TISSEEL
Brand Information
| Brand name | TISSEEL |
| Active ingredient | Aprotinin + Factor XIII + Fibrinogen, Thrombin + Calcium chloride dihydrate |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the TISSEEL.
Summary CMI
TISSEEL
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor, nurse or pharmacist.
1. Why am I receiving TISSEEL?
TISSEEL is a fibrin sealant with the active ingredients fibrinogen, thrombin, aprotinin, factor XIII and calcium chloride dihydrate. It is used to help stop excessive blood loss during surgery, and as a ‘tissue glue’ in certain types of surgery.
For more information, see Section 1. Why am I receiving TISSEEL? in the full CMI.
2. What should I know before I am given TISSEEL?
Do not use if you have ever had an allergic reaction to aprotinin or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am given TISSEEL? in the full CMI.
3. What if I am taking other medicines?
There are currently no medicines known to interact with TISSEEL.
See Section 3. What if I am taking other medicines? in the full CMI.
4. How is TISSEEL used?
- Your surgeon will apply TISSEEL as needed during your surgical operation.
- Your surgeon will decide how much TISSEEL will be applied, depending on your individual need.
- More instructions can be found in Section 4. How is TISSEEL used? in the full CMI.
5. What should I know after having TISSEEL?
| Things you should do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know after having TISSEEL? in the full CMI.
6. Are there any side effects?
Serious side effects include sudden allergic reactions (early symptoms can be flushing, increased or decreased pulse rate, hives, itching, or difficulty breathing). Allergic reactions are more likely if you have received TISSEEL in previous surgery. Allergic reactions can be life-threatening.
Products like TISSEEL can be applied with spray devices. Improper use of spray devices can cause air to get into the blood circulation. This side effect can be life-threatening.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
TISSEEL (frozen) fibrin sealant syringe
Active ingredients: fibrinogen, thrombin, aprotinin, factor XIII, and calcium chloride dihydrate
Consumer Medicine Information (CMI)
This leaflet provides important information about using TISSEEL. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using TISSEEL.
Where to find information in this leaflet:
1. Why am I receiving TISSEEL?
2. What should I know before I am given TISSEEL?
3. What if I am taking other medicines?
4. How is TISSEEL used?
5. What should I know after having TISSEEL?
6. Are there any side effects?
7. Product details
1. Why am I receiving TISSEEL?
TISSEEL is a fibrin sealant containing the active ingredients fibrinogen, thrombin, aprotinin, factor XIII, and calcium chloride dihydrate.
TISSEEL is used to help stop blood loss during surgery by making a clot, which is very similar to a natural blood clot. This clot adheres firmly to tissue, acting as a kind of ‘tissue glue’.
Because of its glue-like properties, TISSEEL can also be used:
- to help close openings the colon from outside the body (colostomies)
- as an aid in some cartilage repair procedures
- in hernia repair procedures.
2. What should I know before I am given TISSEEL?
Warnings
You should not be given TISSEEL if:
- you are hypersensitive to aprotinin, or any of the ingredients listed at the end of this leaflet.
Check with your doctor if you:
- have any other medical conditions
- tend to have allergic reactions
- take any medicines for any other condition
- have been given aprotinin or fibrin sealant in a previous surgical operation.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Use in children
The safety and effectiveness of TISSEEL in children have not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect TISSEEL.
There are currently no medicines known to interact with TISSEEL.
4. How is TISSEEL USED?
How it is used
- Your surgeon will apply TISSEEL as needed during your surgical operation.
How much is given
- Your surgeon will decide how much TISSEEL will be applied, depending on your individual need.
If too much TISSEEL is used
Your surgeon applies TISSEEL only on the tissue surface (local application). It is unlikely that your surgeon will use too much (overdose).
5. What should I know after having TISSEEL?
Things you should do
Talk to your doctor and surgeon about how you feel after your surgical operation.
Since TISSEEL is given in a hospital, your healthcare provider will monitor your progress and any side effects.
Looking after your medicine
You will not have to store or handle this medicine.
This medicine is stored in the hospital pharmacy, kept at minus 18°C or colder and protected from light. Medical professionals handle, prepare and use it according to the manufacturer's instructions.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Skin-related:
Followed about two weeks later by:
Followed by
| Speak to your doctor if you have any of these less serious side effects. |
Serious side effects
| Serious side effects | What to do |
Allergic reaction-related:
| Call the doctor on duty straight away if you notice the symptoms of an allergic reaction. Go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is only available with a doctor's prescription. It is only used in a hospital setting, during specific surgical operations.
What TISSEEL contains
TISSEEL contains synthetic and human plasma proteins. Human proteins have been isolated from the pooled blood of healthy donors according to World Health Organisation (WHO) guidelines.
