Salbutamol Cipla
Brand Information
| Brand name | Salbutamol Cipla |
| Active ingredient | Salbutamol |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Salbutamol Cipla.
Summary CMI
Salbutamol Cipla
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Salbutamol Cipla cipules?
Salbutamol Cipla cipules contains the active ingredient salbutamol sulfate. Salbutamol Cipla cipules helps you to breathe more easily.
For more information, see Section 1. Why am I using Salbutamol Cipla cipules? in the full CMI.
2. What should I know before I use Salbutamol Cipla cipules?
Do not use if you have ever had an allergic reaction to Salbutamol Cipla cipules or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. Do not use in children under 18 months.
For more information, see Section 2. What should I know before I use Salbutamol Cipla cipules? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Salbutamol Cipla cipules and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Salbutamol Cipla cipules?
- For adults, the usual dose is one 5 mg Nebule via nebuliser every 4-6 hours as necessary.
- For children (aged 4-12 years), the usual dose is one 2.5 mg Nebule via nebuliser every 4-6 hours as necessary.
More instructions can be found in Section 4. How do I use Salbutamol Cipla cipules? in the full CMI.
5. What should I know while using Salbutamol Cipla cipules?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Salbutamol Cipla cipules? in the full CMI.
6. Are there any side effects?
Less serious side effects include headache, tremor, feelings of anxiety, restlessness, or excitability, 'warm' feeling or flu shing, restlessness in children, mouth or throat irritation and muscle cramps. Serious side effects include allergic reactions, irregular or fast heartbeat, increased blood flow to extremities, immediate breathing difficulties and wheezing, changes in blood potassium levels and lactic acidosis.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Salbutamol Cipla
Active ingredient: Salbutamol sulfate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Salbutamol Cipla cipules. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Salbutamol Cipla cipules.
Where to find information in this leaflet:
1. Why am I using Salbutamol Cipla cipules?
2. What should I know before I use Salbutamol Cipla cipules?
3. What if I am taking other medicines?
4. How do I use Salbutamol Cipla cipules?
5. What should I know while using Salbutamol Cipla cipules?
6. Are there any side effects?
7. Product details
1. Why am I using Salbutamol Cipla cipules?
Salbutamol Cipla Cipules contains the active ingredient, salbutamol. Salbutamol Cipla cipules belong to a group of medicines called bronchodilators. Because Salbutamol Cipla Cipules gives fast relief from your chest symptoms, it is often called a 'reliever'.
Salbutamol Cipla cipules are used for the relief of bronchospasm in patients with asthma or chronic obstructive pulmonary disease (COPD), and for relief against exercise-induced asthma and other stimuli known to induce bronchospasm.
Bronchodilators relax the muscles of the small air passages in the lungs. This helps to open the airways and makes it easier for air to get in and out of the lungs. When it is used regularly, it helps the small air passages to remain open.
There are other types of medicines that prevent wheezing or chest tightness. These medicines are called 'preventers' and must be used every day. Your doctor may tell you to use a 'preventer' in addition to your Salbutamol Cipla Cipules.
Salbutamol inhalation solution are not the only form of salbutamol available. Your doctor will decide which form of salbutamol is right for you.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Your doctor may have prescribed it for another reason.
2. What should I know before I use Salbutamol Cipla cipules?
Warnings
Do not use Salbutamol Cipla cipules if:
- you are allergic (hypersensitive) to salbutamol sulfate, or any of the ingredients listed at the end of this leaflet.
- the expiry date printed on the pack has passed or if the packaging is torn or shows signs of tampering.
Do not use Salbutamol Cipla cipules to stop a miscarriage or premature labour.
Always check the ingredients to make sure you can take this medicine.Check with your doctor if you:
- have any other medical conditions.
- take any medicines for any other condition.
- are allergic (hypersensitive) to foods, dyes, preservatives or any other medicines.
- have had to stop taking this or any other asthma medicine for any reason.
- have been diagnosed with, or are being treated for, a thyroid problem.
- have been diagnosed with, or are being treated for, high blood pressure.
- have been diagnosed with, or are being treated for, a heart problem.
- have been diagnosed with, or are being treated for, a liver problem.
- have been diagnosed with, or are being treated for, a kidney problem.
- have ever been told you have diabetes or high blood sugar.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
The ingredients of Salbutamol Cipla Cipules are known to cross the placenta during pregnancy. Your doctor can discuss with you the risks and benefits involved.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
It is not known whether the ingredients of Salbutamol Cipla Cipules can pass into breast milk.
