Ursosan
Brand Information
| Brand name | Ursosan |
| Active ingredient | Ursodeoxycholic acid |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Ursosan.
Summary CMI
Ursosan®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Ursosan?
Ursosan contains active ursodeoxycholic acid. Ursosan may be used to treat liver diseases such as primary biliary cirrhosis (PBC), primary sclerosing cholangitis (PSC), and cystic fibrosis (CF)-related cholestasis. For more information, see Section 1. Why am I using Ursosan? in the full CMI.
2. What should I know before I use Ursosan?
Do not use if you have ever had an allergic reaction to ursodeoxycholic acid or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Ursosan? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Ursosan and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Ursosan?
The dose for Ursosan is determined by your body weight. Your doctor will tell you how much Ursosan you should take.
More instructions can be found in Section 4. How do I use Ursosan? in the full CMI.
5. What should I know while using Ursosan?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Ursosan? in the full CMI.
6. Are there any side effects?
Ursodeoxycholic acid is generally well tolerated with few side effects. The common side effect is diarrhoea. Allergic reactions have been reported in some patients. Other adverse reactions reported include increased cholestasis, nausea, vomiting and sleep disturbance. The serious side effects include severe right-sided upper abdominal pain and severe worsening (decompensation) of liver cirrhosis.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Ursosan®
Active ingredient(s): ursodeoxycholic acid
Consumer Medicine Information (CMI)
This leaflet provides important information about using Ursosan.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Ursosan.
Where to find information in this leaflet:
1. Why am I using Ursosan?
2. What should I know before I use Ursosan?
3. What if I am taking other medicines?
4. How do I use Ursosan?
5. What should I know while using Ursosan?
6. Are there any side effects?
7. Product details
1. Why am I using Ursosan?
Ursosan contains ursodeoxycholic acid.
Ursodeoxycholic acid is a bile acid, which may have a protective effect on the liver by reducing the absorption of other potentially toxic bile salts.
Ursosan may be used to treat liver diseases such as primary biliary cirrhosis (PBC), primary sclerosing cholangitis (PSC), and cystic fibrosis (CF)-related cholestasis.
Your doctor may have prescribed this medicine for another reason.
Ask your doctor if you have any questions about why it has been prescribed for you.
Ursosan is only available with a doctor's prescription.
Ursosan is not addictive.
2. What should I know before I use Ursosan?
Warnings
Do not use Ursosan if:
- you are allergic to any medicine containing ursodeoxycholic acid, bile acids or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- you have a bile duct or gall bladder that is swollen, painful or blocked.
- you have certain types of cholesterol stones or bile pigment stones
- your doctor advises you that you will have your gall bladder removed.
- Do not take it after the expiry date printed on the pack.
Check with your doctor if you:
- take any medicines for any other condition.
- have allergies to any other medicines, foods, preservatives or dyes.
- kidneys that do not work properly
- a deficiency in a certain step of metabolism (sulfation)
- calcified gallstones
- frequent cramp-like pains in the upper abdomen (biliary colic).
- have a gall bladder that cannot be seen on X-ray.
- have a gall bladder which is not able to contract properly.
If you are not sure whether you or your child should start taking this medicine, talk to your doctor.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor can discuss with you the risks and benefits involved.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Ursosan may increase the effect of some medicines, including:
- medicines used to lower high levels of cholesterol in the blood, such as rosuvastatin, atorvastatin, fluvastatin, simvastatin, pitavastatin or pravastatin.
Ursosan may decrease the effects / absorption of some medicines, including:
- nitrendipine (used to treat high blood pressure) and other medicines which are metabolised in the same way.
- medicines used to suppress the immune system, such as ciclosporin;
- antibiotics used to prevent certain infections, such as ciprofloxacin or dapsone.
Medicines that may reduce the effect of Ursosan include:
- medicines used to lower high levels of cholesterol in the blood, such as colestyramine or colestipol; an absorbent, such as charcoal;
- medicines such as antacids containing aluminium hydroxide and/or smectite, which are used for indigestion.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Ursosan.
4. How do I use Ursosan?
How much to take
- The dose for Ursosan is determined by your body weight.
- Your doctor will tell you how much Ursosan you should take.
