Timoptol-XE
Brand Information
| Brand name | Timoptol-XE |
| Active ingredient | Timolol |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Timoptol-XE.
Summary CMI
TIMOPTOL-XE®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using TIMOPTOL-XE?
TIMOPTOL-XE contains the active ingredient timolol maleate. TIMOPTOL-XE is used to lower raised pressure in the eye and to treat glaucoma.
For more information, see Section 1. Why am I using TIMOPTOL-XE? in the full CMI.
2. What should I know before I use TIMOPTOL-XE?
Do not use if you have ever had an allergic reaction to TIMOPTOL-XE or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
Tell your doctor if you have a heart condition or any breathing conditions including asthma, emphysema or chronic obstructive pulmonary disease (COPD).
For more information, see Section 2. What should I know before I use TIMOPTOL-XE? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with TIMOPTOL-XE and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use TIMOPTOL-XE?
- Your doctor will tell you how many drops you need to use each day.
- Use TIMOPTOL-XE every day, at about the same time each day, unless your doctor tells you otherwise.
More instructions can be found in Section 4. How do I use TIMOPTOL-XE? in the full CMI.
5. What should I know while using TIMOPTOL-XE?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using TIMOPTOL-XE? in the full CMI.
6. Are there any side effects?
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using TIMOPTOL-XE.
Common side effects include: temporary blurred or double vision, irritated or watery eyes, headache, tiredness, change in mood, upset stomach, dry mouth, cold hands or feet, tingling fingers or toes.
Serious side effects are rare, and may include: fast or irregular heart beat, dizziness, fainting, shortness of breath.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
TIMOPTOL-XE® (tim-opt-ol ex-ee)
Active ingredient(s): timolol maleate
Consumer Medicine Information (CMI)
This leaflet provides important information about using TIMOPTOL-XE. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using TIMOPTOL-XE.
Where to find information in this leaflet:
1. Why am I using TIMOPTOL-XE?
2. What should I know before I use TIMOPTOL-XE?
3. What if I am taking other medicines?
4. How do I use TIMOPTOL-XE?
5. What should I know while using TIMOPTOL-XE?
6. Are there any side effects?
7. Product details
1. Why am I using TIMOPTOL-XE?
TIMOPTOL-XE contains the active ingredient timolol maleate. TIMOPTOL-XE belongs to a family of medicines called beta-blockers.
TIMOPTOL-XE is used, either alone or in combination with other eye drops or medicines, to lower raised pressure within your eye(s) and to treat glaucoma.
Glaucoma is a condition in which the pressure of fluid in the eye may be high. However, some people with glaucoma may have normal eye pressure. Also, some people with raised eye pressure may not have glaucoma.
This raised pressure may damage the back of the eye resulting in gradual loss of sight. Damage can progress so slowly that the person is not aware of this gradual loss of sight. Sometimes even normal eye pressure is associated with damage to the back of the eye. There are usually no symptoms of glaucoma. The only way of knowing that you have glaucoma is to have your eye pressure, optic nerve and visual field checked by an eye specialist or optometrist.
If glaucoma is not treated it can lead to serious problems. You may have no symptoms but eventually glaucoma can lead to total blindness. In fact, untreated glaucoma is one of the most common causes of blindness.
Although TIMOPTOL-XE helps control your glaucoma it does not cure it.
For more information about glaucoma, contact Glaucoma Australia Inc., PO Box 420, Crows Nest 1585, telephone 1800 500 880, or at www.glaucoma.com.au
2. What should I know before I use TIMOPTOL-XE?
Warnings
Do not use TIMOPTOL-XE if:
- you are allergic to timolol maleate, or any of the ingredients listed at the end of this leaflet
- Always check the ingredients to make sure you can use this medicine
- you have, or have had in the past certain serious breathing problems such as asthma, a history of asthma, chronic obstructive lung disease (COPD), emphysema, or other breathing problems
- you have certain heart conditions, such as a slow heart rate, an irregular heart beat, or heart failure
- the seal around the cap is broken
- the bottle shows signs of tampering
- the expiry date on the pack has passed. If you use this medicine after the expiry date has passed, it may not work
Check with your doctor if you:
- have any other medical conditions, especially the following:
- heart problems (such as coronary heart disease, heart failure or low blood pressure)
- heart rate disturbances (such as slow or irregular heart beats)
- poor blood circulation problems (such as Raynaud's syndrome)
- lung or breathing problems (such as asthma or chronic obstructive lung disease)
- diabetes or other blood sugar problems
- thyroid disease
- are already using another beta-blocker eye drop. It is not recommended to use two beta-blockers at the same time.
