduction in these risk factors.
Orlistat produced weight loss greater than diet alone only when used in conjunction with a hypocaloric diet.
One year results. Weight loss, weight maintenance, risk factors and quality of life. Weight loss was observed within 2 weeks of initiation of therapy and continued for 6 to 12 months. Weight loss was evident even in those patients where diet alone had failed to induce a significant weight loss. The weight loss was maintained with continued therapy.
During the weight loss and weight maintenance period, a well balanced mildly hypocaloric diet that provided 30% calories from fat was recommended. The diet was calculated using initial body weight to provide a caloric deficit of 2.1 to 3.3 kilojoules (500 to 800 calories) per day, which represents an average caloric decrease of 20%. In addition, all patients were offered nutritional counselling.
The percentages of patients achieving a > 5% and > 10% weight loss after 1 year in the largest multicentre study for the intent to treat population are shown in Table 2.

Subpopulations with fasting insulin ≥ 120 picomol/L or LDL cholesterol ≥ 3.362 mmol/L had clinically and statistically significant improvements in these risk factors compared to placebo at the end of 1 year of treatment.
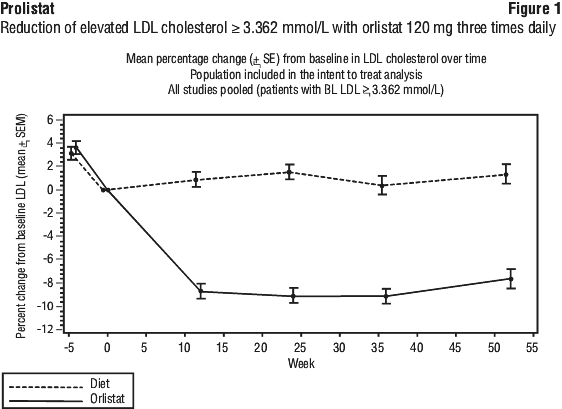
In addition, anthropometric measurements, including waist circumference and measurements of body composition, showed significant decreases in body fat including a decrease of up to 30% in visceral adipose tissue. A statistically significant difference in the satisfaction with treatment aspect of the quality of life questionnaire was observed over 1 year in favour of orlistat compared to placebo, although both groups showed worsening.
Two year results. Long-term weight control, risk factors and quality of life. Orlistat was shown to be more effective than placebo in long-term weight control in four large, multicentre, 2 year double blind, placebo controlled studies. At the end of year one the patients' diets were reviewed and changed where necessary. The diet prescribed in the second year was designed for weight maintenance rather than to produce additional weight loss.
Fifty two percent (52%) of all patients who were treated with 120 mg three times daily of orlistat and completed 2 years of the same therapy had > 5% weight loss. The weight loss advantage between orlistat 120 mg three times daily and diet alone treatment groups was the same after 2 years as for 1 year, indicating that the pharmacologic advantage of orlistat was maintained over 2 years. In the same study cited in the one year results, the percentages of patients achieving a > 5% and > 10% weight loss after 2 years are shown in Table 3.

In addition, in patients treated with orlistat, anthropometric measurements, including waist circumference and measurements of body composition, showed significant decreases in body fat. A statistically significant difference in quality of life (overweight distress and satisfaction with treatment) was observed over 2 years in favour of orlistat compared to diet alone.
Ancillary studies conducted showed that the majority of weight loss was loss in fat mass, which consistently accounted for more than 75% of total weight lost.
Prevention of weight regain. Three separate studies evaluated the effect of orlistat on the prevention of weight regain in patients who previously lost at least 5% of their initial body weight.
In two of the studies, by the end of the second year the patients receiving dietary and counselling treatment alone regained a mean of 56% of their lost weight after 1 year, while patients treated with orlistat regained a mean of 31%. The third study was conducted to evaluate the effect of orlistat on prevention of weight regain in patients who lost 8% or more of their initial body weight with diet alone. There was significantly less weight regain in patients treated with orlistat than with diet alone.