- The standard adult dose, depending on your weight and the condition being treated, is between two to nine capsules per day.
- Ask your doctor or pharmacist if you are unsure of the correct dose for you.
- Swallow Ursosan whole with a full glass of water, because the content of the capsules is bitter.
- If you take the wrong dose, Ursosan may not work as well, and your problem may not improve.
When to take Ursosan
- Ursosan should be taken regularly and at about the same time(s) each day. Taking it at the same time(s) each day will have the best effect. It will also help you to remember when to take it.
- If you need to take cholesterol lowering medicines or an antacid, take it at least two hours before or two hours after the dose of Ursosan.
- Ursosan capsules should be swallowed whole with a full glass of water because the content is bitter.
- Take Ursosan capsules regularly.
How long to take Ursosan
- Continue taking the capsules for as long as your doctor tells you to.
- Ursosan helps to control your condition but does not cure it. It is important to keep taking the medicine even if you feel well. You may need to take Ursosan for many months for it to work.
- If you are unsure whether you should stop taking Ursosan, talk to your doctor or pharmacist.
If you forget to take Ursosan
Ursosan should be used regularly at the same time each day. If you miss your dose at the usual time, take it as soon as you remember, and then go back to taking it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for hints.
If you take too much Ursosan
If you think that you have used too much Ursosan, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Symptoms of an overdose may include diarrhoea. If you suffer from diarrhoea, make sure you drink enough liquids to replace the fluid and electrolyte balance.
5. What should I know while using Ursosan?
Things you should do
If you are about to be started on any new medicine, tell your doctor and pharmacist that you are taking Ursosan. Likewise, tell any other doctors, dentists and pharmacists who are treating you that you are taking this medicine.
If you are going to have surgery, tell the surgeon and anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you are about to have any blood tests, tell your doctor that you are taking Ursosan.
Keep all of your doctor's appointments so that your progress can be checked.
Your doctor may perform liver function tests. During the first three months of taking Ursosan, your doctor should monitor your liver function every four weeks. After the first three months of taking this medicine, your doctor should monitor your liver function every three months.
See your doctor if you feel that your condition is not improving or it is getting worse.
Call your doctor straight away if you:
- become pregnant while taking this medicine.
Remind any doctor, dentist you visit that you are using Ursosan.
Things you should not do
- Do not give your medicine to anyone else, even if they have the same condition as you. This medicine is only intended for the person it has been prescribed for.
- Do not take Ursosan to treat any other complaints unless your doctor tells you to.
- Do not stop taking Ursosan or change the dosage without checking with your doctor.
Driving or using machines
Ursosan does not cause any negative effect on driving ability and operating machinery.
Drinking alcohol
Tell your doctor if you drink alcohol.
Looking after your medicine
Keep your capsules in the pack until it is time to take them. If you take the capsules out of the pack they may not keep well.
Keep Ursosan in a cool dry place where the temperature stays below 25°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; do not store it:
- in the bathroom or near a sink, or
- in the car or on windowsills.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice this serious side effect (During treatment of PBC). |
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice these serious side effects. |
| Severe worsening (decompensation) of liver cirrhosis. | Stop taking Ursosan immediately if you notice this serious side effect (During treatment of PBC). |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Ursosan contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
| Potential allergens | Ursosan does not contain lactose, sucrose, tartrazine or any other azo dyes. |
Do not take this medicine if you are allergic to any of these ingredients.
What Ursosan looks like
Ursosan is available in blister packs of 100 capsules.
The capsules are white in colour.
Australian Registration Number: AUST R 218444
Who distributes Ursosan
Boucher & Muir Pty Ltd
Level 9, 76 Berry Street
North Sydney NSW 2060
This leaflet was prepared in Aug 2025.
Brand Information
| Brand name | Ursosan |
| Active ingredient | Ursodeoxycholic acid |
| Schedule | S4 |
MIMS Revision Date: 01 September 2019
1 Name of Medicine
Ursodeoxycholic acid.
2 Qualitative and Quantitative Composition
One capsule contains ursodeoxycholic acid (UDCA) 250 mg.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Ursosan capsules are white, hard gelatin capsules.
4 Clinical Particulars
4.1 Therapeutic Indications
Ursosan is indicated in the treatment of chronic cholestatic liver diseases.
