RiaSTAP
Brand Information
| Brand name | RiaSTAP |
| Active ingredient | Fibrinogen |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the RiaSTAP.
Summary CMI
RiaSTAP®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using RiaSTAP®?
RiaSTAP® contains the active ingredient human fibrinogen. RiaSTAP® is used for the treatment of acute bleeding in people with an absence or low level of human fibrinogen.
For more information, see Section 1. Why am I using RiaSTAP®? in the full CMI.
2. What should I know before I use RiaSTAP®?
Do not use if you have ever had an allergic reaction to RiaSTAP® or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding or are on a sodium controlled diet.
For more information, see Section 2. What should I know before I use RiaSTAP®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with RiaSTAP® and affect how it works.
For more information, see in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use RiaSTAP®?
- Treatment should be started and initiated by a doctor experienced in the treatment of coagulation conditions.
- If it is not injected immediately it must be stored below 25°C and used within 6 hours of reconstitution. The reconstituted solution should not be stored in the refrigerator.
More instructions can be found in Section 4. How do I use RiaSTAP®? in the full CMI.
5. What should I know while using RiaSTAP®?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using RiaSTAP®? in the full CMI.
6. Are there any side effects?
All medicines can have side effects. sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects. The most common side effect is fever. The more serious side effects include rash, or reddening of the skin, itching, difficulty breathing, feeling faint, dizziness and shortness of breath.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
RiaSTAP®
Active ingredient(s): Human Fibrinogen
Consumer Medicine Information (CMI)
This leaflet provides important information about using RiaSTAP®. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using RiaSTAP®.
Where to find information in this leaflet:
1. Why am I using RiaSTAP®?
2. What should I know before I use RiaSTAP®?
3. What if I am taking other medicines?
4. How do I use RiaSTAP®?
5. What should I know while using RiaSTAP®?
6. Are there any side effects?
7. Product details
1. Why am I using RiaSTAP®?
RiaSTAP® contains the active ingredient human fibrinogen. RiaSTAP® is made from human plasma (this is the liquid part of the blood).
RiaSTAP® for intravenous (into the vein) injection is used for the treatment of acute bleeding in people with an absence or low level of human fibrinogen (congenital lack of fibrinogen).
Human fibrinogen is a protein which is important for blood clotting (coagulation). If you have missing or malfunctioning fibrinogen the blood does not clot as quickly as it should which results in an increased tendency of bleeding. The replacement of human fibrinogen with RiaSTAP® will help to temporarily correct the clotting in patients with fibrinogen deficiency.
2. What should I know before I use RiaSTAP®?
Warnings
Do not use RiaSTAP® if:
- you are allergic to human fibrinogen, or any of the ingredients listed at the end of this leaflet.
Talk to your doctor before using RiaSTAP®:
There is an increased risk of blood clots (thrombosis), particularly
- in case of a high dose or repeated doing
- if you have had a heart attack (a history of coronary heart disease or myocardial infarction)
- if you have just had surgery
- if you suffer from liver disease
- if you will be having surgery soon
- in newborn infants
- if you are more likely to suffer from blood clots than normal.
Your doctor will consider carefully the benefit of treatment with RiaSTAP® compared with the risk of these complications.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
This medicine should only be used if clearly needed during pregnancy or breastfeeding.
Virus safety
RiaSTAP® is made from human plasma (this is the liquid part of blood). When products are made from human blood or plasma and injected into you, it is possible that viruses or other substances could be present in the product and cause an illness. These could be viruses such as hepatitis, human immunodeficiency virus (HIV), or parvovirus B19. There could also be other infectious agents some of which may not yet have been discovered.
To reduce the risk of infection extra steps are taken when manufacturing this product. Strict controls are applied when selecting blood donors and donations. The product is specially treated to kill and remove viruses. These special treatments are considered effective against certain viruses known as enveloped viruses (such as HIV and hepatitis B and C) and also for the non-enveloped virus hepatitis A.
The measures taken may be of limited value against non-enveloped viruses such as parvovirus B19. Despite these measures, the risk of transmitting infection cannot be totally eliminated.
Vaccines are available against some of these viruses and your doctor will be able to help you decide whether it is worthwhile having any of those vaccines.
Please discuss the risks and benefits of this medicine with your doctor.
