Sudafed Sinus + Anti-inflammatory Pain Relief Caplets
Brand Information
| Brand name | Sudafed Sinus + Anti-inflammatory Pain Relief Caplets |
| Active ingredient | Ibuprofen + Pseudoephedrine hydrochloride |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Sudafed Sinus + Anti-inflammatory Pain Relief Caplets.
Full CMI
SUDAFED Sinus + Anti-inflammatory Pain Relief
Active ingredient(s): Pseudoephedrine hydrochloride and Ibuprofen
Consumer Medicine Information (CMI)
This leaflet provides important information about using Sudafed Sinus + Anti-inflammatory Pain Relief. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Sudafed Sinus + Anti-inflammatory Pain Relief.
Where to find information in this leaflet:
1. Why am I using Sudafed Sinus + Anti-inflammatory Pain Relief?
2. What should I know before I use Sudafed Sinus + Anti-inflammatory Pain Relief?
3. What if I am taking other medicines?
4. How do I use Sudafed Sinus + Anti-inflammatory Pain Relief?
5. What should I know while using Sudafed Sinus + Anti-inflammatory Pain Relief?
6. Are there any side effects?
7. Product details
1. Why am I using Sudafed Sinus + Anti-inflammatory Pain Relief?
Sudafed Sinus + Anti-inflammatory Pain Relief contains the active ingredient Pseudoephedrine hydrochloride and Ibuprofen. Pseudoephedrine hydrochloride belongs to a group of medicines called sympathomimetic decongestants. It works by reducing congestion in the upper respiratory tract, including the nose, nasal passages and sinuses, and making it easier to breathe. Ibuprofen belongs to a group of medicines called non-steroidal anti-inflammatory drugs (NSAIDs).
Sudafed Sinus + Anti-inflammatory Pain Relief is used for the temporary relief of symptoms of sinus pain congestion occurring as a result of cold and flu, allergic rhinitis or sinusitis.
2. What should I know before I use Sudafed Sinus + Anti-inflammatory Pain Relief?
Warnings
Do not use Sudafed Sinus + Anti-inflammatory Pain Relief if:
- you are allergic to Pseudoephedrine hydrochloride, or Ibuprofen, aspirin or other NSAIDs, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you are younger than 12 years
- have very high blood pressure or high blood pressure not controlled by your medication
- renal impairment
- have severe coronary artery disease (heart disease caused by poor blood flow or narrowing of the blood vessels of the heart)
- have taken monoamine oxidase inhibitors, medicines used to treat depression, in the last 14 days
- have asthma that is sensitive to aspirin or NSAIDs
- have a stomach or duodenal ulcer
- have recently vomited blood or material that looks like coffee grounds
- have recently bled from the back passage (rectum), had black sticky bowel motions or bloody diarrhoea
- have alcohol dependence.
Check with your doctor or pharmacist if you:
- have any other medical conditions
- high blood pressure
- overactive thyroid gland
- diabetes
- heart disease and poor blood flow in the blood vessels of the heart
- glaucoma (high pressure in the eyes)
- fluid retention
- prostate problems
- liver disease
- asthma
- gastrointestinal disease
- stomach or duodenal ulcer
- signs of gastrointestinal bleeding, such as vomiting blood or bleeding from the back passage (rectum) - take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
Small amounts of pseudoephedrine pass into the breast milk. Your pharmacist or doctor will discuss the potential benefits and risks of taking the medicine if you are breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Sudafed Sinus + Anti-inflammatory Pain Relief and affect how it works.
These include
- medicines used to treat depression especially monoamine oxidase inhibitors and tricyclic antidepressants
- medicines used to treat high blood pressure
- medicines used to treat urinary tract infections and bladder problems
- medicines used to treat behavioral disorders
- phenylephrine, a medicine used to treat congestion
- appetite suppressants
- warfarin, a medicine used to prevent blood clots
- lithium, a medicine used to treat mood swings and some types of depression
- medicines used to lower blood pressure
- diuretics
- methotrexate, a medicine used to treat arthritis and some types of cancer
- medicines used to treat heart conditions
- medicines such as prednisone, prednisolone and cortisone, which reduce the activity of your immune system
- zidovudine, a medicine used to treat HIV infection
- aspirin, salicylates and other non-steroidal anti-inflammatory drugs (NSAIDs)
- medicines used to treat diabetes.
