PYtest
Brand Information
| Brand name | PYtest |
| Active ingredient | Urea (14C) |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the PYtest.
Summary CMI
PYTEST®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using PYTEST?
The PYtest capsule contains the active ingredient Urea C14. The PYtest capsule is used to detect gastric urease as an aid in the diagnosis of Helicobacter pylori infection in the stomach.
For more information, see Section 1. Why am I using PYTEST? in the full CMI.
2. What should I know before I use PYTEST?
Do not use if you have ever had an allergic reaction to the PYtest capsule or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use PYTEST? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with the PYtest and affect how it works and produce false results.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use PYTEST?
- The PYtest capsule will be administered under the supervision of a doctor, pathologist or healthcare professional who will instruct you on how to use the PYtest capsule
- The PYtest capsule must be taken on an empty stomach, therefore you must not eat or drink anything (including water) for at least 4 hours prior to the test.
- The dosage for adults is one capsule, swallowed whole with 20 mL water initally. Do not chew the capsule.
More instructions can be found in Section 4. How do I use PYTEST? in the full CMI.
5. What should I know while using PYTEST?
| Things you should do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using PYTEST? in the full CMI.
6. Are there any side effects?
There have been no reported side effects in patients using the PYtest capsule. If you notice anything that may be making you feel unwell tell your doctor, pathologist pharmacist or healthcare professional.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
PYTEST®
Active ingredient(s): Urea C14
Consumer Medicine Information (CMI)
This leaflet provides important information about using the PYtest. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using PYtest.
Where to find information in this leaflet:
1. Why am I using PYTEST?
2. What should I know before I use PYTEST?
3. What if I am taking other medicines?
4. How do I use PYTEST?
5. What should I know while using PYtest?
6. Are there any side effects?
7. Product details
1. Why am I using PYTEST?
PYtest capsule contains the active ingredient Urea C14. The PYtest capsule is a gelatin capsule which contains C14 labelled urea adsorbed onto sugar spheres and coloured yellow with a dye called fluorescein sodium.
PYtest is used to detect gastric urease as an aid in the diagnosis of Helicobacter pylori (H. pylori) infection in the human stomach.
The urease enzyme is not present in human cells, so the presence of urease in the stomach is evidence that bacteria are present. The presence of urease is not specific for H. pylori, but other bacteria are not usually found in the stomach.
The presence of H.pylori infection of the stomach has been linked with inflammation of the stomach, which can give rise to stomach ulcers and even cancer of the stomach..
2. What should I know before I use PYTEST?
Warnings
Do not use PYTEST if:
- you are allergic to urea C14, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- take any medicines for any other condition, particularly in the last 4 weeks.
- have taken any medication for stomach problems or reflux in the past 7 days.
- have achlorhydria which is a condition where your stomach does not produce hydrochloric acid.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
PYtest has not been studied in children.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with PYtest and affect how it works and produce false results.
Tell your doctor is you have taken any antibiotics or medicines containing bismuth in the past 4 weeks before taking the test. The test will need to be postponed until you have been off these medicines for four weeks.
Tell your doctor if you have been taking Sucralfate in the 2 weeks before the test.
Tell your doctor if you are taking a proton pump inhibitor such as Losec (Omeprzaole), Somac (Pantaprazole) or Zoton (Lansoprazole). You will need to stop taking these medicines for 1 week before the test. Consult your doctor before stopping any medicines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect PYtest.
4. How do I use PYTEST?
How much to take
- The doctor, pathologist or healthcare practitioner administering the test will explain how the PYtest capsule is administered.
- The PYtest capsule must be taken on an empty stomach, therefore you must not eat or drink anything (including water) for at least 4 hours prior to the test.
- The dosage for adults is one capsule, swallowed hole with 20 mL water. Do not chew the capsule.
- During the test, avoid handling the capsule as this may interfere with the test result.
- After a few minutes, you will be instructed to drink another 20 mL water to ensure the PYtest capsule has reached your stomach.
When to take the PYtest capsule
- The doctor, pathologist or healthcare practitioner administering the test will explain when to take the PYtest capsule.
- You should be sitting at rest when swallowing the capsule.