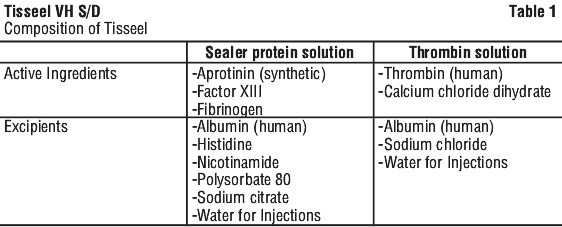
TISSEEL contains two components, the Sealer Protein Solution and the Thrombin Solution (see table below). The two components are filled in two separate chambers of a double-chamber syringe.
| Active ingredients - (main ingredients) | Sealer Protein Solution
|
| Other ingredients (inactive ingredients) | Sealer Protein Solution Human Albumin, Histidine, Sodium Citrate, Polysorbate 80, Nicotinamide, Water for Injections Thrombin Solution Sodium Chloride, Human Albumin, Water for Injections |
| Potential allergens | Aprotinin |
Do not take this medicine if you are allergic to any of these ingredients.
What TISSEEL looks like
TISSEEL is a frozen, two-component fibrin sealant, supplied in a single-use double-chamber syringe packed in two bags and one device set with 2 joining pieces and 4 application cannulas.
TISSEEL is available in 2.0 mL, 4.0 mL, and 10.0 mL package sizes. (Aust R 147141).
Who distributes TISSEEL
Baxter Healthcare Pty Ltd
1 Baxter Drive
Old Toongabbie
NSW 2146
This leaflet was prepared in June 2025.
Baxter and TISSEEL are trademarks of Baxter International Inc.
Brand Information
| Brand name | TISSEEL |
| Active ingredient | Aprotinin + Factor XIII + Fibrinogen, Thrombin + Calcium chloride dihydrate |
| Schedule | Unscheduled |
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Aprotinin (synthetic), factor XIII, fibrinogen, thrombin and calcium chloride dihydrate.
2 Qualitative and Quantitative Composition
The active ingredients of Tisseel VH S/D are formulated as two sterile, deep-frozen solutions, the Sealer Protein Solution and Thrombin Solution (see Table 1 for composition of Tisseel). Each solution is presented in a separate pre-filled chamber of one double-chamber syringe. The active ingredients are fractionated from pooled human plasma. See Table 2.

3 Pharmaceutical Form
Two-Component Fibrin Sealant, Deep-Frozen, Vapour Heated (VH) and Solvent Detergent (S/D) treated, Tisseel VH S/D1. The 2 components of Tisseel are colourless to pale yellow, opalescent when frozen and clear to slightly turbid solutions once defrosted.
1 The term 'Vapour Heated (VH) and Solvent Detergent (S/D) treated' is abbreviated as VH S/D.
4 Clinical Particulars
4.1 Therapeutic Indications
Tisseel is indicated:
as adjunct to haemostasis during surgical procedures, when control of bleeding by conventional surgical techniques is ineffective or impractical;
as a sealant as an adjunct for closure of colostomies;
as a sealant and/or adhesive for use in autologous chondrocyte implantation (ACI) or matrix induced autologous chondrocyte implantation (MACI) procedures;
for mesh fixation in noniatrogenic abdominal wall hernia repair, as an alternative to sutures, staples or tacks.
4.2 Dose and Method of Administration
Dosage. Tisseel should only be administered topically. Do not inject. Tisseel must not be applied intravascularly. The required dose depends upon the size of the surface to be covered. To avoid the formation of excess granulation tissue, and to ensure gradual absorption of the solidified fibrin sealant, only a thin layer of Tisseel should be applied. Excessive thickness of the fibrin layer may interfere with the product's efficacy and the wound healing process.
The application can be repeated, if necessary. However, avoid re-application of Tisseel to a pre-existing polymerized Tisseel layer as Tisseel will not adhere to a polymerised layer. If used for tissue adherence, it is recommended that the initial application cover the entire intended application area.
The approximate surface areas covered by each package size of Tisseel are listed in Table 3.

The quantity of Tisseel required for mesh fixation depends on the mesh size selected and the recommended amount is the same for different application techniques. For example, 2 - 4 mL of reconstituted Tisseel applied as a thin layer is suitable to adequately fix a standard size mesh of approximately 10 x 15 cm.
When using the drop technique, surgeons should apply Tisseel at key anchor points for fixing the mesh (e.g. pubic tubercle in inguinal hernia repair) and at the margins of the mesh. Application by spray, either alone or in combination with drops, should cover the mesh uniformly with a thin layer.
In inguinal hernia repair the mesh covering vascular structures and nerves can be fixed with Tisseel alone using drops and/or spray.
Method of preparation of Tisseel pre-filled syringe (frozen). General. Do not use Tisseel unless it is completely thawed and warmed to 33°C - 37°C (liquid consistency).
Tisseel must not be exposed to temperatures above 37°C and must not be microwaved.
The sealer protein and the thrombin solutions should be clear or slightly opalescent.
Do not use solutions that are cloudy, discolored, have deposits or other changes in their appearance.
The protective syringe cap should not be removed until storage, thawing and warming is complete and application tip is ready to be attached.
To facilitate removal of the tip cap from the syringe, rock the tip cap by moving it backward and forward, then pull the protective cap off the syringe.