4.2 Dose and Method of Administration
For patients under 34 kg or patients who are unable to swallow Ursosan capsules an oral suspension 250 mg/5 mL is available in another brand.
Dosage for adults and the elderly. For PBC and chronic cholestatic liver diseases other than CF and PSC, the dosage of 12-16 mg/kg body weight/day of UDCA is recommended.
For CF-related cholestasis, the recommended dose is 20 mg/kg/day of UDCA.
For PSC, the dosage of 10-15 mg/kg body weight/day of UDCA is recommended. A dosage of 20 mg/kg body weight/day has also been shown to improve histology and liver function tests in PSC patients.
Dosage for children. Data on use in children are very limited. In the few available studies, dosages used have generally been up to 15-20 mg/kg/day.
Administration. For PBC patients: In the first 3 months of treatment, Ursosan capsules should be taken in 2 to 3 doses over the day. With improvement of the liver function parameters, the daily dose may be taken as a single dose in the evening.
For other cholestatic liver diseases, Ursosan capsules should be taken in 2 to 3 doses over the day.
The capsules should be swallowed whole with some liquid.
Care should be taken to ensure that Ursosan is taken regularly.
In patients with PBC, there may, in rare cases, be an initial deterioration in symptoms, e.g. itching. If this is the case, therapy can be continued with 1 capsule of Ursosan daily, and the daily dose gradually increased weekly until the recommended daily dose has been reached.
For PSC patients, dominant stenoses of the bile ducts should be dilated before and during treatment with Ursosan.
4.3 Contraindications
Ursosan must not be used if there is hypersensitivity to the active ingredient or any of the excipients.
4.4 Special Warnings and Precautions for Use
During the first 3 months of therapy, it is advisable to monitor the liver parameters of AST (serum glutamic oxaloacetic transaminase (SGOT)), ALT (serum glutamic pyruvic transaminase (SGPT)), and GGT every 4 weeks, subsequently every 3 months. Apart from allowing for identification of responders and non-responders in patients being treated for PBC, this monitoring would also enable early detection of potential hepatic deterioration, particularly in patients with advance stage PBC.
The effect of UDCA in patients with renal impairment has not been studied.
Ursosan is not recommended in patients with dominant stenoses of the bile ducts unless the obstructed bile ducts are dilated (see Section 4.2 Dose and Method of Administration).
If diarrhoea occurs, the dose must be reduced and in cases of persistent diarrhoea, the therapy should be discontinued.
Treatment of patients with primary sclerosing cholangitis. Long periods of UDCA therapy (28 - 30 mg/kg) in patients with PSC may be associated with a higher rate of serious adverse events.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Some drugs, such as colestyramine, charcoal, colestipol and certain antacids (containing aluminium hydroxide and/or smectite (aluminium oxide)) bind to UDCA in the intestine and thereby inhibit its absorption and efficacy. Should the use of a preparation containing one of these substances be necessary, it must be taken at least 2 hours before or after Ursosan.
UDCA may increase the absorption of ciclosporin in transplantation and non-transplant patients. Therefore, monitoring of ciclosporin plasma concentrations is recommended and ciclosporin dose should be adjusted if necessary.
UDCA has been reported to decrease the absorption of ciprofloxacin in a few cases.
In a clinical study in 12 healthy volunteers with the Organic Anion Transporting Polypeptide 1B1 (OATP1B1) *1b/*1b genotype, predicting high OATP1B1 activity, it was demonstrated that concomitant use of UDCA (500 mg/day) and rosuvastatin (20 mg/day) resulted in a significant increase in the plasma levels of rosuvastatin. UDCA increased the AUC of rosuvastatin by approximately 60%, from 145.5 nanogram/mL per hour to 231.9 nanogram/mL per hour (p=0.004). Administration of UDCA for 14 days also significantly increased total bilirubin by 139 ± 39% (p=0.003), conjugated bilirubin by 127 ± 29% (p=0.005) and unconjugated bilirubin by 151 ± 52% (p=0.004). The proposed biological mechanism for this interaction is that bilirubin and rosuvastatin are both metabolites of OATP1B1. OATP1B1 expression is regulated by transcription factor hepatic nuclear factor (HNF) 1α. UDCA acts as an inhibitor of HNF1α and consequently may decrease expression of OATP1B1. A dose reduction in rosuvastatin should be considered in any individuals exposed to both rosuvastatin and UDCA. The clinical relevance of this interaction with regard to other statins is unknown. However, it is biologically possible that this interaction may also occur between UDCA and other statins which are known substrates of OATP1B1, such as atorvastatin, fluvastatin, simvastatin acid, pitavastatin and pravastatin.
