Retrieve Cream
Brand Information
| Brand name | Retrieve Cream |
| Active ingredient | Tretinoin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Retrieve Cream.
Summary CMI
Retrieve® Cream
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Retrieve?
Retrieve contains the active ingredient tretinoin. Retrieve is used for the treatment of dry, sun-damaged skin and acne.
For more information, see Section 1. Why am I using Retrieve? in the full CMI.
2. What should I know before I use Retrieve?
Do not use if you have ever had an allergic reaction to Retrieve or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Retrieve? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Retrieve and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Retrieve?
- The usual dose is to apply a thin layer to the affected skin area(s).
- Apply once a day before going to bed.
- Carefully follow the exact instructions given to you by your doctor or pharmacist.
More instructions can be found in Section 4. How do I use Retrieve? in the full CMI.
5. What should I know while using Retrieve?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Retrieve? in the full CMI.
6. Are there any side effects?
Common and less-serious side effects include redness of skin, feeling of warmth on skin, peeling of skin, slight stinging and darkening or lightening of the treated areas.
Serious side effects include excessive irritation, severe burning and the skin becomes very sensitive to light.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Retrieve® Cream
Active ingredient: Tretinoin 0.5 mg/g
Consumer Medicine Information (CMI)
This leaflet provides important information about using Retrieve. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Retrieve.
Where to find information in this leaflet:
1. Why am I using Retrieve?
2. What should I know before I use Retrieve?
3. What if I am taking other medicines?
4. How do I use Retrieve?
5. What should I know while using Retrieve?
6. Are there any side effects?
7. Product details
1. Why am I using Retrieve?
Retrieve contains the active ingredient Tretinoin.
Tretinoin is the acid form of Vitamin A.
Retrieve is used for the treatment of:
- dry, sun-damaged skin.
- acne
Retrieve works by regulating the growth and regeneration of the outer layer of the skin.
Retrieve can help to:
- make your skin less oily
- loosen blackheads and whiteheads so that they come out more easily
- stop new blackheads, whiteheads and spots from forming
- lower the number of red, inflamed acne spots.
Your doctor may have prescribed Retrieve for another reason. Ask your doctor if you have any questions why Retrieve has been prescribed for you.
2. What should I know before I use Retrieve?
Warnings
Do not use Retrieve if:
- You are allergic to tretinoin, isotretinoin, vitamin A, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty in breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
- The packaging is torn or shows signs of tampering
Check with your doctor if you have:
- any other medical conditions including:
- sunburn. Delay the use of Retrieve until the skin is fully recovered.
- inflammatory skin conditions like perioral dermatitis and rosacea which are red, sometimes pus-filled bumps on your face and neck.
- other forms of skin disease such as eczema which is characterised by inflamed, itchy, red, dry and scaly skin. Delay use of Retrieve until the eczema has cleared or use with caution. Retrieve can cause severe irritation on eczema.
- sensitive skin. - take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
- Do not use Retrieve if you are pregnant or planning a pregnancy.
- Stop using Retrieve immediately if you fall pregnant and see your doctor.
- Tell your doctor if you are breastfeeding or wish to breastfeed. It is not known whether Retrieve passes into breastmilk.
- Before using Retrieve seek advice from a doctor or pharmacist on an appropriate contraceptive method to prevent pregnancy.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Retrieve and affect how it works.
These include:
- Medicines and skin preparations used to treat acne.
- Skin preparations containing peeling agents (e.g., resorcinol, sulfur, salicylic acid, or benzoyl peroxide).
- Skin preparations containing vitamin A.
- Medicines which can increase sun sensitivity including some antibiotics, diuretics, and antipsychotic medicines
- Cosmetics, perfumes, or astringents (e.g., aftershave, skin toner) especially those containing high concentration of alcohol, spices, lime, or menthol.
- Abrasive powders, scrubbing agents, soaps, shampoos, or cleansers.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Retrieve.
4. How do I use Retrieve?
How much to use
- Apply a thin layer to the affected skin area(s). For example, a pea-sized amount should be enough to cover the entire face.
- Follow the instructions provided below and use Retrieve until your doctor tells you to stop.
