Takhzyro
Brand Information
| Brand name | Takhzyro |
| Active ingredient | Lanadelumab |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Takhzyro.
Summary CMI
TAKHZYRO®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
▼ This medicine is new or being used differently. Please report side effects. See the full CMI for further details.
1. Why am I using TAKHZYRO?
TAKHZYRO contains the active ingredient lanadelumab. TAKHZYRO is used in patients aged 2 years and older to prevent angioedema attacks in hereditary angioedema (HAE). For more information, see Section 1. Why am I using TAKHZYRO? in the full CMI.
2. What should I know before I use TAKHZYRO?
Do not use if you have ever had an allergic reaction to TAKHZYRO or any of the ingredients listed at the end of the CMI. Do not give TAKHZYRO to a person below 2 years of age.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. Tell your doctor if you are using TAKHZYRO before you have laboratory tests to measure how well your blood is clotting. For more information, see Section 2. What should I know before I use TAKHZYRO? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with TAKHZYRO and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use TAKHZYRO?
- TAKHZYRO should be used exactly as your doctor has told you
- TAKHZYRO must be injected under the skin (subcutaneously).
More instructions can be found in Section 4. How do I use TAKHZYRO? in the full CMI.
5. What should I know while using TAKHZYRO?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using TAKHZYRO? in the full CMI.
6. Are there any side effects?
Serious side effects: allergic reactions which may result in shortness of breath; wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; or rash, itching or hives on the skin. Common side effects include injection site reactions (including pain, skin redness, bruising, discomfort, swelling, bleeding, itching, hardening of skin, tingling, warmth and rash), dizziness or feeling faint, muscle pain, raised skin rash, allergic reactions (including itching, discomfort, tingling, of the tongue), abnormal liver-related blood test results.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
▼ This medicine is subject to additional monitoring due to approval of an extension of indications. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems.
TAKHZYRO®
Active ingredient(s): lanadelumab
Consumer Medicine Information (CMI)
This leaflet provides important information about using TAKHZYRO. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using TAKHZYRO.
TAKHZYRO is a new medicine. Please check with your doctor whether there is any additional information about this medicine that you should know since you were last treated with this medicine.
Where to find information in this leaflet:
1. Why am I using TAKHZYRO?
2. What should I know before I use TAKHZYRO?
3. What if I am taking other medicines?
4. How do I use TAKHZYRO?
5. What should I know while using TAKHZYRO?
6. Are there any side effects?
7. Product details
1. Why am I using TAKHZYRO?
TAKHZYRO contains the active ingredient lanadelumab. It is a type of protein that blocks the activity of plasma kallikrein. This helps to reduce the amount of bradykinin in your bloodstream and prevents symptoms associated with HAE.
TAKHZYRO is used in patients aged 2 years and older to prevent angioedema attacks in hereditary angioedema (HAE).
2. What should I know before I use TAKHZYRO?
Warnings
Do not use TAKHZYRO if:
you are allergic to lanadelumab, or any of the ingredients listed at the end of this leaflet. When using TAKHZYRO for the first time, check the ingredients to make sure you can use the medicine.
Do not give this medicine to a person below 2 years of age.
Check with your doctor if you:
- are using TAKHZYRO before you have laboratory tests to measure how well your blood is clotting. TAKHZYRO in the blood may interfere with some laboratory tests, leading to inaccurate results.
- take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
TAKHZYRO may affect other medicines or be affected by them. Your doctor and pharmacist have more information on medicines to be careful with or avoid while using this medicine.
4. How do I use TAKHZYRO?
How much to use
- Adult patients: the recommended starting dose is 300 mg every 2 weeks. If you have not had an attack for a long period, your doctor may change the dose to 300 mg every 4 weeks, especially if you have a low body weight
- 12 to <18 years patients: the recommended dose is 300 mg every 2 weeks. If you have not had an attack for a long period, your doctor may change the dose to 300 mg every 4 weeks
- 6 to <12 years patients: the recommended dose is 150 mg every 2 weeks. If you have not had an attack for a long period, your doctor may change the dose to 150 mg every 4 weeks
- 2 to <6 years patients: the recommended dose is 150 mg every 4 weeks.
- If you are not sure how much and how frequently you need to inject TAKHZYRO, ask your doctor, pharmacist, or nurse
How to use TAKHZYRO
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor, pharmacist, or nurse for help.
TAKHZYRO should be injected under the skin (subcutaneously).
The injection can be self-administered or given by another person, for example your carer, your doctor, his/her assistant or your nurse.
If you are injecting the medicine yourself, you must receive adequate training by your doctor or nurse. You will find detailed instructions for injections at the end of this leaflet.
Instructions for administering TAKHZYRO
Important information
Read the leaflet carefully before using TAKHZYRO.
TAKHZYRO is for injection under the skin (subcutaneous injection). Do not inject it into a vein or muscle.
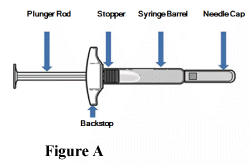
Parts of your TAKHZYRO pre-filled syringe before use (Figure A):
STEP 1: Prepare for your injection
1.1 Gather a sharps disposal container, alcohol swab, cotton ball/gauze pad and adhesive bandage. Place them on a clean, flat surface in a well-lighted area. These items are not included in the TAKHZYRO pack.
1.2 Take out the TAKHZYRO pack from the refrigerator, open the carton box and remove the TAKHZYRO pre-filled syringe from the tray.