Thaw and warm the pre-filled syringes in one of the following options:
Quick thawing/ warming methods (preparation in a single step). An overview of quick thawing/ warming methods is provided in Table 4.

Remove the pre-filled syringe from the inner pouch and place the syringe directly into the sterile water heated to 33°C - 37°C ensuring the syringe is completely immersed in the water (see Table 4 for minimum thawing/warming times).
To monitor the specified temperature range, control the water temperature using a thermometer and change the water as necessary.
Non-sterile water bath. Place the pre-filled syringe packed in both pouches, in a water bath heated to 33°C - 37°C outside the sterile area, ensuring the pouches remain immersed in the water (see Table 4 for minimum thawing/warming times).
Remove the pouches from the water bath after thawing and warming.
Dry the outer pouch and transfer the pre-filled syringe in the inner pouch, onto the sterile area.
Incubator. Place the pre-filled syringe, packed in both pouches, in an incubator outside the sterile area (see Table 4 for minimum thawing/warming times).
After thawing/warming in the incubator, remove the outer pouch and transfer the prefilled syringe, inside the inner pouch, into the sterile area.
Thawing at room temperature (not above +25°C) followed by warming in incubator. Thaw the pre-filled syringe, packed in both pouches, at room temperature outside the sterile area (see Table 5 for minimum thawing times).
Warm the pre-filled syringe, packed in both pouches, in an incubator at 33°C - 37°C outside the sterile area (see Table 5 for minimum warming times).
After thawing/warming in the incubator, remove the outer pouch and transfer the prefilled syringe, inside the inner pouch, into the sterile area.

The solutions must be used within 72 hours after thawing at 25°C or below.
Any unused product and/or devices should be disposed of in accordance with local requirements.
Method of application. Application of Tisseel must be completed within 4 hours after opening the pre-filled frozen double chamber syringe. Discard any unused product. Separate, sequential application of the two components of Tisseel must be avoided.
Dry the site of application. Before application, the surface area of the wound needs to be dried using standard techniques. Do not use pressurised air or gas for drying the site.
If application is interrupted, clogging occurs immediately in the cannula. Replace the application cannula with a new one only immediately before application is resumed. If the aperture of the joining piece (Y connector) facing the cannula is clogged, use the spare joining piece provided in the package.
To prevent Tisseel from adhering to gloves and instruments, wet these with sodium chloride solution before contact.
In cases where very small volumes (1 to 2 drops) of Tisseel are administered, expel and discard the first several drops from the application cannula immediately before application, to ensure use of adequately mixed product.
Caution must be used when applying fibrin sealant using pressurized air or gas.
Any application of pressurized air or gas is associated with a potential risk of air embolism, tissue rupture, or gas entrapment with compression, which may be life-threatening.
Tisseel with the spray set must not be used in enclosed body areas.
Tisseel must be sprayed only onto application sites that are visible.
The user must follow the instructions and precautions in the Easy Spray device user manual, for example regarding the need to limit the gas pressure to a maximum of 2 bars and not be sprayed if the distance is closer than 10 cm from the tissue surface. When using the DuploSpray MIS spray device (for minimally invasive/laparoscopic surgery) it should not be sprayed closer than 2 cm. In minimally invasive/laparoscopic procedures, use a pressure regulator device (DuploSpray MIS spray device) that delivers a maximum pressure of no more than 1.5 bar (22 psi) using only carbon dioxide (CO2) gas. Only use application devices licensed/CE Marked for the administration of Tisseel.
When spraying Tisseel, changes in blood pressure, pulse, oxygen saturation and end tidal CO2 should be monitored because of the possibility of occurrence of air or gas embolism.
Application beyond the intended area of application should be avoided.
After the two components have been applied, fix or hold the sealed parts in the desired position for at least three to five minutes to ensure the setting Tisseel adheres firmly to the surrounding tissue.
It is strongly recommended that every time Tisseel is applied to a patient, the name and batch number of the product are recorded in order to maintain a link between the patient and the batch of the product.
Non-spray application. For application, connect the double chamber ready-to-use syringe with the sealer protein solution and the thrombin solution to a joining piece and an application cannula - both are provided in the set with the application devices. The common plunger of the double chamber ready-to-use syringe ensures that equal volumes of the two sealant components are fed through the joining piece into the application cannula where they are blended and then applied.
Operating instructions. Expel all air from the syringe prior to attaching any application device.
Do not expel the air remaining inside the joining piece and inside the application cannula until you start the actual application because this may clog the application cannula.
Align the joining piece and tether to the side of the syringe with the tether strap hole.
Connect the nozzles of the double chamber ready-to-use syringe to the joining piece, ensuring that they are firmly attached.
Secure the joining piece by fastening the tether strap to the double chamber ready to-use syringe.
If the tether strap tears, use the spare joining piece provided in the kit.
If a spare joining piece is not available, the system can still be used if care is taken to ensure that the connection is secure and leak-proof.