RiaSTAP® contains sodium
RiaSTAP® contains up to 164 mg sodium (approximately 7.1 mmol) per 1g fibrinogen (vial). Please take this into account if you are on a sodium controlled diet.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop. Some medicines may affect the way other medicines work.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect RiaSTAP®.
4. How do I use RiaSTAP®?
How much to use
The dose of RiaSTAP® you need and the duration of your treatment will depend on:
- the severity of the disease
- the site and intensity of the bleeding
- your clinical condition.
Your doctor will determine the dose suitable for you.
When to use RiaSTAP®
- Treatment should be started and initiated by a doctor experienced in the treatment of coagulation conditions.
- Your doctor will discuss with you when you should be given RiaSTAP®.
Preparing RiaSTAP® for administration
The following steps are used as a guide only. Please contact your doctor if you have any questions.
|
|
|
|
|
|
Instructions for RiaSTAP® reconstitution
|
|
|
|
|
|
|
|
|
|
Instructions for administration preparation
|
|
|
|
|
|
|
|
|
|
|
|
After reconstitution the solution should be clear or slightly opalescent, that is it might sparkle when held up to the light but must not contain any obvious particles.
Do not mix RiaSTAP® with other medicinal products or diluents either before or during administration.
RiaSTAP® should be used for one person on one occasion only.
RiaSTAP® should be administered by slow intravenous injection / infusion and should not exceed 5 mL per minute.
If it is not injected immediately it must be stored below 25°C and used within 6 hours of reconstitution. The reconstituted solution should not be stored in the refrigerator.
Any unused portion remaining in the vial must be discarded appropriately.
Every time that RiaSTAP® is injected, the date of administration, batch number and injected volume should be recorded.
If you forget to use RiaSTAP®
RiaSTAP® should only be used as instructed by your doctor. The dosage and duration will depend on the severity of your condition.
Consult your doctor is you have missed a dose to avoid overdose risks (blood clots).
If you use too much RiaSTAP®
If you think that you have used too much RiaSTAP®, you may need urgent medical attention.
Overdose may enhance the risk of blood clots (thrombosis).
Your doctor should regularly check your blood clot status during your treatment.
If you have any questions consult your doctor.
5. What should I know while using RiaSTAP®?
Things you should do
Call your doctor straight away if you:
Notice signs or symptoms of a serious side effect. This may be a sign of a serious allergy or anaphylactic reaction. The administration of RiaSTAP® should be stopped immediately.
Remind any doctor, dentist or pharmacist you visit that you are using RiaSTAP®.
Things you should not do
- Do not stop using this medicine suddenly unless instructed by your doctor.
- Do not give or share your medicine with anyone else, even if they have the same condition as you.
- Do not mix RiaSTAP® with other medicines or diluent either before or during administration.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how RiaSTAP® affects you.
RiaSTAP® has no or negligible influence on the ability to drive and use machines.
Looking after your medicine
- Store at 2°C to 8°C (Refrigerate. Do not freeze).
- Keep the vial in the carton in order to protect it from light.
- Do not use after the expiry date.
- RiaSTAP® does not contain a preservative so the made-up solution should preferably be used immediately.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep it out of the sight and reach of children.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
RiaSTAP® is usually given in a hospital and is only available with a doctor's prescription.
What RiaSTAP® contains
| Active ingredient (main ingredient) | Human fibrinogen |
| Other ingredients (inactive ingredients) | Albumin Arginine hydrochloride Sodium chloride Sodium citrate |
Do not take this medicine if you are allergic to any of these ingredients.
What RiaSTAP® looks like
RiaSTAP® is a white powder and is supplied with water for injections as solvent.
RiaSTAP® comes in a pack which also contains:
- one vial of diluent (50 mL Water for Injections)
- one transfer set
- one dispensing pin
- one syringe filter.
1 g: AUST R 162828
Who distributes RiaSTAP®
RiaSTAP® is supplied in Australia by
CSL Behring (Australia) Pty Ltd
ABN 48 160 734 761
189-209 Camp Road
Broadmeadows VIC 3047
Australia
For Medical / Technical Enquiries
TOLL FREE: 1800 642 865
For Customer Service Enquiries
TOLL FREE: 1800 063 892
customerservice@cslbehring.com.au
This leaflet was prepared in March 2025.
