Rikodeine Oral Liquid
Brand Information
| Brand name | Rikodeine Oral Liquid |
| Active ingredient | Dihydrocodeine tartrate |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Rikodeine Oral Liquid.
Full CMI
WARNINGS
Limitations of use
Rikodeine should only be used when your doctor or pharmacist decides that other treatment options are not able to effectively manage your cough, or you cannot tolerate them.
Hazardous and harmful use
Rikodeine poses risks of abuse, misuse and addiction which can lead to overdose and death.
Life threatening respiratory depression
Rikodeine can cause life-threatening or fatal breathing problems (slow, shallow, unusual or no breathing) even when used as recommended. These problems can occur at any time during use, but the risk is higher when first starting Rikodeine and after a dose increase, if you are older, or have an existing problem with your lungs.
Use of other medicines while using Rikodeine
Using Rikodeine with other medicines that can make you feel drowsy such as sleeping tablets (e.g., benzodiazepines), other pain relievers, antihistamines, antidepressants, antipsychotics, gabapentinoids (e.g., gabapentin and pregabalin), cannabis and alcohol may result in severe drowsiness, decreased awareness, breathing problems, coma and death.
You must not drink alcohol while using Rikodeine.
Rikodeine® Oral Liquid (rye-co-deen)
Active ingredient(s): Dihydrocodeine tartrate and sorbitol (die-hydro-co-deen tahr-treyt + sore-be-tol)
Consumer Medicine Information (CMI)
This leaflet provides important information about using Rikodeine. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Rikodeine.
Where to find information in this leaflet:
1. Why am I taking Rikodeine?
2. What should I know before I take Rikodeine?
3. What if I am taking other medicines?
4. How do I take Rikodeine?
5. What should I know while taking Rikodeine?
6. Are there any side effects?
7. Product details
1. Why am I taking Rikodeine?
Rikodeine is a cough suppressant and contains the active ingredients, dihydrocodeine tartrate and sorbitol.
Rikodeine is used to give temporary relief for an unproductive and dry cough, which will not go away.
2. What should I know before I take Rikodeine?
Warnings
Do not give Rikodeine to children aged under 6 years.
Only give Rikodeine to children aged 6 – 11 years on the advice of a doctor, pharmacist, or nurse practitioner.
Do not take Rikodeine if:
- You are allergic to dihydrocodeine, other opioids, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine
- You have severe lung or breathing problems
- The expiry date printed on the packaging has passed
- The packaging shows signs of tampering.
Check with your doctor or pharmacist if you:
- Have any of the following medical conditions:
- Kidney or liver disease
- Impaired function of the adrenal or thyroid gland
- Asthma, emphysema, or any breathing problems
- Increased prostate size
- Narrowing of urinary tract
- Biliary tract disease or inflammation of the pancreas
- A condition associated with fits or convulsions
- Diarrhoea
- Muscle disorders - serious muscle weakness
- Inflammatory or obstructive bowel disorders
- Low blood pressure
- Take any medicines for any other condition
- Are allergic to foods, dyes, preservatives, or any other medicines
- Have had a head injury or increased pressure in the head
- Regularly drink large amounts of alcohol.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Taking Rikodeine regularly during the last trimester of pregnancy may risk causing breathing problems or withdrawal symptoms in the newborn baby.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
Addiction
You can become addicted to Rikodeine even if you take it exactly as prescribed. Rikodeine may become habit forming causing mental and physical dependence. If abused, it may become less able to provide temporary relief of an unproductive and dry cough.
Dependence
As with all other opioid containing products, your body may become used to you taking Rikodeine. Taking it may result in physical dependence. Physical dependence means that you may experience withdrawal symptoms if you stop taking Rikodeine suddenly, so it is important to take it exactly as directed by your doctor or pharmacist.
Tolerance
Tolerance to Rikodeine may develop, which means that the effect of the medicine may decrease. If this happens, more may be needed to maintain the same effect.