Do not expel the air remaining inside the joining piece.
Attach an application cannula on to the joining piece.
Administration. Prior to applying Tisseel the surface of the wound needs to be dried by standard techniques (e.g. intermittent application of compresses, swabs, use of suction devices). Do not use pressurized air or gas for drying the site.
Ensure that all parts of the body outside the desired application area are sufficiently covered to prevent tissue adhesion at undesired sites.
Apply the mixed sealer protein - thrombin solution on to the recipient surface or on to the surfaces of the parts to be glued by slowly pressing on the back of the common plunger.
In surgical procedures that require the use of minimal volumes of fibrin sealant, it is recommended to expel and discard the first few drops of product.
After Tisseel has been applied, allow at least 2 minutes to achieve sufficient polymerization.
Note. If application of the fibrin sealant is interrupted, clogging may occur quickly in the cannula. In this case, replace the application cannula with a new one immediately before application is resumed. If the openings of the joining piece are clogged, use the spare joining piece (Y connector) provided in the package.
Application with spray device. The pressure regulator should be used in accordance with the manufacturer's instructions.
When applying Tisseel using a spray device be sure to use a pressure and a distance from tissue within the ranges recommended by the manufacturer as follows, see Table 6.

For the application of Tisseel in enclosed thoracic and abdominal spaces the DuploSpray MIS applicator and regulator system is recommended. Please refer to the instruction manual of the DuploSpray MIS device.
Equivalent spray devices, intended for specific use with Tisseel, may also be used. When using other spray devices, follow the instructions for use that are provided with the device.
See Section 6.2 Incompatibilities.
4.3 Contraindications
Known hypersensitivity to aprotinin or known hypersensitivity to any other component of Tisseel.
Injection of Tisseel into tissues is contraindicated. Such use has been associated with inadvertent intravascular injection and may result in life-threatening thromboembolic complications, can lead to intravascular coagulation, and may increase the likelihood and severity of acute hypersensitivity reactions in susceptible patients.
Tisseel should be applied with caution to minimise any risk of intravascular application, for example in coronary bypass surgery. Tisseel should only be applied topically.
Additionally, soft tissue injection of Tisseel carries the risk of an anaphylactic reaction and/or local tissue damage.
4.4 Special Warnings and Precautions for Use
Viral and prion risk. Sealer protein concentrate and thrombin are made from human plasma. Products made from human plasma may contain infectious agents which can cause disease, such as viruses and theoretically, the agent that causes Creutzfeldt-Jakob Disease (CJD) in humans. Standard measures to prevent infections resulting from the use of medicinal products prepared from human blood or plasma include selection of donors, screening of individual donations and plasma pools for specific markers of infection and the inclusion of effective manufacturing steps for the inactivation/removal of viruses. Despite this, when medicinal products prepared from human blood or plasma are administered, the possibility of transmitting infective agents cannot be totally excluded. This also applies to unknown or emerging viruses or other pathogens.
The measures taken (including double virus inactivation by vapour heat treatment and solvent detergent treatment) are considered effective for enveloped viruses such as HIV, HBV, and HCV, and for the non-enveloped virus HAV. The measures taken may be of limited value against small non-enveloped viruses such as parvovirus B19. Parvovirus B19 infection may be serious for pregnant women (foetal infection) and for individuals with immunodeficiency or increased red blood cell turnover (e.g. hemolytic anaemia).
All infections thought by a clinician possibly to have been transmitted by Tisseel should be reported by the clinician or other healthcare provider to Baxter.
Patients should be instructed to consult their clinician if symptoms of B19 virus infection appear (fever, drowsiness, chills and runny nose, followed about two weeks later by a rash and joint pain).
General. Administration of Tisseel may result in allergic reactions in some patients. For patients with a known allergic diathesis, a history of hypersensitivity to medical products or a history of having previously received aprotinin-containing products (including previous use of Tisseel) a careful risk-benefit assessment should be carried out prior to administration. The risk of immunisation against proteins such as aprotinin is increased if repeated exposure occurs within six months. If it is decided to proceed with treatment in such patients, prior administration of antihistamines should be considered.
Manifestations of hypersensitivity reactions to Tisseel observed include: bradycardia, tachycardia, hypotension, flushing, bronchospasm, wheezing, dyspnea, nausea, urticaria, angioedema, pruritus, erythema, paresthesia. Fatal anaphylactic reactions, including anaphylactic shock, have also been reported with Tisseel (see Section 4.8 Adverse Effects (Undesirable Effects)). Intravascular application might increase the likelihood and severity of acute hypersensitivity reactions in susceptible patients. Because of the risk of intravascular injection, the product must not be injected into highly vascularised tissue, such as nasal mucosa.
The new formulation of Tisseel contains synthetic aprotinin. As synthetic aprotinin is structurally identical to bovine aprotinin, the use of Tisseel in patients with allergies to bovine proteins should be carefully evaluated.
