Trisequens
Brand Information
| Brand name | Trisequens |
| Active ingredient | Estradiol, Estradiol + Norethisterone acetate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Trisequens.
Summary CMI
TRISEQUENS®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
WARNING: Important safety information is provided in a boxed warning in the full CMI. Read before using this medicine.
1. Why am I using Trisequens®?
Trisequens® contains the active ingredients estradiol hemihydrate and norethisterone acetate.
Trisequens® is hormone replacement therapy (HRT) used for short-term relief of some of the symptoms of menopause.
For more information, see Section 1. Why am I using Trisequens®? in the full CMI.
2. What should I know before I use Trisequens®?
Do not use if you have ever had an allergic reaction to Trisequens® or any of the ingredients listed at the end of the CMI.
Talk to your doctor about your medical history and your family's medical history, and if you take any other medicines, before starting Trisequens®. Do not use if pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Trisequens®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Trisequens® and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Trisequens®?
- Trisequens® must be taken once a day, at about the same time each day.
- It is supplied in a 28-day calendar dial pack.
More instructions can be found in Section 4. How do I use Trisequens®? in the full CMI.
5. What should I know while using Trisequens®?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Trisequens®? in the full CMI.
6. Are there any side effects?
Serious side effects may include the potential for blood clots (in veins, lungs or brain), severe allergic reaction (anaphylaxis), sudden onset headache or migraine, changes to breasts suggestive of breast cancer, unexpected vaginal bleeding.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
WARNING:
Estrogens and progestogens should not be used for the prevention of heart attacks, stroke or dementia.
A large study called the Women's Health Initiative reported that women who took these hormones in combination or alone after menopause, compared to a placebo (a medicine with no active ingredient), had an increased risk of heart attacks and strokes, breast cancer, blood clots in the lungs or legs, and a higher risk of developing dementia (memory loss) in older women.
Any estrogen with or without progestogen should be prescribed at the lowest effective dose and for the shortest duration determined by considering the risk and benefits for you as an individual. Speak to your doctor if you have any questions.
TRISEQUENS®
Active ingredients: estradiol hemihydrate and norethisterone acetate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Trisequens®. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Trisequens®
Where to find information in this leaflet:
1. Why am I using Trisequens®?
2. What should I know before I use Trisequens®?
3. What if I am taking other medicines?
4. How do I use Trisequens®?
5. What should I know while using Trisequens®?
6. Are there any side effects?
7. Product details
1. Why am I using Trisequens®?
Trisequens® contains the active ingredient estradiol hemihydrate (an estrogen) and norethisterone acetate (a progestogen).
Trisequens® is a hormone replacement therapy (HRT). It is used for short-term relief of some of the symptoms of menopause.
2. What should I know before I use Trisequens®?
Medical history and regular check-ups
The use of HRT carries risks which need to be considered when deciding whether to start taking it, or whether to carry on taking it.
Before you start (or restart) HRT, your doctor must ask about your own and your family´s medical history. Your doctor may decide to perform a physical examination. This may include an examination of your breasts and/or an internal examination, if necessary.
Note: Trisequens® is not a contraceptive. If it is less than 12 months since your last menstrual period or you are under 50 years' old, you may still need to use additional contraception to prevent pregnancy.
Speak to your doctor before starting Trisequens® if you have any questions or concerns.
Warnings
Do not use Trisequens® if:
- you have, have had or suspect having breast cancer
- you have, have had or suspect having cancer of the uterus lining (endometrial cancer), or any other estrogen dependent cancer
- you have any unexplained vaginal bleeding
- you have excessive thickening of the uterus lining (endometrial hyperplasia) that is not being treated
- you have or have ever had a blood clot in a vein (venous thromboembolism), such as in the legs (e.g. deep vein thrombosis), or the lungs (pulmonary embolism)
- you have a blood clotting disorder (such as protein C, protein S or antithrombin deficiency)
- you have or previously have had a disease caused by blood clots in the arteries, such as a heart attack, stroke or angina
- you have or have ever had a liver disease, and your blood test results have not returned to normal
- you have a rare blood problem called 'porphyria' which is passed down in families (inherited)
- you are pregnant or suspect you may be pregnant, or you are breastfeeding
- you are allergic to estradiol or norethisterone, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have premature menopause
- have fibroids or a growth of the uterus lining outside your uterus (endometriosis)
- have a history of excessive growth of the uterus lining (endometrial hyperplasia)
- have an increased risk of developing blood clots (see Blood clots in a vein (venous thromboembolism)
- are to be hospitalised or undergoing surgery, particularly where you are or will be off your feet for a long time. You may need to stop taking Trisequens® for several weeks before your operation, to reduce the risk of a blood clot
- have an increased risk of getting an estrogen-sensitive cancer (such as having a mother, sister or grandmother who has had breast cancer)
- have high blood pressure
- have a liver disorder, such as a benign liver tumour
- have diabetes
- have or have had gallstones
- have migraines or severe headaches
- have systemic lupus erythematosus (SLE)
- have epilepsy
- have asthma
- have otosclerosis (hearing loss caused by changes to the bones in your ear)
- have very high levels of fat in your blood (triglycerides)
- have fluid retention due to heart or kidney problems
- have a condition where your thyroid gland fails to produce enough thyroid hormone (hypothyroidism) and you are treated with thyroid hormone replacement therapy
- have a hereditary condition causing recurrent episodes of severe swelling (hereditary angioedema) or if you have had episodes of rapid swelling of the hands, face, feet, lips, eyes, tongue, throat (airway blockage) or digestive tract
- have a lactose intolerance
- have any other medical conditions
- take any medicines for any other condition.
Cancer risk
Endometrial hyperplasia and cancer
Taking estrogen-only HRT will increase the risk of excessive thickening of the lining of the uterus (endometrial hyperplasia) and cancer of the uterus lining (endometrial cancer).
The progestogen in Trisequens® reduces this extra risk.
In women who still have a uterus and who are not taking HRT, on average, 5 in 1,000 will be diagnosed with endometrial cancer between the ages of 50 and 65.
For women aged 50 to 65 who still have a uterus and who take estrogen-only HRT, between 10 and 60 women in 1,000 will be diagnosed with endometrial cancer, depending on the dose and for how long it is taken.
Irregular bleeding
You may have irregular bleeding or drops of blood (spotting) during the first 3-6 months of taking Trisequens®. However, see your doctor as soon as possible if the irregular bleeding:
- carries on for more than 6 months
- starts after you have been taking Trisequens® for more than 6 months
- carries on after you have stopped taking Trisequens®.
Breast cancer
Evidence shows that taking combined estrogen-progestogen or estrogen-only HRT increases the risk of breast cancer. The extra risk depends on how long you use HRT. The additional risk becomes clear within 3 years of use. After stopping HRT the extra risk will decrease with time, but the risk may persist for 10 years or more if you have used HRT for more than 5 years.