When to use Retrieve
- Retrieve should be used once a day before going to bed.
How to use Retrieve
Carefully follow the exact instructions given to you by your doctor or pharmacist. These may differ from the information contained in this CMI.
- If you are using a new tube, unscrew the cap and check that the aluminium seal is intact. To break the seal, reverse the cap and pierce through.
- Wash the affected areas with a mild soap-free cleanser and lukewarm water. Pat dry.
- Apply the cream to the affected skin. Massage in gently.
- Wash hands thoroughly with water after each application.
- Only use Retrieve on intact skin. Be careful to avoid contact with your eyes, mouth, lips, angles of the nose and open wounds. Do not swallow.
For dry, sun-damaged skin, and acne treatment, the treatment program is to be started slowly. The usual dosage regimen is as follows:
- First night - Apply, leave on for 5 minutes, then wash off.
- Second night - Apply, leave on for 10 minutes, then wash off.
- Third night - Apply, leave on for 30 minutes, then wash off.
- Fourth night - Apply, leave on for one hour, then wash off.
- Fifth night - Apply, leave on for one and a half hours, then wash off.
- Sixth night - Apply, leave on for two hours, then wash off.
- If after a two-hour application, no redness or irritation has developed on the skin the following day, then the application may be left on overnight and washed off next morning.
- If there is redness or irritation, check with your doctor.
For acne treatment, it can take several weeks before you start noticing improvement of applying Retrieve to the affected area. Treatment should normally be continued for three months.
Take care not to apply too much Retrieve, especially where it could run into your eyes, the angles of your nose, any skin folds or other areas of the skin that do not require treatment.
Applying too much Retrieve or applying it too frequently will not help your acne clear up faster and it may cause redness, peeling and discomfort.
Your doctor will assess how your skin responds to treatment, and how well it tolerates the medication. If excessive skin reactions occur, your doctor may adjust the application to alternate nights or temporarily discontinue treatment.
At your next visit, your doctor will review how your skin is responding to treatment. Your doctor will decide on the best course of treatment according to your skin condition and response. This may change from time to time depending on your medical condition.
Ask your doctor or pharmacist on how to apply Retrieve if you are not sure.
If you forget to use Retrieve
Use Retrieve regularly at the same time each day. If you miss your dose at the usual time, apply the medicine when you remember, and then go back to using it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you use too much Retrieve
If you think that you have applied too much Retrieve, remove any excess cream.
Do not take this medicine by mouth. If you or someone else accidentally swallows some Retrieve, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre by calling 13 11 26 in Australia, or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Retrieve?
Things you should do
- Contact your doctor or pharmacist immediately if you notice any unusual symptoms (such as a rash) during treatment.
- Tell your doctor if, for any reason, you have not used your medicine exactly as directed.
- Protect your skin when you are in the sun. If you are outdoors, wear protective clothing and apply a broadspectrum high protection sunscreen. Reapply every two hours or more frequently when sweating, and after swimming or toweling.
Call your doctor straight away if you:
- Become pregnant while you are using Retrieve.
- Notice any excessive skin reaction and unusual symptoms (such as a rash).
Remind any doctor, dentist or pharmacist you visit that you are using Retrieve.
Things you should not do
- Do not apply to the eyes, around eyelids, mouth, lips, mucosa, or angles of the nose.
- Do not apply other skin preparations on top of the nightly application of Retrieve. Suitable sunscreens and moisturisers may be used during the day.
- Do not apply more often than or longer time than what your doctor has told you.
- Do not use Retrieve to treat any other complaint unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the similar symptoms to yours.
- Do not use any skin treatments, such as skin peels or medicated dressing on the same affected areas unless your doctor has told you to.
- Do not mix Retrieve with any other preparations. They may prevent Retrieve from working.
- Do not use on skin that is irritated or damaged such as cuts, grazes, abrasions and sunburn.
- Do not use on skin that has had cosmetic treatment such as depilation, chemical hair treatment, chemical peel, dermabrasion or laser resurfacing.
- Do not swallow this medicine.
