Valganciclovir Sandoz
Brand Information
| Brand name | Valganciclovir Sandoz |
| Active ingredient | Valganciclovir |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Valganciclovir Sandoz.
Summary CMI
VALGANCICLOVIR SANDOZ®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Valganciclovir Sandoz?
Valganciclovir Sandoz contains the active ingredient valganciclovir. Valganciclovir Sandoz acts against a virus called cytomegalovirus or CMV. Valganciclovir Sandoz is used to treat CMV eye infections in AIDS patients and to prevent CMV infection in patients following organ transplantation.
For more information, see Section 1. Why am I using Valganciclovir Sandoz? in the full CMI.
2. What should I know before I use Valganciclovir Sandoz?
Do not use if you have ever had an allergic reaction to valganciclovir or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Valganciclovir Sandoz? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Valganciclovir Sandoz and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Valganciclovir Sandoz?
- Induction treatment for adults (for active CMV retinitis): 900 mg twice daily with food for 21 days.
- Maintenance treatment for adults (after induction treatment or for inactive CMV retinitis): 900 mg once daily with food.
- Prevention of CMV disease in transplantation for adults: 900 mg once daily with food, starting within 10 days post transplantation until 100 days post transplantation. If you have received a kidney transplant, the same daily dose is required until 200 days post transplantation.
More instructions can be found in Section 4. How do I use Valganciclovir Sandoz? in the full CMI.
5. What should I know while using Valganciclovir Sandoz?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Valganciclovir Sandoz? in the full CMI.
6. Are there any side effects?
Go to your nearest hospital if you experience: swelling of the tongue, lips or throat, any sign of infection such as fever, chills, sore throat or mouth ulcers, unexplained bruising or bleeding, hallucinations, confusion, agitation; or fits.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
VALGANCICLOVIR SANDOZ®
Active ingredient(s): valganciclovir
Consumer Medicine Information (CMI)
This leaflet provides important information about using Valganciclovir Sandoz. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Valganciclovir Sandoz.
Where to find information in this leaflet:
1. Why am I using Valganciclovir Sandoz?
2. What should I know before I use Valganciclovir Sandoz?
3. What if I am taking other medicines?
4. How do I use Valganciclovir Sandoz?
5. What should I know while using Valganciclovir Sandoz?
6. Are there any side effects?
7. Product details
1. Why am I using Valganciclovir Sandoz?
Valganciclovir Sandoz contains the active ingredient valganciclovir.
In the body valganciclovir rapidly changes to ganciclovir.
Valganciclovir Sandoz belongs to a group of medicines used to prevent the growth of viruses.
Valganciclovir Sandoz acts against a virus called cytomegalovirus or CMV (a type of herpes virus). It prevents this virus from growing and multiplying in the body.
CMV causes infections, mainly in people with poor immunity. Poor immunity can be caused by HIV/AIDS or by medications taken after an organ transplant.
Valganciclovir Sandoz is used to treat CMV eye infections (known as CMV retinitis) in AIDS patients, which, if left untreated can cause blindness. It is not a cure for CMV eye infections.
Valganciclovir Sandoz is not effective against any underlying HIV infection.
Valganciclovir Sandoz is also used to prevent CMV infection in patients following organ transplantation.
Valganciclovir Sandoz is not addictive.
This medicine is available only with a doctor's prescription.
Your doctor, however, may have prescribed Valganciclovir Sandoz for another purpose.
2. What should I know before I use Valganciclovir Sandoz?
Animal and other laboratory studies have shown Valganciclovir Sandoz causes infertility, birth defects and cancer. It is possible that these effects may also occur in humans.
Warnings
Do not use Valganciclovir Sandoz if:
- you are allergic to valganciclovir, ganciclovir, valaciclovir or aciclovir, or any of the ingredients listed at the end of this leaflet. Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing o swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Always check the ingredients to make sure you can use this medicine. - you have very low blood counts for platelets (which help clotting), neutrophils (a type of white blood cell which defends against infection) or low haemoglobin (oxygen carrying substance in the blood).