Risk with 5 years of use
For women aged 50 who start taking estrogen-progestogen HRT for 5 years, it is estimated that 21 cases of breast cancer in 1000 users are diagnosed, compared with 13 to 17 cases per 1000 in those who do not take HRT.
Risk with 10 years of use
For women aged 50 who start taking estrogen-progestogen HRT for 10 years, it is estimated at 48 cases of breast cancer in 1,000 users are diagnosed, compared with 27 cases per 1,000 in those who did not take HRT.
Ovarian cancer
Ovarian cancer is rare – much rarer than breast cancer. The use of estrogen-only or combined estrogen-progestogen HRT has been associated with a slightly increased risk of ovarian cancer.
Blood clots in a vein (venous thromboembolism)
The risk of blood clots in the veins is about 1.3 to 3 times higher in HRT users than in non-users, especially during the first year of taking it.
Blood clots can be serious, and if one travels to the lungs, it can cause chest pain, breathlessness, fainting or even death.
Inform your doctor if any of these risks apply to you:
- you are unable to walk for a long time because of major surgery, injury or illness
- you have had one or more miscarriages
- you are overweight or obese (BMI >30 kg/m²)
- you have any blood clotting problem that needs long-term treatment with a medicine used to prevent blood clots
- if any of your close relatives has ever had a blood clot in the leg, lung or another organ
- you have systemic lupus erythematosus (SLE)
- you have cancer.
Heart disease (heart attack)
There is no evidence that HRT will prevent a heart attack. Women over the age of 60 years who use estrogen-progestogen HRT are slightly more likely to develop heart disease than those not taking any HRT.
Stroke
The risk of having as stroke is about 1.5 times higher in HRT users than in non-users. The number of extra cases of stroke due to use of HRT will increase with age.
Other things that can increase the risk of stroke include:
- high blood pressure
- smoking
- drinking too much alcohol
- an irregular heartbeat.
Other conditions
HRT will not prevent memory loss. There is some evidence of a higher risk of memory loss in women who start using HRT after the age of 65.
Tell your doctor or the laboratory staff that you are taking Trisequens® if you need a blood test. This is because this medicine can affect the results of some tests.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not take Trisequens® if you are pregnant or suspect you are pregnant.
Do not take Trisequens® if you are breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Trisequens® and affect how it works; Trisequens® may interfere with some medicines and affect how they work.
Medicines that may increase the effect of Trisequens® include:
- other medicines containing estrogens
- some anti-infectives, e.g. ketoconazole
Medicines that may reduce the effect of Trisequens® include:
- medicines to help you sleep, including barbiturates
- medicines for epilepsy e.g. phenytoin, carbamazepine, lamotrigine
- some antibiotics and other anti-infective medicines, e.g. - rifampicin, rifabutin, nevirapine, efavirenz, ritonavir, telaprevir and nelfinavir
- medicines for hepatitis C infections, e.g. telaprevir
- St John's Wort - used to treat depression, low mood
- antihistamines - used to treat allergies
Trisequens® may increase the effectiveness of some medicines. These include:
- corticosteroids - used to treat inflammatory conditions
- Imipramine
- pethidine
- cyclosporin
Trisequens® may reduce the effectiveness of some medicines. These include:
- lamotrigine - a medicine to treat epilepsy
- some medicines used to treat depression
- blood thinners used to prevent clots
- medicines used to treat diabetes
- thyroid hormones - used to treat poorly functioning thyroid gland.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Trisequens®.
4. How do I use Trisequens®?
How much to take
- Carefully read the instructions below, in order to correctly use the calendar pack.
- Take one tablet a day, preferably at the same time each day, until all 28 tablets have been taken.
Days 1–12:
- Take one blue tablet every day for 12 days.
Days 13–22:
- Take one white tablet every day for 10 days.
Days 23–28:
- Take one red tablet every day for 6 days.
Swallow each tablet with a glass of water. When you have finished each pack, start the next pack immediately.
When to take Trisequens®
- Your doctor will tell you when to start taking the tablets.
- If you are still having periods, you would usually take the first tablet on the fifth day after the start of your period.
- If your periods have stopped or have become infrequent, the first tablet can be taken as soon as you get the product.
How to use the calendar pack
- Set the day reminder
Turn the inner disk to set the day of the week opposite the little plastic tab.
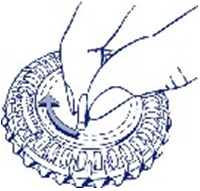
- Take the first day's tablet
Break the plastic tab and tip out the first tablet.
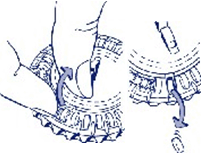
- Move the dial every day
On the next day simply move the transparent dial clockwise one space as indicated by the arrow. Tip out the next tablet. Remember to take only one tablet once a day.
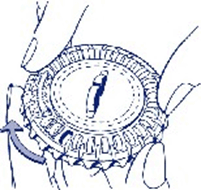
You can only turn the transparent dial after the tablet in the opening has been removed.
If you forget to use Trisequens®
You can always see if you have taken your tablet by looking at the day on the calendar pack.
If you forget to take a tablet at the usual time, throw away the tablet you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
You may have vaginal bleeding or spotting if you forget to take your tablets.
If you take too much Trisequens®
If you think that you or anyone else may have taken too many Trisequens® tablets you may need medical attention.
You should:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Trisequens®?
Things you should do
- Regularly check your breasts
See your doctor if you notice changes, such as:
- dimpling of the skin
- changes to the nipple
- any lumps you can feel. - Go for regular breast screening and pap smear tests.
- Once you've started on HRT, you should see your doctor for regular check-ups (at least once a year). At these check-ups, your doctor may discuss with you the benefits and risks of continuing to take HRT.
- If you need to have surgery, tell your surgeon you are taking HRT, and specifically Trisequens®. You may need to stop taking your tablets a few weeks prior to your surgery.
- If you have stopped therapy, ask your doctor when you can start taking Trisequens® again.
Call your doctor straight away if you:
- Become pregnant while taking Trisequens®. Trisequens® is for use in postmenopausal women, only.
Remind any doctor, dentist or pharmacist you visit that you are taking Trisequens®.
Things you should not do
- Do not stop using this medicine suddenly.
- Do not change the way you are taking Trisequens®
- Do not give Trisequens® to anyone else, even if you think they may have the same condition as you.
- Do not take Trisequens® if you are breastfeeding.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Trisequens® affects you.
Drinking alcohol
Tell your doctor if you drink alcohol.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep the calendar pack in the outer carton in order to protect from light.
Do not put Trisequens® in the refrigerator.
Keep it where young children cannot reach it.
When to discard your medicine
Discard all medicine if it is after the expiry date printed on the pack. The expiry date refers to the last day of that month.