- have a history of allergic problems including eczema, hives or hay fever
- you have had an allergy to any other medicines or any other substances, including foods, preservatives or dyes.
- take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. Your doctor will discuss the possible risks and benefits of using TIMOPTOL-XE during pregnancy and a decision can be made if you should or should not use it.
Do not use TIMOPTOL-XE if you are breast-feeding or intend to breastfeed. Your baby may absorb this medicine from breast milk and therefore there is a possibility of harm to the baby.
Using TIMOPTOL-XE with contact lenses
Do not put the eye drops into your eye(s) while you are wearing soft contact lenses. The preservative in TIMOPTOL-XE(benzalkonium chloride) may be deposited in soft contact lenses. Wait at least 15 minutes after you have used TIMOPTOAL before you put your soft contact lenses back into your eyes.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and TIMOPTOL-XE may interfere with each other. These include:
- medicines for high blood pressure or heart conditions, including a group of medicines called betablockers
- digoxin, a medicine used to treat heart failure
- quinidine, a medicine used to treat irregular heart beats
- medicines to treat depression
- medicines to treat diabetes
These medicines may be affected by TIMOPTOL-XE, or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect TIMOPTOL-XE.
4. How do I use TIMOPTOL-XE?
How much to use
- Follow the instructions provided by your doctor and use TIMOPTOL-XE until your doctor tells you to stop.
- The usual dose for adults is one drop of TIMOPTOL-XE a day, in either one or both eyes.
When to use TIMOPTOL-XE
- TIMOPTOL-XE should be used once a day, every day at the same time each day unless your doctor tells you otherwise. Using your eye drops at the same time each day will have the best effect on your eye pressure. It will also help you remember when to use the eye drops.
How to use TIMOPTOL-XE drops
- Please refer to the instruction leaflet inside the TIMOPTOL-XE box for directions on how to use your eye drops.
- You may find it easier to put drops in your eye(s) while you are sitting or lying down.
- If using TIMOPTOL-XE with another eye drop, use the other eye drop at least 10 minutes before using TIMOPTOL-XE.
- If you are wearing contact lenses remove them before putting the drops in your eye(s), and wait at least 15 minutes after you have used the drops before putting your contact lenses back in.
- Be careful not to touch the dropper against eye, eyelid or anything else to avoid contaminating the eye drops. Contaminated eye drops may give you an eye infection.
- You may experience blurred vision in the eye(s) for 30 seconds to 5 minutes immediately after using TIMOPTOL-XE drops.
If you forget to use TIMOPTOL-XE
If it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
Otherwise, use the drops as soon as you remember and then go back to using them as you would normally.
If you are not sure whether to skip a dose, talk to your doctor or pharmacist.
Do not take a double dose to make up for the dose you missed.
If you have trouble remembering to use your eye drops, ask your pharmacist for some hints.
If you use too much TIMOPTOL-XE
If you think that you have used too much TIMOPTOL-XE, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre (by calling 13 11 26), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using TIMOPTOL-XE?
Things you should do
Have your eye pressure checked when your eye specialist says, to make sure TIMOPTOL-XE is working.
Tell your doctor before you have an operation that you are using TIMOPTOL-XE. TIMOPTOL-XE may change the effects of some medicines during anaesthesia.
Remind any doctor, dentist or pharmacist you visit that you are using TIMOPTOL-XE.
If you are about to be started on any new medicine, tell your doctor and pharmacist that you are using TIMOPTOL-XE.
Call your doctor straight away if you:
- Develop an eye infection, receive an eye injury or have eye surgery. Your doctor may tell you to use a new bottle of TIMOPTOL-XE because of possible contamination of the old one, or may advise you to stop y our treatment with TIMOPTOL-XE.
Things you should not do
- Do not stop using TIMOPTOL-XE without first talking with your doctor. If you stop using your eye drops, your eye pressures may rise again and damage to your eye may occur.
- Do not give TIMOPTOL-XE to anyone else, even if they have the same condition as you.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how TIMOPTOL-XE affects you.