UDCA reduces the peak plasma concentrations (Cmax) and the area under the curve (AUC) of the calcium channel blocker nitrendipine. On the basis of this, together with a single case report of an interaction with the substance dapsone (reduction of the therapeutic effect) and in vitro findings, it may be assumed that UDCA induces the medicinal product-metabolising enzyme cytochrome P450 3A4. Caution should therefore be exercised in cases of co-administration of medicinal products metabolised by this enzyme, and a dose adjustment may be necessary. Induction has, however, not been observed in a well-designed interaction study with budesonide, which is a known cytochrome P450 3A substrate.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. In a fertility study in Sprague-Dawley rats at oral doses up to 2700 mg/kg/day (25 times the maximum recommended human dose of 20 mg/kg/day based on body surface area (BSA)), no adverse effects on male or female fertility or pregnancy outcome were observed. However, in an oral fertility study in Wistar rats, there was evidence of a reduction in female mating behaviour at doses ≥ 250 mg/kg/day (2.5 times the maximum recommended human dose based on BSA) and of embryolethality (resulting in a reduction in number of live foetuses) at doses ≥ 1000 mg/kg/day (9 times the maximum recommended human dose based on BSA). Human data on fertility effects following treatment with ursodeoxycholic acid are not available.
Use in pregnancy. (Category B3)
UDCA has been shown to cross the placenta in rats. Animal studies have provided evidence of a teratogenic effect of UDCA during the early phase of gestation. In studies in rats, tail malformations occurred after a dose of 2000 mg per kg of body weight (18 times the maximum recommended human dose based on BSA). In one of two studies in rats, there was evidence of embryolethality, with a reduction in number of live foetuses and live births at oral doses of 2000 mg/kg/day. In studies in rabbits, embryotoxic effects from a dose of 100 mg per kg of body weight were found (2 times the maximum recommended human dose). No teratogenic effects were found in the study of UDCA following oral administration to mice or rabbits at doses of up to 1500 and 300 mg/kg/day, respectively (at least 5 times the maximum recommended human dose).
There are no adequate or well-controlled studies in pregnant women during the first trimester. Therefore, UDCA should not be used during the first three months of pregnancy. Women of childbearing potential should be advised to avoid becoming pregnant while receiving treatment with UDCA.
In women with Intrahepatic Cholestasis of Pregnancy (ICP), UDCA reduces pruritus when given in the second or third trimesters of pregnancy. Data are insufficient to determine the effect of UDCA on neonatal outcomes.
Use in lactation. It is not known whether UDCA is excreted in human milk, but small amounts of UDCA or its metabolites were excreted in milk of lactating rats following oral administration of 30 mg/kg. In an oral peri-postnatal study in rats, there was a slight transient reduction in postnatal body weight gain of pups at 2000 mg/kg/day (18 times the maximum recommended human dose based on BSA).
According to few documented cases of breastfeeding women, UDCA was secreted into the breast milk of lactating mothers. The possibility of adverse reactions on the infant should be considered if UDCA is administered to a nursing mother. Alternatively, breastfeeding can be discontinued.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
UDCA is generally well tolerated with few side effects. Diarrhoea is the main reported side effect. The incidence of diarrhoea in controlled studies was up to 3%.
Some patients may experience increased pruritus in the early weeks of treatment. In such cases a dose reduction, and thereafter a slow (weekly) increase of dose to the recommended dose, may help.
Severe right upper abdominal pain has occurred during the treatment of PBC (≤ 1 in 10,000 patients). During advanced stages of PBC, in very rare cases (≤ 1 in 10,000 patients), decompensation of hepatic cirrhosis has been observed, which partially regressed after the treatment was discontinued.
Calcification of gallstones can occur in ≤ 1 in 10,000 patients.
