Soluvit N
Brand Information
| Brand name | Soluvit N |
| Active ingredient | Thiamine nitrate + Riboflavine sodium phosphate + Nicotinamide + Pyridoxine hydrochloride + Pantothenic acid + Sodium ascorbate + Biotin + Folic acid + Cyanocobalamin |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Soluvit N.
Summary CMI
Soluvit® N
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Soluvit N?
Soluvit N contains the active ingredients thiamine nitrate, riboflavine sodium phosphate, nicotinamide, pyridoxine hydrochloride, sodium pantothenate, sodium ascorbate, folic acid, biotin and cyanocobalamin. Soluvit N is a water-soluble vitamins supplement to parenteral nutrition which provides your body with nutrition by the intravenous route. It is usually given together with carbohydrates, amino acids and salts to provide an intravenous diet.
For more information, see Section 1. Why am I using Soluvit N? in the full CMI.
2. What should I know before I use Soluvit N?
Do not use if you have ever had an allergic reaction to any of the ingredients listed at the end of the CMI. Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Soluvit N? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Soluvit N and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Soluvit N?
This medicine is given an infusion into a vein. The dose of Soluvit N which you or your child will require will be determined by your doctor or pharmacist. Your doctor will supervise your or your child's treatment with Soluvit N.
More instructions can be found in Section 4. How do I use Soluvit N? in the full CMI.
5. What should I know while using Soluvit N?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Soluvit N? in the full CMI.
6. Are there any side effects?
Adverse reactions may be expected based on experience with other water-soluble vitamins compounds administered intravenously which include allergic reactions.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Soluvit® N
Active ingredient(s): thiamine nitrate, riboflavine sodium phosphate, nicotinamide, pyridoxine hydrochloride, sodium pantothenate, sodium ascorbate, folic acid, biotin and cyanocobalamin
Consumer Medicine Information (CMI)
This leaflet provides important information about using Soluvit N. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Soluvit N.
Where to find information in this leaflet:
1. Why am I using Soluvit N?
2. What should I know before I use Soluvit N?
3. What if I am taking other medicines?
4. How do I use Soluvit N?
5. What should I know while using Soluvit N?
6. Are there any side effects?
7. Product details
1. Why am I using Soluvit N?
Soluvit N contains the active ingredients thiamine nitrate, riboflavine sodium phosphate, nicotinamide, pyridoxine hydrochloride, sodium pantothenate, sodium ascorbate, folic acid, biotin and cyanocobalamin.
When the intake of nutrients or food into the mouth or directly into the gut is not possible or not enough to supply the needs, then intravenous nutrients or foods can be given. This is especially important for people under physical stress from illness or recent surgery.
Soluvit N is intended as a supplement in intravenous nutrition to meet the daily requirements of the water-soluble vitamins in adults, adolescents, children and infants. Soluvit N is usually given together with fats, amino acids, glucose and trace elements to provide a complete intravenous diet. Fat-soluble vitamins should also be administered to patients receiving prolonged parenteral nutrition.
2. WHAT SHOULD I KNOW BEFORE I USE SOLUVIT N?
Warnings
Do not use Soluvit N if your or your child:
- are allergic or has known hypersensitivity to to the ingredients of this medicine..
Always check the ingredients to make sure you can use this medicine
Check with your doctor if you or your child:
- have any other medical conditions such as liver or kidney disease
- have severe vitamin deficiency as Soluvit N dose may be insufficient
- have allergies are or your child is allergic to any other medicines or any other substances, such as foods, preservatives or dyes
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Soluvit N can be used by pregnant and breast-feeding women. Check with your doctor if Soluvit N is sufficient to meet the altered requirements in pregnancy and breastfeeding,
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Use in elderly
No data is available for use of Soluvit N in elderly patients. Your doctor will decide what to do.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Soluvit N and affect how it works. These include levodopa and phenytoin.
Other drugs should not be added to Soluvit N dissolved in Intralipid, SMOFlipid or Vitalipid N, due to the possibility of physical incompatibilities.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Soluvit N.
4. How do I use Soluvit N?
How much is given
Your doctor will determine the dose of Soluvit N which you or your child require and will supervise your or your child's treatment with Soluvit N.
