Siguent Hycor 1% Eye ointment
Brand Information
| Brand name | Siguent Hycor 1% Eye ointment |
| Active ingredient | Hydrocortisone acetate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Siguent Hycor 1% Eye ointment.
Summary CMI
SIGUENT HYCOR 1% eye ointment
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using SIGUENT HYCOR?
SIGUENT HYCOR contains the active ingredient hydrocortisone acetate. SIGUENT HYCOR is used to treat inflammatory conditions of the eye.
For more information, see Section 1. Why am I using SIGUENT HYCOR? in the full CMI.
2. What should I know before I use SIGUENT HYCOR?
Do not use if you have ever had an allergic reaction to hydrocortisone acetate or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use SIGUENT HYCOR? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with SIGUENT HYCOR and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use SIGUENT HYCOR?
- Gently pull out the lower eyelid to form a sac. Run the ointment into the sac, then allow the eye to close.
- Apply gentle pressure on the closed eye for about one minute.
- Try not to let the nozzle of the tube contact the eye when applying.
More instructions can be found in Section 4. How do I use SIGUENT HYCOR? in the full CMI.
5. What should I know while using SIGUENT HYCOR?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using SIGUENT HYCOR? in the full CMI.
6. Are there any side effects?
The most common side effect of SIGUENT HYCOR is temporary blurred vision (especially after application).
Other side effects are uncommon to rare. Serious side effects of SIGUENT HYCOR include secondary eye infection and sudden changes in vision.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
SIGUENT HYCOR 1% eye ointment
Active ingredient: hydrocortisone acetate
Consumer Medicine Information (CMI)
This leaflet provides important information about using SIGUENT HYCOR 1% eye ointment.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using SIGUENT HYCOR 1% eye ointment.
Where to find information in this leaflet:
1. Why am I using SIGUENT HYCOR?
2. What should I know before I use SIGUENT HYCOR?
3. What if I am taking other medicines?
4. How do I use SIGUENT HYCOR?
5. What should I know while using SIGUENT HYCOR?
6. Are there any side effects?
7. Product details
1. Why am I using SIGUENT HYCOR?
SIGUENT HYCOR contains 1% hydrocortisone acetate as the active ingredient.
This medicine is a type of glucocorticoid and belongs to the group of medicines called corticosteroids.
It is used in inflammatory (red, swollen) conditions of the eye including:
- allergic conjunctivitis
- conjunctivitis or swollen eyelids
- shingles (herpes zoster ophthalmicus)
- recurring eye ulcers.
SIGUENT HYCOR is for use in the eyes only. It should not be taken by mouth.
Ask your doctor if you have any questions about why it has been prescribed for you.
Your doctor may have prescribed this medicine for another purpose.
2. What should I know before I use SIGUENT HYCOR?
Warnings
Do not use SIGUENT HYCOR if:
- you are allergic to hydrocortisone acetate or any of the ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue or any other parts of the body; rash, itching or hives on the skin. - you wear contact lenses.
Wearing contact lenses while using this medicine may increase the risk of infection. - you have any of the following health conditions:
- acute herpes infection of the eye (herpes simplex), vaccinia (a special form of chicken pox), varicella, or other viral infections of the eye
- ocular tuberculosis
- fungal diseases of the eye
- pus producing infections of the eye. - the expiry date on the pack has passed.
- the packaging is torn or shows signs of tampering.
Check with your doctor if you:
- have any other medical conditions, especially the following:
- glaucoma or a family history of glaucoma
- cataracts
- diabetes
- any disease in which the cornea is thinned (perforation may occur). - are a wearer of contact lenses
- take any medicines for any other condition
- have had any allergy to any other medicines, or any other substances, such as foods, preservatives, or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with SIGUENT HYCOR and affect how it works. These include:
- drugs used to treat glaucoma.
- atropine and related compounds, these include medicines used to relieve stomach cramps or spasms, to prevent travel sickness and to treat Parkinson's disease.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect SIGUENT HYCOR.
4. How do I use SIGUENT HYCOR?
How to use it
- Gently pull out the lower eyelid to form a sac. Run the ointment into the sac, then allow the eye to close.
- Apply gentle pressure on the closed eye for about one minute.
- Try not to let the nozzle of the tube contact the eye when applying.
If handled incorrectly, eye ointments such as SIGUENT HYCOR can become contaminated by bacteria which can cause further infections when applied to the eye.
How much to use
- Apply two to four times a day or as directed by your doctor.
- Do not use more than the amount advised or use this medicine more frequently than directed.
It is important to use the ointment exactly as directed. If you use less than you should, it may not work as well and your medical condition may not improve. Using it more frequently than you should may not improve your condition any faster and may cause or increase side effects.
How long to use it for
SIGUENT HYCOR should be used as long as your doctor thinks it is necessary.
If you forget to use it
Use it as soon as you remember and then return to your normal dosing time. If your next dose is almost due, leave using it until that time.
Do not try to make up for missed doses b using more than one dose at a time.
This may increase the chance of getting an unwanted side effect.