TIMOPTOL-XE generally does not cause any problems with your ability to drive a car or operate machinery. However, TIMOPTOL-XE may cause temporary blurred vision or dizziness in some people. Make sure you know how you react to TIMOPTOL-XE and that your vision is clear before driving a car or operating machinery.
Looking after your medicine
Store TIMOPTOL-XE drops below 30°C.
Keep the drops away from direct light.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
- In the pockets of your clothes
Keep it where young children cannot reach it.
Do not leave the lid off the bottle for any length of time to avoid contaminating the eye drops.
When to discard your medicine
Write the date on the bottle when you open the eye drops and throw out any remaining solution after 4 weeks.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Problems with your eyes such as:
| Speak to your doctor if you have any of these less serious side effects and they worry you. These are usually mild side effects of TIMOPTOL-XE |
Serious side effects
| Serious side effects | What to do |
Heart related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. Serious side effects are rare. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What TIMOPTOL-XE contains
| Active ingredient (main ingredient) | timolol maleate |
| Other ingredients (inactive ingredients) | gellan gum mannitol tromethamine benzododecinium bromide (as preservative) |
Do not use TIMOPTOL-XE eye drops if you are allergic to any of these ingredients.
What TIMOPTOL-XE looks like
TIMOPTOL-XE is a clear colourless solution in a plastic 5 mL bottle. It comes in 2 strengths : 0.25% and 0.5%.
TIMOPTOL-XE 0.25% - AUST R 50304
TIMOPTOL-XE 0.5% - AUST R 50306
Who distributes TIMOPTOL-XE
Mundipharma Pty Limited
10 Carrington Street
Sydney NSW 2000
This leaflet was prepared in August 2025
® TIMOPTOL-XE is a registered trade mark.
TIMOPTOL-XE-CMI v2 (IPC-MK0950-GFS-022014)
Brand Information
| Brand name | Timoptol-XE |
| Active ingredient | Timolol |
| Schedule | S4 |
MIMS Revision Date: 01 August 2022
1 Name of Medicine
Timolol maleate.
2 Qualitative and Quantitative Composition
Each mL of Timoptol-XE 0.25% contains 2.5 mg of timolol (3.4 mg of timolol maleate). Each mL of Timoptol-XE 0.5% contains 5.0 mg of timolol (6.8 mg of timolol maleate).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Timoptol-XE is a sterile, colourless to nearly colourless, slightly opalescent, slightly viscous, aqueous ophthalmic (eye drop) solution containing gellan gum.
Gellan gum is a highly purified anionic heteropolysaccharide. Aqueous solutions of gellan gum form a clear transparent gel at low polymer concentrations in the presence of cations. When Timoptol-XE contacts the precorneal tear film, it becomes a gel. The concentration of sodium cation in tears is ideally suited to cause gelation of the material when topically instilled in the conjunctival sac.
4 Clinical Particulars
4.1 Therapeutic Indications
Timoptol-XE is indicated for the reduction of elevated intraocular pressure in patients with: ocular hypertension; chronic open-angle glaucoma; aphakia and glaucoma.
4.2 Dose and Method of Administration
The usual starting dose is one drop of 0.25% Timoptol-XE in the affected eye(s) once a day. If the clinical response is not adequate, the dosage may be changed to one drop of 0.5% Timoptol-XE in the affected eye(s) once a day. Invert the closed container and shake once energetically before instillation. Depress the finger-press area on the side of the bottle to dispense one drop.
Dosages higher than one drop of 0.5% Timoptol-XE once a day have not been studied.
If needed, concomitant therapy with other agent(s) for lowering intraocular pressure may be given with Timoptol-XE. Other topically applied medications should be administered no less than 10 minutes before Timoptol-XE. The use of two topical beta-adrenergic blocking agents is not recommended (see Section 4.4 Special Warnings and Precautions for Use).
Systemic absorption of drugs from ophthalmic solutions may be minimised by pressure on the tear-duct immediately after application. This may result in an increase in local activity.
How to transfer patients from other therapy. When a patient is transferred from Timoptol to Timoptol-XE, Timoptol should be discontinued after proper dosing on one day, and treatment with the same concentration of Timoptol-XE started on the following day.