® Registered trademark of CSL Limited Group of Companies
Brand Information
| Brand name | RiaSTAP |
| Active ingredient | Fibrinogen |
| Schedule | Unscheduled |
MIMS Revision Date: 01 July 2020
1 Name of Medicine
Human fibrinogen.
2 Qualitative and Quantitative Composition
RiaSTAP is a sterile, freeze-dried fibrinogen (coagulation factor I) concentrate derived from human plasma.
RiaSTAP contains up to 164 mg (7.1 mmol) sodium per vial. For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Powder and diluent for solution for injection.
Powder: white.
Diluent (WFI): clear, colourless.
4 Clinical Particulars
4.1 Therapeutic Indications
Treatment of acute bleeding episodes in patients with congenital fibrinogen deficiency, including afibrinogenaemia and hypofibrinogenaemia.
4.2 Dose and Method of Administration
Treatment should be initiated under the supervision of a physician experienced in the treatment of coagulation disorders.
Dosage. The dosage and duration of the substitution therapy depend on the severity of the disorder, location and extent of bleeding and the patient's clinical condition.
The (functional) fibrinogen level should be determined in order to calculate individual dosage and the amount and frequency of administration should be determined on an individual patient basis by regular measurement of plasma fibrinogen level and continuous monitoring of the clinical condition of the patient and other replacement therapies used.
Only general dosage guidelines are given below. Normal plasma fibrinogen level is in the range of 2.0-4.5 g/L. The critical plasma fibrinogen level below which haemorrhages may occur is approximately 1.0 g/L.
Clinical data on the use of RiaSTAP in patients undergoing surgical procedures are very limited. In case of surgical intervention, precise monitoring of replacement therapy by coagulation assays is essential.
Initial dose. If the patient's fibrinogen level is not known, the recommended dose is 70 mg per kg of body weight (BW) administered intravenously.
Subsequent dose. The target level (1 g/L) for minor events (e.g. epistaxis, intramuscular bleeding or menorrhagia) should be maintained for at least three days. The target level (1.5 g/L) for major events (e.g. head trauma or intracranial haemorrhage) should be maintained for seven days. See Equation 1.
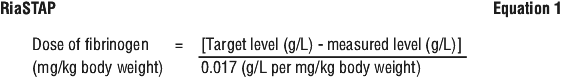
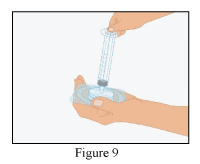
Reconstitution. The procedures below are provided as general guidelines for preparation and reconstitution of RiaSTAP.
Use aseptic technique when preparing and reconstituting RiaSTAP.
Warm both the water for injections and RiaSTAP in unopened vials to room or body temperature (not above 37°C).
Wash hands or use gloves before reconstituting the product.
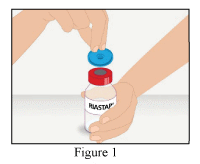
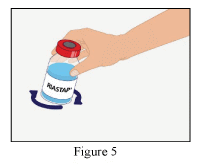
1. Remove the cap from the RiaSTAP vial to expose the central portion of the rubber stopper.
2. Clean the surface of the rubber stopper with an antiseptic solution and allow it to dry.
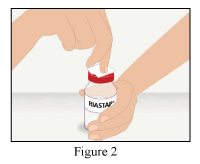
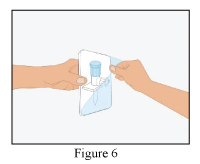
3. Remove the safety cap from one end of the provided transfer set and pierce the stopper of the RiaSTAP vial.
4. Remove the safety cap from the other end of the transfer set, invert the Water for Injections (WFI) vial, apply gentle pressure to pierce the stopper and transfer the contents into the RiaSTAP vial.
5. Discard the WFI vial and remove the transfer set from the RiaSTAP vial.
6. Gently swirl the RiaSTAP vial to ensure the product is fully dissolved.
Avoid shaking which causes formation of foam.
The powder should be completely reconstituted within 15 minutes (generally 5 to 10 minutes).