You may need different amounts of your medicines, or you may need to take different medicines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Sudafed Sinus + Anti-inflammatory Pain Relief.
4. How do I use Sudafed Sinus + Anti-inflammatory Pain Relief?
How much to take
- The recommended dose of Sudafed Sinus + Anti-inflammatory Pain Relief for adults and children over 12 years is 1-2 tablets every 4 to 6 hours, as necessary.
- Follow the instructions provided with the medicine.
- Do not exceed 6 tablets in 24 hours.
- Do not exceed the recommended dosage.
When to take Sudafed Sinus + Anti-inflammatory Pain Relief
- Sudafed Sinus + Anti-inflammatory Pain Relief should be taken every 4 to 6 hours, as necessary.
How to take Sudafed Sinus + Anti-inflammatory Pain Relief
- Swallow the tablets whole.
- Do not use for more than a few days at a time except on medical advice.
- Follow all directions given to you by your pharmacist or doctor carefully. This may differ from the information contained in this leaflet.
- If you forget to take it and it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Otherwise, you may take a dose as soon as you remember if you think you need it. Do not take a double dose to make up for the dose that you missed.
- If you do not understand the instructions on the pack, ask your pharmacist or doctor for help.
If you use too much Sudafed Sinus + Anti-inflammatory Pain Relief
If you think that you have used too much Sudafed Sinus + Anti-inflammatory Pain Relief, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Sudafed Sinus + Anti-inflammatory Pain Relief?
Things you should do
Stop use and talk to your doctor or pharmacist if your symptoms persist, worsen, or if new symptoms occur. Your doctor or pharmacist will assess your condition and decide if you should continue to take the medicine.
Remind any doctor, dentist or pharmacist you visit that you are using Sudafed Sinus + Anti-inflammatory Pain Relief.
Things you should not do
- Do not take Sudafed Sinus + Anti-inflammatory Pain Relief to treat any other complaints unless your pharmacist or doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Do not take more than the recommended dose unless your pharmacist or doctor tells you to.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Sudafed Sinus + Anti-inflammatory Pain Relief affects you.
Sudafed Sinus + Anti-inflammatory Pain Relief may cause dizziness in some people.
If this happens, do not drive or operate machinery.
Drinking alcohol
Tell your doctor or pharmacist if you drink alcohol.
Looking after your medicine
- Keep your medicine in the original pack until it is time to take.
- Keep your medicine in a cool dry place where the temperature stays below 30°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people. Children and people over 65 years if age may have an increased chance of getting side effects.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What Sudafed Sinus + Anti-inflammatory Pain Relief contains
| Active ingredient (main ingredient) | Pseudoephedrine hydrochloride Ibuprofen |
| Other ingredients (inactive ingredients) | Candelilla wax Croscarmellose sodium Microcrystalline cellulose Opadry aqueous film coating YS-1-7034 clear UK Opadry aqueous film coating YS-1-7717 white UK Sodium lauryl sulfate Stearic acid |
Do not take this medicine if you are allergic to any of these ingredients.
What Sudafed Sinus + Anti-inflammatory Pain Relief looks like
White, capsule-shaped tablet.
(Aust R 80163).
Who distributes Sudafed Sinus + Anti-inflammatory Pain Relief
Kenvue Pacific
Australia and New Zealand
Sydney, NSW, Australia and Auckland New Zealand
Phone: 1800 029979
This leaflet was prepared in January 2026.
Brand Information
| Brand name | Sudafed Sinus + Anti-inflammatory Pain Relief Caplets |
| Active ingredient | Ibuprofen + Pseudoephedrine hydrochloride |
| Schedule | S3 |
MIMS Revision Date: 01 January 2026
Notes
Sponsored by Kenvue Pacific Australia New Zealand
1 Name of Medicine
Pseudoephedrine hydrochloride; ibuprofen.