When a patient is transferred from another topical ophthalmic beta-adrenergic blocking agent, that agent should be discontinued after proper dosing on one day and treatment with Timoptol-XE started on the following day with 1 drop of 0.25% Timoptol-XE in the affected eye once a day. The dose may be increased to one drop of 0.5% Timoptol-XE once a day if the clinical response is not adequate.
When a patient is transferred from a single antiglaucoma agent, other than a topical ophthalmic beta-adrenergic blocking agent, continue the agent and add one drop of 0.25% Timoptol-XE to each affected eye once a day. On the following day, discontinue the previously used antiglaucoma agent and continue Timoptol-XE. If a greater response is required, substitute one drop of 0.5% Timoptol-XE for the 0.25% dosage.
4.3 Contraindications
Timoptol-XE is contraindicated in patients with:
Reactive airway disease, bronchial asthma or with a history of bronchial asthma, or severe chronic obstructive pulmonary disease.
Sinus bradycardia; sino-atrial block; second or third degree atrioventricular block; overt cardiac failure; cardiogenic shock.
Hypersensitivity to any component of this product.
4.4 Special Warnings and Precautions for Use
As with other topically applied ophthalmic drugs, this drug may be absorbed systemically. The same adverse reactions found with systemic administration of beta-adrenergic blocking agents may occur with topical administration (see Section 4.8 Adverse Effects (Undesirable Effects), Potential adverse effects).
Cardio-respiratory reactions. Sympathetic stimulation may be essential for support of the circulation in individuals with diminished myocardial contractility, and its inhibition by beta-adrenergic receptor blockade may precipitate more severe failure.
In patients without a history of cardiac failure, continued depression of the myocardium with beta-blocking agents over a period of time can, in some cases, lead to cardiac failure. At the first sign or symptom of cardiac failure, Timoptol-XE should be discontinued.
Cardiac failure should be adequately controlled before beginning therapy with Timoptol-XE. Patients with a history of cardiovascular disease, including cardiac failure should be watched for signs of deterioration of these diseases, and pulse rates should be monitored.
Due to its negative effect on conduction time, beta-blockers should be given with caution to patients with first degree heart block.
Because of potential effects of beta-adrenergic blocking agents relative to blood pressure and pulse, these agents should be used with caution in patients with cerebrovascular insufficiency. If signs or symptoms suggesting reduced cerebral blood flow develop following initiation of therapy with Timoptol-XE, alternative therapy should be considered.
Respiratory complications, including exacerbation of asthma and death due to bronchospasm in patients with asthma, and cardiac complications, including rarely death in association with cardiac failure, have been reported following administration of beta-adrenergic blocking agents. These are potential complications of therapy with Timoptol-XE.
In patients with mild/ moderate chronic obstructive pulmonary disease (COPD), Timoptol-XE should be used with caution, and only if the potential benefit outweighs the potential risk.
Muscle weakness. Beta-adrenergic blockage has been reported to increase muscle weakness consistent with certain myasthenic symptoms (e.g. diplopia, ptosis, and generalised weakness). Timolol has been reported rarely to increase muscle weakness in some patients with myasthenic symptoms.
Vascular disorders. Patients with severe peripheral circulatory disturbance/ disorders (e.g. severe forms of Raynaud's disease or Raynaud's syndrome) should be treated with caution.
Masking of hypoglycemic symptoms in patients with diabetes mellitus. Beta-adrenergic blocking agents should be administered with caution in patients subject to spontaneous hypoglycemia or to diabetic patients (especially those with labile diabetes) who are receiving insulin or oral hypoglycemic agents. Beta-adrenergic blocking agents may mask the signs and symptoms of acute hypoglycemia.
Masking of thyrotoxicosis. Beta-adrenergic blocking agents may mask certain clinical signs of hyperthyroidism (e.g. tachycardia). Patients suspected of developing thyrotoxicosis should be managed carefully to avoid abrupt withdrawal of beta-adrenergic blocking agents which might precipitate a thyroid storm.
Surgical anesthesia. The necessity or desirability of withdrawal of beta-adrenergic blocking agents prior to major surgery is controversial. If necessary during surgery, the effects of beta-adrenergic blocking agents may be reversed by sufficient doses of adrenergic agonists (see Section 4.9 Overdose).