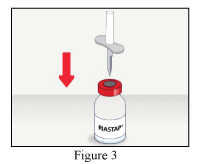
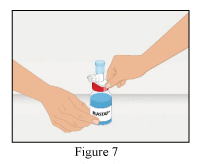
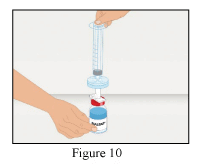
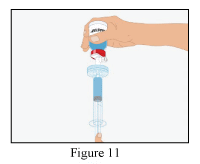
7. Open the plastic blister containing the dispensing pin.
8. Take the provided dispensing pin and insert into the stopper of the vial with the reconstituted RiaSTAP.
9. After the dispensing pin is inserted, remove the cap. After the cap is removed, do not touch the exposed surface.
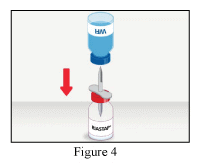
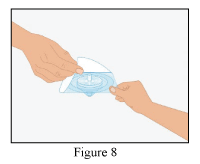
10. Open the blister with the provided syringe filter.
11. Screw the syringe (not supplied) onto the filter.
12. Screw the syringe with the mounted filter onto the dispensing pin.
13. Draw the reconstituted RiaSTAP into the syringe.
14. When completed, remove the syringe filter, dispensing pin and empty vial from the syringe, dispose of appropriately, and proceed with administration as usual.
After reconstitution, the RiaSTAP solution should be colourless and clear to slightly opalescent. Inspect visually for particulate matter and discoloration prior to administration. Do not use if the solution is cloudy or contains particulates.
Administration. Do not mix RiaSTAP with other medicinal products or intravenous solutions. RiaSTAP should be administered through a separate injection site.
Use aseptic technique when administering RiaSTAP.
Administer RiaSTAP at room temperature by slow intravenous injection at a rate not exceeding 5 mL per minute.
It is strongly recommended that every time RiaSTAP is administered to a patient, the name and batch number of the product are recorded in the patient notes in order to maintain a link between the patient and the batch of product.
This product does not contain an antimicrobial preservative. To reduce microbiological hazard, the product should be used as soon as practicable after reconstitution. If it is not administered immediately, it must be stored below 25°C and used within 6 hours of reconstitution. The reconstituted solution should not be stored in the refrigerator. Any unused solution must be discarded appropriately. Use in one patient on one occasion only.
4.3 Contraindications
Hypersensitivity to the active substances or to any of the excipients.
4.4 Special Warnings and Precautions for Use
Thrombosis. There is a risk of thrombosis when patients with congenital fibrinogen deficiency are treated with human fibrinogen particularly with high dose or repeated dosing. Thrombosis may occur spontaneously in patients with congenital fibrinogen deficiency with or without the use of fibrinogen replacement therapy. Patients given human fibrinogen should be observed closely for signs or symptoms of thrombosis.
Some patients with congenital dysfibrinogenaemia may have an increased tendency to thrombosis. The safety and efficacy of RiaSTAP in the treatment of congenital dysfibrinogenaemia have not been established.
In patients with a history of coronary heart disease, or myocardial infarction, in patients with liver disease, in peri- or postoperative patients, in neonates, or in patients at risk of thromboembolic events (TEE) or disseminated intravascular coagulation, the potential benefit of treatment with RiaSTAP should be weighed against the risk of thromboembolic complications. Caution and close monitoring should also be performed.
Allergic reactions. If allergic or anaphylactic-type reactions occur, the injection/ infusion should be stopped immediately. In case of anaphylactic shock, standard medical treatment for shock should be implemented.
In the case of replacement therapy with coagulation factors in other congenital deficiencies, antibody reactions (including inhibitor formation) have been observed, but there is currently no data with fibrinogen.
Sodium. RiaSTAP contains up to 164 mg (7.1 mmol) sodium per vial. This correlates with 11.5 mg (0.5 mmol) sodium per kg body weight of the patient if the recommended initial dose of 70 mg/kg body weight is applied. This is to be taken into consideration by patients on a controlled sodium diet.
See Section 4.2 Dose and Method of Administration for further precautions regarding administration of RiaSTAP.
Pathogen safety. This product is made from human plasma. Products made from human plasma may contain infectious agents such as viruses that can cause disease. The risk that such products will transmit an infectious agent has been reduced by screening plasma donors for prior exposure to certain infectious agents and by testing for the presence of certain viral markers. In addition, the RiaSTAP manufacturing process includes pasteurisation (60°C for 20 hours) and multiple precipitation and absorption steps as dedicated virus removal and inactivation procedures to reduce the possibility of virus transmission.