Withdrawal
Continue taking Rikodeine for as long as your doctor or pharmacist tells you. If you stop taking this medicine suddenly, you may experience some or all of the following withdrawal symptoms:
- Nervousness, restlessness, agitation, trouble sleeping or anxiety
- Body aches, weakness, or stomach cramps
- Loss of appetite, nausea, vomiting or diarrhoea
- Increased heart rate, breathing rate or pupil size
- Watery eyes, runny nose, chills, or yawning
- Increased sweating.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins, or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Rikodeine may increase the effects of alcohol.
Some medicines may interfere with Rikodeine and affect how it works.
These include:
- Any medicine causing sleepiness or drowsiness
- Sedating antihistamines (medicines used to treat hay fever and allergies)
- Tranquillisers (medicines for anxiety or nerves)
- Benzodiazepines (medicines used as sedatives or to treat anxiety)
- Medicines containing alcohol (ethanol), e.g., some cough syrups
- Medicines used to treat depression
- Medicines used to treat mental illness
- Medicines to treat high blood pressure
- Medicines to treat diarrhoea, nausea, or vomiting
- Monoamine oxidase inhibitors (medicines used to treat depression) taken within the last 10 days
- Pain relief medication such as codeine, morphine, and pethidine
- Other cough suppressants
- Quinidine (used to treat abnormal heart rhythms)
- Medicines used to treat alcohol and/or opioid dependence (e.g., naltrexone, buprenorphine, or methadone)
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Rikodeine.
4. How do I take Rikodeine?
How much to take
- Adults and children 12 years and over: take 5 to 10 mL every four to six hours. If you are elderly take a 5 mL dose until you know how the medicine affects you.
- For children between 6 to 11 years: give 2.5 to 5 mL every four to six hours (only on the advice of a doctor, pharmacist, or nurse practitioner).
When to take it
- Only use Rikodeine when you need to stop your cough
- Do not take more than 6 doses over a 24 hour period.
How to take it
Accurately measure the dose with a medicine measure. Using a medicine measure ensures that you will get the correct dose. You can buy a medicine measure from your pharmacist.
How long to take Rikodeine
Stop using this medicine when the cough has gone. If the coughing continues or gets worse, talk to your doctor.
If you take too much Rikodeine
If you or someone else receive too much (overdose), and experience one or more of the symptoms below, immediately call triple zero (000) for an ambulance. Keep the person awake by talking to them or gently shaking them every now and then.
Symptoms of overdose include:
- Slow, unusual or difficult breathing
- Extreme sleepiness progressing to unconsciousness
- Slow or weak heartbeat
- Cold, clammy skin
- Pinpoint pupils
- Flabby muscles
You should follow the above steps even if someone other than you have accidentally used Rikodeine that was prescribed for you.
If you think you or someone else may have used too much Rikodeine, you should immediately:
- Phone the Poisons Information Centre (by calling 13 11 26), or
- Contact your doctor, or
- Go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
When seeking medical attention, take this leaflet and remaining medicine with you to show the doctor. Also tell them about any other medicines or alcohol which have been taken.
Depending on your body's individual ability to break down dihydrocodeine, you may experience signs of overdose even when you take Rikodeine as stated on the labelling. If overdose symptoms occur, seek immediate medical advice.
5. What should I know while taking Rikodeine?
Things you should do
- Take Rikodeine exactly as your doctor or pharmacist has prescribed
- Tell your doctor if you become pregnant while taking Rikodeine
- Remind your doctor, dentist, or pharmacist you visit that you are taking Rikodeine
- If you are having surgery, tell the surgeon or anaesthetist that you are taking Rikodeine as it may affect other medicines used during surgery.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Rikodeine affects you.
Rikodeine may cause drowsiness, dizziness, or light-headedness and problems with vision in some people.
Drinking alcohol
Do not drink alcohol while taking Rikodeine.
Drinking alcohol increases the likelihood of becoming drowsy.
Looking after your medicine
Keep Rikodeine in the bottle until it is time to use it.
Store Rikodeine below 30°C in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep medicines where children cannot reach them. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
If you are over 65 years of age you may have an increased chance of getting side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
As with many other opiates, abuse of dihydrocodeine has been reported.
If you think you are having an allergic reaction to Rikodeine, tell your doctor immediately or go to Accident and Emergency at your nearest hospital.