Things to be careful of:
- Sunlight/Solarium - avoid exposure of the treated area to excessive sunlight or overuse of a sunlamp because your skin may be more prone to sunburn whilst using Retrieve. When exposed to sunlight use a high protection SPF broad-spectrum sunscreen and wear protective clothing over the treated areas.
- Strong Wind/Dry Air - These may cause skin discomfort. Apply moisturiser or emollient if this occurs. Check with your doctor or pharmacist about suitable wind protection if you notice any unusual rash or redness.
- Cosmetics - Check with your doctor or pharmacist which cosmetics or perfumed toiletries you can use. Wash any cosmetics off any affected areas before you apply Retrieve.
- Washing your face - Avoid using harsh soaps or exfoliants. Use a mild bland soap, or any suitable soap free cleansers.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place where the temperature is below 25°C, away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side affects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems.
By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Retrieve contains
| Active ingredient (main ingredient) | Tretinoin |
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not take this medicine if you are allergic to any of these ingredients.
What Retrieve looks like
Retrieve is a smooth off-white to pale yellow cream contained in an aluminium tube with an outer carton and is available in the following pack sizes:
- 5g tube
- 50g tube
(AUST R 15553).
Who distributes Retrieve
iNova Pharmaceuticals (Australia) Pty Limited
Level 10, 12 Help Street
Chatswood NSW 2067
Tel: 1800 630 056
This leaflet was prepared 29 January 2026
Brand Information
| Brand name | Retrieve Cream |
| Active ingredient | Tretinoin |
| Schedule | S4 |
MIMS Revision Date: 01 March 2026
1 Name of Medicine
Tretinoin.
2 Qualitative and Quantitative Composition
ReTrieve Cream contains tretinoin 0.05% w/w (0.5 mg/g).
Excipients with known effect. Contains hydroxybenzoates and diazolidinyl urea.
3 Pharmaceutical Form
Cream, topical.
ReTrieve is a soft, smooth off-white to pale yellow homogenous glossy cream.
4 Clinical Particulars
4.1 Therapeutic Indications
ReTrieve Cream is indicated for:
Adjunctive treatment of dry photoaged skin and related conditions.
Treatment of acne vulgaris, in particular forms where comedones, papules and pustules predominate in patients 12 years of age and older. It is not generally effective in most cases of severe pustular or nodulocystic acne.
4.2 Dose and Method of Administration
Treatment with tretinoin should be individualised according to tolerance and response. No other topical preparations should be applied over the nightly inunction, but suitable moisturisers may be used during the day.
Certain types of skin could be too sensitive to use ReTrieve Cream. Patients with very sensitive skin should consult a dermatologist before commencing treatment.
Dose. Dry photoaged skin. ReTrieve should be applied sparingly to the affected areas once daily at bedtime.
Acne vulgaris. ReTrieve should be applied sparingly to the affected areas once daily at bedtime. Therapeutic effects may not be seen until several weeks after the start of treatment. Treatment should normally be continued for three months. Treatment should not exceed three months of continuous use without careful evaluation.
Method of administration. Begin the treatment program slowly, as follows:
1. Wash the affected areas prior to any application with mild soap free cleansers and pat dry.
2. First night: apply, leave for five minutes, then wash off.
3. Second night: apply, leave for ten minutes, then wash off.
4. Third, fourth, fifth and sixth nights: increase the treatment time each night by 30 minutes until the application is left on for two hours.
5. If, after a two hour application, no redness or irritation has developed on the skin the following day, then the application may be left on overnight and washed off next morning.
6. If excessive skin reactions occur, adjust the schedule to alternate nights until the skin accommodates.
4.3 Contraindications
Pregnancy (see Section 4.6 Fertility, Pregnancy and Lactation).
Women planning a pregnancy.
Hypersensitivity to tretinoin or any of the ingredients in the formulation.
Retrieve should only be given to women of childbearing potential that are using effective contraception correctly during treatment and for 1 month after discontinuation of treatment.
4.4 Special Warnings and Precautions for Use
Identified precautions. Concomitant use with other topical preparations. Concomitant application of other topical preparations including cosmetics should be avoided because of possible incompatibility and interaction with tretinoin.