- Do not use the TAKHZYRO pre-filled syringe if the tamper evident seal is open or broken.
- Before you prepare your injection, allow the pre-filled syringe to reach room temperature for at least 15 to 30 minutes.
- Your medicine is sensitive to warm temperature. Do not use external heat sources such as hot water to warm your TAKHZYRO pre-filled syringe.
- Do not remove the needle cap until you are ready to inject the medicine.
1.3 Wash your hands with soap and water (Figure B). Dry your hands completely.
1.4 Check the expiry date on the label (Figure C).
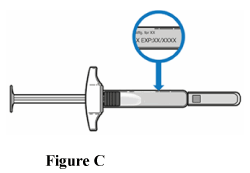
- Do not use the TAKHZYRO pre-filled syringe if the expiry date has passed.
1.5 Visually inspect the TAKHZYRO pre-filled syringe for any damage and make sure the medicine is colourless to slightly yellow.
- Do not use it if the syringe is damaged (e.g., cracked syringe)
- Do not use it if the solution is discoloured, cloudy, or has flakes or particles in it
- You might see air bubbles in the TAKHZYRO pre-filled syringe. This is normal and will not affect your dose.
STEP 2: Select and prepare your injection site
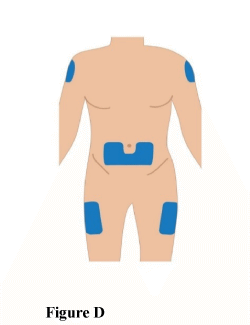
2.1 The TAKHZYRO pre-filled syringe should be injected into your stomach (abdomen), thigh, or the back of the upper arm (only if a caregiver is giving you the injection) (Figure D).
- It is important to rotate injection sites to keep skin healthy. Each new injection should be given at least 2.5 cm (1 inch) from the last site you used
- Do not inject into an area of your body where the skin is irritated, reddened, bruised, or infected
- The area you choose for injection should be at least 5 cm (2 inches) away from any scars or your belly button (navel)
2.2 Clean the injection site with an alcohol swab and allow it to dry
- Do not fan or blow on the cleaned site
- Do not touch this area again before giving your injection.
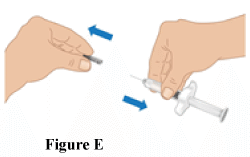
2.3 Remove needle cap from the TAKHZYRO pre-filled syringe. Gently pull the needle cap straight off with one hand and firmly hold the middle of TAKHZYRO pre-filled syringe with the other hand (Figure E). Throw away the needle cap.
- Do not recap your TAKHZYRO pre-filled syringe
- Do not use the TAKHZYRO pre-filled syringe if it has been dropped without the needle-cap on or if the needle looks damaged or bent
- Do not touch the needle or allow the needle to touch anything.
STEP 3: Inject TAHKZYRO
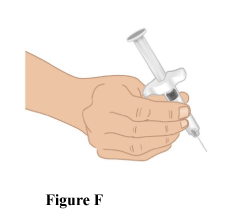
3.1 Grip the TAKHZYRO pre-filled syringe in one hand like a pencil. Avoid touching the needle or pushing on the plunger (Figure F).
3.2 With your hand, gently pinch a 2.5 cm (1 inch) fold of skin at the cleaned injection site.
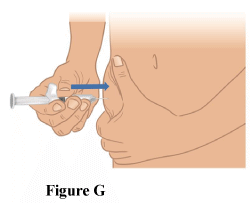
3.3 Using a quick dart-like motion, insert the needle. Make sure to keep the needle in place (Figure G).
3.4 Slowly push the plunger until all of the liquid is injected and the syringe is empty, then gently let go of your skin.
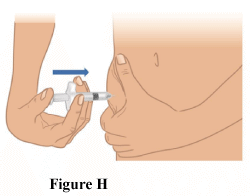
3.5 Slowly withdraw the needle while maintaining the syringe at the same angle (Figure H).
3.6 Press cotton ball or gauze pad over the injection site if needed and hold for 10 seconds.
- Do not rub the injection site. You may have a minor bleeding. This is normal
- Cover the injection site with an adhesive bandage if needed.
3.7 Throw away your used TAKHZYRO pre-filled syringe.
- Do not touch the needle
- To avoid a needle-stick injury, do not recap the needle
- Put your used TAKHZYRO pre-filled syringe in a sharps disposal container right away after use
- Do not reuse the TAKHZYRO pre-filled syringe and any of your injection items.
Important: Always keep the sharps disposal container out of reach of children.
If you forget to use TAKHZYRO
If you forget to use this medicine (or cannot inject it at your usual time), use it as soon as possible but there must be at least 10 days between each dose for adult and 6 to <12 years patients, and at least 24 days for patients aged 2 to <6 years.
Do not inject a double dose to make up for the dose that you missed.
If you have trouble remembering to use your medicine, ask your pharmacist or nurse for some hints.
If you use too much TAKHZYRO
If you think that you have used too much TAKHZYRO, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(Australia: 13 11 26; New Zealand: 0800 POISON or 0800 764766) for advice, or - go to the Accident and Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using TAKHZYRO?
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are using TAKHZYRO.
Tell any other doctors, dentists, and pharmacists who treat you that you are using this medicine.
If you become pregnant while using this medicine, tell your doctor immediately.
It is strongly recommended that every time you inject a dose of TAKHZYRO, write down the date and batch number of the medicine. This is so that you keep a record of the batches used in case you have a severe allergic reaction to TAKHZYRO.