The measures taken are considered effective for enveloped viruses such as human immunodeficiency virus (HIV), hepatitis B virus (HBV) and hepatitis C virus (HCV), and for the non-enveloped virus hepatitis A virus (HAV).
The measures taken may be of limited value against non-enveloped viruses such as parvovirus B19. Parvovirus B19 infection may be serious for pregnant women (foetal infection) and for individuals with immunodeficiency or increased erythropoiesis (e.g. haemolytic anaemia).
Despite these measures, such products may still potentially transmit disease. There is also the possibility that other known or unknown infectious agents may be present in such products.
Vaccination for patients in receipt of medicinal products from human plasma should be considered where appropriate.
Use in the elderly. The efficacy and safety of RiaSTAP in the elderly population has not been established in controlled clinical trials.
Paediatric use. The efficacy and safety of RiaSTAP in the paediatric population has not been established in controlled clinical trials.
Effects on laboratory tests. Human fibrinogen is an endogenous plasma protein so no specific effects on laboratory tests are anticipated.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The interaction of RiaSTAP with other drugs has not been established in appropriate studies.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There are no data on fertility available.
Use in pregnancy. Animal reproduction studies have not been conducted with RiaSTAP. Since the active substance is of human origin, it is catabolised in the same manner as the patient's own protein. These physiological constituents of the human blood are not expected to induce adverse effects on reproduction or on the foetus.
The safety of human plasma fibrinogen products for use in human pregnancy has not been established in controlled clinical trials.
Clinical experience with fibrinogen products in the treatment of obstetric complications suggests that no harmful effects on the course of the pregnancy or health of the foetus or the neonate are to be expected.
Use in lactation. It is unknown whether RiaSTAP is excreted in human milk. The safety of human plasma fibrinogen products for use during lactation has not been established in controlled clinical trials.
A risk to the suckling child cannot be excluded. A decision must be made whether to discontinue breast-feeding or to discontinue/ abstain from RiaSTAP therapy taking into account the benefit of breast-feeding for the child and the benefit of therapy for the woman.
4.7 Effects on Ability to Drive and Use Machines
RiaSTAP has no or negligible influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile. Allergic or anaphylactic type reactions have been uncommonly observed. The events reported in association with allergic/ anaphylactic reactions include generalised urticarial, rash, dyspnoea, tachycardia, nausea, vomiting, chills, pyrexia, chest pain, cough, blood pressure decreased, and anaphylactic shock (see Section 4.4 Special Warnings and Precautions for Use).
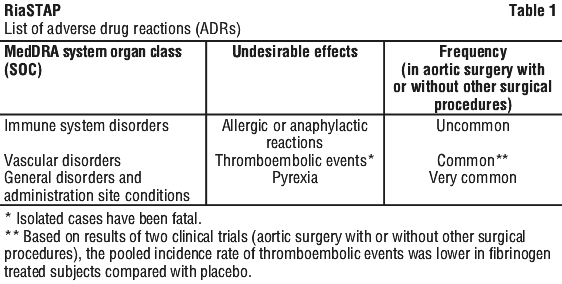
The risk of TEE following the administration of fibrinogen concentrate (see Section 4.4 Special Warnings and Precautions for Use) as determined in clinical trials is further described in Table 1.
Pyrexia has been very commonly observed.
Tabulated list of adverse reactions. Table 1 combines the adverse reactions identified from clinical trials and postmarketing experience. Frequencies presented in Table 1 have been based on pooled analyses across two company sponsored clinical trials performed in aortic surgery with or without other surgical procedures according to the following convention: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1000 to < 1/100); rare (≥ 1/10,000 to < 1/1000); very rare (< 1/10,000) or unknown (cannot be estimated from the available data).
The calculated frequency is based on crude incidence rates without considering the frequency of the comparator arm. It should be noted that in the clinical trials included in the analysis, the incidence of TEEs was higher in the placebo arm. In view of the fact that these trials were conducted in only the narrow population of aortic surgery with or without other surgical procedures, adverse drug reaction rates observed in these trials may not reflect the rates observed in clinical practice and are unknown for clinical settings outside the studied indication.

Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
In order to avoid overdose, regular monitoring of the plasma level of fibrinogen during therapy is indicated.
In case of overdose, the risk of development of thromboembolic complications is enhanced.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Human fibrinogen in the presence of thrombin, activated coagulation factor XIII (FXIIIa) and calcium ions is converted into a stable and elastic three-dimensional fibrin haemostatic clot.