Do not use your medicine if the packaging is torn or shows signs of tampering.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
When you start taking Trisequens® your body has to adjust to new hormone levels. See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Breast and gynaecological:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Breast and gynaecological:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Trisequens® contains
| Each red tablet contains: | |
| Active ingredient (main ingredient) | 1 mg estradiol hemihydrate |
| Other ingredients (inactive ingredients) | lactose monohydrate maize starch purified talc magnesium stearate hyprolose hypromellose titanium dioxide (E171) propylene glycol iron oxide red CI77491 (E172) |
| Potential allergens | lactose monohydrate |
| Each blue tablet contains: | |
| Active ingredient (main ingredient) | 2 mg estradiol hemihydrate |
| Other ingredients (inactive ingredients) | lactose monohydrate maize starch purified talc magnesium stearate hyprolose hypromellose titanium dioxide (E171) macrogol 400 indigo carmine CI73015 (E132) |
| Potential allergens | lactose monohydrate |
| Each white tablet contains: | |
| Active ingredient (main ingredient) | 2 mg estradiol hemihydrate 1 mg norethisterone acetate |
| Other ingredients (inactive ingredients) | lactose monohydrate maize starch purified talc magnesium stearate hyprolose hypromellose triacetin |
| Potential allergens | lactose monohydrate |
Do not take this medicine if you are allergic to any of these ingredients.
What Trisequens® looks like
Trisequens® comes in a calendar dial pack. Each pack holds 28 tablets.
Trisequens® calendar dial pack contains:
12 blue film-coated, biconvex tablets engraved with 'NOVO 280'.
10 white film-coated, biconvex tablets engraved with 'NOVO 281'.
6 red film-coated, biconvex tablets engraved with 'NOVO 282'.
AUST R 192476
Who distributes Trisequens®
Novo Nordisk Pharmaceuticals Pty Ltd
Level 10
118 Mount Street
North Sydney NSW 2060
Australia
Trisequens® is registered trademark of Novo Nordisk Health Care AG.
© 2025
Novo Nordisk A/S
Further information
For further information call Novo Nordisk Medical Information on 1800 668 626.
Always check the following websites to ensure you are reading the most recent version of the Consumer Medicine Information:
This leaflet was prepared in December 2025.
Brand Information
| Brand name | Trisequens |
| Active ingredient | Estradiol, Estradiol + Norethisterone acetate |
| Schedule | S4 |
MIMS Revision Date: 01 June 2024
The Women's Health Initiative (WHI) study reported increased risks of myocardial infarction, stroke, invasive breast cancer, pulmonary emboli, and deep vein thrombosis in postmenopausal women (50 to 79 years of age) during 5 years of treatment with conjugated estrogens (0.625 mg) combined with medroxyprogesterone acetate (2.5 mg) relative to placebo (see Section 5.1 Pharmacodynamic Properties, Clinical trials; Section 4.4 Special Warnings and Precautions for Use).
The WHI study reported increased risks of stroke and deep vein thrombosis in postmenopausal women (50 to 79 years of age) during 6.8 years of treatment with conjugated estrogens (0.625 mg) relative to placebo (see Section 5.1 Pharmacodynamic Properties, Clinical trials; Section 4.4 Special Warnings and Precautions for Use).
The Women's Health Initiative Memory Study (WHIMS), a substudy of WHI, reported increased risk of developing probable dementia in postmenopausal women 65 years of age or older during 4 to 5.2 years of treatment with conjugated estrogens, with or without medroxyprogesterone acetate, relative to placebo. It is unknown whether this finding applies to younger postmenopausal women (see Section 5.1 Pharmacodynamic Properties, Clinical trials; Section 4.4 Special Warnings and Precautions for Use).
Other doses of conjugated estrogens and medroxyprogesterone acetate, and other combinations and dosage forms of estrogens and progestagens were not studied in the WHI clinical trials and, in the absence of comparable data, these risks should be assumed to be similar. Because of these risks, estrogens with or without progestagens should be prescribed at the lowest effective doses and for the shortest duration consistent with treatment goals and risks for the individual woman.
1 Name of Medicine
Estradiol hemihydrate and norethisterone acetate.
2 Qualitative and Quantitative Composition
Trisequens is a sequential estrogen/ progestagen hormone replacement therapy (HRT) preparation formulated with the natural human estrogen, estradiol, and norethisterone acetate. The first 12 tablets in the calendar dial pack contain estradiol alone. In the next 10 tablets, norethisterone acetate is added to induce endometrial shedding. The last 6 tablets contain a low dose of estradiol. This regimen provides adequate estrogen replacement throughout the cycle and reduces the risk of endometrial hyperplasia. The composition of the tablets is shown in Table 1.

3 Pharmaceutical Form
Film coated tablets.
Blue film-coated, biconvex tablets engraved with 'NOVO 280'. Diameter: 6 mm.
White film-coated, biconvex tablets engraved with 'NOVO 281'. Diameter: 6 mm.
Red film-coated, biconvex tablets engraved with 'NOVO 282'. Diameter: 6 mm.
4 Clinical Particulars
4.1 Therapeutic Indications
Short-term symptomatic treatment of estrogen deficiency associated with natural or artificial menopause (see Section 4.2 Dose and Method of Administration; Section 5.1 Pharmacodynamic Properties, Clinical trials).
4.2 Dose and Method of Administration
Dosage. Trisequens is a continuous sequential HRT product. The estrogen is dosed continuously. The progestagen is added for 10 days of every 28 day cycle, in a sequential manner.
In menstruating women, treatment with Trisequens should be started on the fifth day of menstrual bleeding. All other patients may start at any time.
Regular withdrawal bleeding will be established, usually during the administration of the red low estrogen tablets, or possibly even during the last phase of the white tablets.
If symptoms such as hot flushes have ceased, consideration of transferring to local vaginal treatment should be given if troublesome local symptoms remain.
Duration of therapy. HRT should be prescribed at the lowest effective dose and for shortest duration (see Section 4.4 Special Warnings and Precautions for Use). The continuation of the treatment should be re-evaluated annually. Women who have undergone a premature menopause may require longer-term treatment.
Method of administration. Trisequens is administered orally one tablet daily without interruption. Treatment should begin with the administration of the blue tablets (2 mg estradiol) for 12 days, followed by the white tablets (2 mg estradiol, 1 mg norethisterone acetate) for 10 days, and then the red tablets (1 mg estradiol) for 6 days. When all the 28 tablets in the pack have been taken, another pack is started.
If the patient has forgotten to take a tablet, the tablet is to be discarded. Forgetting a dose may increase the likelihood of breakthrough bleeding and spotting.
Use of the calendar pack. The first tablet to be taken is under the sealed opening in the transparent outer rim of the pack. Turn the inner disc of the pack until the day of the week on which the first tablet is to be taken is opposite the sealed opening. The seal is then lifted off with a finger nail and the first tablet removed from the pack. Each day turn the transparent outer rim of the pack in the direction of the arrow to obtain the next tablet. Continue until all tablets have been taken.