2 Qualitative and Quantitative Composition
Sudafed Sinus + Anti-inflammatory Pain Relief caplets contain pseudoephedrine hydrochloride 30 mg and ibuprofen 200 mg.
Sudafed Sinus + Anti-inflammatory Pain Relief caplets also contain: methyl hydroxybenzoate, propyl hydroxybenzoate.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Sudafed Sinus + Anti-inflammatory Pain Relief caplets are white, capsule-shaped, film-coated tablets.
4 Clinical Particulars
4.1 Therapeutic Indications
Sudafed Sinus + Anti-inflammatory Pain Relief provides relief of symptoms of sinus pain with sinus congestion occurring as a result of cold and flu, allergic rhinitis or sinusitis.
4.2 Dose and Method of Administration
The recommended dosage of Sudafed Sinus + Anti-inflammatory Pain Relief for adults and children over 12 years is 1 or 2 caplets with fluid every four to six hours when necessary. Do not exceed 6 caplets in 24 hours.
Sudafed Sinus + Anti-inflammatory Pain Relief should not be used for children under 12 years of age.
Sudafed Sinus + Anti-inflammatory Pain Relief should not be used for more than a few days at a time except on medical advice, in which case the patient should be reviewed regularly with regards to efficacy, risk factors and ongoing need for treatment. Excessive use can increase the risk of heart attack, stroke or liver damage.
4.3 Contraindications
Sudafed Sinus + Anti-inflammatory Pain Relief is contraindicated for use in patients:
with known hypersensitivity or idiosyncratic reaction to pseudoephedrine or ibuprofen (or any of the other ingredients in the product), other nonsteroidal anti-inflammatory drugs or other salicylates;
with severe or uncontrolled hypertension or severe coronary artery disease;
taking monoamine oxidase inhibitors (MAOIs) or who have taken MAOIs within the previous 14 days;
known hypersensitivity to aspirin and other NSAIDs;
asthma that is aspirin or NSAID sensitive;
active gastrointestinal bleeding or peptic ulceration;
renal impairment;
heart failure;
severe liver impairment;
undergoing treatment of perioperative pain in setting of coronary artery bypass surgery (CABG);
right before or after heart surgery.
Use of ibuprofen is contraindicated during the third trimester of pregnancy.
Use of ibuprofen is contraindicated right before or after heart surgery.
Use of pseudoephedrine is contraindicated in patients with severe acute or chronic kidney disease/renal failure.
Sudafed Sinus + Anti-inflammatory Pain Relief should not be taken with other products containing ibuprofen or with other anti-inflammatory medicines.
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions for additional information.
4.4 Special Warnings and Precautions for Use
Posterior reversible encephalopathy syndrome (PRES) and reversible cerebral vasoconstriction syndrome (RCVS). Cases of PRES and RCVS have been reported with the use of pseudoephedrine-containing products (see Section 4.8). The risk is increased in patients with severe or uncontrolled hypertension, or with severe acute or chronic kidney disease/renal failure (see Section 4.3).
Pseudoephedrine should be discontinued and immediate medical assistance sought if the following symptoms occur: sudden severe headache or thunderclap headache, nausea, vomiting, confusion, seizures and/or visual disturbances. Most reported cases of PRES and RCVS resolved following discontinuation and appropriate treatment.
Identified precautions. Sudafed Sinus + Anti-inflammatory Pain Relief should be used with caution in patients with hypertension, hyperthyroidism or thyroid disease, diabetes mellitus, coronary heart disease, ischaemic heart disease, glaucoma, prostatic hypertrophy, previous history of gastrointestinal haemorrhage or ulcers, asthma who have not previously taken an NSAID, cardiac impairment or heart disease, fluid retention, alcohol dependence, pregnancy (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy) and patients: taking a diuretic, taking anticoagulants, taking corticosteroids.
Due to the ibuprofen component, this medicine should be taken with caution when using other products containing aspirin and salicylates.