Symptoms usually include some or all of the following:
- Shortness of breath
- Wheezing or difficulty breathing
- Swelling of the lips/mouth face, lips, tongue, or other parts of the body
- Hay fever
- Rash, itching or hives on the skin
- Fainting
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side affects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What Rikodeine contains
| Active ingredients (main ingredient) | Dihydrocodeine tartrate Sorbitol |
| Other ingredients (inactive ingredients) | Sucrose Citric acid anhydrous Methyl hydroxybenzoate Amaranth CI16185 Strawberry flavour Water |
What Rikodeine looks like
Rikodeine is a clear, red, strawberry flavoured liquid. It is packed in 100 mL and 200 mL bottles.
Registration number: AUST R 10587.
Who distributes Rikodeine
iNova Pharmaceuticals (Australia) Pty Limited
Level 10, 12 Help Street
Chatswood NSW 2067
Tel: 1800 630 056
This leaflet was prepared in September 2021.
Brand Information
| Brand name | Rikodeine Oral Liquid |
| Active ingredient | Dihydrocodeine tartrate |
| Schedule | S3 |
MIMS Revision Date: 01 October 2021
Life threatening respiratory depression. Serious, life-threatening or fatal respiratory depression may occur with the use of Rikodeine Oral Liquid. Be aware of situations which increase the risk of respiratory depression, modify dosing in patients at risk and monitor patients closely, especially on initiation or following a dose increase (see Section 4.4 Special Warnings and Precautions for Use).
Concomitant use of benzodiazepines and other central nervous system (CNS) depressants, including alcohol. Concomitant use of opioids with benzodiazepines, gabapentinoids, antihistamines, tricyclic antidepressants, antipsychotics, cannabis or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Limit dosages and durations to the minimum required; and monitor patients for signs and symptoms of respiratory depression and sedation. Caution patients not to drink alcohol while taking Rikodeine Oral Liquid.
1 Name of Medicine
Dihydrocodeine tartrate and sorbitol.
2 Qualitative and Quantitative Composition
Each 10 mL of Rikodeine Oral Liquid contains dihydrocodeine tartrate 19 mg, and sorbitol 4.4 g. It also contains sucrose, citric acid, methyl hydroxybenzoate, amaranth, strawberry flavour 73858 and water.
Excipients with known effects. Sugars, hydroxybenzoates, sorbitol (26.4 g per 60 mL (maximum recommended daily dose).
3 Pharmaceutical Form
Oral liquid - pink coloured solution.
4 Clinical Particulars
4.1 Therapeutic Indications
For relief of stubborn, unproductive cough.
4.2 Dose and Method of Administration
See Table 1.

4.3 Contraindications
1. Hypersensitivity to dihydrocodeine, other opioids, or other components listed.
2. Opioid derivatives such as dihydrocodeine are generally but not always contraindicated in the following disorders: acute alcoholism, convulsive disorders, paralytic ileus, head injuries and conditions in which the intracranial pressure is raised. It should not be given to a comatose patient.
3. Severe respiratory disease, acute respiratory disease and respiratory depression.
4.4 Special Warnings and Precautions for Use
Hazardous and harmful use. Rikodeine Oral Liquid contains the opioid dihydrocodeine tartrate and is a potential drug of abuse, misuse and addiction. Addiction can occur in patients appropriately prescribed Rikodeine Oral Liquid at recommended doses. Abuse of dihydrocodeine has been reported.
The risk of addiction is increased in patients with a personal or family history of substance abuse (including alcohol and prescription and illicit drugs) or mental illness. The risk also increases the longer the drug is used and with higher doses. Patients should be assessed for their risks for opioid abuse or addiction prior to being prescribed Rikodeine Oral Liquid.
All patients receiving opioids should be routinely monitored for signs of misuse and abuse. Opioids are sought by people with addiction and may be subject to diversion. Strategies to reduce these risks include prescribing the drug in the smallest appropriate quantity and advising the patient on the safe storage and proper disposal of any unused drug (see Section 6.4 Special Precautions for Storage; Section 6.6 Special Precautions for Disposal). Caution patients that abuse of oral or transdermal forms of opioids by parenteral administration can result in serious adverse events, which may be fatal.