4.3 Contraindications
Known, suspected or past history of carcinoma of the breast.
Known, suspected or past history of estrogen dependent neoplasia e.g. endometrial carcinoma.
Untreated endometrial hyperplasia.
Genital bleeding of unknown aetiology.
Previous or current venous thromboembolism (deep venous thrombosis, pulmonary embolism).
Known thrombophilic disorders (e.g. protein C, protein S, or antithrombin deficiency (see Section 4.4 Special Warnings and Precautions for Use)).
Active or previous arterial thromboembolic disease (e.g. angina, myocardial infarction).
Acute liver disease or a history of liver disease as long as liver function tests have failed to return to normal.
Porphyria.
Known hypersensitivity to the active ingredients or any of the excipients.
Known or suspected pregnancy.
4.4 Special Warnings and Precautions for Use
HRT should not be initiated or continued to prevent or treat cardiovascular disease.
The benefits and risks of HRT must always be carefully weighed, including consideration of the emergence of risks as therapy continues. A careful appraisal of the risks and benefits should be undertaken at least annually.
HRT should be used only in women with menopausal symptoms and ordinarily not for long-term use. HRT should be prescribed at the lowest effective doses and for the shortest duration consistent with the treatment goals and risks for the individual women.
Trisequens tablets contain lactose monohydrate. Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency, or glucose-galactose malabsorption should not take this medicine.
The toxicity profiles of estradiol and norethisterone acetate are well known. There are no preclinical data of relevance to the prescriber which are additional to that already included in other sections of the product information.
Medical examination/ follow-up. Before initiating or reinstituting HRT, a complete personal and family medical history should be taken. Physical (including pelvic and breast) examinations should be guided by this and by the contraindications and warnings for use.
There is a need for caution when prescribing estrogens in women with known or past history of breast nodules or fibrocystic disease or family history of breast cancer and in women with established coronary artery disease.
During treatment, periodic check-ups are recommended of a frequency and nature adapted to the individual woman including gynaecological examinations.
Women should be advised what changes in their breasts should be reported to their doctor or nurse (see Breast cancer below). Investigations including appropriate imaging tools e.g. mammography, should be carried out in accordance with currently accepted screening practices, modified to the clinical needs of the individual.
Conditions which need supervision. If any of the following conditions are present, have occurred previously, and/or have been aggravated during pregnancy or previous hormone treatment, the patient should be closely supervised. It should be taken into account that these conditions may recur or be aggravated during treatment with Trisequens, in particular:
cardiac failure;
leiomyoma (uterine fibroids) or endometriosis;
risk factors for thromboembolic disorders (see below);
risk factors for estrogen dependent tumours e.g. 1st degree heredity for breast cancer;
hypertension;
liver disorders (e.g. liver adenoma);
diabetes mellitus with or without vascular involvement;
cholelithiasis;
migraine or (severe) headache;
systemic lupus erythematosus;
a history of endometrial hyperplasia (see below);
epilepsy;
asthma;
otosclerosis.
Reasons for immediate withdrawal of therapy. Therapy should be discontinued in case a contraindication is discovered or in the following situations:
Jaundice or deterioration in liver function.
Significant increase in blood pressure.
New onset of migraine type headache.
Pregnancy.
Sudden visual disturbance.
Venous thromboembolism. HRT is associated with a 1.3 to 3-fold risk of developing venous thromboembolism (VTE), i.e. deep venous thrombosis or pulmonary embolism. The occurrence of such an event is more likely in the first year of HRT than later (see Section 4.8 Adverse Effects (Undesirable Effects)). The risk versus the benefits should therefore be carefully weighed in consultation with the individual woman when prescribing HRT to women with a risk factor for VTE.
Patients with known thrombophilic states have an increased risk of VTE, and HRT may add to this risk. HRT is therefore contraindicated in these patients (see Section 4.3 Contraindications). Generally recognised risk factors for VTE include a personal history, a family history (the occurrence of VTE in a direct relative at a relatively early age may indicate genetic predisposition), use of estrogens, major surgery, prolonged immobilisation, pregnancy/ postpartum period, systemic lupus erythematosus (SLE), obesity, and cancer. The risk of VTE also increases with age. There is no consensus about the possible role of varicose veins in VTE.
The risk of VTE may be temporarily increased with prolonged immobilisation, major elective or post-traumatic surgery or major trauma. Depending on the nature of the event and the duration of the immobilisation, consideration should be given to a temporary discontinuation of HRT. If feasible, estrogens should be discontinued at least 4 to 6 weeks before surgery of the type associated with an increased risk of thromboembolism, or during periods of prolonged immobilisation. Treatment should not be restarted until the woman is completely mobilised.
In women with no personal history of VTE but with a first degree relative with a history of VTE at a young age, screening may be offered after careful counselling regarding its limitations (only a proportion of thrombophilic defects are identified by screening).
If a thrombophilic defect is identified which segregates with thrombosis in family members, or if the defect is severe (e.g. antithrombin, protein S or protein C deficiencies, or a combination of defects), HRT is contraindicated.
Women already on chronic anticoagulant treatment require careful consideration of the benefit/ risk of using HRT.
If VTE develops after initiating therapy, the drug should be discontinued. Patients should be told to contact their doctors immediately when they are aware of a potential thromboembolic symptom (e.g. painful swelling of a leg, sudden pain in the chest, dyspnoea).
Scarabin and others(4) reported the results of a case control study conducted during 1999-2002 in France. The investigators recruited 155 consecutive cases with a first documented episode of VTE of unknown cause (92 with pulmonary embolisms and 63 with deep venous thrombosis) and 381 controls (women admitted to hospital for other reasons) matched for centre, age and time of recruitment. Overall, 32 (21%) cases and 27 (7%) controls were current users of oral estrogen replacement therapy (this was defined in this study as estrogen only therapy or combined HRT), whereas 30 (19%) cases and 93 (24%) controls were current users of transdermal estrogen replacement therapy. After adjustment for potential confounding variables, the odds ratio for VTE in current users of oral and transdermal estrogen replacement therapy compared with nonusers was 3.5 (95% CI 1.8-6.8) and 0.9 (0.5-1.6), respectively. Estimated risk for VTE in current users of oral estrogen replacement therapy compared with transdermal estrogen replacement therapy users was 4.0 (1.9-8.3). These results may be interpreted as meaning that (i) the higher risk of VTE as shown in the WHI study has been further supported; and (ii) current use but not past use was a risk factor for VTE. Use in the first year was also more risky than later use, a finding that is also consistent with the WHI study.
Coronary artery disease (CAD). There is no evidence from randomised controlled trials of cardiovascular benefit with continuous combined conjugated estrogens and medroxyprogesterone acetate (MPA). Two large clinical trials (WHI and HERS i.e. heart and estrogen/ progestin replacement study) showed a possible increased risk of cardiovascular morbidity in the first year of use and no overall benefit. For other HRT products there are only limited data from randomised controlled trials examining effects in cardiovascular morbidity or mortality. Therefore, it is uncertain whether these findings also extend to other HRT products.
