Rhophylac
Brand Information
| Brand name | Rhophylac |
| Active ingredient | Immunoglobulin (anti-RhD) |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Rhophylac.
What is in this leaflet
This leaflet answers some common questions about Rhophylac®. It does not contain complete information about Rhophylac®. It does not take the place of talking to your doctor.
If you have any concerns about using this medicine, ask your doctor. Follow your doctor’s advice even if it is different from what this leaflet says.
Please read this leaflet carefully and keep it for future reference.
The information in this leaflet is subject to change. Please check with your doctor whether there is any new information about this medicine that you should know since you were last treated.
What Rhophylac® is used for
What is Rhophylac®?
Rhophylac® is a ready-to-use solution for injection, which comes in a pre-filled syringe. The solution contains special proteins, isolated from human blood plasma. These proteins belong to the class of so called 'immunoglobulins', also called antibodies. The active ingredient of Rhophylac® is a specific antibody called 'anti-D (Rh) immunoglobulin'. This antibody works against Rhesus factor type D.
What is Rhesus factor type D?
Rhesus factors are special characteristics of human red blood cells. About 85% of the population carry the so called Rhesus factor type D (abbreviated 'Rh(D)'). These people are called Rh(D)-positive. People who do not carry Rhesus factor type D are called Rh(D)-negative.
What is anti-D (Rh) immunoglobulin?
Anti-D (Rh) immunoglobulin is an antibody, which works against Rhesus factor type D and is produced by the human immune system. When a Rh(D)-negative person receives Rh(D)-positive blood, her/his immune system will recognise the Rh(D)-positive red blood cells as 'foreign' to her/his body, and will attempt to destroy them. For this purpose, the immune system will build specific antibodies against Rhesus factor type D. This process is called 'immunisation' and it usually takes some time (2-3 weeks). Therefore, the Rh(D)-positive red blood cells will not be destroyed upon the first contact, and no signs or symptoms are usually seen then. But when the same Rh(D)-negative person receives Rh(D)-positive blood a second time, the antibodies will be 'ready at hand' and her/his immune system will destroy the foreign red blood cells immediately.
How Rhophylac® works
If a Rh(D)-negative person is given a sufficient amount of human anti-D (Rh) immunoglobulin, immunisation against Rhesus factor type D can be prevented. To achieve this, treatment with Rhophylac® should commence before or early enough after the first contact to Rh(D)-positive red blood cells. The anti-D (Rh) immunoglobulins contained in Rhophylac® will then destroy the foreign Rh(D)-positive red blood cells immediately. Thus, the person’s immune system will not be prompted to build-up its own antibodies.
When is Rhophylac® used
Rhophylac® is used in two distinct situations:
- You are a Rh(D)-negative pregnant woman, who carries a Rh(D)-positive baby.
In this special situation you may be immunised by red blood cells from your baby passing over into your own blood circulation. If this happens, the first baby is not usually affected and fully healthy. But in the next Rh(D)-positive baby, the mother’s antibodies would destroy the baby’s red blood cells already during pregnancy. This may lead to complications with the baby, including his/her possible death.
As a Rh(D)-negative pregnant woman, you may receive anti-D (Rh) immunoglobulins in the following situations:
- when you carry or have just delivered a Rh(D)-positive baby
- when you lose a Rh(D)-positive baby (miscarriage, threatened miscarriage or abortion)
- when your pregnancy is severely complicated (ectopic pregnancy or hydatidiform mole)
- when it is likely that your baby’s red blood cells have passed over into your own blood circulation (transplacental haemorrhage resulting from antepartum haemorrhage). This may, for example, happen when you experience vaginal bleedings during pregnancy
- when your doctor needs to perform testing methods for foetal deformities (amniocentesis, chorionic biopsy)
- when your doctor or midwife needs to try moving the baby from outside (e.g. external version of the baby or other obstetric manipulative procedures)
- when you have an accident hurting your stomach or gut (abdominal trauma). - You are a Rh(D)-negative person, who has accidentally received infusions (transfusions) of Rh(D)-positive blood (mismatched transfusion). This also applies to any blood products containing Rh(D)-positive red blood cells.