Things you must not do
Do not use TAKHZYRO to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop using your medicine or change the dosage without checking with your doctor.
Driving or using machines
Be careful driving or operating machinery until you know how TAKHZYRO affects you.
Do not drive or use machines if you feel dizzy after using TAKHZYRO.
Looking after your medicine
Keep your medicine in the original carton to protect the pre-filled syringe from light until it is time to use it.
If you take the medicine out of the pack it may not keep well.
Keep the medicine in the refrigerator at 2°C to 8°C. Do not freeze it.
TAKHZYRO pre-filled syringe removed from refrigeration should be stored below 25°C and used within 14 days. Do not return pre-filled syringes to refrigerated storage after storage at room temperature.
Do not shake TAKHZYRO.
Do not store TAKHZYRO or any other medicine in the bathroom or near a sink. Do not leave it on a windowsill or in the car.
Heat and dampness can destroy some medicines.
Keep it where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If your doctor tells you to stop using this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Contact your doctor or go to the Accident and Emergency Dept. at the nearest hospital immediately if any of these serious side effects occur |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What TAKHZYRO contains
| Active ingredient | Lanadelumab |
| Other ingredients |
|
Do not take this medicine if you are allergic to any of these ingredients.
What TAKHZYRO looks like
TAKHZYRO, a ready-to-use solution for injection under the skin (subcutaneous injection), is supplied in a single-dose, pre-filled glass syringe.
Each TAKHZYRO pre-filled syringe contains either 300 mg of lanadelumab in 2 mL solution or 150 mg of lanadelumab in 1 mL solution.
TAKHZYRO solution is colourless to slightly yellow in colour.
Each pack contains one pre-filled syringe fitted with a 27-gauge needle for injection (see 'Instructions for administering TAKHZYRO' above).
(AUST R 330280 and 409942).
Who distributes TAKHZYRO
Australia:
Takeda Pharmaceuticals Australia Pty Ltd
Level 39, 225 George Street
Sydney NSW 2000
Australia
Telephone: 1800 012 612
www.takeda.com/en-au
New Zealand:
Takeda New Zealand Limited
Level 10, 21 Queen Street
Auckland 1010
New Zealand
Telephone: 0508 169 077
www.takeda.com/en-au
This leaflet was prepared in July 2024.
TAKHZYRO® is a registered trademark of Dyax Corp., a Takeda company. TAKEDA and the TAKEDA Logo are registered trademarks of Takeda Pharmaceutical Company Limited.
Brand Information
| Brand name | Takhzyro |
| Active ingredient | Lanadelumab |
| Schedule | S4 |
▼ This medicinal product is subject to additional monitoring in Australia. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse events at www.tga.gov.au/reporting-problems.
MIMS Revision Date: 01 September 2024
1 Name of Medicine
Lanadelumab.
2 Qualitative and Quantitative Composition
Ready-to-use solution, for subcutaneous injection only.
Takhzyro is a sterile, preservative-free solution supplied in a single-dose pre-filled syringe or a single-dose vial.
Each pre-filled syringe contains either 300 mg of lanadelumab in 2 mL solution or 150 mg of lanadelumab in 1 mL solution. Each vial contains 300 mg of lanadelumab in 2 mL solution.
Excipient with known effect. Each mL of solution contains 3.45 mg (0.150 mmol) of sodium.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection.
The solution is colourless to slightly yellow, appearing either clear or slightly opalescent.
4 Clinical Particulars
4.1 Therapeutic Indications
Takhzyro is indicated for routine prevention of recurrent attacks of hereditary angioedema (C1-esterase-inhibitor deficiency or dysfunction) in patients aged 2 years and older.
4.2 Dose and Method of Administration
Takhzyro therapy should be initiated under supervision of a physician experienced in the care of patients with hereditary angioedema (HAE).
Dosage. Adult patients. The recommended starting dose is 300 mg lanadelumab every 2 weeks administered subcutaneously. In patients who are well-controlled (e.g. attack free) on treatment, a dose reduction of 300 mg lanadelumab every 4 weeks may be considered, especially in patients with low weight.
Elderly. Limited information is available on patients above 65 years of age. Available data indicates that no dose adjustment is required for patients above 65 years of age.
Hepatic impairment. No studies have been conducted in patients with hepatic impairment.
Renal impairment. No studies have been conducted in patients with renal impairment.
Paediatric patients. 12 to < 18 years. The recommended dose is 300 mg administered subcutaneously every 2 weeks. A dosing interval of 300 mg every 4 weeks may be considered if the patient is well-controlled (e.g. attack-free) for 26 weeks (see Section 5.1 Pharmacodynamic Properties).
6 to < 12 years. The recommended dose is 150 mg administered subcutaneously every 2 weeks. A dosing interval of 150 mg every 4 weeks may be considered if the patient is well-controlled (e.g. attack free) for 26 weeks (see Section 5.1 Pharmacodynamic Properties).
2 to < 6 years. The recommended dose is 150 mg administered subcutaneously every 4 weeks (see Section 5.1 Pharmacodynamic Properties).
The safety and efficacy of Takhzyro in children aged < 2 years has not been established and therefore treatment in children aged < 2 years is not recommended.
The product is for single use in one patient only. Discard any residue.
Method of administration. Takhzyro is administered subcutaneously only.