If you accidentally swallow it
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning. You should also report any other medicine or alcohol which has been taken.
5. What should I know while using SIGUENT HYCOR?
Things you should do
- Tell your doctor if you feel it is not helping your condition.
- Tell your doctor if, for any reason, you have not used this medicine as prescribed.
Otherwise, your doctor may think that it was not effective and change your treatment unnecessarily. - If you develop any other eye condition such as injury, eye surgery or infections while using this medicine, seek your doctor's advice concerning the continued use.
- Remind any doctor, dentist or pharmacist you visit that you are using SIGUENT HYCOR.
Things you should not do
- Do not wear contact lenses while using SIGUENT HYCOR as it may increase the risk of infection.
- Do not give it to anyone else even though their symptoms seem like yours.
- Do not use it for other conditions unless your doctor tells you.
SIGUENT HYCOR has been prescribed for you for a specific condition. If you use it for another reason, it may not work or may make the condition worse. - Do not give this ointment to children unless specifically directed to by your doctor.
Things to be careful of
Do not use SIGUENT HYCOR for a long time.
If you use it for longer than recommended or use more than the prescribed dose, the chances of absorption of the drug into the body and the chance of side effects increases.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how SIGUENT HYCOR affects you.
SIGUENT HYCOR may temporarily blur vision after application.
Looking after your medicine
- Keep SIGUENT HYCOR below 25°C.
Do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
Discard any remaining ointment 4 weeks from first using it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| This side effect is common, especially after application of the medicine. |
| Speak to your doctor if you have any of these less serious side effects and they worry you. These side effects are uncommon to rare. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor immediately or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
| Usually associated if this medicine is used for a long time. Your doctor will decide if there is a need for a test to monitor the hormone levels in the blood. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What SIGUENT HYCOR 1% eye ointment contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
SIGUENT HYCOR 1% eye ointment does not contain any preservatives.
Do not take this medicine if you are allergic to any of these ingredients.
What SIGUENT HYCOR looks like
SIGUENT HYCOR 1% eye ointment is a soft, yellowish-white sterile ointment with a faint odour of paraffin and lanolin. Available in a 5 g tube with an elongated nozzle.
Australian Registration number: AUST R 139082.
Who distributes SIGUENT HYCOR
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australia
This leaflet was revised in October 2025.
Brand Information
| Brand name | Siguent Hycor 1% Eye ointment |
| Active ingredient | Hydrocortisone acetate |
| Schedule | S4 |
MIMS Revision Date: 01 April 2020
1 Name of Medicine
Hydrocortisone acetate.
2 Qualitative and Quantitative Composition
Siguent Hycor eye ointment contains 1% hydrocortisone acetate as the active ingredient in a sterile oculentum base. It is a preservative-free preparation.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
A soft, yellowish-white, clear eye ointment with an odour faintly of paraffin and lanolin.
4 Clinical Particulars
4.1 Therapeutic Indications
Inflammatory conditions of the eye, including allergic conjunctivitis, nonpurulent conjunctivitis and/or blepharitis, iritis, herpes zoster ophthalmicus, recurrent marginal ulceration.
4.2 Dose and Method of Administration
Apply two to four times daily.
4.3 Contraindications
Hypersensitivity to any ingredient including excipients.
Acute herpes simplex, vaccinia, varicella and most other viral diseases of the cornea and conjunctiva; ocular tuberculosis; fungal diseases of the eye; purulent conjunctivitis and blepharitis and other acute purulent untreated infections of the eye which may be masked or enhanced by the presence of a steroid.
4.4 Special Warnings and Precautions for Use
Extended use of the topical steroid therapy may cause increased intraocular pressure in certain individuals. Ophthalmologic examinations, especially tonometry and slit lamp examinations should be performed 2 to 3 weeks following onset of chronic therapy; subsequent examinations should be performed at intervals as determined by patient status or risk factors.
Risk/ benefit should be considered when the following medical problems exist: cataracts (may be exacerbated); diabetes mellitus (patient may be predisposed toward increase in intraocular pressure and/or cataract formation); diseases causing thinning of the cornea or sclera (use may result in perforation); glaucoma, chronic open angle or family history of glaucoma (may be precipitated or exacerbated); infections of the cornea or conjunctiva (risk of exacerbation or development of secondary infections).
There have been reports of bacterial keratitis associated with the use of multiple dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface.
Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye or the surrounding structures.
Patients should be instructed that ocular solutions, if handled improperly, can become contaminated by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions.
Patients should also be advised that if they develop an intercurrent ocular condition (e.g. trauma, ocular surgery or infection) or any ocular reactions, particularly conjunctivitis and lid reactions, they should immediately seek their physician's advice concerning the continued use of the product.
If more than one topical ophthalmic drug is being utilised, the drugs should be administered at least 10 minutes apart.
To minimise systemic absorption of the active ingredient, pressure should be applied to the tear duct for one minute immediately after application.