Risk from anaphylactic reaction. While taking beta-blockers, patients with a history of atopy or a history of severe anaphylactic reaction to a variety of allergens may be more reactive to repeated challenge with such allergens, either accidental, diagnostic, or therapeutic. Such patients may be unresponsive to the usual doses of adrenaline (epinephrine) used to treat anaphylactic reactions.
Other. Patients who are already receiving a beta-adrenergic blocking agent orally and who are given Timoptol-XE should be observed for a potential additive effect either on the intraocular pressure or on the known systemic effects of beta-blockade. The use of two topical beta-adrenergic blocking agents is not recommended.
In patients with angle-closure glaucoma, the immediate objective of treatment is to reopen the angle. This requires constricting the pupil with a miotic. Timolol maleate has little or no effect on the pupil. Should Timoptol-XE be used to reduce elevated intraocular pressure in angle-closure glaucoma, it should be used with a miotic and not alone.
Choroidal detachment has been reported with the administration of aqueous suppressant therapy (e.g. timolol, acetazolamide) after filtration procedures.
Timoptol-XE has not been studied in patients wearing contact lenses. In a clinical study, the time required to eliminate 50% of the gellan solution from the eye was up to 30 minutes.
Hypersensitivity to any of its ingredients, including the preservative, may develop. Contact irritancy due to the preservative may occur.
Patients should be advised that if they develop an intercurrent ocular condition (e.g. trauma, ocular surgery or infection) or any ocular reactions, particularly conjunctivitis and lid reactions they should immediately seek their physician's advice concerning the continued use of the product.
Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye or surrounding structures. Ocular solutions, if handled improperly, can become contaminated by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions.
There have been reports of bacterial keratitis associated with the use of multiple-dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or disruption of the ocular epithelial surface.
Use in the elderly. No data available.
Paediatric use. Safety and efficacy in children have not been established by adequate and well controlled studies.
Effects on laboratory tests. Clinically important changes in standard laboratory parameters were rarely associated with the administration of systemic timolol maleate. Slight increases in blood urea nitrogen, serum potassium, serum uric acid and triglycerides and slight decreases in haemoglobin, haematocrit, and HDL-cholesterol occurred, but were not progressive or associated with clinical manifestations.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Close observation of the patient is recommended when a beta-blocker is administered to patients receiving catecholamine-depleting drugs such as reserpine or another beta-adrenergic blocking agent, because of possible additive effects and the production of hypotension and/or marked bradycardia, which may produce vertigo, syncope, or postural hypotension.
The potential exists for hypotension, atrioventricular (AV) conduction disturbances and left ventricular failure to occur in patients receiving a beta-blocking agent when an oral calcium channel blocker is added to the treatment regimen. The nature of any cardiovascular adverse effect tends to depend on the type of calcium channel blocker used. Dihydropyridine derivatives, such as nifedipine, may lead to hypotension, whereas verapamil or diltiazem have a greater propensity to lead to AV conduction disturbances or left ventricular failure when used with a beta-blocker.
The potential exists for additive effects and production of hypotension and/or marked bradycardia when Timoptol-XE is administered together with an oral calcium-channel blocker, catecholamine-depleting drugs, antiarrhythmics, parasympathomimetics or beta-adrenergic blocking agents.
The concomitant use of beta-adrenergic blocking agents and digitalis with either diltiazem or verapamil may have additive effects in prolonging AV conduction time.
Oral calcium-channel antagonists may be used in combination with beta-adrenergic blocking agents when heart function is normal, but should be avoided in patients with impaired cardiac function.
Intravenous calcium-channel blockers should be used with caution in patients receiving beta-adrenergic blocking agents.
Potentiated systemic beta-blockade (e.g. decreased heart rate, depression) has been reported during combined treatment with CYP2D6 inhibitors (e.g. quinidine and SSRIs) and timolol.
Although timolol maleate used alone has little or no effect on pupil size, mydriasis resulting from concomitant therapy with adrenaline (epinephrine) has been reported occasionally. The potential for mydriasis exists from concomitant therapy with Timoptol-XE and adrenaline (epinephrine).
Beta-adrenergic blocking agents may exacerbate the rebound hypertension which can follow the withdrawal of clonidine. Caution should be exercised in patients using these drugs concomitantly. There have been no reports of exacerbation of rebound hypertension with ophthalmic timolol maleate.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Reproduction and fertility studies in rats demonstrated no adverse effect on male or female fertility at doses up to 100 mg/kg/day.