Use in children
There is limited information on the safety and effectiveness of the use of valganciclovir in children. Your doctor will advise you whether Valganciclovir Sandoz is suitable for your child.
Check with your doctor if you:
- are allergic to any other medicines, foods, dyes or preservatives, especially any medicine which you have taken previously to treat your current condition
- have any other health problems, especially the following:
- you have a history of low blood counts for platelets (thrombocytopenia), neutrophils (neutropenia) or anaemia
- you have, or previously have had, poor kidney function - are pregnant or intend to become pregnant
- Valganciclovir Sandoz is not recommended for use during pregnancy. Valganciclovir may affect your developing baby if you take it during pregnancy. Your doctor will discuss the risks and benefits of using Valganciclovir Sandoz if you are pregnant. - are breast-feeding or intend to breast-feed
- It is not known whether Valganciclovir Sandoz passes into breast milk. Breast-feeding is not recommended during therapy with Valganciclovir Sandoz - are a woman who could become pregnant and you are not using contraception
- You must use a reliable form of contraception during Valganciclovir Sandoz therapy, and for at least 30 days after stopping Valganciclovir Sandoz, unless you are not sexually active. - are a sexually active man
- You should use condoms during and for at least 90 days following treatment with Valganciclovir Sandoz unless it is certain that your female partner is not at risk of pregnancy.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Make sure your doctor is aware that you are pregnant or plan to become pregnant or are breastfeeding or intend to breastfeed. You must use a reliable form of contraception during Valganciclovir Sandoz therapy, and for at least 30 days after stopping Valganciclovir Sandoz, unless you are not sexually active.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Valganciclovir Sandoz may interfere with each other. These medicines include:
- imipenem/cilastatin (Primaxin®) a combination of medicines used to treat some infections
- probenecid (e.g. Benemid®), a medicine used to treat gout
- zidovudine (AZT), Retrovir®, Combivir®), didanosine (ddI or Videx®), stavudine, other medicines used to treat HIV infection
- medicines for the treatment of cancer such as doxorubicin, vinblastine, vincristine, hydroxyurea
- mycophenolate mofetil (CellCept®), and other medicines used to prevent organ rejection after a transplant e.g. ciclosporin, tacrolimus.
- anti-infective agents such as trimpethroprim / sulphonamides, dapsone, pentamidine, flucytosine pegylated interferons plus ribavirin and amphotericin B
These medicines may be affected by Valganciclovir Sandoz, or may affect how well it works. You may need to use different amounts of your medicine, or you may need to take different medicines. Your doctor or pharmacist have more information on medicines to be careful with or avoid while taking Valganciclovir Sandoz.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Valganciclovir Sandoz.
4. How do I use Valganciclovir Sandoz?
Follow all directions given to you by your doctor or pharmacist carefully.
They may differ from the information contained in this leaflet.
If you do not understand the instructions, ask your doctor or pharmacist for help.
How much to take
Take Valganciclovir Sandoz exactly as your doctor has prescribed. Your doctor will tell you how many Valganciclovir Sandoz tablets to take each day.
Treatment of CMV Retinitis in AIDS (adult patients)
- Induction Treatment for adults (for active CMV retinitis)
The usual dose is 900 mg (two 450 mg tablets) twice daily with food for 21 days.
- Maintenance Treatment for adults (after induction treatment or for inactive CMV retinitis)
The usual dose is 900 mg (two 450 mg tablets) once daily with food.
Prevention of CMV Disease in Transplantation for adults
The usual dose is 900 mg (two 450 mg tablets) once daily with food, starting within 10 days post transplantation until 100 days post transplantation.
If you have received a kidney transplant, the same daily dose is required until 200 days post transplantation.
Your dose may have to be reduced or stopped if you have or develop low blood counts, have kidney disease, or if you are older than 65 years.