Before you are given Rhophylac®
Rhophylac® must not be used if you have a history of allergy to this product. Tell your doctor if you have allergies to any other medicines or if you have ever had an allergic reaction to an injection.
Tell your doctor also if you:
- have a blood type Rh(D)-positive
- have a blood type Rh(D)-negative but have been previously exposed to Rh(D)-positive blood
- are allergic to any of the components of the product
- have previously been advised that you have immunoglobulin A (IgA) deficiency
- are taking or using any other medicines. These include medicines bought from pharmacies, supermarkets and health food stores
- suffer from a blood disorder or blood clotting problem
- are pregnant or breast-feeding
- have had any vaccination during the last two weeks or intend to have any vaccination for the next three months
- have any other medical conditions.
Information on the starting material of Rhophylac®
Rhophylac® is made from human blood plasma (this is the liquid part of the blood).
When medicines are made from human blood or plasma, certain measures are put in place to prevent infections being passed on to patients. These include:
- careful selection of blood and plasma donors to make sure those at risk of carrying infections are excluded, and
- the testing of each donation and pools of plasma for signs of virus/infections.
Manufacturers of these products also include steps in the processing of the blood or plasma that can inactivate or remove viruses. Despite these measures, when medicines prepared from human blood or plasma are administered, the possibility of passing on infection cannot be totally excluded. This also applies to any unknown or emerging viruses and other types of infections.
Vaccines are available against some of these viruses and your doctor will be able to help you decide whether it is worthwhile having any of those vaccines.
Please discuss the risks and benefits of this product with your doctor.
The measures taken are considered effective for enveloped viruses such as human immunodeficiency virus (HIV), hepatitis B virus and hepatitis C virus, and for the non-enveloped hepatitis A and parvovirus B19 viruses.
Immunoglobulins like Rhophylac® have not been associated with hepatitis A or parvovirus B19 infections. Rhophylac® also contains antibodies against hepatitis A or B19 virus and it is assumed that these antibodies contribute to the viral safety, too.
How Rhophylac® is used
Your doctor will give you Rhophylac® as an infusion, that is, an injection given slowly into the vein or it will be injected into the muscle. Your doctor will determine the dose(s) of Rhophylac® that you are to receive and which is the appropriate route of administration. If your body mass index (BMI) is greater or equal to 30 (calculated by dividing your body mass by the square of your height), the injection of Rhophylac® into a muscle may not be fully effective. In this case, your doctor or healthcare professional should inject this medicine into a vein.
If a large volume of product is required, you may receive more than one injection.
If you are treated with Rhophylac® after a mismatched transfusion, you may receive quite a large amount of the product (up to 3000 mg, equivalent to 20 mL or 10 syringes). In this case there is an increased risk for a special complication called haemolytic reaction. This results from the intended destruction of the foreign Rh(D)-positive red blood cells. For this reason your doctor or health care professional will monitor you closely and may need to do special blood tests.
If you want further information, consult your doctor.
Side effects
Along with their intended effects, blood products occasionally cause unwanted effects, some of which are serious. Furthermore, individual patients may react differently to the same dose of the same product. This applies to Rhophylac®.
Reactions to Rhophylac® are rare in Rh(D)-negative individuals. If Rhophylac® is given into a muscle, local pain and tenderness at the injection site may be felt.
Tell your doctor as soon as possible if you notice any of the following serious side effects:
- shortness of breath
- chest pain
- skin becoming yellow
- dark urine
In rare cases this type of medicine may cause a sudden fall in blood pressure or a condition called anaphylactic shock, which is an allergic reaction that has symptoms such as low blood pressure (feeling faint) and difficulty breathing.
Contact your doctor immediately if you experience any of the mentioned effects or any other abnormal signs after treatment.