How to take Soluvit N
Soluvit N should be dissolved first and then diluted in a bag of amino acids, glucose and/or fats or in clear standard solution. This should be done immediately before the start of the infusion and used within 24 hours. An electronic pump may be used to control the speed of the infusion of drip. The bag containing Soluvit N should be protected from light.
If you forget to use Soluvit N
This rarely happens as Soluvit N is usually administered under the care of a trained health care professional in a hospital or clinic setting.
If you use too much Soluvit N
This rarely happens as Soluvit N is usually administered under the care of a trained professional in a hospital or clinic setting. However, if you think that you have received too much Soluvit N, inform your doctor or nurse immediately.
Your doctor has information on how to recognize and treat an overdose. Ask your doctor if you have any concerns.
If you think that you have used too much Soluvit N, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Soluvit N?
Things you should do
Remind any doctor, dentist, pharmacist or nurse you visit that you are being treated with Soluvit N.
If you are about to have any blood or urine test, tell your health care professional that you are being treated with Soluvit N. It may interfere with the results of some laboratory tests.
Call your doctor straight away if you have signs and symptoms of allergic reactions. In case of an allergic reaction, the infusion should be stopped immediately, and appropriate measures need to be taken.
Looking after your medicine
Soluvit N is usually administered under the care of a trained health care professional in a hospital or clinic setting.
Soluvit N is recommended to be stored below 25°C in a cool dry place, protected from light and away from moisture or heat. Do not freeze.
Do not use after the expiry date which is stated on the carton label and the vial..
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
Information below are adverse reactions that may be expected based on other intravenous water-soluble vitamins. Since Soluvit N has to be diluted with other nutrient solutions, these also may cause unwanted effects in some people. If you need to, ask your doctor or nurse if you have any further questions about side effects
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or nurse if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Soluvit N contains
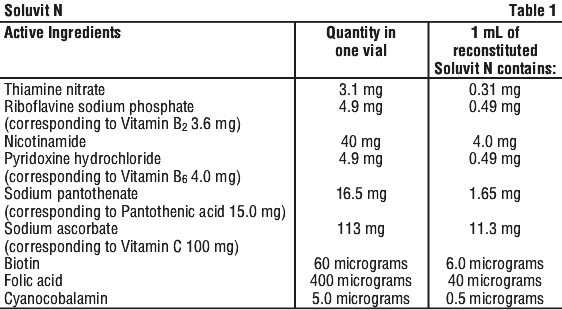
| Active ingredient (main ingredient) (quantity in one vial) | thiamine nitrate 3.1 mg, riboflavine sodium phosphate 4.9 mg (corresponding to Vitamin B2 3.6 mg) nicotinamide 40 mg, pyridoxine hydrochloride 4.9 mg (corresponding to Vitamin B6 4.0 mg) sodium pantothenate 16.5 mg (corresponding to pantothenic acid 15 mg) sodium ascorbate 113 mg (correspondingto Vitamin C 100 mg) biotin 60 µg, folic acid 400 µg, cyanocobalamin 5 µg, |
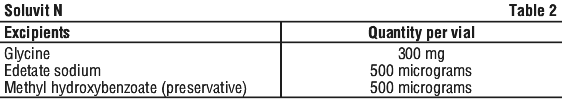
| Other ingredients (inactive ingredients) | glycine edetate sodium methyl hydroxybenzoate (preservative) |
| Potential allergens | methyl hydroxybenzoate |
Do not take this medicine if you are allergic to any of these ingredients.
Soluvit N does not contain gluten, lactose, sucrose, tartrazine or any other azo dyes.
Soluvit N comes in carton of 10 glass vials. (Aust R 40145)
What Soluvit N looks like
Soluvit N is a yellow lyophilised powder in a glass vial
Who distributes Soluvit N
Fresenius Kabi Australia Pty Limited
Level 2, 2 Woodland Way
Mount Kuring-gai NSW 2080
Australia
Telephone: 1300 361 004
This leaflet was prepared in March 2023
Brand Information
| Brand name | Soluvit N |
| Active ingredient | Thiamine nitrate + Riboflavine sodium phosphate + Nicotinamide + Pyridoxine hydrochloride + Pantothenic acid + Sodium ascorbate + Biotin + Folic acid + Cyanocobalamin |
| Schedule | Unscheduled |
MIMS Revision Date: 01 May 2022
1 Name of Medicine
Thiamine nitrate, riboflavine sodium phosphate, nicotinamide, pyridoxine hydrochloride, sodium pantothenate, sodium ascorbate, folic acid, biotin, cyanocobalamin.