Particular caution should be exercised in the use of keratolytic agents such as sulphur, salicylic acid, benzoyl peroxide or resorcinol and chemical abrasives. If the patient has been treated with such preparations, the effect of the peeling agents must subside before any commencement of topical ReTrieve therapy.
Some medicated cleansers and scrubbing solutions have a strong drying effect. They should not be used in patients receiving tretinoin topical therapy.
In patients whose skin has been subjected to procedures such as depilation, chemical hair treatment, chemical peels, dermabrasion or laser resurfacing, the skin should be allowed to recover before application is considered.
Skin and mucous membrane irritation. Particular caution is indicated for patients with eczema, since tretinoin has been reported to cause severe irritation on eczematous skin.
Extreme weather conditions, such as strong wind or cold dry air may cause skin irritation to patients receiving tretinoin treatment.
Do not swallow and avoid contact with mucous membranes or open wounds.
ReTrieve should not be applied to the eyes, mouth, lips, mucosa, or angles of the nose.
Should any of these occur, rinse the affected areas thoroughly with water to avoid local irritation.
The hands should be washed thoroughly with water after each application.
Over enthusiastic use or too frequent application may cause redness, stinging and discomfort. If severe irritation occurs, especially in the early stage of therapy, patient should be advised to discontinue temporarily or reduce the frequency of application.
Use in patients with a history of acute eczemas, rosacea and perioral dermatitis. Tretinoin preparations have been reported to cause severe irritation of eczematous skin and should only be used with the utmost caution in patients with this condition.
Use in patients with a personal or familial history of skin cancer. Patients receiving tretinoin are more susceptible to the effect of UV irradiation (see Section 4.4 Special Warnings and Precautions for Use, Ultraviolet exposure). A 91-week dermal study in mice using topical tretinoin was associated with the development of squamous cell carcinomas and papillomas in female mice at the site of application (see Section 5.3 Preclinical Safety Data). The risks and benefits of use of ReTrieve in patients with a personal or family history of skin cancer should be carefully considered.
Ultraviolet exposure. Exposure of the treated areas to sunlight including sunlamps should be minimised during the course of topical treatment with ReTrieve.
Patients receiving tretinoin treatment are more susceptible to the effect of UV irradiation especially at the start of the therapy. Animal studies suggest that tretinoin may accelerate the tumorigenic potential of ultraviolet radiation in hairless albino mice, especially at high concentrations of the drug. Although the significance to human is unknown, patients undergoing tretinoin treatment should exercise utmost caution.
Patients with sunburn should be advised to use ReTrieve only after the skin is fully recovered.
Exposure to ultraviolet irradiation increases the intensity of inflammatory reaction. Patients receiving ReTrieve therapy should avoid exposure to artificial sunlamps or solarium.
Patients should be counselled to routinely use high SPF sunscreens as well as protective clothing while undergoing ReTrieve topical treatment, especially those individuals at risk of chronic sun exposure or having a family history of light sensitivity.
Use in hepatic impairment. No data available.
Use in renal impairment. No data available.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Concomitant use of other topical medications (especially those containing keratolytic agents such as resorcinol, sulphur, salicylic acid, benzoyl peroxide and abrasive chemicals etc.) should be avoided in patients undergoing treatment with ReTrieve because of possible interactions with tretinoin. The application of ReTrieve should only commence after the effect of the peeling agents has completely subsided (see Section 4.4 Special Warnings and Precautions for Use). Tretinoin is an unstable compound that is often incompatible with substances found in topical preparations.
Some topical products and certain cosmetics contain high concentrations of alcohol, spices, lime or menthol. They should be used with caution especially in the early phase of treatment due to stinging action of these chemicals.
ReTrieve Cream should not be administered if the patient is also taking medicines known to be photosensitisers (e.g. thiazides, tetracyclines, flluoroquinolones, phenothiazines, sulphonamides) because of the possibility of augmented phototoxicity.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category D)
There have been isolated reports of birth defects in babies born to women using topical tretinoin in pregnancy. To date, there have been no adequate and well controlled prospective studies in women using topical tretinoin in pregnancy. A retrospective cohort study of babies born to 215 women exposed to topical tretinoin during the first trimester of pregnancy found no more birth defects among these babies than those born to 430 women in the same cohort who were not similarly exposed.