Stroke. One large randomised clinical trial (WHI trial) found, as a secondary outcome, an increased risk of ischaemic stroke in healthy women during treatment with continuous combined conjugated estrogens and MPA. For women who do not use HRT, it is estimated that the number of cases of stroke that will occur over a 5 year period is about 3 per 1000 women aged 50-59 years and 11 per 1000 women aged 60-69 years. It is estimated that for women who use conjugated estrogens and MPA for 5 years, the number of additional cases will be between 0 and 3 (best estimate = 1) per 1000 users aged 50-59 years and between 1 and 9 (best estimate = 4) per 1000 users aged 60-69 years. It is unknown whether the increased risk also extends to other HRT products.
Endometrial hyperplasia and carcinoma. There is some evidence that obesity and possibly hypertension or diabetes mellitus, are predisposing factors to endometrial carcinoma. Patients with these conditions and those with a family history of endometrial carcinoma should be monitored closely.
The risk of endometrial hyperplasia and carcinoma is increased when estrogens are administered alone for prolonged periods. The reported increase in endometrial cancer risk among estrogen only users varies from 2 to 12-fold compared with nonusers, depending on the duration of treatment and estrogen dose (see Section 4.8 Adverse Effects (Undesirable Effects)). After stopping treatment, the risk may remain elevated for more than 10 years.
The addition of a progestagen for at least 10 days per cycle in nonhysterectomised women greatly reduces this risk. Patients who are, or have previously been, treated with unopposed estrogens should be examined with special care to exclude endometrial stimulation before commencing Trisequens therapy.
Breakthrough bleeding and spotting may occur during the first months of treatment. If breakthrough bleeding or spotting appears after some time on therapy, or continues after treatment has been discontinued, the reason should be investigated, which may include endometrial biopsy to exclude endometrial malignancy.
Breast cancer. The use of estrogen-only and combined estrogen-progestagen by women has been shown to increase the risk of breast cancer, that is dependent on the duration of taking HRT.
Combined estrogen-progestagen therapy. The randomised placebo-controlled trial, the Women's Health Initiative study (WHI), and a meta-analysis of prospective epidemiological studies are consistent in finding an increased risk of breast cancer in women taking combined estrogen-progestogen for HRT that becomes apparent after about 3 (1-4) years (see Section 4.8 Adverse Effects (Undesirable Effects)).
Estrogen only therapy. The WHI trial found no increase in the risk of breast cancer in hysterectomised women using estrogen-only HRT. Observational studies have mostly reported a small increase in risk of having breast cancer diagnosed that is lower than that found in users of estrogen-progestagen combinations (see Section 4.8 Adverse Effects (Undesirable Effects)).
Results from a large meta-analysis showed that after stopping treatment, the excess risk will decrease with time and the time needed to return to baseline depends on the duration of prior HRT use. When HRT was taken for more than 5 years, the risk may persist for 10 years or more.
Women using estrogen/ progestagen combined HRT had a higher risk as compared with women who used estrogens alone. Although obese women are at an increased risk of having breast cancer, HRT did not further increase this risk.
A randomised clinical trial (WHI study) with conjugated estrogens and MPA showed that the risk of breast cancer increases with the number of years of HRT usage and gets significant in year 5 of HRT use. The absolute increase in risk is small to moderate (see Section 5.1 Pharmacodynamic Properties, Clinical trials). The Million Women Study demonstrated that this risk increase is similar for different estrogen/ progestagen combinations, different regimens and routes of hormone administration. Whether the low dose HRT has a different effect on breast cancer risk is still unclear.
In the WHI study, the continuous combined conjugated equine estrogen and medroxyprogesterone acetate (CEE + MPA) product used was associated with breast cancers that were slightly larger in size and more frequently had local lymph node metastases compared to placebo.
All women should receive yearly breast examinations by a healthcare provider and perform monthly breast self examinations. In addition, mammography examinations should be scheduled based on patient age, risk factors, and prior mammogram results. The use of estrogen plus progestagen has been reported to result in an increase in abnormal mammograms requiring further evaluation. Mammographic density may be increased after the use of combined HRT. This may have implications for the sensitivity and specificity of breast cancer screening. Regular breast examination and, where appropriate, mammography should be carried out in women on HRT. Breast status should also be closely monitored in women with a history of or known breast nodules, fibrocystic disease, or with a family history of breast cancer.
Combination HRT should not be used in hysterectomised women because it is not needed in these women and it may increase the risk of breast cancer.
Ovarian cancer. Ovarian cancer is much rarer than breast cancer.
Epidemiological evidence from a large meta-analysis suggests a slightly increased risk in women taking estrogen only or combined estrogen/ progestagen HRT, which becomes apparent within 5 years of use and diminishes over time after stopping. Some other studies, including the WHI trial, suggest that use of combined HRTs may be associated with a similar or slightly smaller risk (see Section 4.8 Adverse Effects (Undesirable Effects)).
Dementia. See Section 5.1 Pharmacodynamic Properties, Clinical trials, WHIMS substudy. HRT use does not improve cognitive function.
Other conditions. It is advisable to withdraw treatment 4 to 6 weeks before major surgery.
Pre-existing uterine fibromyoma may increase in size with estrogens. If this is observed, estrogen therapy should be discontinued.
Trisequens has no contraceptive effect.
The risks and benefits in younger women receiving treatment for the short-term management of menopausal symptoms of estrogen deficiency or for the management of premature menopause were not examined in the WHI study. As well, the study did not include other formulations or dosage regimens, such as Novo Nordisk's products containing 17-beta-estradiol and norethisterone acetate, or other routes of administration of HRT.
In the absence of data specific to this product, if prescribing any form of hormone replacement therapy as primary prevention of osteoporosis, the potential for increased cardiovascular, thrombotic and neoplastic adverse events must be considered. Combined hormone replacement therapy should not be used for the long-term maintenance of general health, including the primary prevention of cardiovascular disease. Estrogen or estrogenic compounds must not be used alone as estrogen replacement therapy in women who have not had a hysterectomy.
Estrogens may cause fluid retention, and therefore patients with cardiac or renal dysfunction should be carefully observed. Patients with terminal renal insufficiency should be closely observed, since it is expected that the level of circulating active ingredients in Trisequens will increase.
Women with pre-existing hypertriglyceridemia should be followed closely during estrogen replacement or hormone replacement therapy, since rare cases of large increases of plasma triglycerides leading to pancreatitis have been reported with estrogen therapy in this condition.
