Ryzodeg 70/30 FlexTouch 3mL
Brand Information
| Brand name | Ryzodeg 70/30 FlexTouch 3mL |
| Active ingredient | Insulin degludec + Insulin aspart |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Ryzodeg 70/30 FlexTouch 3mL.
Summary CMI
Ryzodeg® 70/30 Penfill®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
▼ This medicine is new or being used differently. Please report side effects. See the full CMI for further details.
1. Why am I using Ryzodeg® 70/30 Penfill®?
Ryzodeg® 70/30 Penfill® contains two types of insulin: a long-acting insulin, called insulin degludec; and a fast-acting insulin, called, insulin aspart. Ryzodeg® 70/30 Penfill® is used to treat diabetes mellitus in patients aged 6 years and above.
For more information, see Section 1. Why am I using Ryzodeg® 70/30 Penfill®? in the full CMI.
2. What should I know before I use Ryzodeg® 70/30 Penfill®?
Do not use if you have ever had an allergic reaction to insulin or any of the ingredients listed at the end of the CMI.
Do not use if you are experiencing low blood sugar ("hypo") when your dose is due.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Ryzodeg® 70/30 Penfill®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with insulin and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Ryzodeg® 70/30 Penfill®?
Carefully follow all instructions given to you by your doctor, nurse educator and pharmacist regarding how to use your medicine, including preparing and injecting it.
More instructions can be found in Section 4. How do I use Ryzodeg® 70/30 Penfill®? in the full CMI.
5. What should I know while using Ryzodeg® 70/30 Penfill®?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Ryzodeg® 70/30 Penfill®? in the full CMI.
6. Are there any side effects?
The most common side effect when using insulin is low blood sugar levels (a hypo). Tell your relatives, friends, close workmates, teachers or carers that you have diabetes. It is important that they can recognise the signs and symptoms of a hypo. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
▼ This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems.
Ryzodeg® 70/30 Penfill®
Active ingredient(s): 70% insulin degludec and 30% insulin aspart
Consumer Medicine Information (CMI)
This leaflet provides important information about using Ryzodeg® 70/30 Penfill®. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Ryzodeg® 70/30 Penfill®
Where to find information in this leaflet:
1. Why am I using Ryzodeg® 70/30 Penfill®?
2. What should I know before I use Ryzodeg® 70/30 Penfill®?
3. What if I am taking other medicines?
4. How do I use Ryzodeg® 70/30 Penfill®?
5. What should I know while using Ryzodeg® 70/30 Penfill®?
6. Are there any side effects?
7. Product details
1. Why am I using Ryzodeg® 70/30 Penfill®?
Ryzodeg® 70/30 Penfill® contains two types of insulin:
a long-acting insulin, called insulin degludec; and
a fast-acting insulin, called insulin aspart.
Ryzodeg® 70/30 Penfill® is used to treat diabetes mellitus in patients aged 6 years and above.
Ryzodeg® 70/30 is a modified insulin, also called an insulin analogue, which is similar to the insulin produced by the pancreas.
There are two types of diabetes mellitus:
Type 1 diabetes
Type 2 diabetes
Patients with type 1 diabetes always require insulin to control their blood sugar levels.
Some patients with type 2 diabetes may also require insulin after initial treatment with diet, exercise and tablets is not enough.
Ryzodeg® 70/30 may be used in combination with other antidiabetic drugs (for example, diabetes tablets) or with a meal-related short- or rapid-acting insulin, such as NovoRapid®.
Ryzodeg® 70/30 can either be used once or twice each day with the main meal(s). When needed, you can change the time of dosing as long as Ryzodeg® 70/30 is dosed with the main meal(s). The effect may last for more than 24 hours.
As with all insulins, the duration of action will vary according to the type of insulin, the dose, injection site, blood flow, temperature and level of physical activity.
Insulin is not addictive. Insulin is available only with a doctor's prescription.
2. What should I know before I use Ryzodeg® 70/30 Penfill®?
Warnings
If you are blind or have poor eyesight and cannot read the dose counter on your insulin delivery device, do not use it without help. Get help from a person with good eyesight who is trained to use the device.
Do not use Ryzodeg® 70/30 Penfill®:
- if you are allergic to insulin, or any product containing insulin, or any of the ingredients listed at the end of this leaflet.
- if you are experiencing low blood sugar level (a "hypo") when your dose is due.
- if the expiry date printed on the pack has passed, or if the packaging is torn or shows signs of tampering.
- in insulin infusion pumps.
Never inject Ryzodeg® 70/30 into a vein or muscle.
Check with your doctor if you:
- are experiencing a lot of hypos, and follow their advice
- have any other medical conditions, such as: kidney liver, adrenal gland, pituitary gland or thyroid gland problems, heart disease or stroke
- take any medicines for any other condition
- have an infection or fever
- are planning to travel.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. Pregnancy can make managing your diabetes more difficult, and may require adjustment of how much insulin you need.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Children
Ryzodeg® 70/30 Penfill® can be used in adolescents and children from the age of 6 years. Ryzodeg® 70/30 Penfill® should be used with special caution in these patients. The risk for very low blood sugar levels may be higher in children and adolescents.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with insulin and affect how it works.
Medicines that may increase the effect of insulin, and therefore reduce insulin requirements include:
- other medicines used to treat type 2 diabetes
- octreotide - used to treat gastrointestinal endocrine tumours and enlargement of parts of the body (e.g. hands, feet, head) caused by abnormal growth hormone levels
- lanreotide - used to treat enlargement of parts of the body (e.g. hands, feet, head) caused by abnormal hormone levels
- monoamine oxidase inhibitors (MAOIs) - used to treat depression
- non-selective beta-blockers - used to treat certain heart conditions and high blood pressure. (They may mask the symptoms of hypoglycaemia and delay recovery from hypoglycaemia)
- alpha-blockers - used to treat high blood pressure and to relieve difficulty in passing urine caused by an enlarged prostate
- angiotensin converting enzyme (ACE) inhibitors - used to treat certain heart conditions, high blood pressure and elevated protein/albumin in the urine
- salicylates, e.g. aspirin - used to relieve pain and lower fever
- anabolic steroids - used to promote growth
- quinine - used to treat malaria and relieve muscle cramps
- quinidine - used to treat heart problems
- sulphonamides - a type of antibiotic used to treat bacterial infections.
Medicines that may reduce the effect of insulin, and increase insulin requirements include:
- oral contraceptives ("the pill") - used for birth control
- thiazides, frusemide or ethacrynic acid - used to treat high blood pressure or fluid retention (oedema)
- glucocorticoids (except when applied locally) - used to treat inflammatory conditions
- thyroid hormones - used to treat malfunction of the thyroid gland
- sympathomimetics - used to treat asthma
- octreotide - used to treat gastrointestinal endocrine tumours and enlargement of parts of the body (e.g. hands, feet, head) caused by abnormal growth hormone levels
- lanreotide - used to treat enlargement of parts of the body (e.g. hands, feet, head) caused by abnormal hormone levels
- growth hormone - used to treat growth disorders
- diazoxide - used to treat high blood pressure
- asparaginase - used to treat leukaemia and lymph gland tumours
- nicotinic acid - used to treat high cholesterol levels in the blood
- oxymetholone - used to treat certain blood disorders
- danazol - used to treat endometriosis, menorrhagia, fibrocystic breast disease and hereditary angioedema.
Beta-blockers may make it harder to recognise the warning signs of too low blood sugar (hypoglycaemia).
Thiazolidinediones – some patients with long-standing type 2 diabetes and heart disease, or previous stroke, treated with thiazolidinediones in combination with insulin experienced the development of symptoms of heart failure. Tell your doctor straight away if you have signs of heart failure such as shortness of breath, tiredness, fluid retention, rapid weight gain and ankle swelling.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Ryzodeg® 70/30 Penfill®
4. How do I use Ryzodeg® 70/30 Penfill®?
Your doctor, diabetes education nurse or pharmacist will have given you advice on how to use your medicine. Carefully follow all the directions.