Orally administered retinoids have been associated with congenital abnormalities. When used in accordance with the prescribing information, topically administered retinoids are generally assumed to result in low systemic exposure due to minimal dermal absorption. However, there could be individual factors (e.g. damaged skin barrier, excessive use) that contribute to an increased systemic exposure.
Oral tretinoin has been shown to be teratogenic in rats when given at doses of 5 mg/kg/day and fetotoxic in rats when given at doses of 2.5 mg/kg/day. Oral doses of tretinoin have caused limb defects in mice.
However, topical tretinoin has not been shown to be teratogenic in rats and rabbits when given at doses of 0.5 mg/kg/day and 1.6 mg/kg/day, respectively.
These latter changes may be considered variants of normal development and are usually corrected after weaning.
ReTrieve is contraindicated in pregnancy, or in women planning a pregnancy (see Section 4.3 Contraindications). If the product is used during pregnancy, or if the patient becomes pregnant while taking this drug, treatment should be discontinued.
Women of childbearing potential. ReTrieve should only be given to women of childbearing potential that are using effective contraception correctly during treatment and for 1 month after discontinuation of treatment. ReTrieve is contraindicated in women of childbearing potential not using an effective method of contraception properly.
Use in lactation. Safe use during lactation has not been established. It is not known whether this drug is excreted in human milk. Therefore, use during lactation is not recommended.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
ReTrieve is generally well tolerated after nightly application. Some patients may experience a transitory sensation of warmth or slight stinging after application of the drug.
If excessive reactions occur, the frequency of application may be reduced or treatment discontinued temporarily until the reactions subside. The dose and frequency may then be adjusted to a level which the patient can tolerate (see Section 4.2 Dose and Method of Administration).
Increased sensitivity to UV light may be experienced in patients undergoing treatment and appropriate measures should be taken (see Section 4.4 Special Warnings and Precautions for Use).
Dry photoaged skin. Side effects have been limited to mild irritation, evidenced by peeling and erythema, especially in the early stage of treatment.
Temporary hyperpigmentation or hypopigmentation has occurred with repeated topical application of the drug.
Contact allergy has been reported in isolated instances.
Reversible changes in liver function tests have been reported after administration of tretinoin topical therapy but do not appear to be of clinical significance. Elevated serum level of bilirubin, alkaline phosphatase, glutamic-pyruvic transaminase, glutamic oxaloacetic transaminase and increase in thymol turbidity and flocculation were observed but in all cases reported, the results reverted to normal on discontinuing treatment.
Acne vulgaris. The following safety information is sourced from an Australian tretinoin cream Product Information.
The following convention is used for the classification of the frequency of an adverse reaction and is based on the CIOMS guidelines:
Very common: ≥ 1/10; common: ≥ 1/100 to < 1/10; uncommon: ≥ 1/1000 to < 1/100; rare: ≥ 1/10000 to < 1/1000; very rare: < 1/10000; not known: (Cannot be estimated from the available data).
Clinical trial data. Skin and subcutaneous tissue disorders. Very common: application site erythema, skin exfoliation, pain of skin, application site pruritus, skin irritation, skin tenderness, skin burning sensation, application site stinging, dry skin.
Post-marketing data. Skin and subcutaneous tissue disorders. Rare: skin hyperpigmentation, skin hypopigmentation, photosensitivity reaction, application site rash, application site oedema/swelling, allergic reaction, skin atrophy.
Post-marketing data for other topical tretinoin creams has reported pyogenic granuloma-like lesions as a rare adverse event.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
No data are available on the consequences of overdosage from accidental ingestion of ReTrieve. Tretinoin is a normal metabolite of vitamin A and has similar toxicity profile. The LD50 of tretinoin in mice and rats has been found to be 4 g/kg and 2 g/kg respectively.
The concentration of tretinoin present in ReTrieve at 0.5 mg/g is unlikely to cause any symptomatic effects. Any acute toxicity arising from accidental ingestion of the preparation will be more related to the toxicity of the vehicle components. Accidental ingestion should be managed clinically.