Children
Do not give this medicine to children under 18 months.
3. What if I am taking other medicines?
Tell your Healthcare Professional (HCP) if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with salbutamol and affect how it works or make it more likely that you'll have side effects.
These medicines include
- xanthine derivatives (such as theophylline) or steroids to treat asthma
- diuretics (also called water tablets), sometimes used to treat high blood pressure or a heart condition
- nebulised anticholinergics
- ephedrine or pseudoephedrine, to relieve a stuffy nose
- beta-blockers, used to treat high blood pressure or a heart condition
Check with your Healthcare Professional (HCP) if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Salbutamol Cipla cipules.
4. How do I use Salbutamol Cipla cipules?
How much to take
Always take Salbutamol Cipla cipules exactly as your doctor has told you to. Check with your Healthcare Professional (HCP) if you are not sure.
- Instructions provided by your HCP may differ from the information contained in this leaflet. The pharmacist's label will usually tell you how many Salbutamol Cipla Cipules should be taken.
- If your condition suddenly gets worse, your doctor may tell you to increase your dose.
Salbutamol Cipla cipules should be used as required rather than regularly.
If you are using Salbutamol Cipla cipules more than twice a week to treat your asthma symptoms, not including preventive use before exercise or other triggers, this indicates poorly controlled asthma and may increase the risk of severe asthma attacks (worsening of asthma). You should contact your doctor as soon as possible to review your asthma treatment.
If you use your asthma anti-inflammatory medicine daily, e.g., "inhaled corticosteroid", it is important to continue using it regularly, even if you feel better.
Adults:
- The dose for adults is one 5 mg cipule via the nebuliser every 4-6 hours as necessary.
- Initial doses in the elderly may be lower than the recommended adult dose.
Children (aged 4-12 years):
- The dose for children (aged 4-12 years) is one 2.5 mg cipule via the nebuliser every 4-6 hours as necessary.
When to take Salbutamol Cipla cipules
- Your doctor will decide how often and for how long you have to take Salbutamol Cipla cipules.
- The pharmacist's label will usually tell you how often to take Salbutamol Cipla cipules.
- Salbutamol Cipla cipules should generally be taken every 4-6 hours as necessary.
How to take Salbutamol Cipla cipules
- Salbutamol Cipla Cipules must only be used by inhalation from a nebuliser and must not be injected or swallowed.
- The nebuliser produces a fine mist which you breathe in through a face mask or a mouthpiece. Make sure you know how to use it properly. If you have problems ask your HCP.
- Do not let the liquid, or the mist produced by the nebuliser get into your eyes.
- You can wear glasses or goggles to protect them.
- Use your nebuliser in a well-ventilated room as some of the mist will be released into the air and may be breathed in by others.
- Any solution remaining in the nebuliser should be discarded after use.
If you forget to use Salbutamol Cipla cipules
Salbutamol Cipla cipules should be taken as required. If you forget to take a dose, do not worry. Just take the next dose at the normal time or earlier if you become wheezy or feel tight in the chest.
Do not take a double dose to make up for the dose you missed. This may increase the chance of you getting unwanted side effects.
Just take your next dose at the usual time. If you become wheezy or develop other symptoms of an asthma attack you may need to take your next dose earlier.
- If you are not sure what to do, ask your Healthcare Professional (HCP).
- If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you use too much Salbutamol Cipla cipules
If you think that you have used too much Salbutamol Cipla cipules, you may need urgent medical attention. You may notice that your heart is beating faster than usual, you feel shaky or have a headache.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26 in Australia), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Salbutamol Cipla cipules?
Things you should do
- Remind any Healthcare Professional (HCP) you visit that you are taking Salbutamol Cipla cipules, especially if you are about to be started on any new medicine.
- If you are about to have surgery, tell the surgeon or anaesthetist that you are taking this medicine.
- If you are about to have any blood tests, inform your HCP that you are taking this medicine.
- Keep all of your doctor's appointments so that your progress can be checked.
- If your asthma worsens (for example you have frequent symptoms or flare ups such as breathlessness, cough, wheezing, tight chest, nighttime awakening or limited physical ability), you should tell your doctor who may start or increase a medicine to control your asthma such as an inhaled corticosteroid.