Patients who require thyroid hormone replacement therapy should have their thyroid function monitored regularly while on HRT to ensure that thyroid hormone levels remain in an acceptable range. Estrogens increase thyroid binding globulin (TBG), leading to increased circulating total thyroid hormone, as measured by protein bound iodine (PBI), T4 levels (by column or by radioimmunoassay) or T3 levels (by radioimmunoassay). T3 resin uptake is decreased, reflecting the elevated TBG. Free T4 and free T3 concentrations are unaltered. Other binding proteins may be elevated in serum, i.e. corticoid binding globulin (CBG), sex hormone binding globulin (SHBG) leading to increased circulating corticosteroids and sex steroids, respectively. Free or biological active hormone concentrations are unchanged. Other plasma proteins may be increased (angiotensinogen/ renin substrate, alpha-I-antitrypsin, ceruloplasmin).
Estrogens may induce or exacerbate symptoms of angioedema, in particular in women with hereditary angioedema.
Use in elderly. The experience of treating women older than 65 years is limited. Pharmacokinetics in the elderly have not been studied.
Of the total number of subjects in the conjugated equine estrogens in combination with medroxyprogesterone acetate substudy of the Women's Health Initiative study(1), 44% (n = 7,320) were 65 years and over, while 6.6% (n = 1,095) were 75 and over. No significant differences in overall safety were observed between subjects 65 years and over compared to younger subjects. There was a higher incidence of stroke and invasive breast cancer in women 75 and over compared to younger subjects.
In the Women's Health Initiative Memory study, including 4,532 women 65 years of age and older, followed up for an average of 4 years, 82% were 65-74 (3,726) while 18% (806) were 75 and over. Most women (80%) had no prior HRT use. Women treated with 0.625 mg conjugated estrogens plus 2.5 mg medroxyprogesterone acetate were reported to have a twofold increase in the risk of developing probable dementia. Ninety percent of cases of probable dementia occurred in the 54% of women that were older than 70.
Paediatric use. No data available.
Effects on laboratory tests. Some laboratory tests may be influenced by estrogen therapy, such as tests for glucose tolerance or thyroid function.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The metabolism of estrogens and progestagens may be increased by concomitant use of substances known to induce drug metabolising enzymes, specifically cytochrome P450 enzymes such as barbiturates (e.g. phenobarbital), anticonvulsants (e.g. phenytoin, carbamazepine), antihistamines, phenylbutazone and some anti-infectives (e.g. rifampicin, rifabutin, nevirapine, efavirenz, telaprevir). Ritonavir, telaprevir and nelfinavir, although known as strong inhibitors, by contrast exhibit inducing properties when used concomitantly with steroid hormones. Herbal preparations containing St. John's wort (Hypericum perforatum) may induce the metabolism of estrogens and progestagens.
Clinically, an increased metabolism of estrogens and progestagens may lead to decreased effect and changes in the uterine bleeding profile.
Reduced estradiol levels have been observed under the simultaneous use of antibiotics e.g. penicillins and tetracycline.
Drugs that inhibit the activity of hepatic microsomal drug metabolising enzymes e.g. ketoconazole, may increase circulating levels of the active substances in Trisequens.
Estrogens can also affect the actions of other drugs, e.g. anticoagulants, antidiabetic agents, antifibrinolytic agents, pethidine, drugs which decrease serum folate, imipramine, thyroid hormones and corticosteroids.
Oral contraceptives (OC) containing ethinylestradiol have been shown to significantly decrease plasma concentrations of lamotrigine when coadministered. Similar interaction may exist between HRT containing estradiol and lamotrigine. Therefore, dosage adjustment of lamotrigine may be necessary for seizure control.
Concomitant administration of cyclosporin may cause increased blood levels of cyclosporin, creatinine and transaminases due to decreased metabolism of cyclosporin in the liver.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category D)
Contraindicated. If pregnancy occurs during medication with Trisequens, treatment should be withdrawn immediately.
In animal studies, maternal administration of high doses of estrogens has produced urogenital malformations in the offspring. The relevance of these animal findings for the clinical use of estradiol is uncertain, but is considered likely to be low. Animal studies have also shown that high doses of progestagens can cause masculinisation of the female foetus.
Clinically, data on a limited number of pregnancies indicate adverse effects of norethisterone on the fetus. At doses higher than normally used in OC and HRT formulations, masculinisation of female fetuses was observed.
The results of most epidemiological studies to date relevant to inadvertent fetal exposure to combinations of estrogens and progestagens indicate no teratogenic or fetotoxic effect.
Use in lactation. Trisequens is not indicated during lactation.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
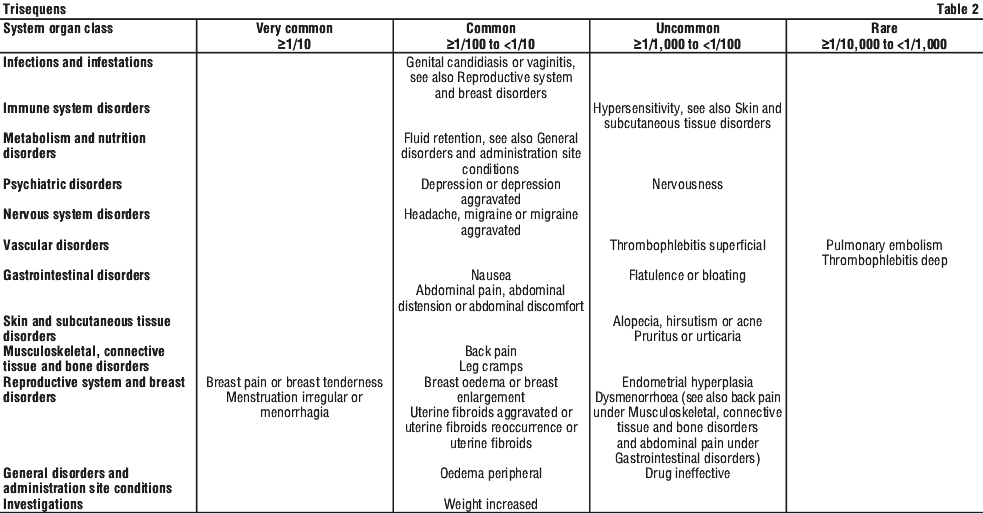
Clinical experience. The most frequently reported adverse events in the clinical trials with Trisequens were vaginal bleeding and breast pain/ tenderness, reported in approximately 10% to 20% of patients. Vaginal bleeding usually occurred in the first months of treatment. Breast pain usually disappeared after a few months of therapy. All adverse events observed in the randomised clinical trials with a higher frequency in patients treated with Trisequens or similar HRT products as compared to placebo and which on an overall judgement are possibly related to treatment are presented in Table 2. Also see Section 5.1 Pharmacodynamic Properties, Clinical trials; Section 4.4 Special Warnings and Precautions for Use.
Postmarketing experience. In addition to the above mentioned adverse drug reactions, those presented below have been spontaneously reported, and are by an overall judgment considered possibly related to Trisequens treatment. The reporting rate of these spontaneous adverse drug reactions is very rare (< 1/10,000, not known (cannot be estimated from the available data)). Postmarketing experience is subject to under reporting especially with regard to trivial and well known adverse drug reactions. The presented frequencies should be interpreted in that light.