2 Qualitative and Quantitative Composition
Soluvit N is a lyophilised, sterile, yellow mixture of water-soluble vitamins for intravenous infusion. See Table 1.
3 Pharmaceutical Form
Powder for Injection, vial.
A yellow, porous freeze-dried cake.
Osmolality in 10 mL of water: approx. 490 mOsm/kg water.
pH in 10 mL of water: 5.8.
4 Clinical Particulars
4.1 Therapeutic Indications
Soluvit N is intended as a supplement in intravenous nutrition in order to meet the daily requirements of the water-soluble vitamins in adults, adolescents, children and infants. Fat-soluble vitamins should also be administered to patients receiving prolonged parenteral nutrition.
4.2 Dose and Method of Administration
Soluvit N may be added to parenteral nutrition admixtures containing carbohydrates, lipids, amino acids, electrolytes, and trace elements, provided that compatibility and stability have been confirmed.
Soluvit N must not be given undiluted. The reconstituted Soluvit N should be added to the infusion solution under aseptic conditions, immediately before the start of the infusion and used within 24 hours. Clear admixtures (e.g. glucose solution or Water for Injections) containing Soluvit N should be protected from light.
The reconstituted mixtures with Vitalipid, Intralipid and SMOFlipid must be added under aseptic conditions to Intralipid only. The reconstituted mixtures with Water, Sodium Chloride 0.9% and Glucose are added under aseptic conditions to Intralipid or glucose solutions for infusion.
Adults and children weighing 10 kg or more. The recommended daily dosage is the contents of one vial.
The contents of one vial are dissolved by the aseptic addition of 10 mL of one of the following: 1. Vitalipid N Adult/Infant*; 2. Intralipid 10%, 20% or 30%# lipid emulsion for infusion; 3. Water for Injections; 4. Sodium chloride 0.9% injection; 5. Glucose solution for infusion; 6. SMOFlipid.
* Vitalipid N Adult is only indicated for use in patients aged 11 years and above.
# Intralipid 30% is not recommended in children.
Children and infants weighing less than 10 kg. Children and infants weighing less than 10 kg should be given 1/10 (1 mL) of the content of one vial per kg body weight per day.
The contents of one vial are dissolved by the aseptic addition of 10 mL of one of the following: 1. Vitalipid N Infant*; 2. Intralipid 10% or 20%; 3. Water for Injections; 4. Sodium chloride 0.9% injection; 5. Glucose solution for infusion; 6. SMOFlipid.
* The mixture Soluvit N and Vitalipid N Infant is not recommended for those weighing less than 10 kg.
4.3 Contraindications
Known hypersensitivity to any of the components, for example, thiamine or methyl hydroxybenzoate.
4.4 Special Warnings and Precautions for Use
Administering folic acid may obscure pernicious anaemia. The Soluvit N doses recommended are insufficient to correct severe vitamin deficiency states and may be insufficient in patients with markedly increased vitamin requirements. In patients receiving total parenteral nutrition (TPN), routine supplementation with both fat-soluble and water-soluble vitamins is recommended to prevent deficiency states and to obviate the need to speculate on individual vitamin status. Daily vitamin requirements must be calculated to avoid overdosage and toxic effects, especially with regards to vitamins A and D, and particularly in paediatric patients. In patients for whom TPN is continued for prolonged periods (months or years), periodic monitoring of blood vitamin levels should be considered.
To prevent excessive excretion of water-soluble vitamins, and for reasons of safety, daily dosage should be administered over a number of hours. See also the product information for Intralipid, SMOFlipid or Vitalipid N if Soluvit N is dissolved in these products.
Use in hepatic impairment. No data available.
Use in renal impairment. No data available.