Use in pregnancy. (Category C)
Timoptol-XE has not been studied in human pregnancy. Beta-adrenergic blocking agents may cause pharmacological effects such as bradycardia in the fetus and newborn infant. During the latter stages of pregnancy and birth, these drugs should be given only after weighing the needs of the mother against the risk to the fetus.
Use in lactation. Timolol is detectable in human milk. Because of the potential for serious adverse reactions from Timoptol-XE in infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Timoptol-XE is usually well tolerated. The most frequent drug-related complaint in the original clinical trials for Timoptol-XE was transient blurred vision (6.0%), lasting from 30 seconds to 5 minutes, following instillation.
In clinical studies, the drug related adverse experiences observed for Timoptol-XE were similar to those of Timoptol, with the exception of a higher incidence of transient blurred vision for Timoptol-XE (6%) compared to Timoptol (1.6%). Drug-related adverse experiences caused 3.4% of patients treated with Timoptol-XE to discontinue compared to 1.4% of patients treated with Timoptol. Less than 1% of patients discontinued Timoptol-XE due to transient blurred vision.
The following possibly, probably, or definitely drug-related adverse reactions occurred with frequency of at least 1% in active treatment controlled clinical trials:
Ocular. Common (≥ 1% and < 10%). Blurred vision, burning and stinging, conjunctival injection, discharge, foreign body sensation, itching.
The following additional adverse reactions have been reported with ocular administration of this or other timolol maleate formulations, either in clinical trials or since the drug has been marketed (also see Potential adverse effects).
Special senses. Signs and symptoms of ocular irritation, including conjunctivitis, blepharitis, keratitis, decreased corneal sensitivity and dry eyes. Visual disturbances, including refractive changes (due to withdrawal of miotic therapy in some cases), diplopia, ptosis, choroidal detachment following filtration surgery (see Section 4.4 Special Warnings and Precautions for Use), tinnitus.
Cardiovascular. Bradycardia, arrhythmia, hypotension, syncope, heart block, cerebrovascular accident, cerebral ischaemia, congestive heart failure, palpitation, cardiac arrest, oedema, claudication, Raynaud's phenomenon, cold hands and feet.
Respiratory. Bronchospasm (predominantly in patients with pre-existing bronchospastic disease), exacerbation of asthma, respiratory failure, dyspnoea, cough.
Body as a whole. Headache, asthenia, fatigue, chest pain.
Integumentary. Alopecia, psoriasiform rash or exacerbation of psoriasis.
Hypersensitivity. Signs and symptoms of allergic reactions including anaphylaxis, angioedema, urticaria, localised and generalised rash.
Nervous system/ psychiatric. Dizziness, depression, insomnia, nightmares, memory loss, paraesthesia.
Neuromuscular. Increase in signs and symptoms of myasthenia gravis.
Digestive. Nausea, diarrhoea, dyspepsia, dry mouth, abdominal pain.
Urogenital. Decreased libido, Peyronie's disease, sexual dysfunction.
Immunologic. Systemic lupus erythematosus.
Musculoskeletal. Myalgia.
Potential adverse effects. The following side effects have been reported in clinical experience with systemic timolol maleate and may be considered potential side effects of ophthalmic timolol maleate.
Body as a whole. Extremity pain, decreased exercise tolerance.
Cardiovascular. AV block (2nd or 3rd degree), sinoatrial block, pulmonary oedema, worsening of arterial insufficiency, worsening of angina pectoris, vasodilation.
Digestive. Vomiting.
Endocrine. Hyperglycaemia, hypoglycaemia.
Integumentary. Pruritus, sweating, exfoliative dermatitis.
Musculoskeletal. Arthralgia.
Nervous system. Vertigo, local weakness.
Psychiatric. Nervousness, diminished concentration, hallucinations, increased dreaming, somnolence.
Haematologic. Nonthrombocytopenic purpura.
Respiratory. Rales.
Urogenital. Impotence, micturition difficulties.