Neoplasms benign and malignant (including cysts and polyps). Endometrial cancer.
Immune system disorders. Generalised hypersensitivity reactions (e.g. anaphylactic reaction/ shock).
Psychiatric disorders. Insomnia, anxiety, libido decreased, libido increased.
Nervous system disorders. Dizziness, stroke.
Eye disorders. Visual disturbances.
Cardiac disorders. Myocardial infarction.
Vascular disorders. Hypertension aggravated.
Gastrointestinal disorders. Dyspepsia, vomiting.
Hepatobiliary disorders. Gall bladder disease, cholelithiasis, cholelithiasis aggravated, cholelithiasis recurrence.
Skin and subcutaneous tissue disorders. Seborrhoea, rash, angioedema.
Reproductive system and breast disorders. Endometrial hyperplasia, vulvovaginal pruritus.
Investigations. Weight decreased, blood pressure increased.
The following adverse reactions have been reported in association with estrogen/ progestagen treatment:
Skin and subcutaneous disorders: alopecia, chloasma, erythema multiforme, erythema nodosum, vascular purpura.
Probable dementia (see Section 4.4 Special Warnings and Precautions for Use).
Gastrointestinal disorders: Crohn's disease, ulcerative colitis.
Dry eyes.
Tear film composition changes.
Breast cancer risk. An up to 2-fold increased risk of having breast cancer diagnosed is reported in women taking combined estrogen/ progestagen therapy for more than 5 years. The increased risk in users of estrogen only therapy is lower than that seen in users of estrogen/ progestagen combinations. The level of risk is dependent on the duration of use (see Section 4.4 Special Warnings and Precautions for Use).
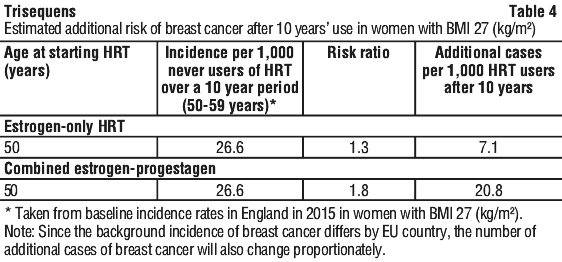
Absolute risk estimations based on results of the largest randomised placebo controlled trial (WHI study) and the largest meta-analysis of prospective epidemiological studies are presented in Tables 3, 4 and 5.


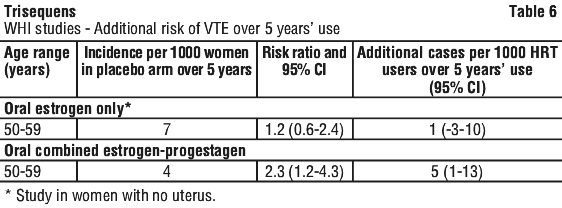
Risk of venous thromboembolism. HRT is associated with a 1.3 to 3-fold increased relative risk of developing venous thromboembolism (VTE), i.e. deep vein thrombosis or pulmonary embolism. The occurrence of such an event is more likely in the first year of using HRT (see Section 4.4 Special Warnings and Precautions for Use). Results of the WHI studies are presented in Table 6.
Risk of ischaemic stroke. The use of estrogen only and estrogen/ progestagen therapy is associated with an up to 1.5-fold increased relative risk of ischaemic stroke. This relative risk is not dependent on age or on duration of use, but as the baseline risk is strongly age dependent, the overall risk of stroke in women who use HRT will increase with age (see Section 4.4 Special Warnings and Precautions for Use). See Table 7.

4.9 Overdose
Overdosage may cause nausea and vomiting, headache, dizziness, endometrial proliferation, sodium and water retention, and breast enlargement. There are no specific recommendations for the management of overdosage.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The pharmacological actions of exogenous estradiol are similar to the physiological effects of the endogenous hormone. Norethisterone acetate given orally has progestational actions similar to those of progesterone and, in addition, has weak estrogenic and androgenic properties.
Trisequens restores plasma estrogen levels and thus relieves or decreases subjective estrogen deficiency symptoms. Trisequens therapy has been demonstrated to suppress gonadotrophin secretion (FSH/LH) and to improve vaginal cytology in peri- and postmenopausal women without adversely affecting the serum lipid profile or inducing liver protein synthesis (e.g. various clotting factors).
A regular withdrawal bleeding is established with Trisequens. The addition of norethisterone acetate in the second series of (white) tablets induces a change in the endometrium to a secretory phase. Endometrial shedding usually occurs during the third series of (red) tablets. The low estradiol content in the last series of tablets prevents the reappearance of menopausal symptoms without affecting withdrawal bleeding.
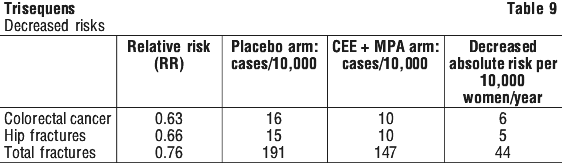
Clinical trials. WHI study(1). In a prospective randomised trial (Women's Health Initiative, WHI) involving 8506 postmenopausal women who received oral hormone replacement therapy (HRT) using a continuous combined regimen of conjugated equine estrogens 0.625 mg/day plus medroxyprogesterone acetate 2.5 mg/day and 8102 women who received placebo for an average of 5.2 years, adverse effects on cardiovascular disease and breast cancer, and beneficial effects on hip and total fractures and colorectal cancer were observed. These results do not necessarily apply to lower dosages of these drugs, to other formulations of oral estrogens and progestins or to estrogen monotherapy.
The WHI study was designed to investigate the efficacy and safety of long-term HRT in preventing coronary heart disease (CHD) in healthy postmenopausal women with an intact uterus. A global index summarising the balance of risks and benefits included an analysis of the 2 primary outcomes, invasive breast cancer and CHD, and the following secondary outcomes: stroke, pulmonary embolism (PE), endometrial cancer, colorectal cancer, hip fracture and death due to other causes. The women enrolled in the study had a mean age at entry of 63.3 years. On average they were overweight (mean body mass index (BMI) = 28.5) and one-third were obese (BMI ≥ 30), 50% were previous or current smokers, one-third had received treatment for high blood pressure and over 10% had raised cholesterol levels requiring medication.
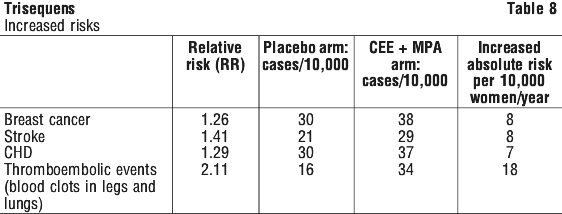
After a mean of 5.2 years of follow-up, the study was prematurely stopped because the risk/ benefit profile was not consistent with the requirements for a viable intervention for primary prevention of chronic diseases. (See Tables 8 and 9.)