Use in the elderly. There have been no specific clinical studies conducted with Soluvit N in the elderly.
Paediatric use. No data available.
Effects on laboratory tests. Biotin may interfere with laboratory tests that are based on a biotin/streptavidin interaction, leading to either falsely decreased or falsely increased test results, depending on the assay. The risk of interference is higher in children and patients with renal impairment and increases with higher doses. When interpreting results of laboratory tests, possible biotin interference has to be taken into consideration, especially if a lack of coherence with the clinical presentation is observed (e.g. thyroid test results mimicking Graves' disease in asymptomatic patients taking biotin or false negative troponin test results in patients with myocardial infarction taking biotin). Alternative tests not susceptible to biotin interference should be used, if available, in cases where interference is suspected.
The laboratory personnel should be consulted when ordering laboratory tests in patients taking biotin.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Vitamin B6 can reduce the effect of levodopa. Folic acid may lower the serum concentration of phenytoin. Other drugs should not be added to Soluvit N dissolved in Intralipid, SMOFlipid or Vitalipid N, due to the possibility of physical incompatibilities (see product information for Intralipid, SMOFlipid and Vitalipid N).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. The potential effects of Soluvit N on fertility and general reproductive performance have not been determined.
Use in pregnancy. The requirement of vitamins in pregnant women may be insufficient due to the patient's altered needs. Soluvit N has been administered to pregnant women with no adverse reactions reported.
Use in lactation. The requirement of vitamins in lactating women may be insufficient due to the patient's altered needs.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Allergic reactions including anaphylactic reactions may occur in patients hypersensitive to any component in the preparation, for example, folic acid, methyl hydroxybenzoate or thiamine (frequency not known). There have been rare reports of anaphylactoid reactions following repeated injection of preparations containing thiamine. Flushing, itching or burning of the skin may occur in patients susceptible to the effects of nicotinamide. Evaluable safety data from clinical trials with Soluvit N are limited. Adverse reactions that may be expected based on experience with other water-soluble vitamin compounds administered intravenously include: allergic reactions, including anaphylaxis; dermatological reactions including flushing, erythema, pruritus, and CNS reactions including headache, dizziness, and agitation.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Adverse effects caused by an overdose of water soluble vitamins have been reported and are unlikely to occur when administered as recommended. In cases of suspected overdose, symptomatic and supportive therapy should be instituted as appropriate, and further administration of the product discontinued.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia) or 0800 764 766 (New Zealand).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. ATC code: B05X C00.
Soluvit N is a mixture of water-soluble vitamins in amounts normally absorbed from the oral diet and should have no pharmacodynamic effect besides maintaining the nutritional status in patients requiring home PN. Additional amounts of some vitamins may be needed e.g. surgery, malnutrition, burns to avoid certain diseases caused by deficiency.
Clinical trials. No clinical data is available.
5.2 Pharmacokinetic Properties
When infused intravenously the water-soluble vitamins in Soluvit N are utilised in a generally similar way to water-soluble vitamins from an oral diet.
Absorption. No data available.
Distribution. No data available.
Metabolism. No data available.
Excretion. No data available.
5.3 Preclinical Safety Data
Genotoxicity. Studies with Soluvit N have not been performed to evaluate the genotoxic potential.
Carcinogenicity. Studies with Soluvit N have not been performed to evaluate the carcinogenic potential.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Table 2.
6.2 Incompatibilities
Soluvit N may only be added to or mixed with other medicinal products for which compatibility has been documented, see Section 4.2 Dose and Method of Administration. Please also see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions for incompatibilities of the product.
6.3 Shelf Life
Approved shelf life: 18 months.
The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
Soluvit N is a sterile, lyophilised powder containing a mixture of the water-soluble vitamin B1, vitamin B2, nicotinamide, vitamin B6, pantothenic acid, vitamin C, biotin, folic acid, and vitamin B12. Methyl hydroxybenzoate and edetate sodium are included as stabilisers.
Glass vials (Type I). Stopper for injection vial, chlorobutyl rubber. Vials: box of 10. AUST R 40145.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. Thiamine nitrate.