They may differ from the information contained in this leaflet.
If you are blind or have poor eyesight and cannot read the dose counter on your insulin delivery device, get help from a person with good eyesight who is trained to use the device.
Any change in dose or type of insulin should be made cautiously and only under medical supervision.
If you change the type of insulin that you use, you may have to use more or less than before. This may happen with the first dose or over a period of time.
If you do not understand the instructions, ask your doctor, diabetes education nurse or pharmacist for help.
How much to use
- Your doctor or diabetes education nurse will tell you how much of this medicine you need to use each day.
- It is very important that you manage your diabetes carefully. Too much or too little insulin can cause serious effects.
Based on your blood sugar level your doctor may change your dose.
When using other medications, ask your doctor if your treatment needs to be adjusted.
When to use Ryzodeg® 70/30 Penfill®
- Your doctor or diabetes education nurse will tell you when and how often to inject your insulin.
- Ryzodeg® 70/30 can either be used once or twice each day
- Use with the main meal(s) - you can change the time of dosing as long as Ryzodeg® 70/30 is dosed with the main meal(s).
- If you want to change your usual diet, check with your doctor, pharmacist or diabetes education nurse first as a change in diet may later your need for insulin.
How to inject Ryzodeg® 70/30 Penfill®
- Penfill® cartridges are designed to be used with Novo Nordisk insulin delivery devices (such as NovoPen®), and NovoFine® needles.
- Make sure you are using the correct type and strength of insulin.
- Carefully follow the instructions on how to use your Penfill® in the Novo Nordisk insulin delivery device.
- If you use more than one type of insulin in a Penfill®, you must use a separate insulin delivery device for each type.
- Do not use the insulin level indicator on the device to measure your dose of insulin.
- Inject insulin under the skin (subcutaneous injection) as shown to you by your doctor or diabetes education nurse.
- Take care not to drop or knock the device that contains Penfill®.
Checking your Penfill® before injection
- Check your Penfill® every time you are preparing your injection to make sure you are using the correct type and strength of insulin.
- Do not use your Ryzodeg® 70/30 Penfill® if the insulin is thickened, coloured, or if it has solid bits in it.
- Do not use your Penfill® if you notice any damage, including any damage to the rubber plunger (stopper) or if there is a gap between the plunger and the white label band.
Preparing your Penfill® for injection
- If Penfill® is already inside your insulin delivery device, check the amount of insulin remaining. If there is insufficient insulin for your dose, follow the relevant instructions in the device user manual.
- Alternatively, or if your insulin delivery device does not already carry a Penfill® cartridge, load a new Ryzodeg® 70/30 Penfill®.
- Disinfect the rubber membrane of Penfill® with an alcohol swab.
- Attach a new NovoFine® needle.
Checking for insulin flow (priming):
- Always check your Novo Nordisk insulin delivery device for insulin flow (priming) before each injection, as described in the device user manual.
- The priming procedure may highlight a malfunction with your insulin delivery device. Priming also removes any air bubbles and helps indicate whether or not a needle is broken.
- Only dial up your required dose after you see a drop of insulin at the needle tip.
- After priming, if you need to put the insulin delivery device down, make sure the needle does not touch anything.
Choose a site for injection
- Inject the medicine into the abdomen, thighs, upper arms or buttocks.
- Change the injection site so that the same position is not used more often than once a month. This reduces the risk of developing lumps or skin pitting.
- Pinch the skin between two fingers, push the needle into the raised skin, and inject the full dose of insulin under the skin.
- Slowly count to 6 before pulling the needle out.
- Keep the push button fully depressed until the needle has been withdrawn.
- Apply gentle pressure over the injection site for several seconds
- Do not rub the area
After injecting:
Using the outer needle cap, remove the used needle and dispose of it safely into a yellow plastic sharps container. If you do not remove it, temperature changes may cause liquid to leak out of the needle.
Health care professionals, relatives and other carers should follow general precautionary measures for removal and disposal of needles, to eliminate the risk of needlestick injury.
Do not share needles, insulin cartridges or insulin delivery devices.
Leave Penfill® in the insulin delivery device until it needs to be replaced.
How long to use Ryzodeg® 70/30
Do not stop using Ryzodeg® 70/30 unless your doctor tells you to.
If you forget to inject your insulin - hyperglycaemia
Ryzodeg® 70/30 Penfill® must be used strictly as advised by your doctor or nurse educator.
If you forget your insulin dose, test your blood sugar level as soon as possible.
If you are not sure what to do, talk to your doctor, diabetes education nurse or pharmacist.
Do not use a double dose of your insulin.
If you forget a dose, inject the missed dose with your next large meal on that day, and then go back to using it as you would normally.
If you use too much insulin - hypoglycaemia
If you think that you have used too much insulin, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Ryzodeg® 70/30 Penfill®?
Hypoglycaemia
Your blood sugar level may become too low (you may experience hypoglycaemia (a hypo)) if you:
- accidentally use too much of this medicine
- have too much or unexpected exercise
- delay eating meals or snacks
- eat too little food
- are ill.
The first symptoms of mild to moderate hypos can come on suddenly. They may include:
- cold sweat, cool pale skin
- fatigue, drowsiness, unusual tiredness and weakness
- nervousness, feeling anxious, tremor, rapid heart beat
- confusion, difficulty concentrating
- excessive hunger
- vision changes
- headache, nausea.
Always carry some sugary food or fruit juice with you.
Do not inject any insulin if you feel a hypo coming on.
Tell your relatives, friends, close workmates or carers that you have diabetes. It is important that they recognise the signs and symptoms of a hypo.
Make sure they know to give you some sugary food or fruit juice for mild to moderate symptoms of a hypo.
If you lose consciousness, make sure they know:
- to turn you on your side and get medical help immediately
- not to give you anything to eat or drink as you may choke.
An injection of the hormone glucagon may speed up recovery from unconsciousness. This can be given by a relative, friend, workmate or carer who knows how to give it.
If glucagon is used, eat some sugary food or have a sugary drink as soon as you are conscious again.
If you do not feel better after this, contact your doctor, diabetes education nurse, or the closest hospital.
See your doctor if you keep having hypo reactions, or if you have ever become unconscious after using insulin.
Your insulin dose may need to be changed.
If a severe hypo is not treated, it can cause convulsions, brain damage and even death.
Hyperglycaemia
Your blood sugar levels may become high (hyperglycaemia) if you:
- miss doses of insulin or use less insulin than you need
- have uncontrolled diabetes
- exercise less than usual
- eat more carbohydrates than usual
- are ill or stressed.
High blood sugar levels over a long period of time can lead to too much acid in the blood (diabetic ketoacidosis).
Contact your doctor immediately if your blood sugar level is high or you recognise any of the following symptoms.
Symptoms of mild to moderate hyperglycaemia include:
- drowsy feeling
- flushed face
- thirst, loss of appetite
- fruity odour on the breath
- blurred vision
- passing larger amounts of urine than usual
- getting up at night more often than usual to pass urine
- high levels of glucose and acetone in the urine.
Symptoms of severe hyperglycaemia include:
- heavy breathing
- fast pulse
- nausea, vomiting
- dehydration
- loss of consciousness.
Severe hyperglycaemia can lead to unconsciousness and in extreme cases death if untreated.
Things you should do
Measure your blood sugar levels regularly.
Make sure all friends, relatives, workmates or carers know that you have diabetes.
If your child has diabetes, it is important to tell their teachers and carers.
Keep using your insulin even if you feel well. It helps to control your diabetes, but does not cure it.
Always carry some sugary food or fruit juice with you. If you experience any of the symptoms of a hypo, immediately eat some sugary food or have a drink, e.g. lollies, biscuits or fruit juice.
Tell your doctor if you are feeling sick, especially if you are experiencing nausea or vomiting.