Symptoms of acute toxicity would be of gastrointestinal disturbance.
Overdosage from excessive dermal application may produce marked erythema and skin inflammatory reactions. Should this occur, discontinue use and if necessary, apply cold compresses and/or mild emollient.
5 Pharmacological Properties
Tretinoin, being a metabolite of retinol, is both pharmacologically and structurally related to vitamin A which regulates cell growth and differentiation. It has been postulated that it acts by enhancing epithelial proliferation and accelerating epithelial differentiation.
5.1 Pharmacodynamic Properties
Mechanism of action. Tretinoin, an all trans retinoic acid, occurs in the body as a tissue metabolite of vitamin A. The precise mechanism of action of topical tretinoin has not been fully elucidated.
For treatment of acne topical tretinoin has comedolytic, anti-comedogenic and anti-inflammatory effects. Tretinoin decreases cohesiveness of follicular epithelial cells resulting in decreased microcomedone formation. Additionally, tretinoin stimulates mitotic activity and increased turnover of follicular epithelial cells, causing extrusion of the comedones. The comedolytic activity is related to a normalisation of the desquamation of the follicular epithelium. Tretinoin exerts anti-inflammatory effects via suppression of toll-like receptors (TLRs).
Clinical trials. Photoaged skin indication. No data available.
Acne vulgaris indication. The results of a number of published studies support the efficacy of tretinoin 0.05% cream in the treatment of acne vulgaris.
Published studies include, Trifu et al.1, Fatum et al.2, Cunliffe3.
Trifu et al. conducted a pilot randomised, double-blind study evaluated the efficacy and safety of cortexolone 17α-propionate (CB-03-01) 1% cream in men with mild-to-moderate facial acne vulgaris, compared with placebo and tretinoin 0.05% cream. Seventy-seven participants were treated once daily for 8 weeks (CB-03-01 n=30; tretinoin n=32; placebo n=15). The primary objective was to compare CB-03-01 with placebo for changes in total lesion count (TLC), inflammatory lesion count (ILC), acne severity index (ASI), and Investigator's Global Assessment (IGA), with secondary comparison versus tretinoin. At week 8, mean percentage reductions were 65.7% (CB-03-01), 52.5% (tretinoin), and 37.0% (placebo) for TLC; 67.3%, 50.7%, and 39.0% for ILC; and 68.4%, 53.1%, and 39.5% for ASI, respectively.
Key limitations of the study included the pilot design, small sample size, and unequal treatment groups, particularly the small placebo arm. The study enrolled only adult men and had a short treatment duration (8 weeks), restricting generalisability and assessment of longer-term outcomes.
Fatum et al. conducted a randomised, double-blind multicentre study which investigated the efficacy and tolerability of topical 0.1% motretinide cream, 0.05% tretinoin cream, and placebo cream in 167 patients with acne vulgaris treated in general practice. After 6-8 weeks, all groups showed a reduction in acne lesions, but tretinoin demonstrated a greater median decrease in total lesion count (11.3) compared to motretinide (8.1) and placebo (6.4). Tretinoin was statistically superior to both motretinide and placebo for reducing pustules and overall lesion count, while all treatments were similarly effective against comedones. Papules responded better to tretinoin than placebo.
Limitations of the study include a smaller sample size than planned, a notable dropout rate, and non-randomised dose reduction in the tretinoin group due to irritation. Local side effects such as erythema, peeling, and burning were significantly more frequent and severe with tretinoin, while motretinide and placebo had similar tolerability profiles.
A multicentre, randomised, investigator-blinded trial by Cunliffe et al. compared the efficacy and tolerability of tretinoin cream 0.05% and adapalene gel 0.1% in 409 patients aged 12-25 years with mild-to-moderate acne vulgaris over a 10-week period. Both treatment groups showed similar reductions in inflammatory lesion counts (tretinoin: 21.0 to 14.0; adapalene: median reduced from 20.5 to 13.0), non-inflammatory lesion counts (both groups reduced from approximately 49-50 to 25), and total lesion counts (both groups reduced from 73.0 to 39.0). The proportion of patients showing marked or moderate improvement, or almost total clearance, was also similar between tretinoin (57.7%) and adapalene (59.6%).