Tell your doctor if you have any of these side effects:
- headache
- skin rashes
- itching
- fever
- chills
- nausea
- vomiting
- joint pain
- dizziness
- rapid heart beat (tachycardia)
- generally feeling unwell
- feeling very tired
- paleness of skin
- abdominal pain
- allergic reactions.
Rhophylac® may interfere with some live vaccines (e.g. measles, mumps, rubella or varicella), even up to three months later. Advise your doctor if you are to receive other vaccines within three months of receiving Rhophylac® or have just received a vaccine in the last two weeks.
Other side effects not listed above may also occur in some people. For a full list and explanation of the possible side effects associated with Rhophylac® please ask your doctor.
If you want further information consult your doctor.
Overdose
The consequences of overdose are not known.
Storing Rhophylac®
Store at 2°C to 8°C (Refrigerate. Do not freeze). Protect from light.
Do not use after the expiry date shown on the label.
Further information
Rhophylac® can only be obtained on a doctor’s prescription. This leaflet does not contain the complete information about Rhophylac®. If you require further information about Rhophylac® and your treatment generally, or if you have any questions or are not sure about something in this leaflet, consult your doctor.
Product description
What it looks like
Rhophylac® is a solution for injection that is clear or opalescent. The product is supplied as a pre-filled syringe.
Ingredients
Each syringe of Rhophylac® contains a solution of gamma globulin (IgG) fraction of human plasma containing antibodies to Rh(D). It also contains glycine, sodium chloride and human albumin.
Distributor
CSL Behring (Australia) Pty Ltd
ABN 48 160 734 761
189-209 Camp Road
Broadmeadows VIC 3047
Australia
Manufacturer
Rhophylac® is manufactured by CSL Behring AG, Switzerland.
Date of most recent amendment
February 2016
Australian Register Number:
1500 IU (300 µg) per 2 mL AUST R 153815
® Registered Trademark of CSL Limited Group of Companies.
Brand Information
| Brand name | Rhophylac |
| Active ingredient | Immunoglobulin (anti-RhD) |
| Schedule | S4 |
MIMS Revision Date: 01 August 2020
1 Name of Medicine
Human anti-D (Rh0) immunoglobulin.
2 Qualitative and Quantitative Composition
Rhophylac is a solution containing 750 IU (150 microgram) per mL human anti-D immunoglobulin corresponding to 1500 IU (300 microgram)/2 mL.
The product contains a maximum of 30 mg/mL of human plasma proteins of which 10 mg/mL is human albumin as stabiliser. At least 95% of the other plasma proteins are IgG. Rhophylac contains not more than 5 microgram/mL IgA.
Rhophylac is a sterile, pyrogen-free liquid solution for intravenous and intramuscular administration. It is purified by means of an ion exchange chromatography isolation procedure resulting in high recovery of anti-D and a high specificity of the product.
Solvent-detergent treatment and virus filtration clearance steps included in the manufacturing process ensure the viral safety of the product.
The solution also contains glycine, sodium chloride and human albumin.
3 Pharmaceutical Form
Solution for injection in pre-filled syringe.
4 Clinical Particulars
4.1 Therapeutic Indications
Rhophylac is indicated for:
prevention of Rh sensitisation in Rh(D)-negative females at or below childbearing age; and
treatment of Rh(D)-negative persons after incompatible transfusions of Rh(D)-positive blood or other products containing red blood cells.
4.2 Dose and Method of Administration
Dosage. It is recommended that prescribed doses of Rhophylac should be expressed as international units written in full.
Prevention of Rh(D) sensitisation in Rh(D)-negative women. Table 1 provides dosing guidelines for the prevention of Rh(D) sensitisation in Rh(D)-negative women.