Ibuprofen may cause a severe allergic reaction, especially in patients allergic to aspirin. Symptoms include hives, facial swelling, asthma (wheezing), shock, skin reddening, rash or blisters with or without pyrexia or erythema. If any of these symptoms occur, patients should stop use and seek medical help right away.
Ibuprofen has very rarely been reported to cause Vanishing Bile Duct Syndrome. Patients should seek medical advice if they develop a sudden onset abdominal pain or chronic abdominal pain associated with loss of appetite and/or new onset itching.
Due to the pseudoephedrine component, this medicine should be discontinued and medical advice sought if sudden abdominal pain, rectal bleeding or other symptoms of ischaemic colitis develop.
If signs and symptoms such as formation of small pustules occur, with or without pyrexia or erythema, then treatment with this medicine should be discontinued and a physician should be consulted.
Ibuprofen treats fever and pain which sometimes can be signs of a serious underlying condition. If symptoms persist or get worse, or if new symptoms occur, patients should stop use and consult a physician.
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions for additional information.
Cardiovascular and cerebrovascular effects. Observational studies have indicated that NSAIDs may be associated with an increased risk of serious cardiovascular events, including myocardial infarction, stroke and Kounis Syndrome, which may increase with dose or duration of use.
Patients with cardiovascular disease, history of atherosclerotic cardiovascular disease or cardiovascular risk factors may also be at greater risk.
Patients should be advised to remain alert for such cardiovascular events, even in the absence of previous cardiovascular symptoms. Patients should be informed about signs and/or symptoms of serious cardiovascular toxicity and the steps to take if they occur.
Fluid retention, hypertension and oedema have been reported in association with NSAID therapy. Patients taking antihypertensives with NSAIDs may have an impaired antihypertensive response.
Sudafed Sinus + Anti-inflammatory Pain Relief should be used with caution in patients with hypertension (see Section 4.3 Contraindications, heart failure).
Use in hepatic impairment. Sudafed Sinus + Anti-inflammatory Pain Relief should be used with caution for patients with severe hepatic dysfunction or impairment.
As with other NSAIDs elevations of one or more liver function tests may occur in up to 15% of patients. These abnormalities may progress, may remain essentially unchanged, or may resolve with continued therapy. Meaningful elevations (three times the upper limit of normal) of ALT or AST occurred in controlled clinical trials in less than 1% of patients.
Patients should be advised to remain alert for hepatotoxicity and be informed about the signs and/or symptoms of hepatotoxicity (e.g. nausea, fatigue, lethargy, pruritus, jaundice, abdominal tenderness in the right upper quadrant and "flu-like" symptoms).
Use in renal impairment. Sudafed Sinus + Anti-inflammatory Pain Relief is contraindicated for use in patients with renal impairment (see Section 4.3 Contraindications).
Use in the elderly. Ibuprofen should not be taken by adults over the age of 65 without careful consideration of co-morbidities and co-medications because of an increased risk of adverse effects, in particular, heart failure, gastro-intestinal ulceration and renal impairment (also see Section 4.3 Contraindications).
Paediatric use. Sudafed Sinus + Anti-inflammatory Pain Relief should not be used for children under 12 years of age.
Effects on laboratory test. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The following interactions with pseudoephedrine have been noted:
Antidepressant medication, e.g. tricyclic antidepressants and monoamine oxidase inhibitors (MAOIs) - may cause a serious increase in blood pressure or hypertensive crisis.
Other sympathomimetic agents, such as decongestants, appetite suppressants and amphetamine-like psychostimulants - may cause an increase in blood pressure and additive effects.
Methyldopa and β-blockers - may cause an increase in blood pressure.
Urinary acidifiers enhance elimination of pseudoephedrine.
Urinary alkalinisers decrease elimination of pseudoephedrine.
The following interactions with ibuprofen have been noted:
Anticoagulants, including warfarin - ibuprofen interferes with the stability of INR and may increase risk of severe bleeding and sometimes fatal haemorrhage, especially from the gastrointestinal tract. Ibuprofen should only be used in patients taking warfarin if absolutely necessary and they must be closely monitored.