Tell your doctor if you have trouble recognising the symptoms of hypos.
Remind any doctor, dentist or pharmacist you visit that you have diabetes and are using insulin.
Tell your doctor if your diet changes or you are exercising more.
Tell your doctor if you notice any skin changes at the injection site.
The injection site should be rotated to help prevent changes to the fatty tissue under the skin, such as skin thickening, skin shrinking or lumps under the skin. The insulin may not work very well if you inject into a lumpy, shrunken or thickened area. Change the injection site with each injection to help prevent these skin changes.
Tell your doctor if you are ill. Illness, especially with nausea and vomiting, may cause your insulin needs to change. Even if you are not eating, you still require insulin. You and your doctor should design an insulin plan for those times when you are sick.
Tell your doctor if you are exercising more than usual. Exercise may lower your need for this medicine. Exercise may also speed up the effect of a dose of it, especially if the exercise involves the area of the injection site (e.g. the leg should not be used for injection prior to jogging or running).
Tell your doctor if your diet changes. Changes in diet may cause your insulin needs to change.
Tell your doctor if you are having trouble with your eyesight. Visual disturbances in uncontrolled diabetes are reversed during the early stages of treatment.
Once established on insulin, if your vision changes, see your doctor as soon as possible.
Tell your doctor, diabetes education nurse or pharmacist if you are travelling. Ask them for a letter explaining why you are taking injecting devices with you. Each country you visit will need to see this letter, so you should take several copies.
You may need to inject your insulin and eat your meals at different times because of time differences in and between countries.
You may not be able to get the same type of insulin in the country you are visiting. Your doctor, diabetes education nurse or pharmacist can provide you with some helpful information.
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are using Ryzodeg® 70/30 Penfill®.
Call your doctor straight away if:
- your blood sugar got so low that you passed out
- you have used glucagon
- you have had too low blood sugar a few times recently
This is because the dosing of your insulin injections, food or exercise may need to be changed.
Things you should not do
- Do not stop using your insulin unless your doctor tells you to.
- Do not give your insulin to anyone else, even if they have diabetes.
- Do not share needles.
- Do not refill your Ryzodeg® 70/30 Penfill®.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how the insulin affects you.
If your blood sugar is low or high your concentration and ability to react might be affected, and therefore also your ability to drive or operate a machine.
Bear in mind that you could endanger yourself or others. Please ask your doctor whether you can drive a car, especially if:
- you have frequent hypos
- you find it hard to recognise hypos.
Drinking alcohol
Tell your doctor if you drink alcohol.
Alcohol may mask the signs of a hypo. Carefully monitor your blood sugar levels when drinking.
Looking after your medicine
- Penfill® cartridges that are not being used should be stored between 2°C and 8°C in the refrigerator (away from the cooling element).
- Penfill® cartridges that you are using, or that you are carrying as a spare, should not be kept in a refrigerator.
- Protect the insulin in Penfill® from light by keeping the cartridges in the carton when not in use.
- Do not allow cartridges to be frozen, or exposed to excessive heat and light.
- Never use Ryzodeg® 70/30 Penfill® if the solution is not clear and colourless.
Keep it where young children cannot reach it.
When to discard your medicine (as relevant)
You can use Ryzodeg® 70/30 Penfill® for up to 4 weeks (28 days) after taking it out of the refrigerator if kept below 30°C.
Discard Penfill® cartridges after 4 weeks even if there is still some insulin left in them.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Never use insulin after the expiry date printed on the Penfill® label and carton after 'Expiry'. The expiry date refers to the last day of that month.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Hypoglycaemia (mild to moderate):
Other effects when you first start using insulin:
| Speak to your doctor if you have any of these less serious side effects and they worry you. They are usually mild and short-lived. |
Serious side effects
| Serious side effects | What to do |
Hypoglycaemia (severe):
Severe allergy - anaphylaxis:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Ryzodeg® 70/30 Penfill® contains
| Active ingredient (main ingredient) | soluble insulin degludec and insulin aspart 100 U/mL, in the ratio of 70:30. |
| Other ingredients (inactive ingredients) | glycerol phenol (as preservative) metacresol (as preservative) zinc acetate sodium chloride Water for injections sodium hydroxide (for pH adjustment) hydrochloric acid (for pH adjustment) |
Do not take this medicine if you are allergic to any of these ingredients.
What Ryzodeg® 70/30 Penfill® looks like?
Ryzodeg® 70/30 is a clear colourless solution for subcutaneous injection. Ryzodeg® 70/30 Penfill® 3 mL is a pre-filled glass cartridge designed to be used with Novo Nordisk insulin delivery systems and NovoFine® needles.
(AUST R 280433)
Who distributes Ryzodeg® 70/30 Penfill®
Ryzodeg® 70/30 Penfill® is supplied in Australia by:
Novo Nordisk Pharmaceuticals Pty Ltd
Level 10
118 Mount Street
North Sydney NSW 2060
Australia
Ryzodeg® 70/30 Penfill® is supplied in in in New Zealand by:
Novo Nordisk Pharmaceuticals Ltd.
PO Box 51-268
Pakuranga
Auckland
New Zealand
Further information
For further information call Novo Nordisk Medical Information on 1800 668 626 (Australia) or 0800 733 737 (New Zealand).
You can also get more information about diabetes from
Diabetes Australia:
- freecall helpline 1300 136 588
- www.diabetesaustralia.com.au
Diabetes New Zealand:
- freecall helpline 0800 342 238
- www.diabetes.org.nz
Ryzodeg® Penfill®, NovoRapid®, NovoPen® and NovoFine® are registered trademarks of Novo Nordisk A/S.
© 2025
Novo Nordisk A/S
This leaflet was prepared in November 2025
Brand Information
| Brand name | Ryzodeg 70/30 FlexTouch 3mL |
| Active ingredient | Insulin degludec + Insulin aspart |
| Schedule | S4 |
▼ This medicinal product is subject to additional monitoring in Australia. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse events at www.tga.gov.au/reporting-problems.
MIMS Revision Date: 01 June 2023
1 Name of Medicine
Insulin degludec (rys) and insulin aspart (rys).
2 Qualitative and Quantitative Composition
Ryzodeg 70/30 is a soluble insulin product consisting of insulin degludec (ultra-long acting basal insulin) and insulin aspart (rapid acting mealtime insulin) administered in one injection.
1 mL of the solution contains 100 U insulin degludec/insulin aspart (70% soluble insulin degludec and 30% soluble insulin aspart) equivalent to 2.56 mg salt-free anhydrous insulin degludec and 1.05 mg salt-free anhydrous insulin aspart. One pre-filled pen or cartridge contains 3 mL, equivalent to 300 U of insulin degludec/insulin aspart.
Insulin degludec and insulin apart are produced by recombinant DNA technology in Saccharomyces cerevisiae.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Ryzodeg 70/30 is a solution for injection.
Ryzodeg 70/30 is a ready-to-use, clear, colourless, neutral solution in pre-filled pen or cartridge. Ryzodeg 70/30 has a pH of approximately 7.4.
4 Clinical Particulars
4.1 Therapeutic Indications
For use in diabetes mellitus in patients aged 6 years and older.
4.2 Dose and Method of Administration
Dosage. Ryzodeg 70/30 is a soluble insulin product consisting of the ultra-long acting basal insulin degludec and the rapid acting prandial insulin aspart in a ratio of 70:30.
Ryzodeg 70/30 can be administered once- or twice-daily with the main meal(s). However, if advised by a healthcare professional, when needed, the patient can change the time of administration as long as Ryzodeg 70/30 is dosed with the main meal when taken once daily.
The potency of insulin analogues, including Ryzodeg 70/30 is expressed in units (U). 1 unit (U) of Ryzodeg 70/30 corresponds to 1 international unit (IU) of human insulin and one unit of most other insulin analogues.