Patients should be advised not to share Rikodeine Oral Liquid with anyone else.
Respiratory depression. Serious, life-threatening or fatal respiratory depression can occur with the use of opioids even when used as recommended. It can occur at any time during the use of Rikodeine Oral Liquid but the risk is greatest during initiation of therapy or following an increase in dose. Patients should be monitored closely for respiratory depression at these times.
The risk of life-threatening respiratory depression is also higher in elderly, frail, or debilitated patients, in patients with renal and hepatic impairment, and in patients with existing impairment of respiratory function (e.g. chronic obstructive pulmonary disease; asthma). Opioids should be used with caution and with close monitoring in these patients (see Section 4.2 Dose and Method of Administration). The use of opioids is contraindicated in patients with severe respiratory disease, acute respiratory disease and respiratory depression (see Section 4.3 Contraindications).
Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol. Concomitant use of opioids and benzodiazepines or other CNS depressants, including alcohol, may result in sedation, respiratory depression, coma and death. Because of these risks, concomitant prescribing of Rikodeine Oral Liquid with CNS depressant medicines, such as other opioid analgesics, benzodiazepines, gabapentinoids, cannabis, sedatives, hypnotics, tricyclic antidepressants, antipsychotics, antihistamines, centrally-active anti-emetics and other CNS depressants, should be reserved for patients for whom other treatment options are not possible. If a decision is made to prescribe Rikodeine Oral Liquid concomitantly with any of the medicines, the lowest effective dose should be used, and the duration of treatment should be as short as possible. Patients should be followed closely for signs and symptoms of respiratory depression and sedation. Patients and their caregivers should be made aware of these symptoms. Patients and their caregivers should also be informed of the potential harms of consuming alcohol while taking Rikodeine Oral Liquid.
Tolerance, dependence and withdrawal. Neuroadaptation of the opioid receptors to repeated administration of opioids can produce tolerance and physical dependence. Tolerance is the need for increasing doses to maintain analgesia. Tolerance may occur to both the desired and undesired effects of the opioid.
Physical dependence, which can occur after several days to weeks of continued opioid usage, results in withdrawal symptoms if the opioid is ceased abruptly or the dose is significantly reduced. Withdrawal symptoms can also occur following the administration of an opioid antagonist (e.g. naloxone) or partial agonist (e.g. buprenorphine). Withdrawal can result in some or all of the following symptoms: dysphoria, restlessness/agitation, lacrimation, rhinorrhoea, yawning, sweating, chills, myalgia, mydriasis, irritability, anxiety, increasing pain, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhoea, increased blood pressure, increased respiratory rate and increased heart rate.
When discontinuing Rikodeine Oral Liquid in a person who may be physically-dependent, the drug should not be ceased abruptly but withdrawn by tapering the dose gradually.
Accidental ingestion/exposure. Accidental ingestion or exposure of Rikodeine Oral Liquid, especially by children, can result in a fatal overdose of dihydrocodeine tartrate. Patients and their caregivers should be given information on safe storage and disposal of unused Rikodeine Oral Liquid (see Section 6.4 Special Precautions for Storage; Section 6.6 Special Precautions for Disposal).
Ceasing opioids. Abrupt discontinuation or rapid decreasing of the dose in a person physically dependent on an opioid may result in serious withdrawal symptoms (see Tolerance, dependence and withdrawal). Such symptoms may lead the patient to seek other sources of licit or illicit opioids. Opioids should not be ceased abruptly in a patient who is physically dependent but withdrawn by tapering the dose slowly. Factors to take into account when deciding how to discontinue or decrease therapy include the dose and duration of the opioid the patient has been taking and the physical and psychological attributes of the patient. During tapering, patients require regular review and support to manage any psychological distress and withdrawal symptoms.
There are no standard tapering schedules suitable for all patients and an individualised plan is necessary. In general, tapering should involve a dose reduction of no more than 10 percent to 25 percent every 2 to 4 weeks (see Section 4.2 Dose and Method of Administration). If the patient is experiencing serious withdrawal symptoms, it may be necessary to go back to the previous dose until stable before proceeding with a more gradual taper.