Ibuprofen may decrease the cardioprotective and antiplatelet activity of aspirin.
Ibuprofen may decrease renal clearance and increase plasma concentration of lithium.
Ibuprofen may reduce the antihypertensive effect of ACE inhibitors, beta-blockers and diuretics and may cause natriuresis and hyperkalemia in patients under these treatments.
Ibuprofen reduces methotrexate clearance.
Ibuprofen may increase plasma levels of cardiac glycoside.
Ibuprofen may increase the risk of gastrointestinal bleeding especially if taken with corticosteroids or with alcohol use.
Ibuprofen may prolong bleeding time in patients treated with zidovudine.
Alcohol use may increase the risk of gastrointestinal bleeding when taking drugs in the NSAID class, including ibuprofen. Therefore, caution should be taken when using ibuprofen with alcohol.
Ibuprofen may also interact with probenecid, antidiabetic medicines and phenytoin.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category C)
Data from epidemiological studies suggest an increased risk of miscarriage after the use of a prostaglandin synthesis inhibitor in early pregnancy.
Pseudoephedrine has been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human foetus having been observed. Studies in animals are inadequate or may be lacking, but available data shows no evidence of an increased occurrence of foetal damage.
Ibuprofen inhibits prostaglandin synthesis and, when given during the latter part of pregnancy, may cause closure of the foetal ductus arteriosus, foetal renal impairment, inhibition of platelet aggregation and may delay labour and birth. Use of ibuprofen is thus contraindicated during the third trimester of pregnancy, including the last few days before expected birth.
Further, there is insufficient experience about the safety of use of ibuprofen in humans during pregnancy. Sudafed Sinus + Anti-inflammatory Pain Relief should therefore not be used during the first six months of pregnancy unless the potential benefits to the patient outweigh the possible risk to the foetus.
Use in lactation. Pseudoephedrine is secreted in breast milk in small amounts. It has been estimated that 0.5% to 0.7% of a single dose of pseudoephedrine ingested by the mother will be excreted in the breast milk over 24 hours. Therefore, it is not recommended for breastfeeding mothers unless the potential benefits to the patient are weighed against the possible risk to the infant.
Ibuprofen appears in breast milk in very low concentrations and is unlikely to affect the breast fed infant adversely.
4.7 Effects on Ability to Drive and Use Machines
It is not known if the combination of ibuprofen and pseudoephedrine has an effect on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
Children and the elderly are more likely to experience adverse effects than other age groups.
Clinical trial data. The safety of the combination of ibuprofen and pseudoephedrine from clinical trial data is based on data from 4 double-blind placebo-controlled single dose randomized studies in the treatment of sinus headache.
Table 1 includes adverse events that occurred where greater than one event was reported, and the incidence was greater than placebo and in 1% of patients or more.
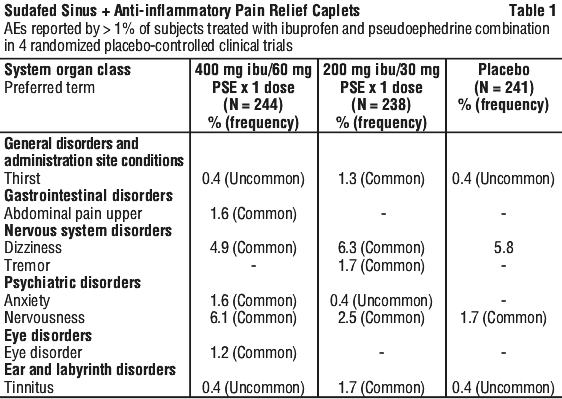

Healthcare professionals are asked to report any suspected adverse reactions at: www.tga.gov.au/reporting-problems.
4.9 Overdose
Ibuprofen. The toxicity of ibuprofen overdose is dependent upon the amount of drug ingested and the time elapsed since ingestion. Individual response may vary, and each case should be evaluated individually. Although uncommon, serious toxicity and death have been reported in association with acute ibuprofen overdose.