Patients receiving dietary and counselling treatment alone regained a mean of 59% of their lost weight, while patients treated with orlistat regained a mean of 33%.
For all three studies, approximately one-half of the patients treated with orlistat regained no more than 25% of their lost weight. For all three studies, approximately one-quarter of patients either did not regain any weight at all or continued to lose weight.
Four year results. Long-term weight control and risk factors. In the XENDOS study (a 4 year multicentre, randomised, double blind, parallel group, placebo controlled trial), the effects of orlistat treatment on long-term weight management and progression to noninsulin dependent diabetes mellitus (type 2 diabetes) in obese patients (BMI ≥ 30 kg/m2 in males and females) were compared to placebo in 3304 obese patients who had either normal or impaired glucose tolerance at baseline. Thirty four percent of the 1655 patients who were randomised to the placebo group and 52% of the 1649 patients who were randomised to the orlistat group completed the 4 year study. Patients were aged between 30-60 years at the time of enrolment.
Orlistat was shown to be more effective than placebo in long-term weight control. Results showed that 73% of the orlistat treated patients lost ≥ 5% of baseline body weight after 1 year of treatment compared with 45% of the placebo treated patients (p < 0.001). In addition, 41% of the orlistat treated patients lost ≥ 10% of body weight after 1 year compared with 21% of the placebo treated patients (p < 0.001). Mean change in body weight was -10.56 kg for the orlistat treatment group compared to -6.19 kg for the placebo treatment group at the end of the first year of treatment (p < 0.001). After 4 years of treatment, 44.8% and 21% of the orlistat treated patients lost ≥ 5% and ≥ 10% of body weight compared to 28.0% and 10% of the placebo treated patients, respectively (p < 0.001). Mean change in body weight after 4 years of treatment was -5.75 kg for the orlistat treatment group compared to -3.03 kg for the placebo treatment group (p < 0.001).
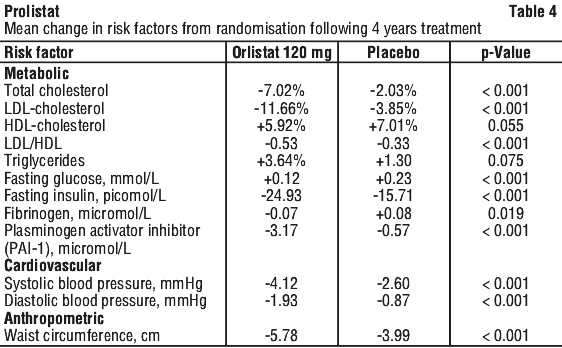
Throughout the entire four year treatment period, orlistat treated compared to placebo treated patients had statistically significant (p < 0.001) reductions in several metabolic risk factors in addition to BMI. These included total cholesterol, LDL cholesterol, LDL: HDL ratio, fasting insulin, fasting glucose, plasminogen activator inhibitor, and waist circumference. Statistically significant improvements over placebo in systolic and diastolic blood pressure were also observed. The relative changes in risk factors associated with obesity following 4 years of therapy are summarised in Table 4.
Glucose tolerance in obese patients. Two year studies that included oral glucose tolerance tests were conducted in obese patients whose baseline oral glucose tolerance test (OGTT) status was either normal, impaired or diabetic. The baseline OGTT status improved in those patients treated with orlistat greater than those on placebo.
The progression from normal at baseline to diabetic status in the group treated with orlistat was 0.0%, as compared to 1.3% of placebo patients. Orlistat prevented or reversed the progression from normal to diabetes. The progression from impaired status at baseline (and thus at greatest risk for developing diabetes) to diabetic status decreased in those treated with orlistat, whose normalisation of glucose status was markedly greater (see Table 4).