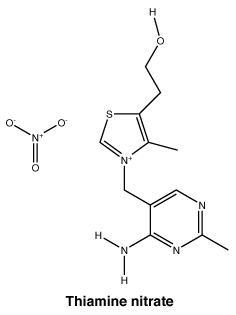
Molecular weight: 327.36.
Chemical name: Thiazolium, 3-[(4-amino-2-methyl-5-pyrimidinyl) methyl]-5-(2-hydroxyethyl)-4-methyl-nitrate.
Riboflavine sodium phosphate.
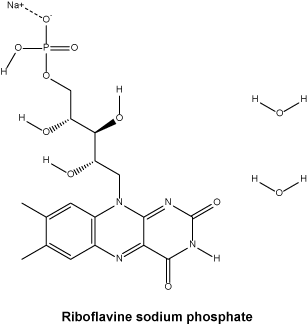
Molecular weight: 514.36.
Chemical name: riboflavin 5'-(dihydrogen phosphate), monosodium salt, dihydrate.
Nicotinamide.
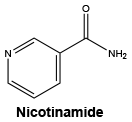
Molecular weight: 122.12.
Chemical name: 3-pyridinecarboxamide.
Pyridoxine hydrochloride.
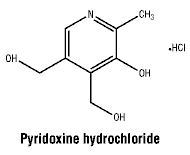
Molecular weight: 205.64.
Chemical name: 3,4-pyridinedimethanol, 5-hydroxy-6-methyl-hydrochloride.
Sodium pantothenate.
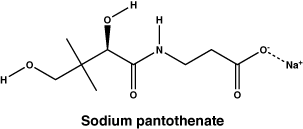
Molecular weight: 241.22.
Chemical name: sodium (R)-3-(2,4-dihydroxy-3,3-dimethylbutanamido) propanoate.
Sodium ascorbate.
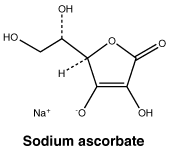
Molecular weight: 198.11.
Chemical name: sodium (2R)-2-[(1S)-1,2-dihydroxyethyl]-4-hydroxy-5-oxo-2H-furan-3-olate.
Biotin.
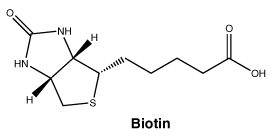
Molecular weight: 244.31.
Chemical name: 5-[(3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d] imidazol-4-yl] pentanoic acid.
Folic acid.
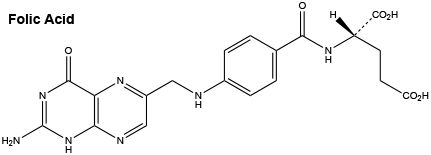
Molecular weight: 441.40.
Chemical name: (2S)-2-[[4-[(2-Amino-4-oxo-1H-pteridin-6 yl) methylamino] benzoyl] amino] pentanedioic acid.
Cyanocobalamin.
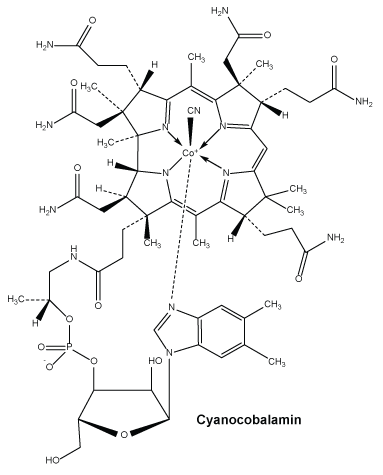
Molecular weight: 1355.37.
Chemical name: cyanocobalamin.
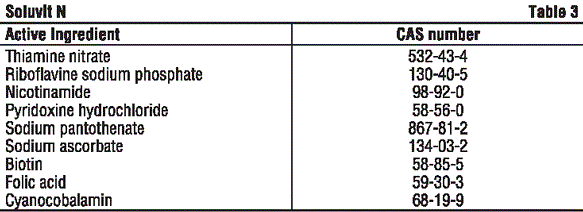
CAS number. See Table 3.
7 Medicine Schedule (Poisons Standard)
Australia: Not Scheduled.
Date of First Approval
21 August 1992
Date of Revision
02 March 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.