Call your doctor straight away if you:
- become pregnant while taking this medicine.
- feel that Salbutamol Cipla cipules do not help your breathing as much as usual.
- feel that the effect of Salbutamol Cipla cipules does not last as long as usual.
Please see Section 6. Are there any side effects? for more instances in which you should contact your doctor.
Things you should not do
- Do not stop using this medicine suddenly or change the dosage without medical advice.
- Do not take Salbutamol Cipla cipules to treat any other complaints unless your doctor tells you to.
- Salbutamol Cipla cipules is only for you. You should not give this medicine to anyone else, even if their symptoms seem similar to yours.
Driving or using machines
Salbutamol Cipla cipules is not expected to have any effect on your ability to drive or use machines.
Action Plan
If you feel that Salbutamol Cipla cipules are not having the same effect as before, this may be a sign that your chest condition is getting worse. Your doctor may decide to prescribe other forms of Salbutamol Cipla cipules, or another medicine, for you to use.
This medicine is only one part of a general plan to help you manage your asthma or other chest condition. You should discuss this plan with your doctor. Ask your doctor to check your treatment regularly.
Looking after your medicine
- Keep your medicine in the pack until it is time to take them.
- If you take the medicine out of the pack they may not keep well.
- Salbutamol Cipla Cipules should be kept in a cool, dry place where the temperature stays below 25°C. Cipule must be protected from light.
- Do not store in direct sunlight or heat. Do not leave in the car on hot days.
- Heat and dampness can destroy some medicines.
- Keep it where children cannot reach it.
- Do not use after the expiry date on the carton.
- Once you have opened each foil pack, you need to note down the date of opening the foil lid. Add three months to this date and write it down in the space provided on the foil pack. Do not use the Salbutamol Cipla Cipules left in the tray after this date.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If your doctor tells you to stop using Salbutamol Cipla or it has passed its expiry date, ask your pharmacist what to do with any that are left over.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
Do not be alarmed by the following list of side effects. You may not experience any of them.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Nervous system disorders:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergic reactions:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Salbutamol Cipla cipules contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
The solution is sterile and preservative free.
Do not take this medicine if you are allergic to any of these ingredients.
What Salbutamol Cipla cipules looks like
Salbutamol Cipla Cipules contain salbutamol solution for inhalation. It is a clear colourless, preservative free solution and comes in plastic unit dose ampoules.
Salbutamol Cipla Cipules comes in two strengths.
AUST R 115652 2.5mg Salbutamol Sulphate in 2.5ml of solution, 30 (6X5) AUSTR 115657 5 mg Salbutamol Sulphate in 2.5ml of solution, 30(6X5)
Who distributes Salbutamol Cipla Cipules
Sponsor:
Cipla Australia Pty Ltd
Level 1 / 132-136 Albert Road
South Melbourne VIC 3205
drugsafety@cipla.com
Phone: 1 800 569 074
Distributor:
Cipla Australia Pty Ltd.
This leaflet was prepared in July 2025.
Brand Information
| Brand name | Salbutamol Cipla |
| Active ingredient | Salbutamol |
| Schedule | S4 |
MIMS Revision Date: 01 December 2025
1 Name of Medicine
Salbutamol sulfate.
2 Qualitative and Quantitative Composition
Each ampoule contains salbutamol 2.5 mg/2.5 mL or 5 mg/2.5 mL as salbutamol sulfate.
Salbutamol sulfate 1.2 mg is approximately equivalent to salbutamol 1 mg.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Conventional inhalation.
Clear colourless to pale yellow solution.
4 Clinical Particulars
4.1 Therapeutic Indications
For the relief of bronchospasm in patients with asthma or chronic obstructive pulmonary disease, and for acute prophylaxis against exercise-induced asthma or in other situations known to induce bronchospasm.
4.2 Dose and Method of Administration
Increasing use of beta-2 agonists may be a sign of worsening asthma. Under these conditions a reassessment of the patient's therapy plan may be required and concomitant glucocorticosteroid therapy should be considered (see Section 4.4 Special Warnings and Precautions for Use).
Salbutamol Cipla ampoule/s are to be used under the direction of a physician.
The solution must not be injected or ingested.
Salbutamol Cipla ampoule/s 2.5 mg/2.5 mL and 5 mg/2.5 mL may be delivered from any efficient nebulising device.