In patients with type 2 diabetes mellitus, Ryzodeg 70/30 can be administered alone, in combination with oral antidiabetic products approved for use with insulin, and in combination with bolus insulin.
In type 1 diabetes mellitus, Ryzodeg 70/30 is combined with short-/rapid-acting insulin at the remaining meals.
Ryzodeg 70/30 is to be dosed in accordance with individual patients' needs. Pre-meal plasma glucose levels should be used to evaluate the adequacy of the preceding dose.
Initiation. Adults with type 2 diabetes. The recommended total daily starting dose of Ryzodeg 70/30 is 10 units with meal(s) followed by individual dosage adjustments.
Patients with type 1 diabetes. The recommended starting dose of Ryzodeg 70/30 is 60-70% of the total daily insulin requirements.
Ryzodeg 70/30 is to be used once daily at mealtime in combination with short-/rapid-acting insulin at the remaining meals followed by individual dosage adjustments.
Transfer from other insulin medicinal products. As with all insulin products, close glucose monitoring is recommended during transfer and in the following weeks. Doses and timing of concurrent rapid-acting or short-acting insulin products or other concomitant antidiabetic treatment may need to be adjusted.
Patients with type 2 diabetes. Patients switching from once daily basal or premix insulin therapy can be converted unit-to-unit to once daily Ryzodeg 70/30 at the same total insulin dose as the patient's previous total daily insulin dose, or twice daily Ryzodeg 70/30 using half the total daily dose twice daily.
Patients switching from more than once daily basal or premix insulin therapy can be converted unit-to-unit to once daily or twice-daily Ryzodeg 70/30 at the same total insulin dose as the patient's previous total daily insulin dose.
Patients switching from basal-bolus insulin therapy to Ryzodeg 70/30 will need to convert their dose based on individual needs. In general, patients are initiated on the same number of basal units.
Patients with type 1 diabetes. The recommended starting dose of Ryzodeg 70/30 is 60-70% of the total daily insulin requirements in combination with short-/rapid-acting insulin at the remaining meals followed by individual dosage adjustments.
Flexibility in dosing time. Ryzodeg 70/30 allows for some flexibility in the timing of insulin administration as long as it is dosed with the main meal(s).
Patients should not take an extra dose to make up for a missed dose. If a dose of Ryzodeg 70/30 is missed, the patient can take the missed dose with the next main meal of that day and thereafter resume the usual dosing schedule.
Avoidance of accidental mix-ups. Patients must be instructed to always check the insulin label before each injection to avoid accidental mix-ups between Ryzodeg 70/30 and other insulin products.
Special populations. Elderly (≥ 65 years old). Ryzodeg 70/30 can be used in elderly patients. As with all insulin products, glucose monitoring is to be intensified and the insulin dose adjusted on an individual basis.
Renal impairment. Ryzodeg 70/30 can be used in renal impaired patients. As with all insulin products, glucose monitoring is to be intensified and the insulin dose adjusted on an individual basis.
Hepatic impairment. Ryzodeg 70/30 can be used in hepatic impaired patients. As with all insulin products, glucose monitoring is to be intensified and the insulin dose adjusted on an individual basis.
Paediatric population. Ryzodeg 70/30 can be used in children and adolescents from the age of 6 years (see Section 5.1, Clinical trials). When changing from another insulin regimen to Ryzodeg 70/30, dose reduction of total insulin needs to be considered on an individual basis in order to minimise the risk of hypoglycaemia (see Section 4.4 Special Warnings and Precautions for Use).
Ryzodeg 70/30 should be used with special caution in paediatric patients because data from the clinical trial indicate that there may be a higher risk for severe hypoglycaemia (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1, Clinical trials).
Method of administration. Ryzodeg 70/30 combines the ultra-long acting basal insulin degludec and the rapid acting prandial insulin aspart in one injection.
Ryzodeg 70/30 is for subcutaneous administration only. Ryzodeg 70/30 must not be administered intravenously as it may result in severe hypoglycaemia. Ryzodeg 70/30 must not be administered intramuscularly as it may change the absorption. Ryzodeg 70/30 must not be used in insulin infusion pumps.
Ryzodeg 70/30 is administered subcutaneously by injection in the abdominal wall, the upper arm or the thigh. Injection sites are always to be rotated within the same region in order to reduce the risk of lipodystrophy and cutaneous amyloidosis (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)).
Ryzodeg 70/30 FlexTouch is a pre-filled pen designed to be used with NovoFine disposable needles. FlexTouch delivers 1-80 units in increments of 1 unit.
Ryzodeg 70/30 Penfill is designed to be used with Novo Nordisk insulin delivery systems and NovoFine disposable needles.
Dosage adjustment. As with all insulin products adjustment of dose may be necessary if patients undertake increased physical activity, change their usual diet or during concomitant illness.
Instructions for use and handling. Ryzodeg 70/30 FlexTouch. The pre-filled pen is designed to be used with NovoFine disposable needles. FlexTouch delivers 1-80 units in increments of 1 unit. The carton contains a Consumer Medicine Information package leaflet with instructions for use and handling.
Ryzodeg 70/30 Penfill. The cartridge is designed to be used with Novo Nordisk delivery systems, such as NovoPen (durable devices for repeated use) and NovoFine disposable needles. The carton contains a Consumer Medicine Information package leaflet with instructions for use and handling. The leaflet refers to the instructions for using the accompanying delivery system.
Ryzodeg 70/30 FlexTouch and Ryzodeg 70/30 Penfill is for use by one person only. The cartridge must not be refilled.
Ryzodeg 70/30 must not be used if the solution does not appear clear and colourless. Ryzodeg 70/30 which has been frozen must not be used.
The patient should discard the needle after each injection.
4.3 Contraindications
Hypersensitivity to insulin degludec, insulin aspart or to any of the excipients.
4.4 Special Warnings and Precautions for Use
Hypoglycaemia. Ryzodeg 70/30 must be taken with a carbohydrate containing meal. Omission of a meal or unplanned strenuous physical exercise may lead to hypoglycaemia.
Dose reductions and increased frequency of glucose monitoring may be required when Ryzodeg 70/30 is co-administered with insulin secretagogues or GLP-1 agonists as they may potentiate the risk of hypoglycaemia.
Hypoglycaemia may occur if the insulin dose is too high in relation to the insulin requirement.
In children, extra care should be taken to match insulin doses with food intake and physical activities in order to minimise the risk of hypoglycaemia.
Patients whose blood glucose control is greatly improved, for example by intensified insulin therapy, may experience a change in their usual warning symptoms of hypoglycaemia and must be advised accordingly. Usual warning symptoms may disappear in patients with long-standing diabetes mellitus.
Concomitant illness, especially infections and fever, usually increases the patient's insulin requirement. Concomitant diseases in the kidney, liver or diseases affecting the adrenal, pituitary or thyroid gland may require changes in the insulin dose.
As with other basal insulin products or insulin products with a basal component, the prolonged effect of Ryzodeg 70/30 may delay recovery from hypoglycaemia.
Hyperglycaemia. Administration of rapid-acting insulin is recommended in situations with severe hyperglycaemia.
Inadequate dosing and/or discontinuation of treatment in patients requiring insulin may lead to hyperglycaemia and potentially to diabetic ketoacidosis. Furthermore, concomitant illness, especially infections, may lead to hyperglycaemia and thereby cause an increased insulin requirement.
Usually, the first symptoms of hyperglycaemia develop gradually over a period of hours or days. They include thirst, increased frequency of urination, nausea, vomiting, drowsiness, flushed dry skin, dry mouth, and loss of appetite as well as acetone odour of breath. In type 1 diabetes mellitus, untreated hyperglycaemic events may lead to diabetic ketoacidosis, which is potentially lethal.
Insulin antibodies. Insulin administration may cause insulin antibodies to form. In rare cases, the presence of such insulin antibodies may necessitate adjustment of the insulin dose in order to correct a tendency to hyper- or hypoglycaemia.