Adverse-effects, causal relationship unknown. The following adverse effects have been reported but a causal relationship to therapy with timolol maleate has not been established: aphakic cystoid macular oedema, nasal congestion, anorexia, CNS effects (e.g. behavioural changes including confusion, anxiety, disorientation and other psychic disturbances), hypertension, retroperitoneal fibrosis and pseudopemphigoid.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There have been reports of inadvertent overdosage with Timoptol resulting in systemic effects similar to those seen with systemic beta-adrenergic blocking agents such as dizziness, headache, shortness of breath, bradycardia, bronchospasm, and cardiac arrest (also see Section 4.8 Adverse Effects (Undesirable Effects)).
The following specific therapeutic measures should be considered:
(1) Symptomatic bradycardia. Administer atropine sulphate intravenously in a dosage of 0.25 to 2 mg to induce vagal blockade. If bradycardia persists, intravenous isoprenaline hydrochloride should be administered cautiously. In refractory cases the use of a transvenous cardiac pacemaker may be considered.
(2) Heart block (second or third degree). Administer isoprenaline hydrochloride or insert a transvenous cardiac pacemaker.
(3) Hypotension. Use sympathomimetic pressor drug therapy, such as dopamine, dobutamine or noradrenaline. In refractory cases the use of glucagon hydrochloride has been reported to be useful.
(4) Acute cardiac failure. Conventional therapy with digitalis, diuretics and oxygen should be instituted immediately. In refractory cases the use of intravenous aminophylline is suggested. This may be followed if necessary by glucagon hydrochloride, which has been reported to be useful.
(5) Bronchospasm. Administer isoprenaline hydrochloride. Additional therapy with aminophylline may be considered.
Timolol does not dialyse readily.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The precise mechanism of action of timolol maleate in lowering intraocular pressure is not clearly established. A fluorescein study and tonography studies indicate that the predominant action may be related to reduced aqueous formation. However, in some studies a slight increase in outflow facility was also observed.
Unlike miotics, timolol maleate reduces intraocular pressure with little or no effect on accommodation or pupil size. Thus, changes in visual acuity due to increased accommodation are uncommon, and the dim or blurred vision and night blindness produced by miotics are not evident. In addition, in patients with cataracts the inability to see around lenticular opacities when the pupil is constricted by miotics is avoided. When changing patients from miotics to Timoptol-XE, refraction may be necessary after the effects of the miotic have passed.
Beta-adrenergic receptor blockade reduces cardiac output in both healthy subjects and patients with heart disease. In patients with severe impairment of myocardial function, beta-adrenergic receptor blockade may inhibit the stimulatory effect of the sympathetic nervous system necessary to maintain adequate cardiac function.
Beta-adrenergic receptor blockade in the bronchi and bronchioles results in increased airway resistance from unopposed parasympathetic activity. Such an effect in patients with asthma or other bronchospastic conditions is potentially dangerous.
Clinical trials. Clinical studies have shown that the intraocular pressure lowering effect of Timoptol-XE administered once a day is equivalent to Timoptol administered twice a day. The vehicle of Timoptol-XE, gellan gum, increases the contact time of the drug with the eye.
Maximum reduction of intraocular pressure occurs in two to four hours with Timoptol-XE. Significant lowering of intraocular pressure has been maintained for 24 hours with both 0.25% and 0.5% Timoptol-XE.
Timoptol-XE has a safety profile similar to that of Timoptol, and both are generally well tolerated. In the three studies comparing Timoptol-XE 0.5% once a day to Timoptol 0.5% twice a day, Timoptol-XE did not reduce mean heart rate as much as Timoptol (see Section 4.4 Special Warnings and Precautions for Use). At trough (24 hours post-dose Timoptol-XE, 12 hours post-dose Timoptol), the mean reduction was 0.8 beats/minute for Timoptol-XE and 3.6 beats/minute for Timoptol; whereas at two hours post-dose, the mean reduction in heart rate was comparable (3.8 beats/minute for Timoptol-XE and 5 beats/minute for Timoptol). There was a higher incidence of transient blurred vision following instillation in patients administered Timoptol-XE.
In clinical studies timolol maleate was generally effective in more patients and produced fewer and less severe side effects than either pilocarpine or adrenaline (epinephrine).
As with other antiglaucoma drugs, diminished responsiveness to timolol maleate after prolonged therapy has been reported in some patients. However, in clinical studies of Timoptol in which 164 patients were followed for at least 3 years, no significant difference in mean intraocular pressure was observed after initial stabilisation. This indicates that the intraocular pressure-lowering effect of timolol maleate is well maintained.