Salbutamol Cipla ampoule/s may be used to achieve bronchodilatation as part of an inhalation therapy regime or for patients requiring assisted ventilation.
There is a large safety margin between therapeutic effects and unpleasant side effects. Nevertheless, because of the possibility of uncontrolled dosage associated with continuous administration, intermittent administration of appropriate amounts of Salbutamol Cipla ampoule/s is preferred.
Use in adults and children. Children 4 to 12 years: 2.5 mg.
Adults: 5 mg.
This dosage may be repeated as necessary every 4-6 hours. Any solution remaining in the nebuliser after completion of therapy should be discarded.
Important. Fresh dilutions should be prepared for each inhalation and any solution remaining in the nebuliser after treatment should be discarded immediately. To avoid contamination, nebulising devices should be thoroughly cleaned after use according to manufacturer's instructions.
Clinical efficacy of nebulised salbutamol in infants under 18 months is uncertain. As transient hypoxaemia may occur, supplemental oxygen therapy should be considered.
Use in the elderly. Initial doses of salbutamol in the elderly should be lower than the recommended adult dosage. The dose may then be gradually increased if sufficient bronchodilatation is not achieved.
Use in hepatic impairment. As about 60% of orally administered salbutamol (this includes not only tablet and syrup preparations but also approximately 90% of an inhaled dose) is metabolised to an inactive form; impairment of liver function may result in accumulation of unchanged salbutamol.
Use in renal impairment. About 60-70% of salbutamol administered by inhalation or intravenous injection is excreted in urine unchanged. Impairment of renal function may therefore require a reduction in dosage to prevent exaggerated or prolonged effects.
4.3 Contraindications
Hypersensitivity to any of the ingredients.
Non-i.v. formulations of salbutamol must not be used to arrest uncomplicated premature labour or threatened abortion.
4.4 Special Warnings and Precautions for Use
The management of asthma should normally follow a stepwise programme, and patient response should be monitored clinically and by lung function tests. Increasing use of short-acting inhaled beta-2 agonists to control symptoms indicates deterioration of asthma control. Under these conditions, the patient's therapy plan should be reassessed as soon as possible by a physician. Patients who are taking salbutamol more than twice a week on an "as needed" basis, not counting prophylactic use prior to a known trigger, may be at risk for overuse of salbutamol. A reassessment of the patient's therapy plan may be required.
Overuse of short-acting beta agonists may mask the progression of the underlying disease which could contribute to deteriorating asthma control, leading to an increased risk of severe asthma exacerbations and possibly mortality.
Sudden and progressive deterioration in asthma control is potentially life-threatening and consideration should be given to starting or increasing corticosteroid therapy. In patients considered at risk, daily peak flow monitoring may be instituted.
Patients who are prescribed regular asthma anti-inflammatory therapy (e.g. inhaled corticosteroids) should be advised to continue taking their anti-inflammatory medication even when symptoms improve, and they no longer require salbutamol.
Patients should be warned that if either the usual relief is diminished or the usual duration of action reduced, they should seek medical advice at the earliest opportunity after increasing the dose.
Animal studies suggest that cardionecrotic effects may occur with high dosages of some sympathomimetic amines. On this evidence the possibility of the occurrence of myocardial lesions cannot be excluded subsequent to long term treatment with these drugs.
Care should be taken with patients who are known to have received large doses of salbutamol or other sympathomimetic drugs, or who are suffering from hypertension, hyperthyroidism, myocardial insufficiency, or diabetes mellitus.
Salbutamol should be administered cautiously to patients with thyrotoxicosis.
In common with other beta-adrenoceptor agonists, salbutamol can induce reversible metabolic changes, for example increased blood sugar levels. The diabetic patient may be unable to compensate for this and the development of ketoacidosis has been reported. Concurrent administration of corticosteroids can exaggerate this effect.
Excessive use may induce a non-responsive state leading to a worsening of hypoxaemia.
Potentially serious hypokalaemia may result from beta-2-agonist therapy, mainly from parenteral and nebulised administration. Particular caution is advised in acute severe asthma as this effect may be potentiated by concomitant treatment with xanthine derivatives, steroids, diuretics and hypoxia. It is recommended that serum potassium levels are monitored in such situations.
The possibility of cardiac arrhythmias arising as a consequence of salbutamol induced hypokalaemia should be borne in mind, especially in digitalised patients, following the administration of salbutamol injection.
Addition of other active substances to Salbutamol Cipla ampoule/s cannot be recommended.
