Rozex Gel
Brand Information
| Brand name | Rozex Gel |
| Active ingredient | Metronidazole |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Rozex Gel.
Summary CMI
ROZEX® GEL
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Rozex gel?
ROZEX GEL contains the active ingredient metronidazole. ROZEX GEL is used to treat the inflammatory papules and pustules of rosacea.
For more information, see Section 1. Why am I using ROZEX GEL? in the full CMI.
2. What should I know before I use ROZEX GEL?
Do not use if you have ever had an allergic reaction to ROZEX GEL or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions including Cockayne Syndrome, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ROZEX GEL? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ROZEX GEL and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ROZEX GEL?
- Apply morning and evening to clean dry skin.
More instructions can be found in Section 4. How do I use ROZEX GEL? in the full CMI.
5. What should I know while using ROZEX GEL?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ROZEX GEL? in the full CMI.
6. Are there any side effects?
Dry skin, reddening of the skin, severe itching, skin discomfort, skin irritation, worsening of rosacea, hyperpigmentation and other pigmentation disorders, contact dermatitis, reduced sense of touch or sensation, tingling or pricking sensation (pins and needles), numbness in the extremities, metallic taste, seborrhoea (red, itchy rash and white scales), sunburn, skin infection, hives, gastritis, bronchitis, stuffy runny nose, skin exfoliation, swelling face, nausea, excess watery fluid collection on the face or eyelids, hypothyroidism, Bursitis (inflammation or irritation of the bursa), Myalgia (muscle pain), osteoporosis, conjunctivitis, abscesses.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using ROZEX GEL?
ROZEX GEL contains the active ingredient metronidazole.
ROZEX GEL is an antiprotozoal and antibacterial medicine.
ROZEX GEL is used for the treatment of inflammatory papules and pustules of rosacea.
2. What should I know before I use ROZEX GEL?
Warnings
Do not use ROZEX GEL if:
- You are allergic to metronidazole, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- You have Cockayne Syndrome.
- You are pregnant.
Check with your doctor if you:
- have any other medical conditions
- take any medicines for any other condition
- are breastfeeding
- are planning to become pregnant
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
With not enough information available on the effect of ROZEX GEL on pregnant women, ROZEX GEL is not recommended during pregnancy.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Oral metronidazole has been shown to boost the effect of warfarin and other coumarin anticoagulants. It is unknown how ROZEX GEL may interact with these products.
Some medicines may interfere with ROZEX GEL and affect how it works.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ROZEX GEL.
4. How do I use ROZEX GEL?
How much to use
- Wash and dry your hands before applying the gel.
- Squeeze a small quantity of ROZEX GEL on to your fingertips and rub a thin film onto the affected areas.
- Avoid getting ROZEX GEL in your eyes.
- Use a sunscreen when exposure to the sun cannot be avoided.
- ROZEX GEL is only for use on the skin.
- Follow the instructions provided and use ROZEX GEL until your doctor tells you to stop.
When to use ROZEX GEL
- ROZEX GEL should be used morning and evening on a clean dry face.
- Continue to use it even though there may not be an immediate effect. Medicines can take several weeks to show an effect. The average period of treatment is usually three to four months. The recommended duration should not be exceeded.
If you forget to use ROZEX GEL
ROZEX GEL should be used regularly at the same times each day. If you miss your dose at the usual time, use it again as directed, as soon as you remember.
If it is almost time for your next dose, skip the dose you missed and apply your next dose when you are meant to.
Do not apply a double dose to make up for forgotten individual doses.
- If you have trouble remembering to use your medicine, ask your pharmacist for some hints.
If you use too much ROZEX GEL
If you think that you have used too much ROZEX GEL, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using ROZEX GEL?
Things you should do
Call your doctor straight away if you:
- Think you have an allergy.
- Symptoms of an allergic reaction may include:
- swelling of the face, lips, tongue, or other parts of the body
- rash, itching, or hives of the skin, especially on the areas where the gel was applied. - if the condition worsens / does not improve, go to the emergency department at your nearest hospital.
Remind any doctor, dentist or pharmacist you visit that you are using ROZEX GEL.
Things you should not do
- Avoid ROZEX GEL coming into contact with eyes or mucous membranes. If accidental contact occurs, rinse thoroughly with water and pat dry.
- Do not use cosmetic products that contain alcohol or are abrasive.
- Do not use medicated soaps. These may have an extra drying or irritant effect.
- Unnecessary or prolonged use of ROZEX GEL should be avoided as the long-term safety is unknown.
- Do not use ROZEX GEL after the expiry date (EXP) printed on the pack has passed, or if the packaging is torn or shows signs of tampering.
- If it has expired or is damaged, return it to your pharmacist for disposal.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ROZEX GEL affects you.
Drinking alcohol
Tell your doctor if you drink alcohol.
Alcohol has been shown to interact with metronidazole when taken orally.