5.2 Pharmacokinetic Properties
Following 8 days of once daily application of 0.5% Timoptol-XE in the eye, peak plasma concentrations of timolol averaged < 0.3 nanogram/mL within 4 hours after the last dose. The maximum plasma levels measured approached 0.5 nanogram/mL at 1 to 2 hours following the last dose.
Timolol maleate is a nonselective beta-adrenergic receptor blocking agent that does not have significant intrinsic sympathomimetic, direct myocardial depressant, or local anaesthetic (membrane-stabilising) activity. Timolol maleate combines reversibly with a part of the cell membrane, the beta-adrenergic receptor, and thus inhibits the usual biologic response that would occur with stimulation of that receptor. This specific competitive antagonism blocks stimulation of the beta-adrenergic receptors by catecholamines having beta-adrenergic stimulating (agonist) activity, whether these originate from an endogenous or exogenous source. Reversal of this blockade can be accomplished by increasing the concentration of the agonist, which will restore the usual biologic response.
Timoptol-XE reduces elevated and normal intraocular pressure whether or not associated with glaucoma. Elevated intraocular pressure is a major risk factor in the pathogenesis of glaucomatous visual field loss. The higher the intraocular pressure, the greater the likelihood of glaucomatous visual field loss and optic nerve damage.
5.3 Preclinical Safety Data
Genotoxicity. In vitro and in vivo studies (Ames test, neoplastic cell transformation assay, cytogenetic assay and micronucleus test in mice) showed no genotoxicity of timolol.
Carcinogenicity. In a 2-year study of timolol maleate administered orally to rats, there was a statistically significant increase in the incidence of adrenal pheochromocytomas in male rats administered 300 mg/kg/day. Similar differences were not observed in rats administered oral doses of 100 mg/kg/day.
In a lifetime oral study in mice, there were statistically significant increases in the incidence of benign and malignant pulmonary tumours, benign uterine polyps and mammary adenocarcinomas in female mice at 500 mg/kg/day, but not at 50 mg/kg/day. In a subsequent study in female mice, in which post-mortem examinations were limited to the uterus and the lungs, a statistically significant increase in the incidence of pulmonary tumours was again observed at 500 mg/kg/day.
The increased occurrence of mammary adenocarcinomas was associated with elevations in serum prolactin which occurred in female mice administered oral timolol at 500 mg/kg, but not at doses of 50 mg/kg/day. An increased incidence of mammary adenocarcinomas in rodents has been associated with administration of several other therapeutic agents that elevate serum prolactin, but no correlation between serum prolactin levels and mammary tumours has been established in humans. Furthermore, in adult human female subjects who received oral dosages of up to 60 mg of timolol maleate (the maximum recommended human oral dosage), there were no clinically meaningful changes in serum prolactin.
6 Pharmaceutical Particulars
6.1 List of Excipients
Timoptol-XE also contains the following inactive ingredients: gellan gum, trometamol, mannitol and water for injections. Benzododecinium bromide 0.012% is added as the preservative.
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at or below 30°C. Avoid freezing. Protect from light. Discard within 28 days of opening.
6.5 Nature and Contents of Container
Timoptol-XE 2.5 mg/mL. 2.5 mL bottle with a controlled dropper tip and white cap.
Timoptol-XE 5.0 mg/mL. 2.5 mL bottle with a controlled dropper tip and white cap.
All strengths may not currently be marketed in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
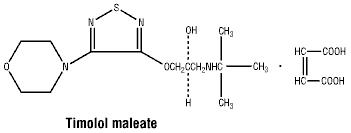
Timolol maleate has a molecular weight of 432.50. It is a white, odourless, crystalline powder which is soluble in water, methanol and alcohol.
Chemical structure. Timolol maleate is described chemically as (S)-1-[(1,1-dimethylethyl) amino]-3-[[4-(4-morpholinyl)-1,2,5-thiadiazol-3-yl] oxy]-2-propanol, (Z)-2-butenedioate (1:1) (salt). Timolol maleate possesses an asymmetric carbon atom and is provided as the levo isomer. The empirical formula is C13H24N4O3S.C4H4O4 and the structural formula is:
7 Medicine Schedule (Poisons Standard)
(S4) Prescription Only Medicine.
Date of First Approval
29 July 1999
Date of Revision
31 May 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.