How to take Valganciclovir Sandoz
- Swallow the tablets whole with a full glass of water. Tablets must be taken with food.
When to take Valganciclovir Sandoz
- Take your medicine at about the same time each day. Take during or immediately after a meal. If you take it on an empty stomach, it may not work as well.
- Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
How to long to take Valganciclovir Sandoz
- Continue taking your medicine for as long as your doctor tells you.
- This medicine helps to control your condition, but does not cure it. It is important to keep taking your medicine even if you feel well.
- Continue taking your medicine until your doctor tells you to stop.
If you forget to use Valganciclovir Sandoz
Valganciclovir Sandoz should be used regularly at the same time each day. If you miss your dose at the usual time, take your dose as soon as you remember, and continue to take it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you use too much Valganciclovir Sandoz
If you think that you have used too much Valganciclovir Sandoz, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Valganciclovir Sandoz?
Things you should do
Tell any other doctors, dentists and pharmacists who treat you that you are taking Valganciclovir Sandoz.
Call your doctor straight away if you become pregnant while taking Valganciclovir Sandoz.
See your doctor regularly so that your CMV disease, blood cell counts and any other potential side effects may be monitored carefully.
If blood cell counts are low then this may reduce your ability to fight infection, or for your blood to clot efficiently. If left undetected these effects on blood cells may contribute to death or serious illness.
If you have a CMV eye infection, you must also see your doctor regularly to monitor the condition of your retina (part of the eye).
Condoms should be used by sexually active men while taking Valganciclovir Sandoz and for 90 days after stopping treatment.
Women who are of childbearing potential should use contraception during and for at least 30 days after stopping Valganciclovir Sandoz.
Tell your doctor if, for any reason, you have not taken your medicine exactly as prescribed. Otherwise, your doctor may think that it was not effective and change your treatment unnecessarily.
Keep all of your appointments with your doctor so that your progress can be checked.
Things you should not do
- Do not take Valganciclovir Sandoz to treat any other complaints unless your doctor tells you to.
- Do not give Valganciclovir Sandoz to anyone else even if they have the same condition as you.
- Do not stop taking Valganciclovir Sandoz or change the dosage without first checking with your doctor.
- Do not let yourself run out of medicine over the weekend or on holidays.
- Do not take any other medicines whether they require a prescription or not without first telling your doctor or consulting a pharmacist.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Valganciclovir Sandoz affects you.
Valganciclovir Sandoz may cause drowsiness, dizziness, confusion or seizures (fits) in some people and therefore may affect alertness. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Looking after your medicine
- Keep your medicine in the original container.
- If you take it out of its original container it may not keep well.
- Keep your medicine in a cool dry place where the temperature stays below 25°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Valganciclovir Sandoz contains
| Active ingredient (main ingredient) | valganciclovir |
| Other ingredients (inactive ingredients) | microcrystalline cellulose crospovidone stearic acid povidone hypromellose titanium dioxide macrogol 400 iron oxide red polysorbate 80 |
| Potential allergens | none |
Do not take this medicine if you are allergic to any of these ingredients.
What Valganciclovir Sandoz looks like
Valganciclovir Sandoz 450mg - pink, convex oral tablets with 'J' on one side and '156' on the other side.
Available in blisters of 60 tablets (Aust R 227605).
Who distributes Valganciclovir Sandoz
Sandoz Pty Ltd
100 Pacific Highway
North Sydney, NSW 2060
Australia
Tel 1800 726 369
This leaflet was prepared in September 2024.
® Registered Trade Mark. The trade marks mentioned in this material are the property of their respective owners.
Brand Information
| Brand name | Valganciclovir Sandoz |
| Active ingredient | Valganciclovir |
| Schedule | S4 |
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Valganciclovir hydrochloride.