Looking after your medicine
- Keep the medicine in a cool dry place where the temperature stays below 25°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Rare side effects
| Rare side effects | What to do |
| Speak to your doctor, if you notice any of these rare side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ROZEX GEL contains
| Active ingredient (main ingredient) | 7.5 mg per gram metronidazole |
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What ROZEX GEL looks like
ROZEX GEL is a homogeneous gel in an aluminium tube with a screw cap containing 2g, 5g, 15g, 30g, and 50g of gel.
Not all pack sizes may be available.
(AUST R 15211).
Who distributes ROZEX GEL
Galderma Australia Pty Ltd
Level 18, 1 Denison Street,
North Sydney, NSW 2060
Telephone 1800 800 765
Distributed in New Zealand by:
Healthcare Logicstics
58 Richard Pearse Drive
Airport Oaks
Auckland
Telephone 0800 174 104
Made in France
® Registered trademark
This leaflet was prepared in January 2026
Brand Information
| Brand name | Rozex Gel |
| Active ingredient | Metronidazole |
| Schedule | S4 |
MIMS Revision Date: 01 March 2026
1 Name of Medicine
Metronidazole.
2 Qualitative and Quantitative Composition
Rozex Gel contains metronidazole 7.5 mg/g in an aqueous gel.
Excipients with known effect. Contains hydroxybenzoates.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Rozex Gel is a colourless to pale yellow viscous and homogeneous gel with no foreign matter.
4 Clinical Particulars
4.1 Therapeutic Indications
Treatment of inflammatory papules, pustules and erythema of rosacea.
4.2 Dose and Method of Administration
Adults. Apply and rub in a thin film of gel twice daily, morning and evening, to entire affected areas of the skin after washing.
Elderly. The dosage recommended in the elderly is the same as that recommended in adults.
Children. Not recommended.
Areas to be treated should be cleansed before application of gel. Patients may use cosmetics after application of the product.
Significant therapeutic results should be noted within three weeks. Clinical studies have demonstrated continuing improvement over nine weeks of therapy. In the absence of a clear clinical improvement, therapy should be stopped.
The average period of treatment is usually of three to four months. The recommended duration of treatment should not be exceeded.
4.3 Contraindications
Contraindicated in individuals with a history of hypersensitivity to metronidazole, hydroxybenzoates or other ingredients of the formulation.
Metronidazole must not be used in patients with Cockayne syndrome. Cases of severe hepatotoxicity/acute hepatic failure, including cases with a fatal outcome with very rapid onset after treatment initiation in patients with Cockayne syndrome have been reported with products containing metronidazole for systemic use.
4.4 Special Warnings and Precautions for Use
Rozex has been reported to irritate the eyes (watering), therefore contact with the eyes and mucous membranes should be avoided. If a reaction suggesting local irritation occurs, patients should be directed to use the medication less frequently, discontinue use temporarily or discontinue use until further instructions. Metronidazole is a nitroimidazole compound and should be used with care in patients with evidence or a history of blood dyscrasia.
Metronidazole transforms into inactive metabolite due to UV exposure, therefore its efficacy decreases significantly. Phototoxic side-effects haven't been reported in clinical trials in relation to metronidazole.
Patients should be advised to avoid or minimise exposure of areas treated with topical metronidazole to sunlight or other sources of UV light (see Section 5.3 Preclinical Safety Data, Carcinogenicity). Unnecessary or prolonged use of this medication should be avoided, as the long-term safety of topical metronidazole is unknown.
Use in the elderly. No data available.
Paediatric use. Rozex (metronidazole 0.75%) gel has not been studied in children. Rosacea is a skin disorder which principally affects adults. Rozex is not recommended for use in children due to a lack of data on safety and efficacy.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Drug interactions are less likely with topical administration but should be kept in mind when Rozex is prescribed for patients who are receiving anticoagulant treatment. Nevertheless, it should be mentioned that disulfiram-like reactions has been reported in small number of patients taking metronidazole and alcohol concomitantly.
Oral metronidazole has been reported to potentiate the anticoagulant effect of coumarin and warfarin resulting in a prolongation of prothrombin time. The effect of topical metronidazole on prothrombin is not known.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Oral metronidazole caused hypospermatogenesis, infertility and abnormal spermatozoa in mice and rats with a NOEL in rats being about 200 times the estimated human metronidazole dose contained in the Rozex Gel, based on body surface area.
Use in pregnancy. (Category B2)
There is no experience to date with the use of Rozex in pregnancy. In case of oral administration, metronidazole crosses the placental barrier and rapidly enters the foetal circulation. There is inadequate evidence of the safety of metronidazole in human pregnancy. In animal studies metronidazole was not teratogenic or embryotoxic unless administered at extremely high doses. Because there are no well-controlled studies of therapy with Rozex Gel in pregnant women, Rozex Gel should not be used during pregnancy.