Takhzyro is provided as a ready-to-use solution that does not require additional reconstitution or dilution for administration. Each Takhzyro pre-filled syringe or vial is intended for single use only. Do not use the pre-filled syringe or vial if the solution appears discoloured or contains visible particles. Avoid vigorous agitation of the pre-filled syringe or vial.
Takhzyro may be administered by a healthcare professional or by the patient/caregiver. The decision on the use of home treatment for an individual patient should be made by the treating physician, who should ensure that appropriate training is provided. The patient or caregiver should receive clear instructions and adequate training on how to perform subcutaneous administration. A healthcare professional should review the self-administration method at intervals to ensure the continued appropriate administration.
Detailed instructions for administration are provided in the Consumer Medicine Information that is included as a package insert and may be used as a training guide.
If a dose of Takhzyro is missed, instruct the patient or caregiver to administer the dose as soon as possible ensuring at least 10 days between the doses.
For paediatric patients, if a dose of Takhzyro is missed, instruct the caregiver to administer the dose as soon as possible, ensuring at least 10 days (6 to < 12 years) and at least 24 days (2 to < 6 years) between the doses.
Administration steps. Adult and paediatric patients (12 to < 18 years). For single-dose 2 mL pre-filled syringe. Inject Takhzyro subcutaneously into the abdomen, thigh, or upper arm. Patients should inject the complete dose as prescribed by their physician.
For single-dose 2 mL vial. An aseptic technique must be used. The 18 gauge needle is used to withdraw the Takhzyro dose from the vial and the 27 gauge needle is used to administer the complete dose as prescribed subcutaneously. Takhzyro may be administered into the abdomen, thigh, or upper arm. Takhzyro should be administered within 2 hours of preparing the dosing syringe at room temperature. After the dosing syringe is prepared, it can be refrigerated at 2°C to 8°C and must be used within 8 hours.
Discard any unused portions of drug remaining in the vial and syringe.
Paediatric patients (2 to < 12 years). For single-dose 1 mL prefilled syringe. Inject Takhzyro subcutaneously into the abdomen, thigh or upper arm. The complete dose should be injected as prescribed.
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special Warnings and Precautions for Use
General. Takhzyro should not be used to treat an acute attack. Patients and caregivers should continue to be prepared to treat attacks with acute HAE treatments when necessary.
There are no available clinical data on the use of Takhzyro in HAE patients with normal C1 esterase inhibitor activity.
In order to improve the traceability of biological medicinal products, the name and the batch number of the administered product should be clearly recorded.
Hypersensitivity reactions. Hypersensitivity reactions have been observed. In case of a severe hypersensitivity reaction, discontinue Takhzyro administration and institute appropriate treatment.
Use in hepatic impairment. No dedicated study has been conducted in subjects with hepatic impairment.
Use in renal impairment. No dedicated study has been conducted in subjects with renal impairment (see Section 5.2 Pharmacokinetic Properties, Special populations).
Use in the elderly. The clinical studies included 11 subjects aged ≥ 65 years, with 5 included in the main efficacy study. Results of the subgroup analysis by age were consistent with overall study results (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Paediatric use. The safety of Takhzyro 300 mg/2 mL was evaluated in a subgroup of 23 subjects aged 12 to < 18 years (HELP studies), with 10 included in the main efficacy study. The safety of Takhzyro was also evaluated at 150 mg/1 mL in a study with 21 subjects aged 2 to < 12 years (SPRING study). Safety and tolerability results for paediatric subjects (2 to < 18) were consistent with overall study results for all subjects (see Section 5.1 Pharmacodynamic Properties; 5.2 Pharmacokinetic Properties).
Effects on laboratory tests. Coagulation tests. Takhzyro can increase activated partial thromboplastin time (aPTT) due to an interaction of Takhzyro with the aPTT assay. The reagents used in the aPTT laboratory test initiate intrinsic coagulation through the activation of plasma kallikrein in the contact system. Inhibition of plasma kallikrein by Takhzyro can increase aPTT in this assay. None of the increases in aPTT in patients treated with Takhzyro were associated with abnormal bleeding adverse events. There were no differences in international normalised ratio (INR) between treatment groups.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No dedicated drug interaction studies have been conducted. Based on the characteristics of lanadelumab, no pharmacokinetic interactions with co-administered medicinal products are expected.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Takhzyro effect on fertility has not been evaluated in humans.
In a 13-week study, once weekly subcutaneous administration at doses of 10 or 50 mg/kg (highest dose tested) lanadelumab had no effects on semen sample weight, total sperm count, sperm density, motility and morphology, testicular measurements, spermatogenesis staging, menstrual cycle length, or reproductive organs (organ weights, macroscopic and microscopic findings). Exposures in sexually mature cynomolgus monkeys were approximately 18-fold greater than that noted at 300 mg every 2 weeks based on AUC.
Use in pregnancy. (Category B1)
Takhzyro has not been studied in pregnant women. There are no or limited amount of data from the use of lanadelumab in pregnant women. A risk to the pregnant woman or developing fetus cannot be excluded. A decision should be made whether to initiate or discontinue treatment with Takhzyro, taking into account the risk/benefit of therapy.
The effects of lanadelumab were evaluated in an enhanced pre- and postnatal developmental (ePPND) toxicity study. In the ePPND study in pregnant cynomolgus monkeys administered once weekly doses of 10 or 50 mg/kg (highest dose tested) from gestation day 20 to delivery, there were no lanadelumab-related effects on pregnancy and parturition, embryo-fetal development, as well as survival, growth, and/or postnatal development of offspring. Exposures in the ePPND study were approximately 28-fold greater than that noted at 300 mg every 2 weeks based on AUC.