Air or gas embolism, tissue rupture, or gas entrapment with compression, which may be life-threatening, have occurred with the use of spray devices employing a pressure regulator to administer Tisseel. These events appear to be related to the use of the spray device at higher than recommended pressures and/or in close proximity to the tissue surface. The risk appears to be higher when Tisseel sprayed with air, as compared to carbon dioxide (CO2) and therefore cannot be excluded with Tisseel when sprayed in open wound surgery.
When using the DuploSpray MIS spray device (for minimally invasive/laparoscopic surgery) it should not be sprayed closer than 2 cm. In minimally invasive/laparoscopic procedures, a pressure regulator device (DuploSpray MIS spray device) that delivers a maximum pressure of no more than 1.5 bar (22 psi) and uses carbon dioxide gas only should be used.
To reduce the risk of a potentially life-threatening gas embolism, when applying Tisseel using a spray device, be sure to use the pressure within the pressure range recommend by the spray device manufacturer. In the absence of a specific recommendation avoid using pressure above 1.4-1.7 bars (20-25 psi). Do not spray if the distance is closer than the distance recommended by the spray device manufacturer. In the absence of a specific recommendation avoid spraying closer than 10-15 cm from the surface of the tissue. When spraying Tisseel, changes in blood pressure, pulse, oxygen saturation and end tidal CO2 should be monitored because of the possibility of occurrence of air or gas embolism.
As sealer protein and thrombin solutions can be denatured following contact with solutions containing alcohol, iodine or heavy metals (e.g. in disinfectants), any such substances should be removed before application (see Section 6.2 Incompatibilities).
Tisseel alone is not indicated for the treatment of severe or brisk arterial or venous bleeding. When used in these situations, Tisseel is likely to be washed away in the flow of blood before haemostasis can be attained.
If possible, cover all tissue adjacent to the site of sealing before applying Tisseel.
Tisseel should not be used for the sealing of neuroanastomoses, as the high aprotinin content of the Tisseel solution delays absorption of the fibrin seal and it cannot be ruled out that this may cause fibrosis.
Injection into the nasal mucosa must be avoided, as severe allergic/anaphylactoid reactions have been observed and thromboembolic complications may occur in the area of the ophthalmic artery.
Apply Tisseel as a thin layer. Excessive clot thickness may negatively interfere with the product's efficacy and the wound healing process.
The safety and effectiveness of Tisseel used alone or in combination with biocompatible carriers in neurosurgical procedures or other surgeries involving confined spaces have not been established. There have been rare reports of serious adverse events such as paralysis and other compressive complications possibly related to the use of fibrin sealants in combination with resorbable haemostatic agents.
If fibrin sealants are applied in confined bodily spaces, the risk of compressive complications should be taken into account.
Use in hernia. Nonclinical data indicate that Tisseel is unlikely to be effective when used alone for mesh fixation to peritoneum (e.g. with an intraperitoneal approach for laparoscopic ventral hernia repair). In such situations Tisseel should only be used as an adjunct to sutures or staples/tacks.
Tisseel is not effective when used with omega 3 fatty acid-containing and non-porous ePTFE meshes. Tisseel should not be used with these meshes. Furthermore, the efficacy of Tisseel has not been demonstrated in meshes with other coatings, including with beta glucan.
Use in the elderly. Of the total number of subjects in a clinical study of Tisseel, 71 out of 144 subjects were 65 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experiences have not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Paediatric use. Safety and effectiveness of Tisseel in paediatric patients have not been established. There has been a single report of disseminated intravascular coagulation occurring in a premature infant who received Tisseel 3 mL during a laparotomy for peritoneal adhesions.
Effects on laboratory tests. The effect of this medicine on laboratory tests has not been established.
4.5 Interactions with Other Medicines and Other Forms of Interactions
There are no known interactions between Tisseel and other drugs. Efficacy has been demonstrated in fully heparinised patients undergoing cardiopulmonary bypass.
See Section 6.2 Incompatibilities for more detailed information on interactions with substances other than drugs.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Studies of the effect of Tisseel on fertility have not been performed.
Use in pregnancy. (Category B2)
Animal reproduction studies have not been conducted with Tisseel. There are no adequate and well-controlled studies in pregnant women. Tisseel should be used during pregnancy only if clearly needed and potential benefit justifies the potential risk to the fetus.
Use in lactation. Studies on Tisseel in lactating animals or women have not been conducted. Tisseel should be used during lactation only when strictly indicated.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Anaphylactic and anaphylactoid reactions may occur in patients who have previously received a fibrin-based sealant, in those with a known hypersensitivity to aprotinin and those who have previously received aprotinin systemically. Even if the second treatment with Tisseel was well tolerated, a subsequent administration of Tisseel or systemic administration of aprotinin may result in severe anaphylactic reactions.
Symptoms associated with allergic/anaphylactic reactions include flushing, urticaria, pruritus, nausea, drop in blood pressure, tachycardia or bradycardia, dyspnoea, severe hypotension, and anaphylactic shock. In the event of hypersensitivity reactions, administration of Tisseel should be discontinued, the topical clot removed, and appropriate treatment instituted.