Eye disorder. Intensification of insulin therapy with abrupt improvement in glycaemic control may be associated with temporary worsening of diabetic retinopathy, while long-term improved glycaemic control decreases the risk of progression of diabetic retinopathy.
Weight gain. Weight gain can occur with any insulin therapy, including Ryzodeg 70/30, and has been attributed to the anabolic effects of insulin and the decrease in glycosuria.
Administration. Ryzodeg 70/30 is for subcutaneous administration only. Ryzodeg 70/30 must not be administered intravenously as it may result in severe hypoglycaemia. Ryzodeg 70/30 must not be administered intramuscularly as it may change the absorption. Ryzodeg 70/30 must not be used in insulin infusion pumps.
Skin and subcutaneous tissue disorders. Patients must be instructed to perform continuous rotation of the injection site to reduce the risk of developing lipodystrophy and cutaneous amyloidosis. There is a potential risk of delayed insulin absorption and worsened glycaemic control following insulin injections at sites with these reactions. A sudden change in the injection site to an unaffected area has been reported to result in hypoglycaemia. Blood glucose monitoring is recommended after the change in the injection site from an affected to an unaffected area, and dose adjustment of antidiabetic medications may be considered.
Transfer of patients between insulin types. Transferring a patient to another type, brand or manufacturer of insulin must be done under medical supervision and may result in the need for a change in dosage.
Use in hepatic impairment. Ryzodeg 70/30 can be used in hepatic impaired patients. As with all insulin products, glucose monitoring is to be intensified and the insulin dose adjusted on an individual basis.
Use in renal impairment. Ryzodeg 70/30 can be used in renal impaired patients. As with all insulin products, glucose monitoring is to be intensified and the insulin dose adjusted on an individual basis.
Use in elderly. Ryzodeg 70/30 can be used in elderly patients. As with all insulin products, glucose monitoring is to be intensified and the insulin dose adjusted on an individual basis.
Paediatric use. Safety and efficacy of Ryzodeg 70/30 in children less than 2 years have not been established.
Avoidance of accidental mix-ups. Ryzodeg 70/30 has a unique PK/PD profile and cannot be automatically substituted for any other insulin. Patients must be instructed to always check the insulin label before each injection to avoid accidental mix-ups between Ryzodeg 70/30 and other insulin products.
Patients must visually verify the dialled units on the dose counter of the pen. Therefore, the requirement for patients to self-inject is that they can read the dose counter on the pen. Patients who are blind or have poor vision, must be instructed to always get help/assistance from another person who has good vision and is trained in using the insulin device.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
A number of medicinal products are known to interact with glucose metabolism. Possible interactions must therefore be taken into account by the physician.
The following substances may reduce the patient's insulin requirement: oral antidiabetic drugs (OADs), GLP-1 receptor agonists, monoamine oxidase inhibitors (MAOIs), nonselective beta-adrenergic blocking agents, angiotensin converting enzyme (ACE) inhibitors, salicylates, anabolic steroids (except danazol and oxymetholone), alpha-adrenergic blocking agents, quinine, quinidine and sulphonamides.
The following substances may increase the patient's insulin requirement: Oral contraceptives, thiazides, glucocorticoids, thyroid hormones, sympathomimetics, growth hormone, diazoxide, asparaginase, nicotinic acid, oxymetholone and danazol.
Beta-blocking agents may mask the symptoms of hypoglycaemia and delay recovery from hypoglycaemia.
Octreotide and lanreotide may either increase or decrease the insulin requirement.
Alcohol may intensify and prolong, or reduce the hypoglycaemic effect of insulin.
Combination of thiazolidinediones and insulin. Cases of cardiac failure have been reported when thiazolidinediones were used in combination with insulin, especially in patients with risk factors for development of cardiac failure. This should be kept in mind if treatment with the combination of thiazolidinediones and Ryzodeg 70/30 is considered. If the combination is used, patients should be observed for signs and symptoms of heart failure, weight gain and oedema. Thiazolidinediones should be discontinued if any deterioration in cardiac function occurs.
Incompatibilities. Substances added to Ryzodeg 70/30 may cause degradation of insulin degludec and/or insulin aspart. Ryzodeg 70/30 must not be added to infusion fluids. Ryzodeg 70/30 must not be mixed with other medicinal products insulin products or solutions.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Animal reproduction studies with insulin degludec have not revealed any adverse effects on fertility.
In a combined fertility and embryofetal study in male and female rats, treatment with subcutaneous doses of insulin degludec up to 21 U/kg/day (yielding 5-6 times the AUC in humans at a clinical dose of 0.8 U/kg/day) prior to mating and in female rats during gestation had no effect on mating performance or fertility.
In reproductive toxicity studies, insulin aspart did not affect the fertility of male and female rats but caused a slight increase in pre-implantation loss at subcutaneous doses greater than 10 U/kg/day. Similar effects were seen with human insulin.
Use in pregnancy. (Category B3)
There is no clinical experience with Ryzodeg 70/30 in pregnant women.
In rats, decreased fetal weight was observed following treatment with insulin degludec and insulin aspart in combination at a subcutaneous dose of 30 U/kg/day (resulting in 8 and 24 times the AUC in humans for the respective insulins at a dose of 1.08 U/kg Ryzodeg 70/30). In addition, an increase in fetal skeletal abnormalities was found in a rat embryofetal study conducted with insulin degludec alone. In both cases, similar effects were seen with human insulin, and are probably secondary to maternal hypoglycaemia.
Animal reproduction studies have not revealed any differences between insulin degludec and human insulin regarding embryotoxicity and teratogenicity.
In general, intensified blood glucose control and monitoring of pregnant women with diabetes mellitus are recommended throughout pregnancy and when contemplating pregnancy. Insulin requirements usually decrease in the first trimester and increase subsequently during the second and third trimester. After delivery, insulin requirements usually return rapidly to pre-pregnancy values.
Use in lactation. There is no clinical experience with Ryzodeg 70/30 during breast-feeding. In rats, insulin degludec and its metabolites were secreted in milk; the peak concentration of insulin degludec in milk was less than half of that in plasma. It is unknown whether Ryzodeg 70/30 is excreted in human milk. No metabolic effects of Ryzodeg 70/30 are anticipated in the breast-fed newborn/infant.
4.7 Effects on Ability to Drive and Use Machines
The patient's ability to concentrate and react may be impaired as a result of hypoglycaemia. This may constitute a risk in situations where these abilities are of special importance (e.g. driving a car or using machines). Patients must be advised to take precautions to avoid hypoglycaemia while driving. This is particularly important in those who have reduced or absent awareness of the warning signs of hypoglycaemia or have frequent episodes of hypoglycaemia. The advisability of driving should be considered in these circumstances.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile. More than 2000 subjects have been exposed to Ryzodeg 70/30 in the clinical development programme.
Hypoglycaemia is the most commonly observed adverse reaction in patients using insulin, including Ryzodeg 70/30.
Tabulated list of adverse events and adverse reactions. See Tables 1, 2 and 3.




Hypoglycaemia. As with any insulin therapy, hypoglycaemia may occur if the insulin dose is too high in relation to the insulin requirement. Severe hypoglycaemia may lead to unconsciousness and/or convulsions and may result in temporary or permanent impairment of brain function or even death. The symptoms of hypoglycaemia usually occur suddenly. They may include cold sweats, cool pale skin, fatigue, nervousness or tremor, anxiousness, unusual tiredness or weakness, confusion, difficulty in concentration, drowsiness, excessive hunger, vision changes, headache, nausea and palpitation.
Injection site reactions. As may occur with any insulin therapy, injection site reactions (including injection site haematoma, pain, haemorrhage, erythema, nodules, swelling, discolouration, pruritus, warmth and injection site mass) occurred in subjects treated with Ryzodeg 70/30. These reactions are usually mild and transitory and they normally disappear during continued treatment.