After you take the PYtest capsule
- After swallowing the capsule, it takes about three minutes for the capsule to dissolve in your stomach.
- If H.pylori is present in the stomach, when the 14C-urea (which is released from the capsule) comes into contact with H. pylori, it is immediately broken down into 14C-carbon dioxide and ammonia.
The carbon dioxide is carried to your lungs through the bloodstream and is breathed out. - Ten minutes after you take the capsule, a breath sample is collected in a balloon.
- The breath sample is analysed to measure the amount of the 14C-carbon dioxide breathed out.
If you use too much PYtest
The PYtest capsule is administered under the supervision of a doctor, pathologist or other healthcare professional so it is unlikely you will use too much. However, if you think that you have used too many PYtest capsules, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using PYTEST?
Tell your doctor, pathologist or healthcare professional straight away if you:
- Feel unwell or are worried about any symptoms or conditions you have.
Remind any doctor, dentist or pharmacist you visit that you have used the PYtest.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how the PYtest affects you.
Drinking alcohol
Tell your doctor if you drink alcohol.
Looking after your medicine
Generally, you will not be asked to store PYtest. However, if you are, you should follow the instructions on the carton on how to take care of the medicine properly.
PYtest should be stored it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
There have been no reported side effects in patients using the PYtest capsule.
Tell your doctor or pharmacist if you notice anything that may be making you feel unwell.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What PYtest contains
| Active ingredient (main ingredient) | Urea [C14] |
| Other ingredients (inactive ingredients) | Starch Sucrose Fluorescein sodium Capsule Shell: Empty Gelatin Capsule size 3 Natural TR-Natural TR |
| Potential allergens | None |
Do not take this medicine if you are allergic to any of these ingredients.
What PYtest looks like
PYtest capsules can be supplied in unit dose packages of 1, 10 and 100 (Aust R 491430).
Not all pack sizes are marketed.
The PYtest capsules can also be supplied as a kit which also includes the breath collection accessories.
Who distributes PYtest
Tri-Med Distributors Pty Ltd
Unit 6/15 Walters Drive,
Osborne Park,
WA 6017, Australia
Phone: +61 (08) 9388 1444
Email: info@trimed.com.au
www.trimed.com.au
This leaflet was prepared in July 2025.
Brand Information
| Brand name | PYtest |
| Active ingredient | Urea (14C) |
| Schedule | Unscheduled |
MIMS Revision Date: 01 November 2025
1 Name of Medicine
Urea [14C].
2 Qualitative and Quantitative Composition
Each clear gelatin capsule contains 1 microgram yellow beads equivalent to 37 kBq 14C.
Each PYtest Kit (C14-urea Breath Test) contains one capsule.
Excipients with known effect. Sucrose.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Clear, hard gelatin capsules containing light yellow sugar spheres.
4 Clinical Particulars
4.1 Therapeutic Indications
PYtest (C14-urea breath test) is indicated for use in the detection of gastric urease as an aid in the diagnosis of H. pylori infection in the human stomach.
4.2 Dose and Method of Administration
PYtest (C14-urea breath test) is a qualitative and non-invasive method for the diagnosis of Helicobacter pylori (H. pylori).
Dosage. Adults. One PYtest capsule.
Children. Clinical studies in children have not been conducted.
Method of administration. For oral administration.
Procedural notes. Inform the patient to fast for 4 hours prior to the test.
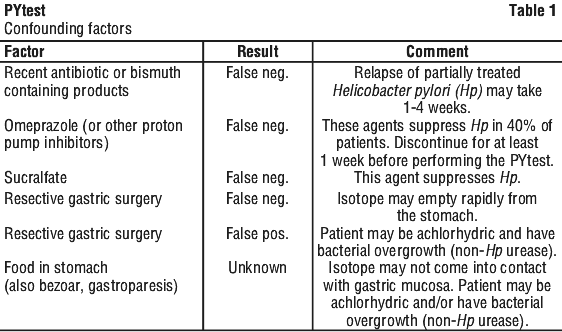
The patient should not have taken antibiotics and bismuth containing products for 1 month, proton pump inhibitors for 1 week and sucralfate for 2 weeks prior to the test.
Have patient sitting at rest while doing the test.