In rare cases, these reactions may also occur in patients receiving aprotinin or Tisseel for the very first time.
Injection of Tisseel into tissues has been associated with inadvertent intravascular administration.
Tisseel must not be applied intravascularly (see Section 4.3 Contraindications).
Intravascular application can lead to intravascular coagulation, may result in life-threatening thromboembolic events, and might increase the likelihood and severity of acute hypersensitivity reactions in susceptible patients.
The adverse reactions presented in this section were reported from clinical trials investigating the safety and efficacy of Tisseel. In these trials, Tisseel was administered for adjunct haemostasis in cardiac, vascular, and total hip replacement surgeries; and for the sealing of lymphatic vessels in patients undergoing axillary lymph node dissection. In these studies, a total of 499 patients were administered Tisseel. The frequencies are based on the number of cases considered possibly/probably related by investigators.
Because clinical trials are conducted under varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug, and may not reflect the rates observed in clinical practice (see Table 7).

Cardiac disorders. Bradycardia, tachycardia.
Gastrointestinal disorders. Nausea, intestinal obstruction.
General disorders and administration site disorders. Hypersensitivity reactions, oedema, pyrexia.
Immune system disorders. Hypersensitivity (including anaphylactic reactions, anaphylactic shock, and the following manifestations: angioedema, paresthesia, bradycardia, tachycardia, flushing, bronchospasm, dyspnoea, wheezing, urticaria, pruritus, and erythema). Anaphylactic reactions and anaphylactic shock have included fatal outcomes.
Injury, poisoning and procedural complications. Anaphylactoid reactions, seroma.
Investigations. Drop in blood pressure.
Skin and subcutaneous tissue disorders. Pruritus, impaired healing.
Vascular disorders. Haematoma, flush, (severe) hypotension, and thromboembolism, including cerebral artery embolism and venous thrombotic cerebral infarction*, air embolism**.
* As a result of intravascular application into the superior petrosal sinus.
** As with other fibrin sealants life-threatening/fatal air or gas embolism when using devices with pressurized air or gas occurred; this event appears to be related to an inappropriate use of the spray device (e.g. at higher than recommended pressure and in close proximity to the tissue surface).
Class reactions. Other adverse reactions associated with the fibrin sealant/haemostatic class include: manifestations of hypersensitivity such as application site irritation, chest discomfort, chills, headache, lethargy, restlessness, and vomiting.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
No symptoms of overdose are known for Tisseel. For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
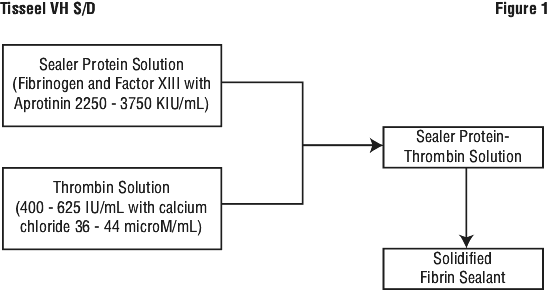
Mechanism of action. Tisseel contains two components, sealer protein solution and thrombin solution. The sealer protein solution contains fibrinogen as the main active ingredient, the active ingredient of the thrombin solution is human thrombin.
Thrombin is a highly specific protease that transforms the fibrinogen contained into fibrin monomers. These fibrin monomers are then polymerized in a linear fashion and stabilised by cross-linking (catalysed by factor XIII) to form an insoluble fibrin clot. Aprotinin (synthetic), is a protease inhibitor which prevents the premature degradation of fibrin.
These reactions simulate the key features of the physiological coagulation process. The resulting fibrin clot appears as a white, elastic mass which firmly adheres to tissue and which can be used to achieve haemostasis or seal tissues.
When the two component solutions come into contact, conversion of fibrinogen to fibrin, and polymerization and cross-linking of fibrin monomers commences immediately and results in the clotting of the fibrin within seconds. Figure 1 illustrates the process.
2 Baxter commercialized several single virus inactivated, predecessor fibrin sealant products, utilizing heat treatment (HT) or vapor heat treatment (VH) for virus inactivation. Predecessor products were manufactured both in frozen or lyophilized presentation.

An earlier formulation of Tisseel VH S/D, Tisseel HT (fibrin sealant heat-treated) was evaluated in an open-label crossover study against control topical haemostatic agents in 489 patients undergoing cardiovascular re-operation or re-sternotomy at 11 institutions. Patients were randomised to Tisseel HT or control haemostatic agents when a topical haemostatic was needed at the conclusion of surgery and after all attempts of surgical haemostasis. Patients were crossed to the alternative therapy if bleeding continued after the 5 minute endpoint. At 10 centres, Tisseel was used after administration of protamine sulfate. At one site, Tisseel could be used before administration of protamine sulfate. 365 of the 489 patients had an eligible bleeding event, for the primary endpoint, successful haemostasis at 5 minutes, Tisseel was statistically significantly superior to control topical haemostatic agents (see Table 9):

In a single centre, open label trial, an earlier formulation of Tisseel was compared to historical controls in patients undergoing laparotomy for blunt or penetrating traumatic injury to the spleen and/or liver. Use of Tisseel resulted in the need for statistically significantly fewer splenectomies than control haemostatic manoeuvres (see Table 10):

In a single centre, prospective open label study of 120 patients randomised to standard of care (59 patients) or standard of care plus fibrin sealant (61 patients) for elective colostomy closure after temporary colostomy placement for treatment of traumatic injury to the colon, the earlier version of Tisseel plus standard of care was shown to be statistically significantly superior to standard of care alone (p = 0.0406, Jonckheere-Terpstra test for ordinal data, two sided) with regard to anastomotic complications (leakage, intra-abdominal abscess formation, re-operation, septic shock, and death).