As with other inhalation therapy, paradoxical bronchospasm may occur, resulting in an immediate increase in wheezing after dosing. This should be treated immediately with an alternative presentation or a different fast-acting inhaled bronchodilator, if immediately available. The specific salbutamol presentation should be discontinued, and if necessary a different fast-acting bronchodilator instituted for ongoing use.
Lactic acidosis. Lactic acidosis has been reported very rarely in association with high therapeutic doses of intravenous and nebulised short-acting beta-agonist therapy, mainly in patients being treated for an acute asthma exacerbation (see Section 4.8 Adverse Effects (Undesirable Effects)). Increase in lactate levels may lead to dyspnoea and compensatory hyperventilation, which could be misinterpreted as a sign of asthma treatment failure and lead to inappropriate intensification of short-acting beta-agonist treatment. It is therefore recommended that patients are monitored for the development of elevated serum lactate and consequent metabolic acidosis in this setting.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Beta-adrenergic blocking drugs inhibit the bronchodilator action of salbutamol and other sympathomimetic bronchodilators. However such drugs should not be used in asthmatic patients as they may increase airway resistance.
Care is recommended if it is proposed to administer salbutamol in concomitant therapy with other sympathomimetic amines as excess sympathetic stimulation may occur.
Animal studies have shown that large doses of salbutamol may interact with imipramine, chlordiazepoxide and chlorpromazine but any practical significance of these results in man remains to be established.
A small number of cases of acute angle closure glaucoma have been reported in patients treated with a combination of nebulised salbutamol and ipratropium bromide. A combination of nebulised salbutamol with nebulised anticholinergics should therefore be used cautiously.
Patients should receive adequate instruction in correct administration and be warned not to let the solution or mist enter the eye.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There is no information on the effects of salbutamol on human fertility.
Use in pregnancy. (Category A)
Salbutamol is known to cross the placental barrier in humans. Safety for use in pregnancy has not been demonstrated, therefore the drug should not be used in pregnant women, or those likely to become pregnant, unless the expected benefits outweigh any potential risk.
Oral administration of salbutamol to rats and rabbits during pregnancy showed no teratogenic effects in offspring.
During worldwide marketing experience, rare cases of various congenital anomalies, including cleft palate and limb defects have been reported in the offspring of patients being treated with salbutamol.
Although intravenous salbutamol and occasionally salbutamol tablets are used in the management of uncomplicated premature labour, salbutamol presentations should not be used for threatened abortion during the first or second trimesters of pregnancy. Intravenous salbutamol is contra-indicated in cases of ante-partum haemorrhage because of the risk of further haemorrhage from an atonic uterus and there is the risk of the same problem arising inadvertently in asthmatics using salbutamol. Profuse uterine bleeding following spontaneous abortion has been reported after the use of salbutamol. Special care is required in pregnant diabetic women.
Use in lactation. It is not known whether salbutamol is excreted in breast milk nor whether it has a harmful effect on the newborn. Therefore it is not recommended for nursing mothers unless the expected benefits outweigh any potential risk.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
A fine tremor of skeletal muscle has been reported in some patients when salbutamol is administered orally or by inhalation, and in about 20% of patients receiving salbutamol injection, the hands being the most obviously affected with a few patients feeling tense. These effects are dose related and are caused by a direct action on skeletal muscle and not by direct CNS stimulation.
Increases in heart rate may occur in patients with normal heart rate after administration of salbutamol respirator solution. These increases are dose dependent and are of the order of 9 beats/minute when 10 mg of salbutamol as 0.5% w/v solution is inhaled by adults over 3 minutes, 13 beats/minute when 20 mg of salbutamol as 0.1% w/v solution is inhaled by adults over 3 minutes. In patients with pre-existing sinus tachycardia, especially those in status asthmaticus, the heart rate tends to fall after the administration of salbutamol respirator solution as the condition of the patient improves.
With higher doses than those recommended, or in patients who are unusually sensitive to beta-adrenergic stimulants, dilatation of some peripheral arterioles may occur leading to a small reduction in arterial pressure; a compensatory increase in cardiac output may then occur.
Cardiac arrhythmias (including atrial fibrillation, supraventricular tachycardia and extrasystoles) have been reported. Peripheral vasodilation and a compensatory small increase in heart rate may occur in some patients. Tachycardia may occur in some patients.