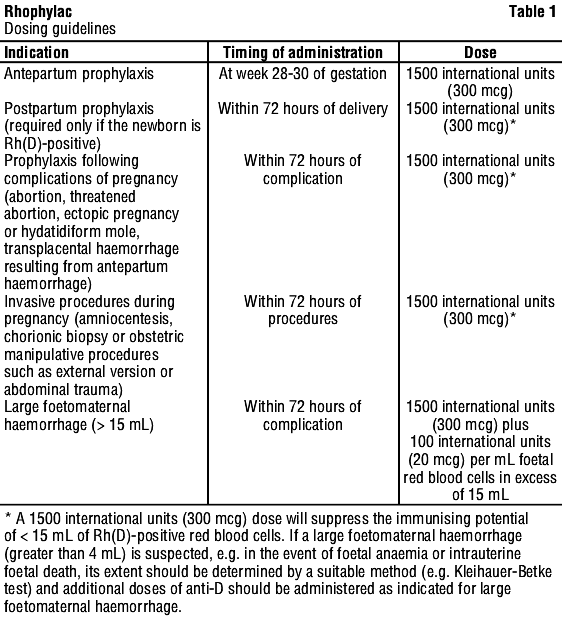
Treatment with an anti-D immunoglobulin can usually be given without a preceding exchange transfusion when the transfused Rh(D)-positive blood represents less than 20% of the total circulating red blood cells. If the volume exceeds 20%, an exchange transfusion should be considered prior to administering Rhophylac.
Method of administration. As with all blood products, patients should be observed for at least 20 minutes following administration of Rhophylac.
Rhophylac can be administered by intravenous or intramuscular injection. In case of haemorrhagic disorders where intramuscular injections are contraindicated, Rhophylac should be administered intravenously. If large doses (more than 5 mL) are required and intramuscular injection is chosen, it is advisable to administer them in divided doses at different sites.
Overweight patients. In patients with a body mass index (BMI) ≥ 30 intravenous administration should be considered (see Section 4.4 Special Warnings and Precautions for Use).
Rhophylac is for single use only and should be brought to room or body temperature before use. The solution should be clear or slightly opalescent. Do not use if solution is cloudy or has deposits.
Rhophylac does not contain an antimicrobial preservative. It must, therefore, be used immediately after opening and any unused product or waste material must be disposed of appropriately.
4.3 Contraindications
Hypersensitivity to any of the components.
Hypersensitivity to human immunoglobulins.
The intramuscular route is contraindicated in persons with severe thrombocytopenia or other disorders of haemostasis.
4.4 Special Warnings and Precautions for Use
In the case of postpartum use, anti-D immunoglobulin is intended for maternal administration. It should not be given to the newborn infant.
The product is neither intended for use in Rh(D)-positive individuals, nor for individuals already immunised to Rh(D) antigen.
Hypersensitivity. Allergic responses to anti-D immunoglobulin may occur even in patients who have tolerated previous administration. Patients should be informed of the early signs of hypersensitivity reactions including hives, generalised urticaria, tightness of the chest, wheezing, hypotension and anaphylaxis. The treatment required depends on the nature and severity of the side effect. In case of shock, the current medical standards for treatment of shock should be observed. If symptoms of allergic or anaphylactic type reactions occur, immediate discontinuation of the administration is required.
The concentration of IgA in Rhophylac was found to be below the detection limit of 5 microgram/mL. Nevertheless, the product may contain trace amounts of IgA. Although anti-D immunoglobulin has been used successfully to treat selected IgA deficient patients, individuals who are deficient in IgA have the potential for developing IgA antibodies and may have anaphylactic reactions after administration of blood components containing IgA. The physician must therefore weigh the benefit of treatment with Rhophylac against the potential risks of hypersensitivity reactions.
Haemolytic reactions. Patients in receipt of an incompatible transfusion who receive very large doses of anti-D immunoglobulin should be monitored clinically and by biological parameters because of the risk of haemolytic reaction.
Obesity. There have been reports that the intramuscular administration of Rhophylac in patients with a body mass index (BMI) ≥ 30 is associated with a risk of lack of efficacy. Therefore, in patients with a BMI ≥ 30 intravenous administration should be considered.