Skin and subcutaneous tissue disorders. As with any insulin therapy, lipodystrophy (including lipohypertrophy and lipoatrophy) and cutaneous amyloidosis may occur at the injection site and delay local insulin absorption. Continuous rotation of the injection site within the given injection area may help to reduce or prevent these reactions (see Section 4.4 Special Warnings and Precautions for Use).
Peripheral oedema. Insulin, including Ryzodeg 70/30, may cause sodium retention and oedema, particularly if previously poor metabolic control is improved by intensified insulin therapy.
Antibody production. There was no clinically relevant development of insulin antibodies after long-term treatment with Ryzodeg 70/30.
Paediatric population. Ryzodeg 70/30 has been administered to children (6-11 years) and adolescents (12-18 years) for the investigation of pharmacokinetic properties (see Section 5.2 Pharmacokinetic Properties). Clinical trials have been performed in children aged 2 to 18 years. The frequency, type and severity of adverse reactions in the paediatric population do not indicate differences to the experience in the general diabetes population with the exception of a signal of higher occurrence of severe hypoglycaemia compared to a basal-bolus regimen in the paediatric population (see Section 4.2 Dose and Method of Administration; Section 4.4 Special Warnings and Precautions for Use; Section 5.1 Pharmacodynamic Properties, Clinical trials).
Other special populations. Based on results from clinical trials, the frequency, type and severity of adverse reactions observed in the elderly patients and in patients with renal or hepatic impairment do not indicate any differences to the broader experience in the general population.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
A specific overdose for insulin cannot be defined; however, hypoglycaemia may develop over sequential stages if doses are administered which are too high relative to the patient's requirements:
Mild hypoglycaemic episodes can be treated by oral administration of glucose. It is therefore recommended that the patient always carries glucose-containing products. Adjustments in drug dosage, meal patterns, or exercise may be needed.
Severe hypoglycaemic episodes, including where the patient is not able to treat themselves, can be treated with glucagon (0.5 to 1 mg) given intramuscularly or subcutaneously by a trained person, or with glucose given intravenously by a healthcare professional. Glucose must be given intravenously, if the patient does not respond to glucagon within 10 to 15 minutes. After apparent clinical recovery from hypoglycaemia, continued observation and additional carbohydrate intake may be necessary to avoid reoccurrence of hypoglycaemia.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
ATC code: A10AD06.
Mechanism of action. Insulin degludec and insulin aspart bind to the human insulin receptor, resulting in the same pharmacological effects as human insulin. The blood glucose-lowering effect of insulin is due to the facilitated uptake of glucose following the binding of insulin to receptors on muscle and fat cells and to the simultaneous inhibition of glucose output from the liver.
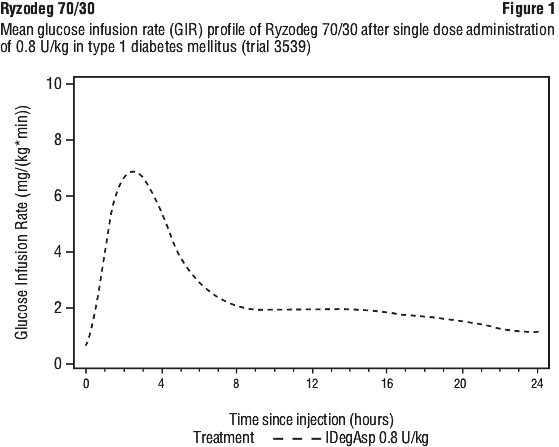
The pharmacodynamic effect of Ryzodeg 70/30 is distinctively separated for the two components (Figure 1) and the resulting action profile reflects the individual components, the rapid acting insulin aspart and the ultra-long acting insulin degludec. The ultra-long acting basal component of Ryzodeg 70/30 (insulin degludec) forms soluble multi-hexamers upon subcutaneous injection of Ryzodeg 70/30 resulting in a depot from which insulin degludec is continuously and slowly absorbed into the circulation leading to a flat and stable glucose-lowering effect. This effect is maintained in the co-formulation with insulin aspart and does not interfere with the rapid acting insulin aspart monomers.
Ryzodeg 70/30 has a rapid onset of action occurring soon after injection providing mealtime coverage while the basal component has a flat, stable and ultra-long action profile providing continuous coverage of the basal insulin requirements. The duration of action of a single dose of Ryzodeg 70/30 is beyond 24 hours.
There is no difference in the pharmacodynamic effect of Ryzodeg 70/30 between elderly and younger subjects.
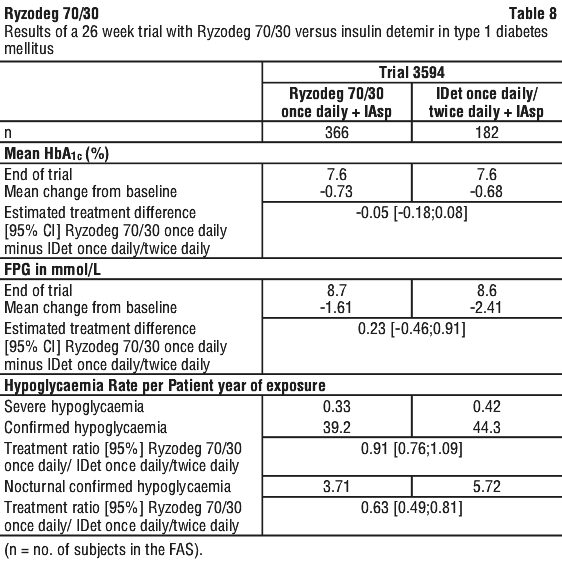
Clinical trials. Five (plus one extension) multi-national, randomised, controlled, open-label, treat-to-target clinical trials of 26 weeks or 52 weeks duration were conducted exposing a total of 1360 subjects with diabetes mellitus (362 subjects in type 1 diabetes mellitus and 998 subjects in type 2 diabetes mellitus) to Ryzodeg 70/30.
Ryzodeg 70/30 administered once daily plus oral antidiabetic drugs (OADs) was compared to insulin glargine (IGlar) once daily plus OADs in two trials in type 2 diabetes mellitus. Ryzodeg 70/30 once daily plus insulin aspart (IAsp) was also compared to once daily or twice-daily insulin detemir (IDet) plus IAsp in type 1 diabetes mellitus. Ryzodeg 70/30 twice daily plus OADs was compared to biphasic insulin aspart 30 (BIAsp 30) twice daily plus OADs in two trials in type 2 diabetes mellitus. BIAsp 30 is a white suspension for subcutaneous injection consisting of 30% soluble insulin aspart and 70% protamine-crystallised insulin aspart, marketed as NovoMix 30.
Efficacy and safety of once daily and twice daily mealtime dosing of Ryzodeg 70/30 either for insulin initiation or insulin intensification were confirmed. Ryzodeg 70/30 effectively improved glycaemic control as measured by HbA1c. Non-inferiority of change from baseline to end-of-trial of HbA1c was confirmed in all trials against all comparators, when treating subjects to target. All trials comparing insulin products were carried out using a treat-to-target design, where titration of basal insulin was based on pre-breakfast glucose values in order to achieve similar degrees of glycaemic control allowing for objective comparison of overall safety profile of the tested insulins, including risk of hypoglycaemia.