2 Qualitative and Quantitative Composition
Each tablet contains valganciclovir 450 mg (as valganciclovir hydrochloride 496 mg).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Valganciclovir Sandoz (valganciclovir HCl) is available as 450 mg pink convex oval tablets with "J" on one side and "156" on the other side. Each tablet contains 450 mg valganciclovir.
4 Clinical Particulars
4.1 Therapeutic Indications
Valganciclovir is indicated for the treatment of cytomegalovirus (CMV) retinitis in adult patients with acquired immunodeficiency syndrome (AIDS).
Valganciclovir is indicated for the prophylaxis of CMV disease in adult solid organ transplantation (SOT) patients who are at risk.
4.2 Dose and Method of Administration
Dosage. Caution. Strict adherence to dosage recommendations is essential to avoid overdose.
Valganciclovir is rapidly and extensively converted to the active ingredient ganciclovir. The bioavailability of ganciclovir from valganciclovir is up to 10-fold higher than from oral ganciclovir, therefore the dosage and administration of valganciclovir tablets or powder for oral solution as described below should be closely followed (see Section 4.4 Special Warnings and Precautions for Use; Section 4.9 Overdose).
Treatment of CMV retinitis. Adult patients. Induction treatment. For patients with active CMV retinitis, the recommended dosage is 900 mg twice daily for 21 days with food. Prolonged induction treatment may increase the risk of bone marrow toxicity (see Section 4.4 Special Warnings and Precautions for Use, Haematologic).
Maintenance treatment. Following induction treatment, or in patients with inactive CMV retinitis the recommended dose is 900 mg once daily with food. Patients whose retinitis worsens may repeat induction treatment (see Induction treatment).
Prevention of CMV disease in transplantation. Adult patients. Kidney transplant. For kidney transplant patients, the recommended dose is 900 mg once daily with food, starting within 10 days of transplantation until 200 days post-transplantation [see Section 5.1 Pharmacodynamic Properties, Clinical trials, IMPACT study (study NT18435)].
Solid organ transplant other than kidney. For all other solid organ transplant patients, the recommended dose is 900 mg once daily with food, starting within 10 days of transplantation until 100 days post-transplantation (see Section 5.1 Pharmacodynamic Properties, Clinical trials, Study PV16000).
Method of administration. Valganciclovir is administered orally, and should be taken with food (see Section 5.1 Pharmacodynamic Properties; Section 5.2 Pharmacokinetic Properties, Absorption).
Dosage adjustment. Renal impairment. Serum creatinine or creatinine clearance levels should be monitored carefully. Dosage adjustment is required based on creatinine clearance as shown in Table 1 (see Section 4.4 Special Warnings and Precautions for Use; Section 5.2 Pharmacokinetic Properties, Special populations).
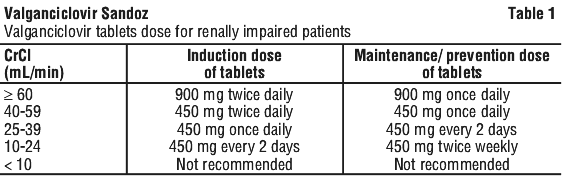
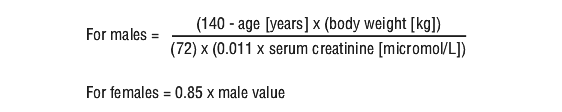
Paediatric patients. (This product is not indicated for paediatric patients. Information has been included for prescriber information only. Valganciclovir powder for oral solution is not available in this brand. It is available in other brands. Refer to the relevant Product Information for more information.)
The safety and efficacy of valganciclovir in paediatric patients have not been established in adequate and well-controlled clinical studies.
In paediatric solid organ transplant patients (paediatric heart transplant patients from 4 weeks and paediatric kidney transplant patients from 4 months (see Section 5.1 Pharmacodynamic Properties, Clinical trials), who are at risk of developing CMV disease, the recommended once daily dose of valganciclovir is based on body surface area (BSA) and creatinine clearance (CrCl) derived from Schwartz formula, and is calculated using the equation below.