Local side effects such as erythema, dryness, desquamation, and stinging or burning sensations were reported. No serious adverse events were reported in either group. Limitations include the absence of a placebo comparator, as both groups received active treatments, and the study population was predominantly Caucasian with mild-to-moderate acne. Results were not stratified by age, sex, or acne severity, and systemic laboratory safety parameters were not assessed.
5.2 Pharmacokinetic Properties
Absorption. Topically applied tretinoin appears to be slightly absorbed from the skin.
Metabolism and excretion. Unlike retinol and its esters, it does not accumulate in the body but metabolises rapidly and excretes in the form of inactive glucuronides or oxidation products. These metabolites are mainly excreted in the faeces and some oxidised metabolites are found in the urine.
In vitro studies in human skin showed that only a small percentage of the applied dose could be detected in urine.
5.3 Preclinical Safety Data
Genotoxicity. Tretinoin was negative in assays for gene mutation in bacteria (Ames test) and mammalian cells (Chinese hamster lung cells). A two-fold increase in sister chromatid exchange (SCE) frequency was found in human diploid fibroblasts, but other chromosomal aberration assays (human lymphocytes in vitro, mouse micronucleus test in vivo) did not show a clastogenic or aneuplodogenic effect.
Carcinogenicity. In a 91-week dermal study in mice, treatment at 0.5 and 1 mg/kg for three days per week was associated with the development of squamous cell carcinomas and papillomas in females at the site of application. These skin tumours occurred in the context of severe dermal irritation; the relevance to humans is unclear. No carcinogenicity was observed at a dose of 0.025 mg/kg (less than the maximum human dose, adjusted for body surface area).
The tumourigenic potential of UV irradiation was increased with concurrent dermal exposure to tretinoin at a dose of 100 mg/kg in hairless albino mice. Although the relevance of this finding to humans is unknown, patients should minimise exposure to sunlight or artificial UV sources (also see Section 4.4 Special Warnings and Precautions for Use, Ultraviolet exposure).
6 Pharmaceutical Particulars
6.1 List of Excipients
ReTrieve Cream contains the following inactive ingredients: cetyl alcohol, diazolidinyl urea, disodium edetate, dl-alpha tocopheryl acetate, glyceryl monostearate, isopropyl palmitate, methyl hydroxybenzoate, polysorbate 60, propylene glycol, propyl hydroxybenzoate, retinol palmitate, sorbitan monostearate and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Contained in a metal (aluminium) tube with an outer carton and is available in the following pack sizes: 5 g tube, 10 g tube*, 50 g tube.
* Not currently distributed in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Tretinoin is a yellow to light orange crystalline powder. It is practically insoluble in water, sparingly soluble in methylene chloride and slightly soluble in ethanol (96 per cent).
Chemical structure.
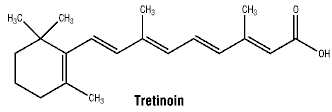
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
20 September 1991
Date of Revision
29 January 2026
Summary Table of Changes

References
1 Cortexolone 17α-propionate 1% cream, a new potent antiandrogen for topical treatment of acne vulgaris. A pilot randomized, double-blind comparative study vs. placebo and tretinoin 0.05% cream - by V. Trifu et al., British Journal of Dermatology, 2011, 165 pp177-183.
2 Topical treatment of acne vulgaris with vitamin A acid derivative motretinide (Tasmaderm), tretinoin (Airol) and placebo cream - by Bjarne Fatum, Hans-Henrik V. Hansen, Erik Mortensen, Birgit Ohrt Mikkelsen, and Kirsten Mollenbach, Weekly Journal for Medical Doctors (Ugeskrift for Laeger), issued by the Danish Medical Association, Volume 142, Issue 51, 15 December 1980, pages 3361-3442.
3 Randomised, controlled trial of the efficacy and safety of adapalene gel 0.1% and tretinoin cream 0.05% in patients with acne vulgaris by W.J. Cunliffe and colleagues, European Journal of Dermatology, Volume 12, Issue 4, July-August 2002.
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.