In patients found to be diabetic at baseline, the glucose status of patients treated with orlistat improved more than placebo. For all patients, the status at baseline and the change over 2 years of treatment are given in Table 5.
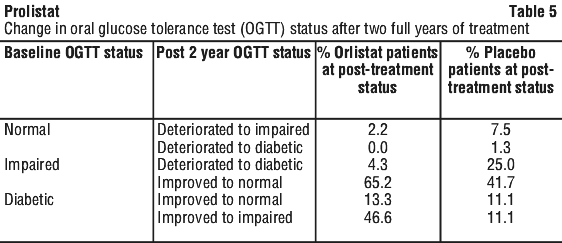
For patients with impaired glucose tolerance (IGT) at baseline, the difference in the cumulative incidence rate was highly significant (p < 0.01). Over the 4 year treatment period, the relative risk of developing noninsulin dependent diabetes mellitus was reduced by 44.9% in the orlistat group compared to the placebo group.
The effect of orlistat in delaying the onset of noninsulin dependent diabetes mellitus is as a consequence of the weight loss due to orlistat.
Treatment in the XENDOS study consisted of orlistat or placebo plus dietary and lifestyle modifications.
Short-term studies. In an 8 week study, orlistat produced a 5% greater decrease in total and LDL cholesterol and a rapid improvement of the LDL/HDL ratio compared to placebo when administered to nonobese patients with moderate familial hypercholesterolemia (LDL cholesterol ≥ 4.2 mmol/L). The patients were on a weight maintaining, lipid lowering diet for 6 weeks prior to treatment with orlistat or placebo. A subgroup of patients with LDL cholesterol ≥ 4.9 mmol/L had a 15% greater decrease in LDL cholesterol compared to placebo following treatment with orlistat. These improvements were independent of weight loss.
In several studies of 6 weeks duration, the effects of therapeutic doses of orlistat on gastrointestinal and systemic physiological processes were assessed in normal weight subjects. There were no clinically significant changes observed in gall bladder motility, bile composition and lithogenicity or colonic cell proliferation rate, and no clinically significant reduction of gastric emptying time and gastric acidity. In addition, no effect on plasma triglyceride metabolism, systemic lipases, plasma and urinary minerals or electrolytes has been observed with the administration of orlistat in these studies.
Obese adolescents. Only limited data on the safety and efficacy of orlistat in adolescents is available. One clinical trial showed that obese adolescents (12-16 years at screening) treated with orlistat for one year had a decreased BMI, while those in the placebo group had an increased BMI. The magnitude of the effect seen with orlistat on adolescents in this study was substantially less than that seen in adults in other studies. The adverse events profile was generally similar to that observed in adults (see Section 4.8 Adverse Effects (Undesirable Effects)). However, there was an unexplained increase in the incidence of bone fractures (6% versus 2.8% in the orlistat and placebo groups, respectively).
5.2 Pharmacokinetic Properties
Absorption. Studies in normal weight and obese volunteers have shown that the extent of absorption of orlistat was minimal. Plasma concentrations of intact orlistat were nonmeasurable (< 5 nanogram/mL) eight hours following oral administration of a single dose of 360 mg orlistat.
In general, after treatment for up to two years at therapeutic doses, detection of intact orlistat in plasma was sporadic and concentrations were extremely low (< 10 nanogram/mL or 0.02 microM), without evidence of accumulation, and consistent with negligible absorption.
Distribution. The volume of distribution cannot be determined because the drug is minimally absorbed. In vitro orlistat is > 99% bound to plasma proteins (lipoproteins and albumin were the major binding proteins). Orlistat minimally partitions into erythrocytes.