Paediatric dose (mg) = 7 x BSA x CrCl (calculated using the Schwartz formula). If the calculated Schwartz creatinine clearance exceeds 150 mL/min/1.73 m2, then a maximum value of 150 mL/min/1.73 m2 should be used in the equation.
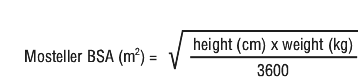
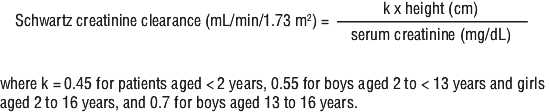
It is recommended to monitor serum creatinine levels regularly and consider changes in height and body weight and adapt the dose as appropriate during prophylaxis period.
4.3 Contraindications
Valganciclovir Sandoz is contraindicated in patients with known hypersensitivity to valganciclovir, ganciclovir or to any component of the product.
Valganciclovir should not be administered if the absolute neutrophil count is less than 500 cells/microL, the platelet count is less than 25,000/microL, or the haemoglobin is less than 8 g/dL.
4.4 Special Warnings and Precautions for Use
This product is not indicated for paediatric patients. Information has been included for prescriber information only. Valganciclovir powder for oral solution is not available in this brand. It is available in other brands. Refer to the relevant Product Information for more information.
Clinical toxicities of valganciclovir, which is metabolised to ganciclovir, include leucopenia and thrombocytopenia. Concomitant administration of valganciclovir and other medicines that are known to be myelosuppressive or associated with renal impairment may result in added toxicity (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
In animal studies, ganciclovir was found to be mutagenic, clastogenic, aspermatogenic, teratogenic and carcinogenic and to impair fertility; therefore, it should be considered a potential teratogen and carcinogen in humans with potential to cause birth defects and cancers. Prior to initiation of valganciclovir treatment, patients should be advised of the potential risks to the foetus and to use contraceptive measures (see Section 4.6 Fertility, Pregnancy and Lactation). Based on clinical and nonclinical studies, valganciclovir causes temporary or permanent inhibition of spermatogenesis in males (see Section 4.2 Dose and Method of Administration; Section 5.3 Preclinical Safety Data, Carcinogenicity and Genotoxicity; Section 6.6 Special Precautions for Disposal). Valganciclovir is indicated in those patients as outlined, see Section 4.1 Therapeutic Indications where the potential benefits to the patient outweighs the risks stated herein.
The diagnosis of CMV retinitis should be made by indirect ophthalmoscopy. Other conditions in the differential diagnosis of CMV retinitis include candidiasis, toxoplasmosis, histoplasmosis, retinal scars and cotton wool spots, any of which may produce a retinal appearance similar to CMV. For this reason, it is essential that the diagnosis of CMV be established by an ophthalmologist familiar with the presentation of these conditions. The diagnosis of CMV retinitis may be supported by culture of CMV in the urine, blood, throat, or other sites, but a negative culture does not rule out CMV retinitis.
Cross hypersensitivity. Due to the similarity of the chemical structure of valganciclovir and that of aciclovir and valaciclovir, a cross-hypersensitivity reaction between these medicines is possible. Caution should therefore be used when prescribing valganciclovir to patients with known hypersensitivity to aciclovir or penciclovir, (or to their prodrugs, valaciclovir or famciclovir respectively).
Haematologic. Severe leucopenia, neutropenia, anaemia, thrombocytopenia, pancytopenia, bone marrow failure and aplastic anaemia have been observed in patients treated with valganciclovir (and ganciclovir) (see Section 4.8 Adverse Effects (Undesirable Effects); Section 4.2 Dose and Method of Administration).
Therapy should not be initiated if the absolute neutrophil count is less than 500 cells/microL; or platelet count is less than 25,000/microL; or the haemoglobin is less than 8 g/dL. It is recommended that complete blood counts and platelet counts be monitored in all patients frequently during therapy; especially in patients with renal impairment in neonates and infants, in patients in whom ganciclovir or other nucleoside analogues have previously resulted in leucopenia, or in whom neutrophil counts are less than 1000 cells/microL at the beginning of treatment.