The capsule should not be handled directly as this may interfere with the test result.
To avoid contamination by bacteria in the mouth, the capsule should be swallowed intact. Do not chew capsule.
Also see Section 4.4 Special Warnings and Precautions for Use.
Step by step procedure for balloon. Materials needed but not provided. 1. Clock or suitable timepiece capable of measuring 10 minutes in minute intervals.
2. Water (40 mL).
Materials provided. As shown in Figure 1, the PYtest kit contains: PYtest capsule, paper cup (x2), straw, balloon, package seals, return carton.
The PYtest Kit (C14-urea breath test) is designed for use with the PYtest capsule, a gelatin capsule for oral administration containing 37 kBq of 14C labelled urea. The urea is absorbed on sugar spheres and coloured yellow with fluorescein.

2. Check that all materials are present.
3. Sit the patient down.
4. Place the capsule into one of the paper cups provided.
5. Fill the second cup 2/3 full (20 mL) with water.
6. Instruct the patient to swallow the capsule whole and to drink the water to wash the capsule down.
7. Three minutes later, fill a paper cup 2/3 full (20 mL) with water and instruct the patient to drink it. This is to ensure the capsule has been 'washed' into the stomach.
8. With the patient still sitting, wait for a further seven minutes.
9. On the neck of the balloon, separate the larger flap from the smaller flap at the black line. This can be achieved by gently squeezing the sides of the flaps together whilst pushing the flaps apart until an opening appears.
10. Insert approximately one third of the straw into the opening.
11. Instruct the patient to take a deep breath, get them to hold it for 5-10 seconds then ask them to blow slowly through the straw into the balloon until it is firm. If the balloon is not filled with the first attempt, a second breath can be used.
12. Hold the neck of the balloon at the base and remove the straw. The balloon will seal.
13. Record the finish time on the balloon.
14. Discard waste (e.g. capsule packaging, used straws) according to your facility's regulations.
Sample preparation. The breath sample is transferred to collection fluid to trap the labelled CO2. The liquid sample is then analysed in a liquid scintillation counter.
Quality control. A minimum of 1 mmol of CO2 is required to perform analysis of a breath sample. The amount of breath required to provide 1 mmol of CO2 varies depending on the amount of CO2 the patient is producing. Since a full balloon typically contains at least 1 mmol of CO2, the balloon should be completely filled.
Post treatment evaluation. A negative PYtest 28 days after completing antibiotic treatment is evidence that the infection has been eradicated.
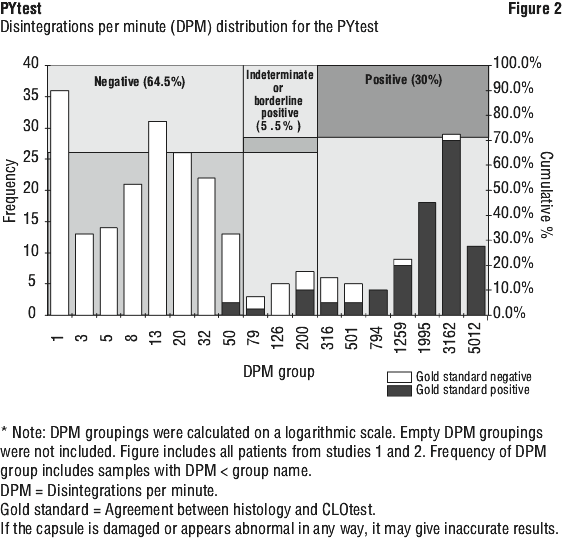
Results. Interpretation of results (10 minute sample). < 50 DPM Negative for H. pylori.
50-199 DPM Borderline positive for H. pylori.
≥ 200 DPM Positive for H. pylori.
Borderline positive results should be evaluated by repeating the PYtest or using an alternative diagnostic method. If repeat breath testing is undertaken, careful history to exclude confounding factors should be obtained. If confounding factors are present, wait an appropriate time (see Table 1) before repeating the PYtest. The cut-off point of 50 DPM was determined to be the mean +3SD of results obtained in-patients who did not have H. pylori.
4.3 Contraindications
Hypersensitivity to the active substance, or any of the excipients.