Metabolism. Based on animal data, it is likely that the metabolism of orlistat occurs mainly within the gastrointestinal wall. Based on a study in obese patients, of the minute fraction of the dose that was absorbed systemically, two major metabolites, M1 (4 member lactone ring hydrolysed) and M3 (M1 with N-formyl leucine moiety cleaved), accounted for approximately 42% of the total plasma concentration. Concentrations of these metabolites were low, averaging 26 nanogram/mL and 108 nanogram/mL, respectively. In view of the extremely weak inhibitory effect on systemic lipases (1000 and 2500-fold lower than orlistat, respectively) and low plasma concentrations, these metabolites are considered to be pharmacologically inconsequential.
Excretion. Studies in normal weight and obese subjects have shown that faecal excretion of the unabsorbed drug was the major route of elimination. This is consistent with the minimal absorption and gastrointestinal site of action of orlistat. Approximately 97% of the administered dose was excreted in faeces and 83% of the total dose excreted was unchanged orlistat.
The cumulative renal excretion of total orlistat related materials was < 2% of the given dose. The time to reach complete excretion (faecal plus urinary) was 3 to 5 days. The disposition of orlistat appeared to be similar between normal weight and obese volunteers. Orlistat and the metabolites M1 and M3 are subject to biliary excretion.
Long-term administration. Evidence from 5 phase III studies demonstrated an extremely low degree of systemic exposure to orlistat and a lack of accumulation in plasma after long-term treatment for up to 2 years.
Special populations. As orlistat is minimally absorbed and has a nonsystemic mode of action, studies in special populations (geriatric, paediatric, different races, and patients with renal and hepatic insufficiency) were not conducted.
5.3 Preclinical Safety Data
Preclinical data reveal no special hazard for humans based on conventional studies of safety pharmacology, repeated dose toxicity, genotoxicity, carcinogenic potential, and toxicity to reproduction.
In animal reproductive studies, no teratogenic effect was observed. In the absence of a teratogenic effect in animals, no malformative effect is expected in man.
Genotoxicity. Orlistat has no detectable genotoxic activity as determined by a bacterial reverse mutation assay, a mammalian forward mutation assay (V79/ HGPRT), an in vitro clastogenesis assay in peripheral human lymphocytes, an unscheduled DNA synthesis assay (UDS) in rat hepatocytes in culture, and an in vivo mouse micronucleus test.
Carcinogenicity. Carcinogenicity studies in rats and mice have not shown a carcinogenic potential for orlistat at oral doses up to 1000 mg/kg/day and 1500 mg/kg/day, respectively. Systemic exposure in these studies, in terms of the plasma Cmax for parent drug, was at least 5 times (mouse) and 270 times (rat) that in humans at the recommended dose. There was a decreased incidence of mammary fibroadenoma in female rats in the high dose group.
6 Pharmaceutical Particulars
6.1 List of Excipients
Prolistat contains the inactive ingredients: microcrystalline cellulose, sodium starch glycollate type A, hydrophobic colloidal silica anhydrous and sodium lauryl sulfate. Each capsule shell contains gelatin, indigo carmine and titanium dioxide.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Store in original container in order to protect from light and moisture.
6.5 Nature and Contents of Container
Available in blister packs of 14, 42 or 84 capsules.
Not all presentations may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Orlistat is a white to off white crystalline powder. Orlistat is practically insoluble in water, freely soluble in chloroform and very soluble in methanol and ethanol. Orlistat is not capable of ionising within the physiological pH range.
Chemical structure. The chemical name for orlistat is (S)-2-formylamino-4-methyl-pentanoic acid (S)-1-[[(2S, 3S)-3-hexyl-4-oxo-2-oxetanyl] methyl]-dodecyl ester.
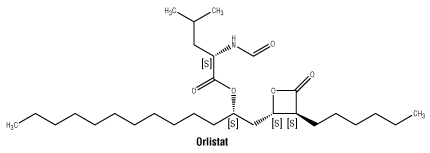
CAS number. 96829-58-2.
7 Medicine Schedule (Poisons Standard)
Schedule 3 - Pharmacist Only Medicine.
Date of First Approval
29 January 2016
Date of Revision
06 December 2021
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.