A review of published literature was conducted studying the repair of defects of the articular cartilage in the knee; (n = 293 patients; 166 patients were treated with either Autologous Chondrocyte Implantation (ACI) or Matrix-Induced Autologous Chondrocyte Implantation (MACI); 127 patients were treated with either mosaicplasty or microfracture or abrasive arthroplasty). In all ACI/MACI procedures, Tisseel Fibrin Sealant was applied topically. The efficacy of Tisseel has been assessed indirectly by the efficacy outcome measures used to assess joint function following repair of cartilage defects. Outcome measures within the first six months of treatment are considered to be of particular importance because treatment failure attributed to graft movement (e.g. periosteal delamination or detachment of the collagen matrix) typically occurs within the first three to six months following implant. In addition, in the first 6 months post-implant, there were no reports by patients of symptoms which may be indicative of graft instability such as "locking" or "catching" of the knee joint. In one study MRI assessments, made at one and two months, showed that there was a high level of graft integration with the surrounding cartilage, and that grafts were present and in their original position in the majority of patients (15/17). These findings suggest that Tisseel is an effective adhesive in this indication. Long term results (≥ 6 months) indicated that treatment with either ACI or MACI was at least as successful as the comparative treatment.
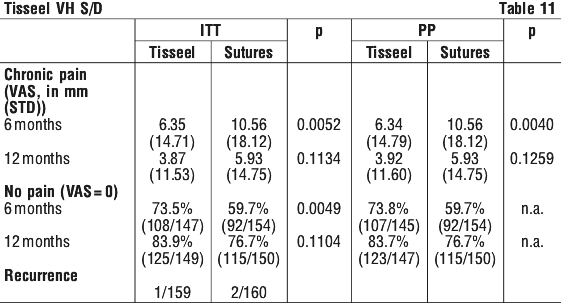
Hernia repair. A prospective, multi-centre, randomized, double-blinded, parallel, controlled clinical trial (Campanelli, 2009) involving 325 male subjects was conducted to evaluate the safety and effectiveness of Tisseel in uncomplicated unilateral or bilateral, direct or indirect primary inguinal hernia using the Lichtenstein technique. Patients were randomized to have mesh fixation either by sutures or by application of Tisseel.
For the clinically important outcomes of chronic pain and recurrence as well as for pain/ no pain the results of the statistical analyses are as follows (see Table 11):
Lau (2005) conducted a single-centre, randomized (1:1), controlled clinical trial to compare the clinical outcomes of simultaneous bilateral endoscopic totally extraperitoneal (TEP) inguinal hernia repair using either an earlier version of Tisseel or staples for mesh fixation in 93 subjects. Efficacy outcomes were recurrence rate and chronic groin pain. At a median follow-up of 1.2 years there were no incidences of hernia recurrence in either treatment group. The difference in incidence of chronic pain for the 78 subjects assessed at median 2 year follow-up was not significant (Tisseel 5/38, 13.2% (95% CI 2.5% - 23.9%), and staples 8/40, 20% (95% CI 7.6% - 32.3%)) (p=0.418).
In a prospective single-centre controlled clinical trial by Lovisetto et al. (2007), 197 subjects with uni- or bilateral inguinal or femoral hernia underwent laparoscopic transabdominal preperitoneal (TAPP) hernioplasty and were randomized to mesh fixation by either staples (n=98) or Tisseel (n=99). Tisseel was applied via a laparoscopic catheter.
The primary efficacy outcomes were early postoperative and late neuralgia recorded using a visual analogue scale (VAS): at 1, 3, and 6 months after surgery, the mean VAS score was significantly lower in the Tisseel group compared with the staples group (see Table 12):


A total of 3 recurrences occurred in the study, all of which occurred in group C (n.s.). The postoperative (24 - 72 hours) pain score in group D (VAS 2) was markedly lower than in groups A (VAS 5-7), B (VAS 4-5), and C (VAS 3-4).
5.2 Pharmacokinetic Properties
Solidified Tisseel VH S/D is intended for local application only, therefore systemic exposure or distribution to other organs or tissues is not expected and pharmacokinetic studies were not conducted.