Use in lactation. Takhzyro has not been studied in lactating women. It is not known whether lanadelumab is present in human milk therefore a risk to the newborns/infants cannot be excluded. The developmental and health benefits of breastfeeding should be taken into consideration along with the mother's medical need for Takhzyro as well as any potential adverse effects on both the infant and the mother.
Available pharmacokinetic data from the ePPND study in cynomolgus monkeys have shown low excretion of lanadelumab in milk at approximately 0.2% of the maternal plasma level.
4.7 Effects on Ability to Drive and Use Machines
Takhzyro has negligible influence on the ability to drive or use machines.
4.8 Adverse Effects (Undesirable Effects)
The safety of lanadelumab was evaluated in 4 clinical studies: a Phase 1a, randomised, double-blind, placebo-controlled study in healthy subjects; a Phase 1b, randomised, double-blind, placebo-controlled, multiple-ascending dose study in subjects with HAE; a pivotal Phase 3, randomised, double-blind, and placebo-controlled study (HELP study) in subjects with HAE; and an open-label extension study (HELP study extension), which includes both subjects from the HELP study (rollover) and additional non-rollover HAE subjects. Two hundred and fifty-seven (257) unique subjects (233 subjects with HAE and 24 healthy subjects) were exposed to at least one dose of lanadelumab.
The safety data described below reflect exposure to Takhzyro in the HELP study and in the HELP study extension; in total, 220 subjects received treatment with lanadelumab in one or both of the studies.
Treatment-emergent adverse events that occurred in ≥ 5% of lanadelumab-treated subjects (overall) in the HELP study are presented in Table 1.
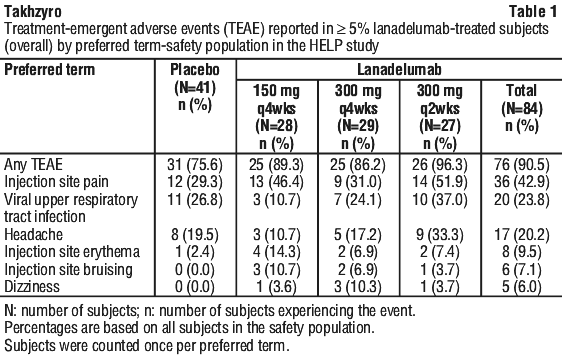
Hypersensitivity reaction (mild and moderate pruritus, discomfort and tingling of tongue) was observed (1.2%) (see Section 4.4 Special Warnings and Precautions for Use, Hypersensitivity reactions).
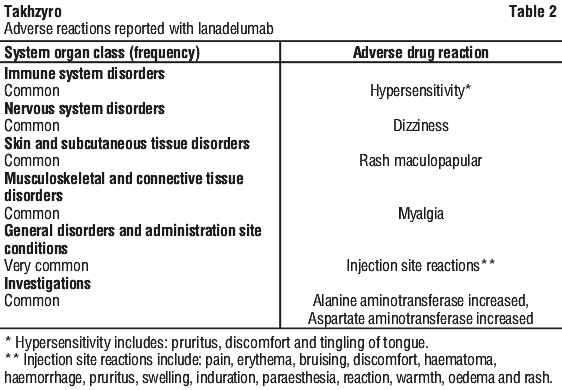
Safety data available from the HELP study extension are consistent with safety data from the HELP study (described in Table 2).
Paediatric population. The safety of Takhzyro 300 mg/2 mL was evaluated in a subgroup of 23 subjects aged 12 to < 18 years old (HELP studies). The safety of Takhzyro was also evaluated at 150 mg/1 mL in a study with 21 subjects aged 2 to < 12 years (SPRING study). No new adverse reactions were identified. Safety and tolerability results for paediatric subjects (2 to < 18 years) were consistent with the overall study results for all subjects (see Section 5.1 Pharmacodynamic Properties; Section 5.2 Pharmacokinetic Properties).
Immunogenicity. In the HELP study, 10 (12%) lanadelumab-treated and 2 (5%) placebo-treated subjects had at least 1 anti-drug antibody (ADA)-positive sample during treatment period (26 weeks); antibody titres were low (range: 20 to 1280). The ADA response observed was transient in 2 of 10 lanadelumab-treated and 1 of 2 placebo-treated subjects. Pre-existing low titre antibodies were observed in 3 lanadelumab-treated subjects and 1 placebo-treated subject with ADAs. Two subjects receiving 150 mg every 4 weeks had low titre antibodies classified as neutralising.
In the SPRING study, 3/20 (15%) of lanadelumab-treated patients developed anti-lanadelumab antibodies (at 52 weeks); all of which were in the 6 to < 12 years group. The ADA response observed was transient in all 3 patients. None of these patients had pre-existing antibodies. One patient had neutralising antibodies.
The development of ADA including neutralising antibodies against Takhzyro did not appear to adversely affect pharmacokinetics, pharmacodynamics, safety or clinical response.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There is no clinical experience with overdosage of Takhzyro.
For information on the management of overdose, contact the Poisons Information Centre on 131126 in Australia, or the National Poisons Centre on 0800 POISON (0800 764766) in New Zealand.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Drugs used in hereditary angioedema, monoclonal antibody, ATC code: B06AC05.