Allergic reactions have been reported in some patients. Urticaria can occur in ≤ 1 in 10,000 patients.
Other adverse reactions reported include increased cholestasis, nausea, vomiting and sleep disturbance.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For overdose in general, the mainstay of treatment is supportive and symptomatic care.
Diarrhoea may occur in cases of overdosage. If diarrhoea occurs, the dose must be reduced and in cases of persistent diarrhoea, the therapy should be discontinued. No specific countermeasures are necessary and the consequence of diarrhoea should be treated symptomatically with restoration of fluid and electrolyte balance.
In general, other symptoms of overdosage are unlikely because the absorption of UDCA decreases with increasing dose and therefore more is excreted with the faeces.
Serious adverse effects are also unlikely to occur in overdosage. However, liver function should be monitored. If necessary, ion-exchange resins may be used to bind bile acids in the intestines.
For information on the management of overdose, contact the Poison Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The mechanism of action of UDCA in liver and cholestatic disorders has not yet been explained totally. However, UDCA alters bile acid composition, resulting in increases in the concentration of UDCA and decreases in the concentrations of the more hydrophobic and potentially toxic bile acids, cholic and chenodeoxycholic acids. UDCA also has a choleretic effect, resulting in increased bile acid output and bile flow. There is some evidence for immunological effects, including a reduction of abnormal expression of HLA Class I antigens on hepatocytes and a suppression of immunoglobulin and cytokine production.
Clinical trials. Primary biliary cirrhosis. Primary biliary cirrhosis (PBC) is an autoimmune disease of the liver marked by the gradual destruction and eventual disappearance of the bile duct epithelial cells. The sustained loss of intralobular bile ducts causes the signs and symptoms of cholestasis, and eventually results in cirrhosis and liver failure.
Eight pivotal randomised, controlled studies examined the efficacy of UDCA in the treatment of PBC. All eight trials were of at least 2 years follow-up. Seven of the eight studies used a dosage in the range of 12-16 mg/kg/day; the eighth trial used a significantly lower dose of 7.7 ± 0.2 mg/kg/day. Significant improvement in some or all biochemical tests of liver function was shown in subjects given UDCA during the treatment period. Symptom improvement or improvement in histology were not consistently reported with UDCA but longer survival without liver transplantation was reported in two long term studies. One of the studies reported that the efficacy of UDCA in patients with PBC was greater in patients with less advanced disease (entry bilirubin < 2 mg/dL; histological stage I or II) compared to patients with more advanced disease.
Primary sclerosing cholangitis. Primary sclerosing cholangitis (PSC) is a chronic cholestatic liver disease characterised by inflammation, fibrosis, and destruction of the large intra- and extra-hepatic bile ducts.
One pivotal randomised, double-blind placebo-controlled study examined the efficacy of UDCA in the treatment of PSC in 105 patients over 2 years. The dosage used was in the range of 13-15 mg/kg/day. Irrespective of initial histological stage, UDCA had no effect on time to treatment failure and survival, without liver transplantation. Serum bilirubin, alkaline phosphatase (ALP) and aspartate transaminase (AST) improved, but UDCA was not associated with a significant improvement in symptoms or histological score.
In three smaller randomised, double-blind, placebo-controlled studies, UDCA similarly showed significant improvement in liver biochemistry (in two of the studies) when compared to placebo, but did not significantly improve symptom scores. One study found significant improvement in some liver histological features in the patients treated with UDCA. These trials used UDCA doses ranging from 10-15 mg/kg/day.
In a small randomised, double-blind, placebo-controlled study, 20 mg/kg/day UDCA treatment in PSC patients showed improvement in liver biochemistry when compared to placebo. Histological progression was significantly reduced in the UDCA-treated group compared to the placebo-treated group.
Cystic fibrosis-related cholestasis. Cystic fibrosis (CF) is a hereditary disease with multi-organ involvement. Clinical liver disease is rare although many patients may have biochemical evidence of cirrhosis.