The most frequently reported symptoms of acute ibuprofen overdose include abdominal pain, nausea, vomiting, lethargy and drowsiness. Other central nervous system symptoms following acute overdose include headache, tinnitus, CNS depression and seizures. Metabolic acidosis, coma, acute renal failure, renal tubular acidosis, rhabdomyolysis, hypothermia, fulminant hepatic failure and apnea (primarily in very young children) may rarely occur, and are more common with severe overdoses of more than 400 mg/kg. Cardiovascular toxicity, including hypotension, bradycardia, tachycardia and atrial fibrillation, also have been reported. Onset of symptoms usually occurs within 4 hours.
Pseudoephedrine. Overdosage may result in nausea, vomiting, sympathomimetic symptoms including central nervous system stimulation, insomnia, tremor, mydriasis, anxiety, agitation, hallucinations, seizures, palpitations, tachycardia, hypertension, and reflex bradycardia. Other effects may include dysrhythmias, hypertensive crisis, intracerebral hemorrhage, myocardial infarction, psychoses, rhabdomyolysis, hypokalemia, and ischemic bowel infarction. Drowsiness has been reported with overdose in children.
In case of overdose, immediately contact the Poisons Information Centre (in Australia, call 13 11 26; in New Zealand call 0800 764 766) for advice.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Pseudoephedrine has direct and indirect sympathomimetic activity and is an effective decongestant in the upper respiratory tract. It is a stereoisomer of ephedrine and has a similar action, but has been found to have less pressor activity and fewer central nervous system (CNS) effects.
Sympathomimetic agents are used as nasal decongestants to provide symptomatic relief. They act by causing vasoconstriction resulting in redistribution of local blood flow to reduce oedema of the nasal mucosa, thus improving ventilation, drainage and nasal stuffiness.
Ibuprofen possesses analgesic, antipyretic and anti-inflammatory properties, similar to other non-steroidal anti-inflammatory drugs (NSAIDs). Its mechanism of action is unknown, but is thought to be through peripheral inhibition of cyclooxygenases and subsequent prostaglandin synthetase inhibition.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Pseudoephedrine is readily absorbed from the gastrointestinal tract. It is largely excreted unchanged in the urine together with small amounts of its hepatic metabolite. It has a half-life of about 5-8 hours; elimination is enhanced and half-life reduced accordingly in acid urine.
Ibuprofen is well absorbed from the gastrointestinal tract. It is highly bound (90-99%) to plasma proteins and is extensively metabolised to inactive compounds in the liver, mainly by glucuronidation. Both the inactive metabolites and a small amount of unchanged ibuprofen are excreted rapidly and completely by the kidney, with 95% of the administered dose eliminated in the urine within four hours of ingestion. The elimination half-life of ibuprofen is in the range of 1.9 to 2.2 hours.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Candelilla wax, microcrystalline cellulose, croscarmellose sodium, sodium lauryl sulfate, stearic acid, methyl hydroxybenzoate, propyl hydroxybenzoate, Opadry Aqueous Film Coating YS-1-7034 Clear UK, Opadry Aqueous Film Coating YS-1-7717 White UK.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
2 years.
6.4 Special Precautions for Storage
Store below 25°C. Keep in a dry dark place.
6.5 Nature and Contents of Container
Sudafed Sinus + Anti-inflammatory Pain Relief caplets are available in blister packs of Alu/PVC/PVDC in the following sizes: 4 caplets; 12 caplets; 24 caplets.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
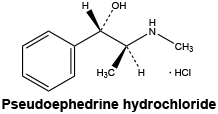
Chemical structure. Pseudoephedrine hydrochloride:
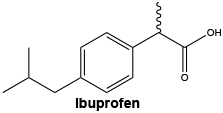
Ibuprofen:
7 Medicine Schedule (Poisons Standard)
Pharmacist Only Medicine (Schedule 3).
Date of First Approval
04 October 2006
Date of Revision
28 November 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.