Visual disturbance. Visual disturbance may be reported with systemic and topical corticosteroid use. If a patient presents with symptoms such as blurred vision or other visual disturbances, the patient should be considered for referral to an ophthalmologist for evaluation of possible causes which may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR) which have been reported after use of systemic and topical corticosteroids.
Use in the elderly. No geriatric specific problems have been documented.
Paediatric use. Corticosteroids should be used with caution in children 2 years of age or younger because the different dose/weight ratio for children increases the risk of adrenal suppression. This risk increases with the length of therapy, which, therefore, should be limited to the shortest possible time (preferably less than 5 days).
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Anti-glaucoma agents. Chronic or prolonged use of corticosteroids may raise intraocular pressure and decrease the efficiency of the anti-glaucoma agents.
Anticholinergics (especially atropine and related compounds). Prolonged corticosteroid therapy may increase risk of intraocular hypertension which may be more likely to occur during treatment with anticholinergics in patients predisposed to acute angle closure.
Interactions with therapeutic devices. Contact lenses: risk of infection is increased.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Systemically absorbed hydrocortisone is classified category A in Australia. Problems in humans have not been documented with ophthalmologically applied hydrocortisone, however, well controlled studies have not been performed. Studies in pregnant mice have shown that hydrocortisone drops, when applied to both eyes five times a day on days 10-13 of gestation, caused a significant increase in foetal cleft palate. Studies in rabbits have shown that corticosteroids produce foetal resorptions and multiple abnormalities, including those of the head, ears, limbs and palate. Infants born to mothers who have received substantial doses of corticosteroids during pregnancy should be carefully observed for signs of hypoadrenalism.
Use in lactation. Problems in humans have not been documented.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Frequent or intensive use of ophthalmic corticosteroids may retard corneal healing.
Common. Ophthalmological. Temporary blurred vision (particularly after application of the eye ointment).
Uncommon to rare. Ophthalmological. Burning, stinging, redness or watering of the eyes; corneal thinning and/or globe perforation (decreased vision, watering of the eyes); glaucoma; ocular hypertension; optic nerve damage; posterior subcapsular cataract; visual acuity and field defects (gradual blurring or loss of vision, eye pain, nausea, vomiting); secondary ocular infection.
Post marketing. Eye disorders. Blurred vision.
4.9 Overdose
Dilution with water and other fluids is the usual response to accidental or deliberate overdose.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Hydrocortisone is the main glucocorticoid secreted by the adrenal cortex. It is an anti-inflammatory hormone. Hydrocortisone acetate is the acetate salt of hydrocortisone.
Mechanism of action. Corticosteroids diffuse across cell membranes and complex with specific cytoplasmic receptors. These then enter the cell nucleus, bind to DNA and stimulate transcription of mRNA and subsequent protein synthesis of enzymes ultimately responsible for anti-inflammatory effects of topical applications of corticosteroids to the eye.
In high concentrations, which may be achieved after topical application, corticosteroids may exert direct membrane effects. Corticosteroids decrease cellular and fibrinous exudation and tissue infiltration, inhibit fibroblastic and collagen-forming activity, retard epithelial regeneration, diminish postinflammatory neovascularization and reduce toward normal levels the excessive permeability of inflamed capillaries.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Hydrocortisone and its acetate salt are absorbed into the aqueous humour, cornea, iris, choroid, ciliary body and retina. Systemic absorption occurs, but may be significant only at higher dosages or in extended paediatric delivery. The biological half-life is about 100 minutes.
Distribution. It is more than 90% bound to plasma proteins.
Metabolism. Hydrocortisone is metabolised in the liver and most body tissues to hydrogenated and degraded forms such as tetrahydrocortisone and tetrahydrocortisol.
Excretion. These are excreted in the urine, mainly conjugated as glucuronides, together with a very small proportion of unchanged hydrocortisone.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. Long-term studies in animals (rats, rabbits, mice) showed no evidence of carcinogenicity or mutagenicity attributable to oral administration of corticosteroids. Long-term animal studies have not been performed to evaluate the carcinogenic potential of topical corticosteroids. Studies to determine mutagenicity with hydrocortisone have revealed negative results.
6 Pharmaceutical Particulars
6.1 List of Excipients
White soft paraffin, liquid paraffin and wool fat.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Discard 4 weeks after opening.
6.5 Nature and Contents of Container
Aluminium tube with a HDPE nozzle. Pack sizes of 2.5 g or 5 g.
(Note: not all pack sizes are available.)
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
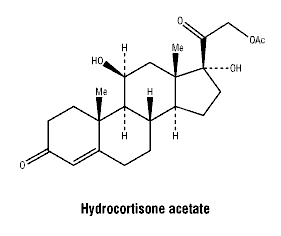
Hydrocortisone acetate is a white or almost white, odourless crystalline powder. 112 mg of hydrocortisone acetate is approximately equivalent to 100 mg of hydrocortisone. It is insoluble in water, soluble 1 in 230 of alcohol, 1 in 200 in chloroform. Formula: C23H32O6. Molecular weight: 404.5.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
04 September 2007
Date of Revision
06 February 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.