One double-blind, placebo-controlled study randomised 55 patients with CF-related cholestasis to UDCA 900 mg/day or placebo for one year. In addition, taurine supplements or placebo were randomly assigned. Efficacy was assessed by improvements in clinically relevant and nutritional parameters, and liver biochemistry. After one year, the UDCA group had significant improvement in gamma-glutamyl transferase (GGT) and 5'-nucleosidase but not AST or alanine aminotransferase (ALT). However, there was a deterioration of overall clinical condition, as measured by the Shwachman-Kulcycki score in those receiving placebo compared to the UDCA group.
In a dose comparison study, UDCA 20 mg/kg/day for 12 months resulted in a more pronounced improvement in GGT and ALT compared to UDCA 10 mg/kg/day.
Improvements in AST and ALP were comparable. Although this study suggested a possible benefit with higher drug doses in resolving liver biochemistry, whether UDCA improves quality of life, histology, or survival is unknown.
5.2 Pharmacokinetic Properties
Absorption. UDCA occurs naturally in the body. After oral administration of a single 500 mg dose of UDCA to healthy volunteers, peak plasma concentrations were 2.7 to 6.3 microgram/mL. Tmax occurs at 60 minutes and a second peak plasma concentration occurs at 180 minutes. After oral administration of 250 mg, 500 mg, 1000 mg and 2000 mg single doses, respective absorption rates were 60.3%, 47.7%, 30.7% and 20.7% based on recovery from bile within 24 hours in patients with external biliary drainage.
Distribution. In plasma, protein binding is 96-98%.
Metabolism. First pass extraction of UDCA from the portal vein by the liver ranges from 50-70%. UDCA is conjugated to glycine and taurine and then excreted into bile, and passes to the small bowel. In the intestine, some conjugates are deconjugated and reabsorbed in the terminal ileum. Conjugates may also be dehydroxylated to lithocholic acid, part of which is absorbed, sulfated by the liver and excreted by the biliary tract. In healthy volunteers given UDCA 500 mg with 14C tracer, 30-44% of the dose was excreted in faeces in the first three days as UDCA (2-4%), lithocholic acid (37%) and 7-ketolithocholic acid (5%).
Excretion. The biological half-life, obtained by radioactive labelling, of orally administered UDCA is 3.5-5.8 days due to the effective enterohepatic circulation of UDCA in the body.
In patients with severe liver disease, renal excretion becomes a major route for elimination of bile acids.
5.3 Preclinical Safety Data
Genotoxicity. UDCA was not genotoxic in the following studies: gene mutation assays (in vitro Ames test, gene mutation assay at the TK locus in mouse lymphoma L5178Y cells), assays of chromosome aberrations (analysis of chromosome aberrations in Chinese hamster bone marrow and in spermatogonia of mice, and micronucleus test in hamsters) and assay of sister chromatid exchanges in cultured human lymphocytes.
Carcinogenicity. In two 24-month oral carcinogenicity studies in mice, UDCA at doses up to 1000 mg/kg/day was not tumourigenic. Based on BSA, this dose represents 4 times the recommended maximum clinical dose of 20 mg/kg/day. In two 2-year oral carcinogenicity studies in rats, UDCA at doses up to 300 mg/kg/day (3 times the recommended maximum human dose based on BSA) was not tumourigenic.
In 103-week oral carcinogenicity studies of lithocholic acid, a metabolite of UDCA, doses up to 250 mg/kg/day in mice and 500 mg/kg/day in rats did not produce any tumours.
6 Pharmaceutical Particulars
6.1 List of Excipients
Ursosan capsules contains UDCA 250 mg, maize starch, pregelatinised maize starch, colloidal anhydrous silica, magnesium stearate, gelatin, titanium dioxide and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Ursosan is supplied in transparent PVC/PVdC/Al blister packs in cardboard cartons. Each carton contains 100 capsules.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Ursodeoxycholic acid (UDCA) is a white or almost white powder. It is practically insoluble in water, readily soluble in alcohol, sparingly soluble in acetone, in chloroform and in ether. It melts at 200-204°C. The IUPAC chemical name of UDCA is 3α, 7β-dihydroxy-5-cholan-24-oic acid.
Chemical structure. The chemical structure of ursodeoxycholic acid is as follows:
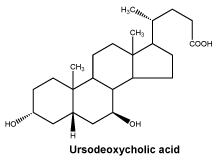
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
07 November 2014
Date of Revision
17 May 2019
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.