When ceasing opioids in a patient who has a suspected opioid use disorder, the need for medication assisted treatment and/or referral to a specialist should be considered.
Lung. As dihydrocodeine may cause the release of histamine, it should be given with caution in asthmatics, in patients with decreased respiratory reserve (e.g. emphysema), cor pulmonale or chronic obstructive respiratory disease (see Section 4.4 Special Warnings and Precautions for Use, Respiratory depression).
Gastrointestinal. Opioids should be given with caution or in reduced doses to patients with inflammatory or obstructive bowel disorders, biliary tract disorders or inflammation of the pancreas. Rikodeine Oral Liquid contains 26.4 g sorbitol in 60 mL (maximum daily dose), which may have a laxative effect or cause diarrhoea in some people.
Others. Dosage of dihydrocodeine should be reduced in hypothyroidism. Opioids should be given with caution or in reduced doses to patients with adrenocortical insufficiency, prostatic hyperplasia, hypotension, shock, or myasthenia gravis.
Use in hepatic impairment. Dosage of dihydrocodeine should be reduced in chronic hepatic disease.
Use in renal impairment. Caution is advised in patients with severe renal impairment as dose accumulation may occur. In the elderly with lower renal clearance, there is marked variability in the pharmacokinetics so small doses may be needed initially (see Section 5.2 Pharmacokinetic Properties, Metabolism and excretion).
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Rikodeine Oral Liquid may enhance the effects of CNS depressants and may result in sedation, respiratory depression, coma and death. Examples of CNS depressants include other opioid analgesics, benzodiazepines, gabapentinoids, cannabis, sedatives, hypnotics, tranquillisers, tricyclic antidepressants, antipsychotics, centrally-active anti-emetics, antihistamines and other CNS depressants, including alcohol (see Section 4.4 Special Warnings and Precautions for Use, Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol).
Quinidine. Two studies (n = 4 and n = 10) and biochemical evidence suggest that the analgesic effects of dihydrocodeine may be reduced or lost if quinidine is administered concurrently in extensive metabolisers (CYP2D6) of codeine. This is the majority of the population. Poor metabolisers of codeine will not be affected. About 6 to 10% of Caucasians lack this enzyme. The incidence of lack of this enzyme is lower in South-East Asians.
The possibility of interactions between dihydrocodeine and other drugs which can inhibit the enzyme CYP2D6, such as phenothiazines and antipsychotic agents should be considered.
Consideration should also be given to potential interactions with other medications, which occur with other opioids:
medications with anticholinergic effects such as antihistamines with anticholinergic effects, or tricyclic antidepressants, which may increase constipation and/or urinary retention;
antihypertensive medications (possible additive hypotensive effects);
antiperistaltic antidiarrhoeals (may increase risk of severe constipation);
monoamine oxidase (MAO) inhibitors (may lead to anxiety, confusion, severe respiratory depression);
neuromuscular blockers (may lead to additive respiratory depressant effects);
opioid agonists (e.g. codeine, morphine, pethidine, etc) may increase the toxic effects of dihydrocodeine;
opioid antagonists (e.g. naloxone).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Medicines have been taken by large numbers of pregnant women and women of child-bearing age for many years without proven increase in the frequency of malformations or other direct or indirect harmful effects on the foetus having been observed. As with all medicines the associated risk to the foetus should be considered against the anticipated benefit to the mother.
Prolonged consumption late in pregnancy may risk causing respiratory depression and withdrawal symptoms in the neonate.
Use in lactation. No information is available. Rikodeine Oral Liquid should not be used in lactation unless the benefits outweigh the possible risk to the infant.
4.7 Effects on Ability to Drive and Use Machines
Dihydrocodeine may cause sedation and may impair the ability of the patient to drive or operate machinery. Patients treated with dihydrocodeine should be cautioned that their ability might be reduced.
4.8 Adverse Effects (Undesirable Effects)
More common reactions. Constipation, although recorded, is less common than with codeine. Drowsiness, nausea, vomiting, headache, respiratory depression and vertigo may occur.