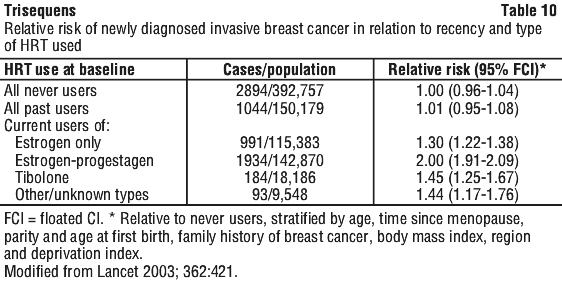
In terms of absolute risk, after ten years use of HRT, it is estimated that there would be 5 (95% CI 3-7) additional breast cancers per 1,000 users of estrogen only preparations and 19 (95% CI 15-23) additional cancers per 1,000 users of estrogen/ progestagen combinations. The elevated risk reduces after discontinuation of hormone replacement therapy and is effectively lost after 5 years.
In the combined HRT subset of WHI, a 26% increase of invasive breast cancer (38 versus 30 per 10,000 person years) after an average of 5.2 years treatment was observed in women receiving the estrogen/ progestagen combination compared to women receiving placebo. The increased risk of breast cancer became apparent after 4 years on study medication. Women reporting prior postmenopausal hormone use had a higher relative risk for breast cancer associated with HRT than those who never used postmenopausal hormones.
WHIMS substudy(3). In a study of women 65 years of age and older (a randomised controlled substudy of the Women's Health Initiative, the Women's Health Initiative Memory Study; n = 4532, 54% older than 70), those treated with 0.625 mg conjugated equine estrogens plus 2.5 mg medroxyprogesterone acetate were reported to have a twofold increase in the risk of developing probable dementia. After an average follow-up of 4 years, the absolute risk of probable dementia was 45 per 10,000 women years in the estrogen plus progestagen group and 22 per 10,000 women years in the placebo group. It is unknown whether these findings apply to younger postmenopausal women. It is unlikely that HRT would be indicated in this age group.
5.2 Pharmacokinetic Properties
Micronised estradiol is rapidly and efficiently absorbed from the gastrointestinal tract following oral administration. Some estradiol is converted to estrone in the intestinal mucosa before absorption into the portal vein. During passage through the liver, a significant proportion of estradiol is metabolised to estrone. Estriol and hydroxyoestrones are also produced, as well as sulfate and glucuronate conjugates. Circulating estrone sulfate may be reconverted to estrone and estradiol in extrahepatic organs like the uterus. Estrogens are partly bound to plasma proteins, including sex hormone binding globulin. Estrogens are excreted into the bile and undergo significant enterohepatic cycling. Conjugates of the various estrogens and their metabolites are excreted in the urine and unconjugated estrogen metabolites appear in the faeces. Estrogens are also secreted in the milk of nursing mothers.
Norethisterone acetate is rapidly absorbed and transformed to norethisterone, then metabolised and excreted as glucuronide and sulfate conjugates. About half the dose is recovered in the urine within 24 hours, the remainder being reduced to less than 1% of the dose within 5-6 days. The terminal plasma half-life is about 10 hours.
Peak plasma concentrations of estradiol occur 2-4 h after tablet ingestion. Thereafter elimination is slow and estradiol levels are maintained above baseline for 24 h. Peak plasma concentrations of norethisterone occur 1-2 h after tablet ingestion. Norethisterone concentrations return to basal levels within 24 h.
5.3 Preclinical Safety Data
Genotoxicity. There is limited evidence available in the literature suggesting that estradiol may be weakly genotoxic. No evidence could be found for an increase in the rate of gene mutation in bacterial or mammalian cells, but there was some evidence for the induction of chromosomal aberrations and aneuploidy and an increased incidence of sister chromatid exchanges (indicative of DNA damage) in mammalian cells. None of these effects were induced by estradiol in human lymphocyte cultures. Importantly, there was no evidence of clastogenicity in rodent bone marrow micronucleus assays.
Carcinogenicity. Supraphysiological doses of estradiol have been associated with the induction of tumours in estrogen dependent target organs in all rodent species tested. The relevance of these findings with respect to humans has not been established.
6 Pharmaceutical Particulars
6.1 List of Excipients
The tablet cores of the blue, white and red tablets contain: lactose monohydrate, maize starch, hyprolose, purified talc, magnesium stearate.
Film-coatings. Blue tablets: hypromellose, purified talc, titanium dioxide (E171), indigo carmine (E132) and macrogol 400.
White tablets: hypromellose, triacetin and purified talc.
Red tablets: hypromellose, purified talc, titanium dioxide (E171), red iron oxide (E172), and propylene glycol.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Keep all medicines out of reach of children. Trisequens should be kept in a cool dry place where the temperature stays below 25°C. Do not store Trisequens in the refrigerator. Keep the container in the outer carton in order to protect it from light.
6.5 Nature and Contents of Container
Trisequens is supplied in a calendar dial pack consisting of a polypropylene base and polystyrene lid containing 28 tablets (12 blue tablets, 10 white tablets and 6 red tablets). See Section 2 Qualitative and Quantitative Composition.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
Estradiol is a white or almost white crystalline powder which is practically insoluble in water and soluble in acetone.
Norethisterone acetate (NETA) is a white or yellowish-white crystalline powder which is practically insoluble in water and soluble in ethanol and acetone.
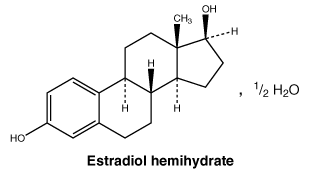
Chemical structure. Estradiol - chemical name: estra-1,3,5(10)-triene-3,17β-diol (as hemihydrate). Estradiol has 5 chiral centres. The molecular formula is C18H24O2. Estradiol hemihydrate has a molecular weight of 281.39.
Norethisterone acetate - chemical name: 17β-acetoxy-19-nor-17α-pregn-4-en-20-yn-3-one. Norethisterone acetate has 6 chiral centres. The molecular formula is C22H28O3, with a molecular weight of 340.5.
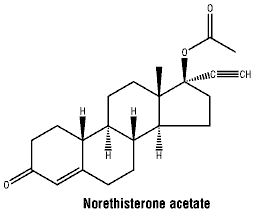
Norethisterone acetate: 51-98-9.
7 Medicine Schedule (Poisons Standard)
S4 Prescription Only Medicine.
Date of First Approval
11 January 2012
Date of Revision
08 January 2024
Summary Table of Changes

References
(1) Writing Group for the Women's Health Initiative. JAMA 2002; 288:321-333.
(2) Million Women Study Collaborators. Lancet 2003; 362:419-427.
(3) Shumaker SA, Legault C, et al. JAMA 2003; 289:2651-2662.
(4) Scarabin P-Y, Oger E, et al. Lancet 2003; 362:428-432.
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.