5.3 Preclinical Safety Data
Genotoxicity. Studies of genotoxic potential of Tisseel have not been performed. Negative results were obtained in bacterial reverse mutation assays (Ames tests) conducted with various components of Tisseel (sealer protein solution containing bovine aprotinin; synthetic aprotinin; human thrombin solution).
Carcinogenicity. Animal studies to evaluate the carcinogenic potential of Tisseel have not been performed.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Solutions containing alcohol, iodine or heavy metals may interfere with the product's performance due to denaturation of proteins or other mechanisms. If any of these substances have been used to clean the wound area, the area must be thoroughly rinsed before application of Tisseel.
Oxidised cellulose-containing preparations may reduce the efficacy of Tisseel and should not be used as carrier materials.
Tisseel must not be mixed with other medicinal products.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
Deep frozen Tisseel has a shelf life of two years when stored at -18°C or colder. The expiry date is stated on the final container and the package.
6.4 Special Precautions for Storage
Store in a freezer at -18°C or colder. The cold storage chain must not be interrupted until preparation for use.
Keep container in the outer carton to protect from light.
Unopened pouches, thawed at 25°C or below, may be stored for up to 72 hours at 25°C or below after removal from the freezer.
If the product is removed from original pouch or warmed to 33-37°C, it must be used within 12 hours.
Special precautions for storage. After thawing, the solutions must not be refrozen or refrigerated.
Store in a freezer (at -18°C or colder); the cold storage chain must not be interrupted until use.
Keep container in the outer carton to protect from light.
Keep out of reach and sight of children.
For single use only. Do not re-sterilise.
6.5 Nature and Contents of Container
Nature of containers. Both sealer protein solution and thrombin solution are contained in two separate chambers of a single use double chamber syringe made of polypropylene.
Contents. Each pack Tisseel contains:
One single use double chamber syringe, each chamber containing:
Chamber number [1]: Sealer Protein Solution (with aprotinin) deep frozen;
Chamber number [2]: Thrombin Solution (with calcium chloride) deep frozen.
One set of devices (see below).
Set of devices. Each pack Tisseel contains a double-sterile set of devices consisting of two joining pieces and four application cannulas. These devices are used for the simultaneous application of the fibrin sealant components. For details on application and complications associated therewith see Section 4.2 Dose and Method of Administration, Operating instructions.
The set of devices is sterile and non-pyrogenic in unopened and undamaged package. Sterilised by exposure to ethylene oxide.
Tisseel is available in the following pack sizes:
Tisseel 2 mL (containing 1.0 mL of sealer protein solution and 1.0 mL of thrombin solution);
Tisseel 4 mL (containing 2.0 mL of sealer protein solution and 2.0 mL of thrombin solution);
Tisseel 10 mL (containing 5.0 mL of sealer protein solution and 5.0 mL of thrombin solution).
Package size. 2 mL, 4 mL, 10 mL.
6.6 Special Precautions for Disposal
Any unused product or waste material should be disposed of in accordance with local requirements.
The Tisseel solutions contain no antimicrobial agent. Tisseel is intended for single use in one patient only and unused solution in the syringe should be discarded.
6.7 Physicochemical Properties
Fibrinogen. Chemical Structure: The major component of the clottable protein (human origin) is fibrinogen. The fibrinogen molecule is a dimer composed of two symmetrical subunits linked by -S-S- bonds. It could be written in a simple formula as (Aα, Bβ, γ)2.
Molecular Weight (MW): about 340,000. The Aα-chain contains 610 amino acids (MW about 68,000), the Bβ-chain 461 amino acids (MW about 57,000), and the γ-chain 411 amino acids (MW about 47,000). Thus, the entire human fibrinogen contains 2964 amino acids.
Thrombin. Chemical Structure: Thrombin (human origin) is a glycosylated protein, consisting of two polypeptide subunits A and B, covalently linked by one -S-S- bond.
Molecular Weight (MW): about 33,800. The human thrombin subunit A chain is made of 36 amino acids, whilst the B chain contains 259 amino acids.
Factor XIII. Chemical Structure: Factor XIII (human origin), also called blood-coagulation factor XIII, is a tetramer composed of two a-chains and two b-chains which are non-covalently associated.
Molecular Weight (MW): about 190,000 (each of chain weight about 80,000).
Aprotinin. Chemical Structure: Aprotinin (synthetic origin) is a protease inhibitor, a polypeptide consisting of one chain of 58 amino acids, also stabilised by -S-S- bonds. Molecular weight (MW): 6511.5.
Calcium chloride dihydrate. Molecular formula: CaCl2.2H2O. Molecular Weight: 147.01. Appearance: a white crystalline powder. Solubility: hygroscopic, freely soluble in water.
CAS number. Fibrinogen. 9001-32-5.
Thrombin. 9002-04-4.
Factor XIII. 9013-56-3.
Aprotinin. 9087-70-1.
Calcium chloride dihydrate. 10035-04-8.
7 Medicine Schedule (Poisons Standard)
Not scheduled.
Date of First Approval
13 March 2009
Date of Revision
01 April 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.