Other reactions which may occur are headaches, nausea, palpitations and sensations of warmth. Hypersensitivity reactions including angioedema, urticaria, bronchospasm, hypotension and collapse have been reported very rarely. There have been rare reports of muscle cramps. Mouth and throat irritation may occur with inhaled salbutamol.
Note. The incidence and severity of particular side effects depends on the dosage and route of administration. Salbutamol does not cause difficulty in micturition because, unlike sympathomimetic drugs such as ephedrine, therapeutic doses have no alpha-adrenergic receptor stimulant activity.
Potentially serious hypokalaemia may result from beta-2-agonist therapy.
Lactic acidosis has been reported very rarely in patients receiving intravenous and nebulised salbutamol therapy for the treatment of acute asthma exacerbation.
As with other inhalation therapy, paradoxical bronchospasm may occur, resulting in an immediate increase in wheezing after dosing (see Section 4.4 Special Warnings and Precautions for Use).
As with other beta-2-agonists hyperactivity has been reported rarely in children.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
The most common signs and symptoms of overdose with salbutamol are transient beta agonist pharmacologically mediated events (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)). The signs of salbutamol overdosage are significant tachycardia and/or significant muscle tremor.
Hypokalaemia may occur following overdosage with salbutamol. Serum potassium levels should be monitored.
Lactic acidosis has been reported in association with high therapeutic doses as well as overdoses of short-acting beta-agonist therapy, therefore monitoring for elevated serum lactate and consequent metabolic acidosis (particularly if there is persistence or worsening of tachypnea despite resolution of other signs of bronchospasm such as wheezing) may be indicated in the setting of overdose.
Consideration should be given to discontinuation of treatment and appropriate symptomatic treatment such as a cardio-selective beta-blocking agent given by intravenous injection in patients presenting with cardiac symptoms (e.g. tachycardia, palpitations). Beta-blocking drugs should be used with caution as they may cause bronchospasm in sensitive individuals.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Salbutamol is a relatively selective beta-2-adrenoreceptor stimulant. It is more specific than both isoprenaline and orciprenaline for adrenergic beta-2 receptors.
After oral and parenteral administration, stimulation of the beta receptors in the body, both beta-1 and beta-2, occurs because (a) beta-2 selectivity is not absolute, and (b) higher concentrations of salbutamol occur in the regions of these receptors with these modes of administration. This results in the beta-1 effect of cardiac stimulation, though not so much as with isoprenaline, and beta-2 effects of peripheral vasodilatation and hypotension, skeletal muscle tremor and uterine muscle relaxation.
Metabolic effects such as hyperinsulinaemia and hyperglycaemia also may occur, although it is not known whether these effects are mediated by beta-1 or beta-2 receptors. The serum potassium levels have a tendency to fall.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Following inhalation, salbutamol acts topically in bronchial smooth muscle and initially the drug is undetectable in the blood. After 2 to 3 hours low concentrations are seen, due presumably to the portion of the dose which is swallowed and absorbed in the gut.
Metabolism. Salbutamol is not metabolised in the lung but is converted to the 4'-o-sulfate ester in the liver.
Excretion. The elimination half-life of inhaled or oral salbutamol is between 2.7 and 5 hours. Salbutamol is excreted in the urine as free drug and as the metabolite. After oral administration 58-78% of the dose is excreted in the urine in 24 hours, approximately 60% as metabolites. A small fraction is excreted in the faeces.
Impairment of liver or renal function may necessitate a reduction in dosage (see Section 4.2 Dose and Method of Administration).
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium chloride, sulphuric acid (for pH adjustment), water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
3 months when removed from foil overwrap.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
Salbutamol Cipla is available in plastic ampoules; Pack sizes: cartons of 30 x 2.5 mL ampoules.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Salbutamol sulfate is a white or almost white, crystalline, odourless powder with a slightly bitter taste. It is freely soluble in water, slightly soluble in alcohol, chloroform and ether, very slightly soluble in methylene chloride.
Chemical structure.
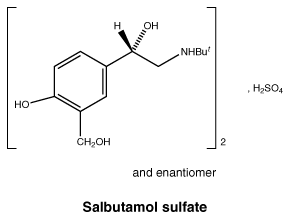
Molecular Formula: (C13H21NO3)2H2SO4.
Molecular weight: 576.7.
CAS number. 51022-70-9.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
18 October 2006
Date of Revision
24 October 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.