Mechanism of action. Lanadelumab is a fully human, monoclonal antibody (IgG1/κ-light chain) produced in Chinese Hamster Ovary (CHO) cells by recombinant DNA technology. Lanadelumab inhibits active plasma kallikrein proteolytic activity without binding prekallikrein, the inactive precursor found in the circulation. Increased plasma kallikrein activity leads to angioedema attacks in patients with HAE through the proteolysis of high-molecular-weight-kininogen (HMWK) to generate cleaved HMWK (cHMWK) and bradykinin, a potent vasodilator that increases vascular permeability resulting in swelling and pain associated with HAE. It has been demonstrated that patients with HAE due to C1 esterase inhibitor deficiency or dysfunction have increased plasma kallikrein activity, as indirectly measured by amount of cHMWK, both during and in between HAE attacks. Lanadelumab provides sustained control of plasma kallikrein activity and thereby limits bradykinin generation in patients with HAE.
Pharmacodynamic effects. At pharmacokinetic steady-state in adult and paediatric (12 to < 18 years) patients, similar inhibition of plasma kallikrein, measured as reduction of cHMWK levels, was demonstrated after subcutaneous administration of Takhzyro 150 mg every 4 weeks, 300 mg every 4 weeks, or 300 mg every 2 weeks in subjects with HAE.
The pharmacokinetic-pharmacodynamic relationship between Takhzyro and cHMWK is described by an indirect exposure-response pharmacological model. The cHMWK formation rate was maximally reduced by 53.7% with an IC50 of 5705 nanogram/mL.
Serial 12 lead ECG monitoring in the clinical studies found that Takhzyro did not prolong the QT/QTc interval.
For paediatric patients 2 to < 6 years (150 mg every 4 weeks) and 6 to < 12 years (150 mg every 2 weeks), the observed mean percent change from baseline in cHMWK levels was similar to that observed in adult and paediatric (12 to < 18 years) patients.
Clinical trials. HELP study. The HELP study investigated efficacy and safety of Takhzyro for routine prevention of attacks of HAE in subjects 12 years of age and older in a multicentre, randomised, double-blind, placebo-controlled parallel-group study.
The study included 125 adult and paediatric (12 to < 18 years) subjects with symptomatic type I or II HAE, including 10 subjects aged 12 to 17 years and 5 subjects aged more than 65 years. Subjects were randomised into 1 of 4 parallel treatment arms, stratified by baseline attack rate, in a 3:2:2:2 ratio (placebo, lanadelumab 150 mg every 4 weeks (q4wks), lanadelumab 300 mg q4wks, or lanadelumab 300 mg every 2 weeks (q2wks) by subcutaneous injection) for the 26-week treatment period. The use of rescue medications for treatment of breakthrough HAE attacks was allowed for subjects receiving placebo or Takhzyro.
The median (range) age of the study population was 42 (12 to 73) years with 88 female subjects (70%). A history of laryngeal angioedema attacks was reported in 65% (81/125) of subjects and 56% (70/125) were on prior long-term prophylaxis (LTP). During the study run-in period, attack rates of ≥ 3 attacks/month were observed in 52% (65/125) of subjects overall.
All Takhzyro treatment arms produced clinically meaningful and statistically significant reductions in the mean HAE attack rate compared to placebo across all primary and secondary endpoints in the Intent-to-Treat population (ITT) (Table 3).
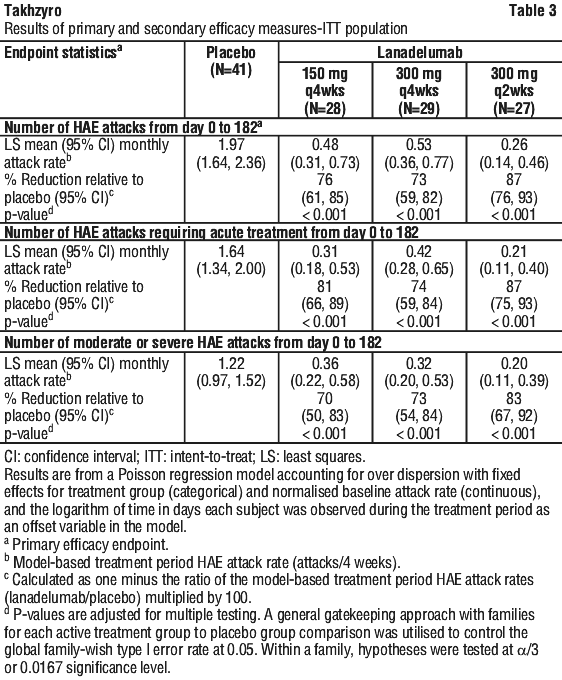
During the anticipated pharmacokinetic steady-state period (Day 70 to Day 182), percentage reductions in the mean monthly HAE attack rate for Takhzyro-treated subjects compared to placebo was 78% in the 150 mg q4wks arm, 81% in the 300 mg q4wks arm, and 91% in the 300 mg q2wks arm.