4.4 Special Warnings and Precautions for Use
After the patient ingests the urea [14C] capsule, the sample collected for test purposes is for in vitro diagnostic use only.
A false positive test could occur in patients who have achlorhydria.
Very rarely, a false positive test may occur due to urease associated with Helicobacters other than H. pylori (e.g. Helicobacter heilmannii).
Information for patients. It is necessary for the patient to fast for 4 hours before the test. The patient should not have taken antibiotics or bismuth containing products for 4 weeks, sucralfate for 2 weeks and proton pump inhibitors for 1 week prior to the test.
Do not handle the capsule directly as this may interfere with the test result. The capsule should be swallowed intact. The capsule should not be chewed.
Limitations of the test. The test has been evaluated in outpatients before elective endoscopy.
Test results should be considered together with clinical signs and patient history when diagnosing H. pylori infection.
A negative result does not completely rule out the possibility of H. pylori infection. If clinical signs and patient history suggest H. pylori infection, repeat the PYtest or use an alternative diagnostic method.
Use in renal and hepatic impairment. No data available.
Use in the elderly. No data available.
Paediatric use. Clinical studies in children have not been conducted. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Antibiotics, proton pump inhibitors, sucralfate, and bismuth containing drug preparations are known to suppress H. pylori. Ingestion of antibiotics or bismuth within 4 weeks, sucralfate within 2 weeks or proton pump inhibitors within 1 week prior to performing the test may give false negative results.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Animal reproduction studies have not been conducted with PYtest (urea [14C]). It is also not known whether PYtest can affect reproduction capacity.
Use in pregnancy. Animal reproduction studies have not been conducted with PYtest (urea [14C]). It is also not known whether PYtest can cause fetal harm when administered to a pregnant woman.
When administration of PYtest to a woman of child-bearing potential is considered, information should always be sought about pregnancy.
Use in lactation. It is not known whether this drug is excreted in human milk.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of this registration.
4.8 Adverse Effects (Undesirable Effects)
There were no adverse reactions reported in clinical trials at a rate > 1%. All adverse reactions reported were felt by the investigators to be unrelated to the PYtest.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
In the event of overdose, the patient should drink one glass of water (150 mL) every hour to hasten excretion of the radioisotope. Maximum excretion of urea is achieved at a urine output of > 2.0 mL/min.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
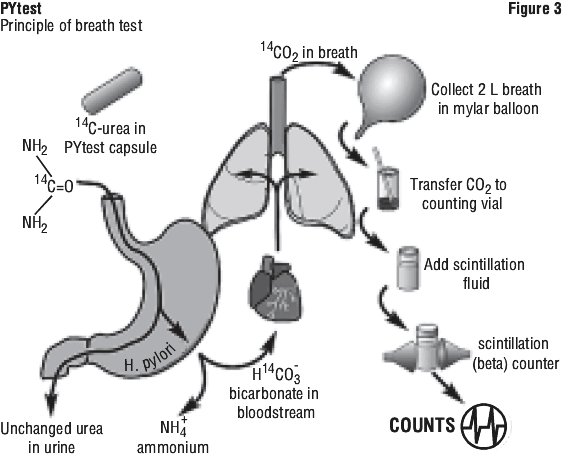
Mechanism of action. The urease enzyme is not present in mammalian cells, so the presence of urease in the stomach is evidence that bacteria are present. The presence of urease is not specific for H. pylori, but other bacteria are not usually found in the stomach. The principle of the breath test is shown in Figure 3.
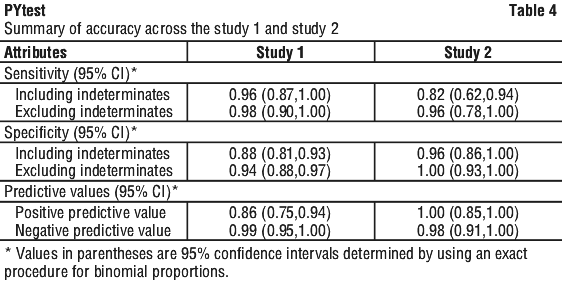
Study 1. Of 186 patients who had histopathology and Campylobacter Like Organism [(CLO)test] (80 men, 106 women), 53 were infected with H. pylori as determined by agreement between histology and CLOtest. The study results are summarized in Table 2.