In 12 healthy volunteers, opiate-related side effects occurred especially with higher steady-state doses: headache, nausea, vomiting, dizziness, constipation, stomach pain, mild urinary retention, dry mouth and mild euphoria. The side effects were mild and reversible, and none of the subjects had to discontinue the study.
In another randomised, placebo controlled, blinded, cross-over study of healthy volunteers (n=100, 95 completed trial), a significant incidence of nausea and dizziness was observed with dihydrocodeine, but other side effects were not significantly different (p=0.05) to the level after administration with placebo.
In a randomised, placebo controlled, double-blind, cross-over study of 18 patients with chronic obstructive airways disease, there were no significant differences in peak flow, drowsiness, nausea, constipation or anxiety.
Less common reactions. Other adverse effects have been infrequently reported include central nervous system effects such as dizziness, visual disturbances and hallucinations, cardiovascular effects such as atrial fibrillation, circulatory failure and syncope and dermal effects such as pruritus, rash and urticaria.
Rikodeine contains sorbitol, which may have a laxative effect or cause diarrhoea in some people.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Symptoms and signs. The blood levels of dihydrocodeine found in impaired individuals and in fatalities show a wide overlap in ranges. In three fatal cases the amount of parent drug always exceeded dihydrocodeine-glucuronide formation and dihydromorphine concentrations ranged from 0.16 to 0.21 mg/mL. Dihydrocodeine overdose is characterised by pinpoint pupils, respiratory depression (reduced respiratory rate and/or tidal volume, Cheynes-Stokes respiration, cyanosis) extreme somnolence progressing to stupor or coma, flaccidity of skeletal muscle, cold or clammy skin and sometimes hypotension and bradycardia. Overdose has been associated with evidence of acute renal failure and hepatic impairment. Continued overdosage may result in apnoea, circulatory collapse, cardiac arrest and death.
Treatment. Primary attention should be given to the re-establishment of adequate respiratory exchange through the provision of a patent airway, supplemental oxygen and controlled or assisted ventilation with maintenance of the fluid balance. The narcotic antagonist, naloxone hydrochloride, is a specific antidote and an appropriate dose should be administered as necessary, in accordance with the patient's response.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Cough suppressant with analgesic properties.
Dihydrocodeine is a semi-synthetic opioid, which is frequently used as an analgesic and antitussive/ cough suppressant drug. It is formulated in Rikodeine Oral Liquid as an antitussive. As an antitussive, the recommended dose for dihydrocodeine tartrate is lower than the usual recommended dose for analgesia. The antitussive effects of dihydrocodeine are mediated through direct action on receptors in the cough centre of the medulla.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Following oral administration of dihydrocodeine tartrate 30 mg and 60 mg to seven healthy volunteers, the serum concentration-time curves indicated a rapid absorption of dihydrocodeine with mean peak serum concentrations were achieved at 1.6 to 1.8 hours. The rate of absorption was independent of administered dose. The mean half-lives varied between 3.3 to 4.5 h. The mean bioavailability of orally administered dihydrocodeine was 21% (range 12-34%). After intravenous administration of 30 mg dihydrocodeine tartrate (n=7 healthy volunteers), the peak serum levels were significantly greater (over 8 times higher) than those achieved after oral dosing (30 mg or 60 mg). The peak serum levels of the acidic metabolites (dihydromorphine and dihydrocodeine glucuronide) occurred between 1.8 h to 2.0 h after oral administration and 2.2 to 2.5 h after intravenous administration and were significantly higher after oral administration. The low oral bioavailability of dihydrocodeine, together with the earlier and higher plasma levels of the acid metabolites after oral dosing compared to IV dosing, is suggestive of substantial first-pass metabolism.
In the dose range 60 to 120 mg as single and multiple doses the pharmacokinetics of dihydrocodeine and its active metabolite dihydromorphine are linear.
Pharmacokinetics in elderly. The effect of age on the pharmacokinetics of dihydrocodeine was investigated in 8 elderly (74 to 90 years) patients and 8 young (21 to 29 years) volunteers. After multiple oral dosing of dihydrocodeine 30 mg four times a day for 3 days, the maximum plasma concentration of dihydrocodeine was significantly higher in the elderly (199 ± 155 nanogram/mL) than in the young (174 ± 53 nanogram/mL); and the area under the plasma concentration-time curve in the elderly was approximately 25% higher than in the young which was considered to be likely to be clinically significant. The greater plasma concentrations in the elderly compared to the young was more likely due to a decrease in first pass effect in the elderly patients. Results from this study suggest that the recommended doses in the elderly may need to be reduced.