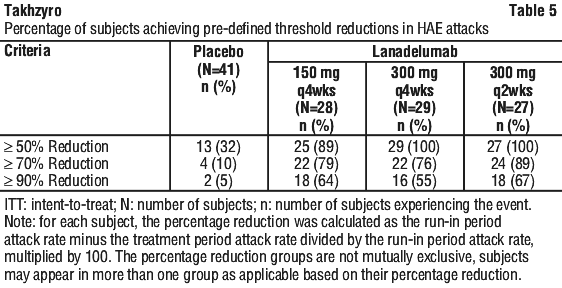
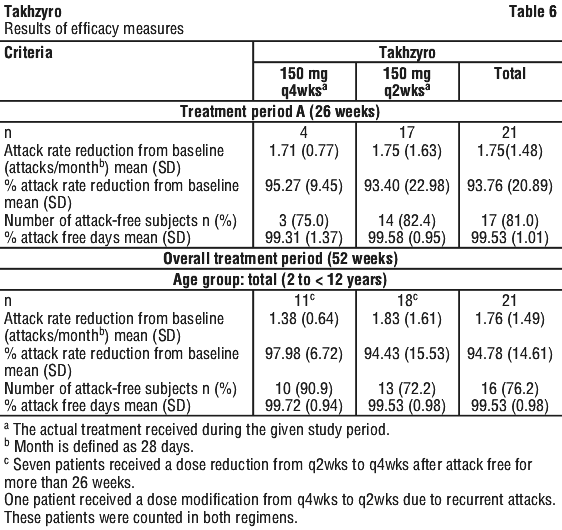
The percentage of subjects who were attack free is provided in Table 4.

HELP study extension. Long-term safety and efficacy, pharmacokinetics, and impact on health-related QoL of Takhzyro for prophylaxis to prevent HAE attacks were evaluated in an open-label uncontrolled HELP study extension.
A total of 212 adult and paediatric (12 to < 18 years) subjects received at least one dose of lanadelumab 300 mg q2wks in this study including 109 subjects who entered as rollover subjects from the HELP study. Rollover subjects, regardless of randomisation group in the HELP study, received a single dose of lanadelumab 300 mg at study entry and did not receive additional treatment until the occurrence of an HAE attack. After the first HAE attack, all subjects received open-label treatment with lanadelumab 300 mg q2wks. The study also included 103 new or non-rollover subjects (including 19 subjects from the Phase 1b study) who had a historical baseline attack rate of ≥ 1 attack per 12 weeks and a confirmed diagnosis of type I or II HAE. The non-rollover subjects received lanadelumab 300 mg q2wks at study entry. The median (range) age of the study population was 43 (12 to 76) years with 67% female subjects. The majority of subjects (173 of 212; 81.6%) who were treated in this study completed at least 30 months of treatment (either as a rollover or non-rollover subjects). The mean (SD) time in the HELP study extension was 29.6 (8.20) months. The majority of subjects self-administered lanadelumab over 10 to 60 seconds (60.6% of 8018 injections).
There was a sustained reduction in attack rates compared to baseline during the HELP study extension, with a similar response to Takhzyro observed in both rollover (92.4%) and non-rollover groups (82.0%) and an overall reduction rate of 87.4%. In the final analysis, mean attack rates for the rollover subjects decreased further from the end of Takhzyro treatment and ranged from 0.08 to 0.26 attacks per month. In addition, the mean (SD) percentage of attack-free days was 97.7 (6.0%) and the mean (SD) duration of the attack-free period was 415.0 (346.1) days. The proportion of patients with a maximum attack-free period of 6 months or more or 12 months or more was 81.8% and 68.9%, respectively.
In the HELP study extension, all Takhzyro treatment groups (rollover and non-rollover groups) observed an improvement in AE-QoL total and domain (functioning, fatigue/mood, fear/shame, and nutrition) scores.
The safety and efficacy of Takhzyro were evaluated in a subgroup of subjects (N=23) aged 12 to < 18 years. Results of the subgroup analysis by age were consistent with overall study results (see Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties; Section 5.2 Pharmacokinetic Properties).
SPRING study. The safety and efficacy of Takhzyro for prophylaxis to prevent HAE attacks in subjects aged 2 to < 12 years were evaluated in an open-label, multicentre, Phase 3 SPRING study. The study enrolled 21 paediatric subjects who had a baseline attack rate of ≥ 1 attacks per 3 months (12 weeks) and a confirmed diagnosis of type I or II HAE. The overall treatment period was 52 weeks, equally divided into Treatment Period A and B. In Treatment Period A, subjects aged 2 to < 6 years (n = 4) and 6 to < 12 years (n = 17) received lanadelumab 150 mg q4wks and 150 mg q2wks, respectively. In Treatment Period B, subjects in the 2 to < 6 years age group would continue the dosing regimen, while subjects in the 6 to < 12 years age group were permitted to switch to 150 mg q4wks if they were well-controlled (e.g. attack free) for 26 weeks with lanadelumab treatment. Seven subjects aged 6 to < 12 years switched to 150 mg q4wks during Treatment Period B, and one subject aged 2 to < 6 years switched to 150 mg q2wks during Treatment B after experiencing recurrent attacks.
The median (range) age of the study population was 8.7 (3.5, 10.9) years with 12 female subjects (57.1%) and the median HAE onset age (range) was 2.0 (0, 9) years. The Takhzyro dose regimen in both age groups produced marked clinically meaningful reduction in mean HAE attack rate compared to baseline and substantial percentage of attack-free subjects (Table 6). Results of the analysis indicate that Takhzyro was safe and effective in paediatric patients (2 to < 12 years) as shown in the Table 6.
5.2 Pharmacokinetic Properties
Pharmacokinetics of lanadelumab showed linear dose-exposure response with doses up to 400 mg and reproducible exposure following subcutaneous administration up to 12 months. The pharmacokinetic (PK) properties and exposure (steady-state) of lanadelumab in adult and paediatric (12 to < 18 years) HAE patients, following subcutaneous administration of 150 mg q4wks, 300 mg q4wks and 300 mg q2wks, are provided in Table 7. The anticipated population time to reach steady-state concentration was approximately 70 days.