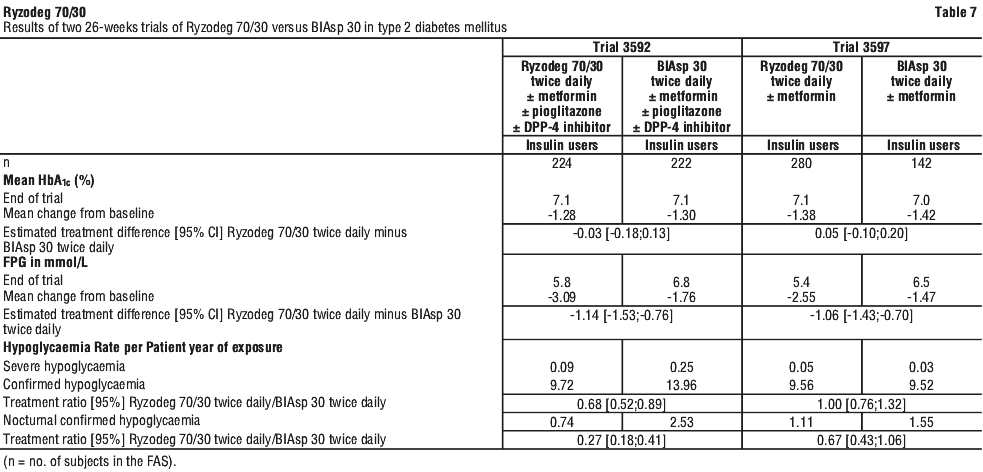
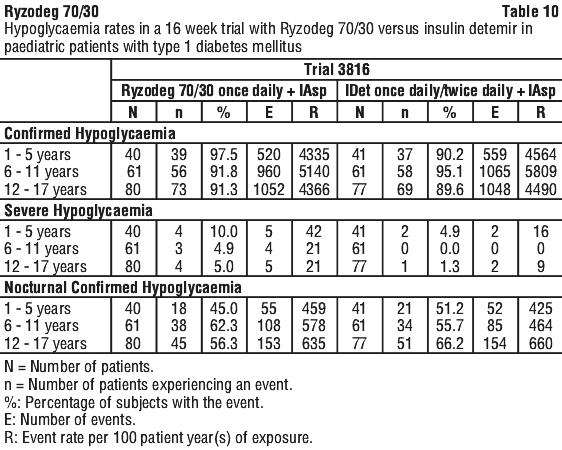
In a non-blinded, treat-to-target clinical trial (trial 3590), insulin-naïve adult subjects with type 2 diabetes mellitus were randomised to 26 weeks of treatment with Ryzodeg 70/30 + metformin (Met) or IGlar + Met. Ryzodeg 70/30 was administered once daily at breakfast. IGlar was administered once daily according to approved labelling. All subjects started on 10 U of either Ryzodeg 70/30 or IGlar. Ryzodeg 70/30 and IGlar groups had similar HbA1c levels with IGlar resulting in lower fasting plasma glucose (FPG) levels. Treatment with Ryzodeg 70/30 resulted in higher rates of confirmed hypoglycaemia but lower rates of nocturnal confirmed hypoglycaemia compared to IGlar (see Table 6).
In a non-blinded, treat-to-target clinical trial (trial 3593), adult subjects with type 2 diabetes mellitus who were inadequately controlled with basal insulin once daily + OADs were randomised to 26 weeks of treatment with Ryzodeg 70/30 once daily or IGlar once daily, both in combination with Met ± pioglitazone (Pio) ± DPP-4 inhibitor. All subjects initiated trial insulin treatment by switching on a unit-to-unit basis from the basal insulin they received before randomisation. Subjects administered Ryzodeg 70/30 once daily with either the evening meal or the largest meal or administered IGlar once daily according to approved labelling. Ryzodeg 70/30 and IGlar groups had similar HbA1c and FPG levels. Treatment with Ryzodeg 70/30 resulted in higher rates of confirmed hypoglycaemia but similar rates of nocturnal confirmed hypoglycaemia compared to IGlar (see Table 6).
The use of Ryzodeg 70/30 in combination with sulphonylureas, GLP-1 agonists, alpha-glucosidase inhibitors and SGLT-2 inhibitors has not been studied.

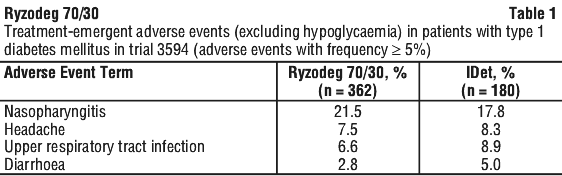
In a non-blinded, treat-to-target clinical trial (trial 3597), Asian adult subjects with type 2 diabetes mellitus who were inadequately controlled on basal insulin, premixed or self-mixed insulin in an once daily or twice daily insulin regimen ± Met were randomised to 26 weeks of treatment with Ryzodeg 70/30 twice daily or BIAsp 30 twice daily, with or without Met. Subjects on once daily insulin split the total dose of their previous insulin treatment into 2 equal doses of trial insulin for twice daily administration. Subjects on twice daily insulin transferred their doses on a unit-to-unit basis to the trial insulin. Ryzodeg 70/30 and BIAsp 30 was administered at the breakfast and main evening meal. Ryzodeg 70/30 and BIAsp 30 groups had similar HbA1c levels with Ryzodeg 70/30 resulting in significantly lower FPG levels. Treatment with Ryzodeg 70/30 resulted in similar rates of confirmed hypoglycaemia and nocturnal confirmed hypoglycaemia compared to BIAsp 30 (see Table 7).

Cardiovascular evaluation. DEVOTE was a randomised, double-blind, active-controlled, treat-to-target and event-driven clinical trial with a median duration of 2 years comparing the cardiovascular safety of insulin degludec versus insulin glargine (100 units/mL) in 7,637 patients with type 2 diabetes mellitus at high risk of cardiovascular events. Patients eligible to enter the trial were 50 years of age or older and had established, stable, cardiovascular, cerebrovascular, peripheral artery disease, chronic kidney disease or NYHA class II and III heart failure (85% of the enrolled population) or were 60 years of age or older and had other specified risk factors for cardiovascular disease (15% of the enrolled population).
The primary analysis was time from randomisation to first occurrence of a 3-component major adverse cardiovascular event (MACE) defined as cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke. The trial was designed as a non-inferiority trial to exclude a pre-specified risk margin of 1.3 for the hazard ratio of MACE comparing insulin degludec to insulin glargine. The cardiovascular safety of insulin degludec as compared to insulin glargine was confirmed (Figure 2).
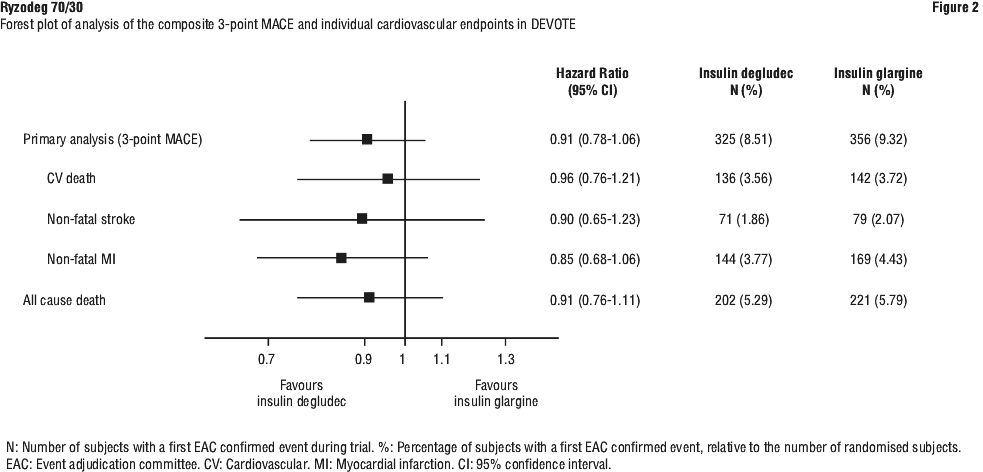
Similar improvements in HbA1c were achieved with insulin degludec and insulin glargine, and a greater reduction in FPG was achieved with insulin degludec.
Insulin degludec was superior compared to insulin glargine in terms of a lower rate of severe hypoglycaemic events and a lower proportion of subjects experiencing severe hypoglycaemia. The rate of nocturnal severe hypoglycaemia was significantly lower for insulin degludec compared to insulin glargine.