Pathogen safety. Standard measures to prevent infections resulting from the use of medicinal products prepared from human blood or plasma include selection of donors, screening of individual donations and plasma pools for specific markers of infection and the inclusion of effective manufacturing steps for the inactivation/ removal of viruses. Despite this, when medicinal products prepared from human blood or plasma are administered, the possibility of transmitting infective agents cannot be totally excluded. This also applies to unknown or emerging viruses and other pathogens.
The measures taken are considered effective for enveloped viruses such as HIV, HBV, and HCV, and for the non-enveloped viruses HAV and parvovirus B19.
Vaccination for patients in receipt of medicinal products from human plasma should be considered where appropriate.
It is strongly recommended that every time Rhophylac is administered to a patient, the name and batch number of the product are recorded in order to maintain a link between the patient and the batch of the product.
Use in the elderly. The safety and efficacy of Rhophylac have not been investigated in subjects 65 years of age and older.
Paediatric use. The safety and efficacy of Rhophylac have not been investigated in paediatric subjects.
Effects on laboratory tests. After injection of immunoglobulin the transitory rise of the various passively transferred antibodies in the patient's blood may result in misleading positive results in serological testing.
When performing red blood cell antibody screening take blood prior to the administration of Rhophylac.
Passive transmission of antibodies to erythrocyte antigens, e.g. A, B, Rh(C), Rh(D) may interfere with some serological tests for red cell antibodies e.g. the antiglobulin test (Coombs' test), particularly in Rh(D)-positive neonates whose mothers have received antepartum prophylaxis.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Active immunisation with live virus vaccines (e.g. measles, mumps, rubella or varicella) should be postponed until 3 months after the last administration of anti-D immunoglobulin, as the efficacy of the live virus vaccine may be impaired. If anti-D immunoglobulin needs to be administered within 2 to 4 weeks of a live virus vaccination, then the efficacy of such a vaccination may be impaired.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No fertility studies have been performed with Rhophylac anti-D immunoglobulin
Use in pregnancy. This medicine is used in pregnancy. No drug related adverse events were reported for the children delivered of 432 patients who received antepartum administration of Rhophylac.
No embryofoetal toxicity studies with Rhophylac have been performed in pregnant animals.
Use in lactation. This medicine is used in nursing mothers. Immunoglobulins are excreted in breast milk. No undesirable effects are expected on nursing infants.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile. When anti-D immunoglobulins are administered by the intramuscular route, local pain and tenderness can be observed at the injection site.
Tabulated list of adverse reactions. The adverse reactions in Table 2 have been reported from 592 patients in clinical studies and from post-marketing experience. The summary table is presented according to the MedDRA system organ classification (SOC and preferred term level).
Frequency has been evaluated using the following criteria: very common (≥ 1/10), common (≥ 1/100, < 1/10), uncommon (≥ 1/1000, < 1/100), rare (≥ 1/10,000, < 1/1000), very rare (< 1/10,000).
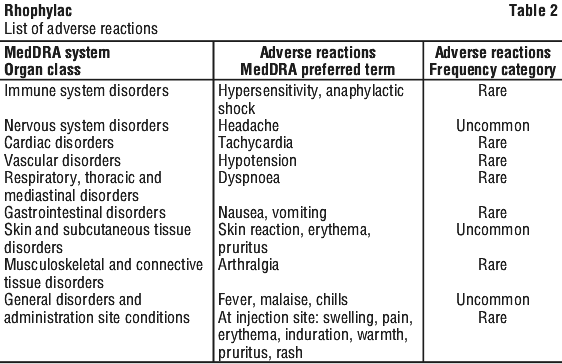
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
No data is available on overdosage.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Rhophylac contains specific IgG antibodies against the Rh(D) antigen of human red blood cells.
Prevention of Rh(D) isoimmunisation. During pregnancy, and especially at the time of childbirth, foetal red blood cells may enter the maternal circulation. When the woman is Rh(D)-negative and the foetus Rh(D)-positive, the woman might become immunised to the Rh(D) antigen and may produce anti-Rh(D) antibodies which cross the placenta and may cause haemolytic disease of the newborn. Passive immunisation with anti-D immunoglobulin prevents Rh(D) immunisation in more than 99% of cases provided that a sufficient dose of anti-D immunoglobulin is administered early enough after exposure to Rh(D)-positive foetal red blood cells.