Pharmacokinetics in chronic renal failure patients. The pharmacokinetics of a single 60 mg oral dose of dihydrocodeine were studied in 9 patients with chronic renal failure treated by haemodialysis and 9 subjects with normal renal function. In patients with chronic renal failure, the mean peak plasma dihydrocodeine concentration occurred later and the area under the plasma concentration-time curve was greater than in the normal subjects. Furthermore, the drug was still detectable after 24 hours in all the patients with renal failure but was only detectable in only three of the normal subjects. Possible explanations for these findings include differences in absorption, the volume of distribution, the rate of metabolism, and the rate of excretion of the drug.
Metabolism and excretion. The metabolism of dihydrocodeine includes O-demethylation to dihydromorphine; N-demethylation to nordihydromorphine; and conjugation of parent drug and hydroxylated metabolites with glucuronic acid.
The cytochrome P450-2D6 (CYP2D6) is the major enzyme mediating O-demethylation of dihydrocodeine to dihydromorphine. In contrast, nordihydrocodeine formation is predominantly catalysed by cytochrome P450-3A (CYP3A).
The pharmacokinetics of dihydrocodeine was studied in subjects with differing rates of metabolism via the enzyme CYP2D6, in six extensive (metabolic ratio < 1), two intermediate (1 < MR < 20) and six poor metabolisers (MR ≥ 20) of sparteine/ debrisoquin, were compared following administration of a single oral dose of dihydrocodeine. Results from this study showed that there were no significant differences in the pharmacokinetics of dihydrocodeine between extensive and poor metabolisers in maximum serum concentration, area under the curve and terminal half-life. However, the area under the serum concentration versus time curve and total urinary recovery of dihydromorphine were significantly lower in poor metabolisers compared with extensive metabolisers. No significant differences between extensive and poor metabolisers were detected in urine for conjugated dihydrocodeine (approximately 30%), unconjugated dihydrocodeine (approximately 31%), conjugated nordihydrocodeine (5-6%) or unconjugated nordihydrocodeine (16-20%). In conclusion, this study demonstrated that following oral administration, dihydrocodeine is mainly excreted in urine as the parent compound or its conjugates in extensive and poor metabolisers. The O-demethylation of dihydrocodeine to dihydromorphine (mainly by CYP2D6) is impaired in poor metabolisers: the dihydromorphine metabolite, only accounts for 0.4-2.8% of the dose in the poor CYP2D6 metabolisers and 3.5-17.1% in the extensive CYP2D6 metabolisers.
Consideration should be given to the possibility that patients who metabolise drugs poorly via CYP2D6 may obtain reduced benefit from dihydrocodeine due to reduced formation of an active metabolite, dihydromorphine.
In healthy adult males, CYP2D6 extensive metabolisers (n=12), the pharmacokinetics of dihydrocodeine and its active metabolite dihydromorphine following multiple oral dosing of 60-120 mg dihydrocodeine are shown to be linear.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine (also see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Amber PET bottle, 30 mL*, 100 mL, 200 mL.
* Not currently marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. Dihydrocodeine tartrate.
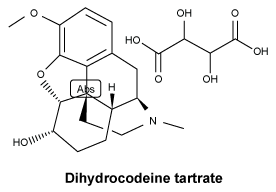
It is odourless or almost odourless colourless crystals or a white crystalline powder. It is freely soluble in water and is sparingly soluble in alcohol.
Molecular formula: C22H29NO9.
CAS number. 5965-13-9.
Sorbitol.
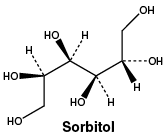
CAS number. 50-70-4.
7 Medicine Schedule (Poisons Standard)
(S3) Pharmacist Only Medicine.
Date of First Approval
04 July 1991
Date of Revision
24 August 2021
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.