Use in lactation. After oral administration metronidazole is excreted in breast milk in concentrations similar to those found in the plasma. Metronidazole blood levels from topical application are significantly lower than those achieved after oral metronidazole. A decision should be made to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Because of the minimal absorption of metronidazole, and consequently its insignificant plasma concentration after topical administration, the adverse experiences reported with the oral form of the drug have not been reported with Rozex. Adverse reactions reported with Rozex include eye irritation (watering) if the gel is applied too closely to this area, transient redness, mild dryness, burning and skin irritation. None of the side effects exceeded an incidence of 2% of patients.
The following spontaneous adverse experiences have been reported, and within each system organ class, are ranked by frequency, using the following convention: very common (≥ 1/10); common (≥ 1/100, < 1/10); uncommon (≥ 1/ 1,000, < 1/100); rare (≥ 1/10,000, < 1/1,000); very rare (< 1/10,000), including isolated reports.
Skin and subcutaneous tissue disorders. Common: dry skin, erythema, pruritus, skin discomfort (burning, pain of skin/stinging), skin irritation, worsening of rosacea. Unknown frequency: contact dermatitis, skin exfoliation, swelling face (*), seborrhoea, skin infection, sunburn, urticaria.
Nervous system disorders. Uncommon: hypoesthesia, paraesthesia, dysgeusia (metallic taste), dizziness.
Gastrointestinal disorders. Uncommon: nausea, gastritis.
Respiratory system disorders. Uncommon: bronchitis, rhinitis.
Endocrine disorders. Rare: hypothyroidism.
Musculoskeletal. Rare: bursitis, myalgia, osteoporosis.
Special senses. Rare: conjunctivitis.
Body as a whole. Uncommon: abscess, accidental injury, flu symptom, infection.
Post-marketing experience. The following non-serious adverse experiences have been reported since 1995: contact dermatitis/ allergic reaction; skin exfoliation, swelling face, local irritation, erythema, pruritus, burning, dryness, tightness, discomfort, rash, hyperpigmentation, pigmentation disorders, hypertrichosis, facial oedema, eyelid oedema, treatment failure (worsening of rosacea); watery eyes, metallic taste, tingling or numbness in the extremities, nausea, other (zoster lesion, pustules on the nose and vesicular bullous eruptions). The causal relationship with topical metronidazole has not been unequivocally established for these adverse experiences.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There is no human experience with overdosage of Rozex. The acute oral toxicity of the Rozex formulation was determined to be greater than 5 g/kg (the highest dose given) in albino rats.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Metronidazole is an antiprotozoal (trichomoniasis, amoebiasis, giardiasis) and anaerobic antibacterial agent. However, the mechanisms by which Rozex acts in reducing inflammatory lesions of rosacea are unknown, but may include an antibacterial and/or anti-inflammatory effect.
Clinical trials.
5.2 Pharmacokinetic Properties
The absorption of metronidazole following topical administration is negligible. Studies on the topical administration of 1 gram of Rozex (7.5 mg of metronidazole) to the face of 10 rosacea patients showed a maximum serum concentration of 66 nanogram/mL in one patient. This concentration is approximately 100 times less than concentrations afforded by a single 250 mg tablet. The serum metronidazole concentrations were below the detectable limits of the assay at the majority of time points in all patients. Three of the patients had no detectable serum concentrations of metronidazole at any time point. The mean dose of gel applied during clinical studies was 600 mg, which represents 4.5 mg of metronidazole per application. Therefore, under normal usage levels, the formulation affords minimal serum concentrations of metronidazole.
5.3 Preclinical Safety Data
Genotoxicity. Metronidazole has shown evidence of mutagenic activity in several bacterial systems. In addition, a dose response increase in the frequency of micronuclei was observed in mice after intraperitoneal injection and an increase in chromosome aberrations has been found in human lymphocyte cultures. The benefit/risk ratio should therefore be carefully assessed in each case particularly in relation to the severity of the disease and the age of the patient.
Carcinogenicity. Animal studies with oral metronidazole showed increased incidences of tumour in the lung, liver, testes, reticulum, mammary gland and pituitary gland in certain rodent species. Evidence of photocarcinogenicity of metronidazole has also been reported in mice. Although there is no evidence to date of a carcinogenic effect in humans it is prudent to avoid unnecessary and prolonged use of Rozex Gel and to avoid or to minimise exposure of sites treated with Rozex Gel to the sun.
6 Pharmaceutical Particulars
6.1 List of Excipients
Methyl and propyl hydroxybenzoates, propylene glycol, carbomer 940, disodium edetate, sodium hydroxide and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Gel. 2 g, 5 g, 15 g, 30 g and 50 g tubes. Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Metronidazole is 1- (2 hydroxyethyl)-2-methyl-5-nitroimidazole. It is a white to brownish crystalline powder that is soluble in water.
Chemical structure.
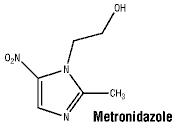
CAS number. 443-48-1.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
08 February 1993
Date of Revision
07 January 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.