The administration of human fibrinogen concentrate provides an increase in plasma fibrinogen level and can temporarily correct the coagulation defect of patients with fibrinogen deficiency. The product is administered intravenously and is immediately available in a plasma concentration corresponding to the dosage administered.
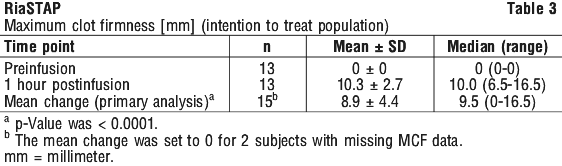
Clinical trials. Efficacy and safety. The pharmacokinetic study evaluated the single-dose pharmacokinetics and maximum clot firmness (MCF) in subjects with afibrinogenaemia. MCF was determined by thromboelastometry (ROTEM) testing. MCF was measured to demonstrate functional activity of replacement fibrinogen when a fixed dose of RiaSTAP was administered. Clot firmness is a functional parameter that depends on: activation of coagulation, fibrinogen content of the sample and polymerisation/ crosslinking of the fibrin network. Thromboelastometry has been shown to be a functional marker for the assessment of fibrinogen content and for the effects of fibrinogen supplementation on clinical efficacy.
For each subject, the MCF was determined before (baseline) and one hour after the single dose administration of RiaSTAP. RiaSTAP was found to be effective in increasing clot firmness in patients with congenital fibrinogen deficiency (afibrinogenaemia) as measured by thromboelastometry. The study results demonstrated that the MCF values were significantly higher after administration of RiaSTAP than at baseline (see Table 3). The mean change from pre-infusion to 1 hour postinfusion was 8.9 mm in the primary analysis (9.9 mm for subjects < 16 years old and 8.5 mm for subjects ≥ 16 to < 65 years old). The mean change in MCF values closely approximated the levels expected from adding known amounts of fibrinogen to plasma in vitro.
5.2 Pharmacokinetic Properties
A pharmacokinetic study evaluated the single-dose pharmacokinetics before and after administration of human fibrinogen in subjects with afibrinogenaemia. This prospective, open label, uncontrolled, multicentre study consisted of 5 females and 10 males, ranging in age from 8 to 61 years (2 children, 3 adolescents, 10 adults). The median dose was 77.0 mg/kg body weight (range 76.6-77.4 mg/kg).
Blood was sampled from 15 subjects (14 evaluable) to determine the fibrinogen activity at baseline and up to 14 days after the infusion was complete. In addition, the incremental in vivo recovery (IVR), defined as the maximum increase in fibrinogen plasma levels per mg/kg body weight dosed, was determined from levels obtained up to 4 hours postinfusion. The median incremental IVR was 17 (range 13.0-27.3) mg/L per mg/kg body weight.
Table 4 provides the pharmacokinetic results.

5.3 Preclinical Safety Data
Genotoxicity. No genotoxicity studies have been conducted with RiaSTAP.
Carcinogenicity. No carcinogenicity studies have been conducted with RiaSTAP.
6 Pharmaceutical Particulars
6.1 List of Excipients
Albumin, arginine hydrochloride, sodium hydroxide (for pH adjustment), sodium chloride, sodium citrate.
6.2 Incompatibilities
Do not mix RiaSTAP with other medicinal products or intravenous solutions. RiaSTAP should be administered through a separate injection site.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
Reconstituted product. This product does not contain an antimicrobial preservative. To reduce microbiological hazard, the product should be used as soon as practicable after reconstitution. If it is not administered immediately, it must be stored below 25°C and used within 6 hours of reconstitution. The reconstituted solution should not be stored in the refrigerator.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze). Protect from light. Do not use after the expiry date.
6.5 Nature and Contents of Container
Each RiaSTAP single pack contains:
one glass vial of 1 g human fibrinogen, with a rubber stopper closed with an aluminium cap and a plastic disc;
one glass vial of 50 mL water for injections, with a rubber stopper closed with an aluminium cap and a plastic disc;
one transfer set;
one dispensing pin;
one syringe filter.
RiaSTAP is packaged in latex-free materials.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
CAS number. 9001-32-5.
7 Medicine Schedule (Poisons Standard)
Unscheduled.
Date of First Approval
02 August 2010
Date of Revision
21 May 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.