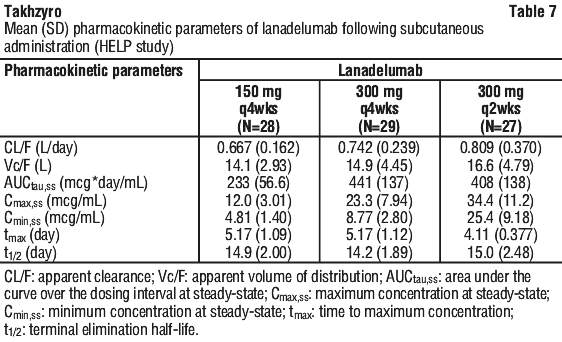
Population PK analyses were performed, using data from rich sampling in two Phase 1 studies and sparse sampling in two Phase 3 (HELP and HELP extension) studies.
The population PK analyses found that patient body weight was an important covariate describing the variability of clearance and volume of distribution, resulting in higher exposure (AUC and Cmax) in lighter patients. After correcting for body weight, no influence of gender was apparent on the clearance or volume of distribution of Takhzyro. No dose adjustment is required.
The population PK analyses for the effects of age, including 22 adolescents [aged 12 to 18], 226 adults [aged > 18 and < 65 years] and 9 elderly [aged > 65 years]. The mean lanadelumab exposure under the same dosing regimen was found to be approximately 37% higher in adolescent patients compared to adult patients, due to lower body weight in adolescent patients.
Although body weight was identified as an important covariate describing the variability of clearance, a 300 mg q2wks dose regimen provided sufficient exposure for the indication.
The population PK analysis of the effect of renal impairment that included estimated GFR: 60 to 89 mL/min/1.73 m2 [mild, N=98], 30 to 59 mL/min/1.73 m2 [moderate, N=9] and < 30 mL/min/1.73 m2 [severe, N=0] found no effect on the clearance or volume of distribution. No dose adjustment is required for mild or moderate renal impairment. No dose recommendation can be made for severe renal impairment.
Paediatric patients. Following subcutaneous administration of 150 mg every 4 weeks (2 to < 6 years) and 150 mg every 2 weeks (6 to < 12 years), the overall exposure (i.e. Cavg,ss) to lanadelumab was similar compared with adult and paediatric (12 to < 18 years) patients who received Takhzyro 300 mg every 2 weeks (ratio to adults ranged from 0.8 to 1.11).
Concomitant medications. There have been no dedicated investigations of PK interactions (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Based on the population PK analysis of the Phase 3 data, the use of analgesic, antibacterial, antihistamine, anti-inflammatory and anti-rheumatic medications had no effect on clearance and volume of distribution of Takhzyro.
For breakthrough HAE attacks, use of rescue medications such as icatibant or plasma-derived C1 esterase inhibitor had no effects on clearance and volume of distribution of Takhzyro.
5.3 Preclinical Safety Data
Genotoxicity. Given that lanadelumab is a monoclonal antibody and therefore is not expected to interact directly with DNA or other chromosomal material, no genotoxicity evaluation has been conducted.
Carcinogenicity. Carcinogenicity has not been evaluated in animals.
6 Pharmaceutical Particulars
6.1 List of Excipients
Dibasic sodium phosphate dihydrate, citric acid monohydrate, histidine, sodium chloride, polysorbate 80, water for injections.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the ARTG. The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store in a refrigerator at 2°C to 8°C. Do not freeze.
Takhzyro pre-filled syringe removed from refrigeration should be stored below 25°C and used within 14 days. Do not return pre-filled syringes to refrigerated storage after storage at room temperature.
Takhzyro vial removed from refrigeration should be stored unopened, below 25°C, and used within 14 days or returned to refrigeration unopened until use. Total period stored out of refrigeration, below 25°C, should not exceed 14 days.
Keep the pre-filled syringe or vial in the original carton in order to protect it from light.
6.5 Nature and Contents of Container
Single-dose pre-filled syringe. Takhzyro is a ready-to-use solution supplied in a glass pre-filled syringe with bromobutyl rubber stopper, 27 gauge ½ inches staked needle with rigid needle cap. Pack size of 1 syringe available as: 150 mg/1 mL solution, 300 mg/2 mL solution.
Single-dose vial. Takhzyro is a ready-to-use solution supplied in a glass vial with chlorobutyl rubber stopper, aluminium crimp seal and violet flip-off cap. Pack size of 1 vial of 300 mg/2 mL.
Each pack also contains the following administration ancillaries: one empty 3 mL syringe, one 18 gauge vial access needle, and one 27 gauge ½ inches needle (for subcutaneous injection).
Note. Not all presentations may be marketed.
6.6 Special Precautions for Disposal
All needles and syringes should be disposed of in a sharps container.
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Takhzyro is a sterile, preservative-free, colourless to slightly yellow solution, appearing either clear or slightly opalescent with a pH of approximately 6.0 and an osmolality of approximately 300 mOsm/kg.
Chemical structure. Based on the amino acid sequence, the molecular weight of the non-glycosylated lanadelumab is 146 kDa. The calculated molecular mass of the fully reduced light chain is 23 kDa. The calculated molecular mass of the fully reduced and non-glycosylated heavy chain is 49 kDa.
CAS number. 1426055-14-2.
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine.
Date of Revision
25 July 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.