5.2 Pharmacokinetic Properties
Absorption. After subcutaneous injection, soluble and stable multi-hexamers of insulin degludec are formed creating a depot in the subcutaneous tissue, while not interfering with the rapid release of insulin aspart monomers into the circulation. Insulin degludec monomers gradually separate from the multi-hexamers resulting in a slow and continuous delivery of insulin degludec into the circulation. Steady-state serum concentrations of the ultra-long acting component (insulin degludec) is reached after 2-3 days of daily Ryzodeg 70/30 administration. The rapid absorption characteristics of the well established insulin aspart are maintained by Ryzodeg 70/30. The pharmacokinetic profile for insulin aspart appears 14 minutes after injection with a peak concentration after 72 minutes.
Distribution. The affinity of insulin degludec to serum albumin corresponds to a plasma protein binding of > 99% in human plasma. Insulin aspart has a low binding to plasma proteins, < 10%, similar to that seen with regular human insulin.
Metabolism. Degradation of insulin degludec and insulin aspart is similar to that of human insulin.
Excretion. The half-life after subcutaneous administration of Ryzodeg 70/30 is determined by the rate of absorption from the subcutaneous tissue. The half-life of the basal component, insulin degludec, at steady-state is approximately 25 hours independent of dose.
Linearity. Total exposure with Ryzodeg 70/30 increases proportionally with increasing dose of the basal component (insulin degludec) and the mealtime component (insulin aspart) in type 1 and type 2 diabetes mellitus.
Special populations. Elderly, race, renal and hepatic impairment. There are no clinically relevant differences in the pharmacokinetics of Ryzodeg 70/30 between elderly and younger adult subjects, between races or between healthy subjects and subjects with renal or hepatic impairment.
Gender. There are no gender differences in the pharmacokinetic properties of Ryzodeg 70/30.
Paediatric population. The pharmacokinetic properties of Ryzodeg 70/30 in type 1 diabetes mellitus were investigated in children (6-11 years) and adolescents (12-18 years) and compared to adults after single dose administration.
The steady state pharmacokinetics properties of the insulin degludec component of Ryzodeg 70/30 were investigated using population pharmacokinetic analysis in children down to 1 year of age.
Total exposure and peak concentration of insulin aspart were higher in children than in adults and were similar for adolescents and adults.
At steady state, the pharmacokinetic properties of insulin degludec in children (1-11 years) and adolescents (12-18 years) were comparable to those observed in adults with type 1 diabetes mellitus. Total exposure of insulin degludec after single dose administration was, however, higher in children and adolescents than in adults with type 1 diabetes mellitus.
5.3 Preclinical Safety Data
Genotoxicity. Genotoxicity studies have not been carried out with insulin degludec. Insulin aspart did not cause gene mutations, chromosomal damage or DNA damage in a range of genotoxicity tests.
Carcinogenicity. Standard 2-year carcinogenicity studies in animals have not been performed with insulin degludec and insulin aspart, either alone or in combination.
In a 52-week study with insulin degludec, rats received subcutaneous doses of up to 10 U/kg/day (resulting in 5 times the AUC in humans at a dose of 0.8 U/kg/day). No treatment-related increases in incidences of hyperplasia, benign or malignant tumours were recorded, and no treatment-related changes in female mammary gland cell proliferation were found using BrdU incorporation. In vitro studies showed the ratio of mitogenic relative to metabolic potency for insulin degludec is unchanged compared to human insulin.
In 52-week repeat dose toxicity studies with insulin aspart in Sprague-Dawley rats at subcutaneous doses up to 50 U/kg/day, the only significant toxicity findings were related to hypoglycaemia. At a higher dose of 200 U/kg/day in female Sprague-Dawley rats, insulin aspart, like human insulin, caused induction of mammary tumours. The clinical relevance of these findings is not known. Neither clinical nor epidemiological studies conducted to date have shown an association between insulin use and carcinogenesis but the available evidence is considered too limited to be conclusive at this time. In vitro studies showed that the mitogenic activity of insulin aspart does not differ from that observed with human insulin.
6 Pharmaceutical Particulars
6.1 List of Excipients
Ryzodeg 70/30 contains the following inactive ingredients: glycerol, metacresol, phenol, sodium chloride, zinc acetate, hydrochloric acid and sodium hydroxide for pH adjustment, and water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Before use. FlexTouch and Penfill. Store at 2°C to 8°C. Refrigerate. Do not freeze. Store Ryzodeg 70/30 away from the freezing element in the refrigerator.
In-use or carried as a spare. FlexTouch. Store below 30°C or in the refrigerator between 2°C to 8°C for up to 28 days. Keep the cap on the pen in order to protect from light. Protect from heat.
Penfill. Store below 30°C for up to 28 days. Do not refrigerate. Keep cartridges in the outer carton in order to protect from light. Protect from heat.
6.5 Nature and Contents of Container
Ryzodeg 70/30 FlexTouch: 3 mL solution in cartridge (type 1 glass) with a plunger (halobutyl) and a stopper (halobutyl/polyisoprene) contained in a pre-filled multidose disposable pen made of polypropylene.
Pack sizes: 1 x 3 mL, 5 x 3 mL.
Ryzodeg 70/30 Penfill: 3 mL solution in cartridge (type 1 glass) with a plunger (halobutyl) and a stopper (halobutyl/polyisoprene) in a carton.
Pack size: 5 x 3 mL.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
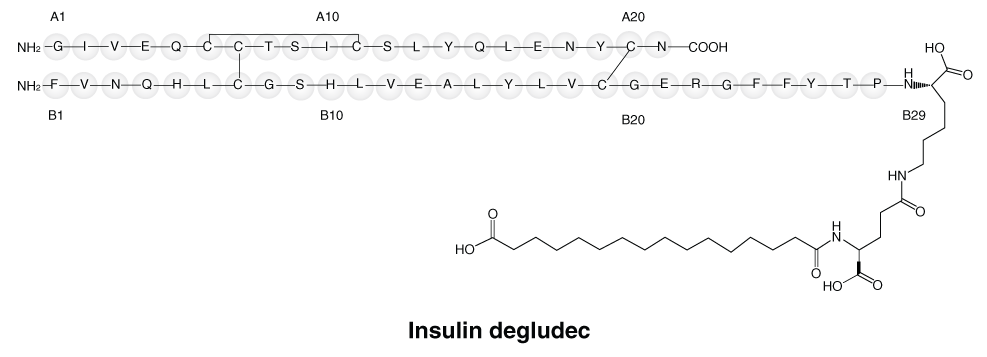
Insulin degludec. Insulin degludec differs from human insulin in that the threonine in position B30 has been omitted and a side-chain consisting of glutamic acid and a C16 fatty acid has been attached (chemical name: LysB29(Nepsilon-hexadecandioyl-γ-Glu) des(B30) human insulin).
Insulin aspart. Insulin aspart is homologous with regular human insulin with the exception of a single substitution of the amino acid proline by aspartic acid in position B28.
Chemical structure. Insulin degludec. Insulin degludec has a molecular formula of C274H411N65O81S6 and a molecular weight of 6103.97 daltons.
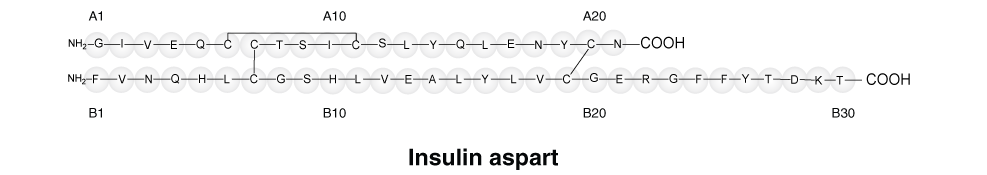
Insulin aspart. 116094-23-6.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
29 November 2017
Date of Revision
18 April 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.