The mechanism by which anti-D immunoglobulin suppresses immunisation to Rh(D)-positive red blood cells is not known. Suppression may be related to the clearance of the Rh(D)-positive red blood cells from the circulation before they reach immunocompetent sites or, it may be due to more complex mechanisms involving recognition of foreign antigen and antigen presentation by the appropriate cells at the appropriate sites in the presence or absence of antibody.
Clinical trials. In a clinical study of Rh(D)-negative healthy male volunteers, both the intravenous and intramuscular administration of a 1000 IU (200 microgram) dose of Rhophylac 24 hours after injection of 15 mL of Rh(D)-positive red blood cells resulted in an effective clearance of Rh(D)-positive red blood cells. On average, 99% of injected Rh(D)-positive red blood cells were cleared within 60 hours following intravenous administration and within 144 hours following intramuscular administration.
In two clinical studies, 446 Rh(D)-negative pregnant women received a 1500 IU (300 microgram) dose of Rhophylac during week 28 of gestation. The women who gave birth to an Rh(D)-positive baby received a second 1500 IU (300 microgram) dose within 72 hours of birth.
Study 1. Eight of the women who participated in the pharmacokinetic study gave birth to an Rh(D)-positive baby and received the postpartum dose of 1500 IU (300 microgram) of Rhophylac. Antibody tests performed 6 to 8 months later were negative for all women. This suggests that no Rh(D) sensitisation occurred.
Study 2. In an open label, single arm clinical study at 22 centres in the US and United Kingdom, 432 pregnant women received the antepartum dose of 1500 IU (300 microgram) of Rhophylac either as an intravenous or intramuscular injection (two randomised groups of 216 women each). Subjects received an additional 1500 IU (300 microgram) dose if an obstetric complication occurred between the routine antepartum dose and birth or if extensive foetomaternal haemorrhage was measured after birth. Of the 270 women who gave birth to an Rh(D)-positive baby, 248 women were evaluated for Rh(D) sensitisation 6 to 11.5 months postpartum. None of these women developed antibodies against the Rh(D) antigen.
5.2 Pharmacokinetic Properties
In a clinical study comparing the pharmacokinetics of intravenous versus intramuscular administration, 14 Rh(D)-negative pregnant women received a single 1500 IU (300 microgram) dose of Rhophylac at week 28 of gestation.
Following intravenous administration, peak serum levels of Rh(D) immunoglobulin ranged from 62 to 84 nanogram/mL after 1 day (i.e. the time the first blood sample was taken following the antepartum dose). Mean systemic clearance was 0.20 ± 0.03 mL/min, and half-life was 16 ± 4 days.
Following intramuscular administration, peak serum levels ranged from 7 to 46 nanogram/mL and were achieved between 2 and 7 days. Mean apparent clearance was 0.29 ± 0.12 mL/min, and half-life was 18 ± 5 days. The absolute bioavailability of Rhophylac was 69%.
Regardless of the route of administration, Rh(D) immune globulin titers were detected in all women up to at least 9 weeks following administration of Rhophylac.
IgG and IgG complexes are broken down in cells of the reticuloendothelial system.
5.3 Preclinical Safety Data
Genotoxicity. No genotoxicity studies have been performed with Rhophylac anti-D immunoglobulin.
Carcinogenicity. No carcinogenicity studies have been performed with Rhophylac anti-D immunoglobulin.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C. (Refrigerate. Do not freeze.) Protect from light. Do not use after the expiry date shown on the label.
6.5 Nature and Contents of Container
Rhophylac solution for injection is available as a single-use pre-filled 2 mL syringe containing 1500 IU (300 microgram) of anti-D immunoglobulin.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
CAS number. None assigned.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
22 December 2009
Date of Revision
09 June 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.