5.2 Pharmacokinetic Properties
Metabolism. To detect H. pylori, urea labelled with 14C is swallowed by the patient. If gastric urease from H. pylori is present, urea is split to form CO2 and NH3 at the interface between the gastric epithelium and lumen.
Excretion. 14CO2 is absorbed into the blood and exhaled in the breath.
Following ingestion of the capsule by a patient with H. pylori, 14CO2 excretion in the breath peaks between 10 and 15 minutes and declines thereafter with a biological half-life of about 15 minutes. 14C-urea that is not hydrolysed by H. pylori is excreted in the urine with a half-life of approximately 12 hours. About 10% of the 14C remains in the body at 72 hours and is gradually excreted with a biological half-life of 40 days.
5.3 Preclinical Safety Data
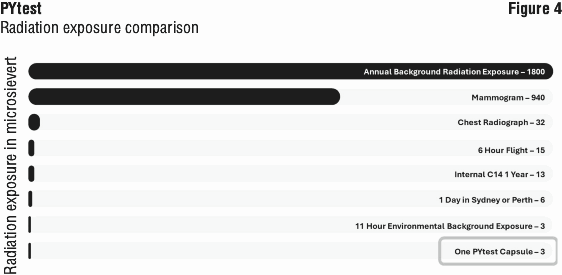
Radiation safety. To allow radiation sources to be effectively compared emissions of various origins are expressed in terms of "effective dose" (ED). The ED of 14C takes into account the fraction of isotope sequestered in each organ tissue (as recommended by the International Commission for Radiological Protection 60 [ICRP-60] which is adopted in Australia), the biological half life of the isotope in that tissue, the effective energy of the beta particles compared with standard ionising radiation (200kV x-ray source) and the administered dose. ED is expressed in Sieverts (1 Sievert [Sv]; 10-6 Sv = 1 microSv). The maximum estimated ED received from a single administration of PYtest capsule (37 kBq of [14C]-urea) is 3 microSv; Figure 4 summarizes radiation exposure from the PYtest in comparison with other common sources of radiation exposure.
The Australian population is subjected to an annual radiation dose of about 1800 microSv to 2400 microSv per caput as a result of natural environmental radiation and the in vivo presence of radionuclides such as natural 14C and 40K in the body. 2400 microSv is equivalent to the radiation dose afforded by the ingestion of 800 PYtest capsules. A single PYtest procedure is the equivalent of about 11 hours of normal and essentially inevitable radiation exposure from the environment.
Carcinogenicity. No data available.
Radiation dosimetry. Maximum effective dose (ED) (H. pylori positive): 3 microSv/37 kBq.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sugar spheres containing sucrose and starch, fluorescein sodium, capsule shell: empty gelatin capsule size 3 Natural TR-Natural TR.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
PYtest capsules and kit have a shelf life of three years.
6.4 Special Precautions for Storage
Store below 30°C in an area designated by each individual institution's regulations.
6.5 Nature and Contents of Container
PYtest capsules are supplied in unit dose packages of 1, 10* and 100*.
*Presentations are not marketed.
PYtest kit (C14-urea breath test) is supplied as a kit containing a PYtest capsule and breath collection accessories.
6.6 Special Precautions for Disposal
Because of the very low radioactivity of individual PYtest capsules, no special precautions are needed for the disposal of small numbers of capsules. Where it is necessary to dispose of large quantities of capsules, the disposal of waste should be carried out in accordance with the NHMRC Code of Practice for Disposal of Radioactive Waste by Users, (1985).
6.7 Physicochemical Properties
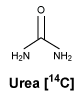
Chemical structure. Molecular formula: NH214CONH2.
Physical characteristics of 14C. Radiation emission: Beta-emission, 49 keV mean, 156 keV max, no other emissions.
External emission: No external radiation hazard. Low-energy beta emissions only. Maximum range of 0.3 mm in water.
Physical half-life: 5730 years.
7 Medicine Schedule (Poisons Standard)
Not scheduled.
Date of First Approval
16 December 1998
Date of Revision
30 September 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.