Valganciclovir should, therefore, be used with caution in patients with pre-existing cytopenias, or who have received or are receiving myelosuppressive medicines or irradiation. Cytopenia may occur at any time during treatment and may increase with continued dosing. In patients with severe leucopenia, neutropenia, anaemia and/or thrombocytopenia, treatment with haematopoietic growth factors and/or the interruption of therapy is recommended. Cell counts usually begin to recover within 3 to 7 days of discontinuing medication. Colony-stimulating factors have been shown to increase neutrophil counts in patients receiving ganciclovir for treatment of CMV retinitis.
Use in hepatic impairment. The safety and efficacy of valganciclovir have not been established in patients with hepatic impairment (see Section 5.2 Pharmacokinetic Properties, Pharmacokinetics in special populations, Patients with hepatic impairment).
Use in renal impairment. In patients with impaired renal function, dosage adjustments based on creatinine clearance are required.
Increased serum creatinine levels have been observed in trials evaluating valganciclovir tablets. Patients should have serum creatinine or creatinine clearance values monitored carefully to allow for dosage adjustments in renally impaired patients (see Section 4.2 Dose and Method of Administration, Dosage adjustment, Renal impairment).
Use in the elderly. The pharmacokinetic profiles of valganciclovir in elderly patients have not been established. Since elderly individuals frequently have a reduced glomerular filtration rate, particular attention should be paid to assessing renal function before and during administration of valganciclovir.
Clinical studies of valganciclovir did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other therapy. Valganciclovir is known to be substantially excreted by the kidney, and the risk of toxic reactions to this medicine may be greater in patients with impaired renal function. Since elderly patients are more likely to have decreased renal function, care should be taken in dose selection. In addition, renal function should be monitored and dosage adjustments should be made accordingly (see Section 4.2 Dose and Method of Administration, Dosage adjustment, Renal impairment; Section 4.4 Special Warnings and Precautions for Use, Use in renal impairment).
Paediatric use. A higher risk of haematological cytopenias in neonates and infants warrants careful monitoring of blood counts in these age groups. Monitoring of liver function abnormalities, renal function and gastrointestinal fluid loss is also recommended in paediatric patients.
The use of valganciclovir in children warrants extreme caution. Valganciclovir should be considered a potential carcinogen in humans. This potential to cause cancers is greater in infants and children than in adults. Valganciclovir is likely to cause temporary or permanent inhibition of spermatogenesis. This could result in permanent male infertility. Administration to children should be undertaken only after careful evaluation and only if, in the opinion of the physician, the potential benefits of treatment outweigh these considerable risks (see Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties, Clinical trials).
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Valganciclovir is rapidly and extensively converted to ganciclovir; therefore, interactions associated with ganciclovir are expected for valganciclovir.
Imipenem-cilastatin. Convulsions have been reported in patients taking ganciclovir and imipenem-cilastatin concomitantly and a pharmacodynamic interaction between these two drugs cannot be discounted. Valganciclovir should not be administered concomitantly with imipenem-cilastatin unless the potential benefits outweigh the potential risks (see Section 4.4 Special Warnings and Precautions for Use).
Clinically significant potential medicine interactions. Toxicity may be enhanced when ganciclovir/valganciclovir is co-administered with other medicines known to be myelosuppressive or associated with renal impairment. This includes nucleoside analogues (e.g. zidovudine, didanosine, stavudine), immunosuppressants (e.g. ciclosporin, tacrolimus, mycophenolate mofetil), antineoplastic agents (e.g. doxorubicin, vinblastine, vincristine, hydroxyurea) and anti-infective agents (e.g. trimethroprim/sulphonamides, dapsone, amphotericin B, flucytosine, pentamidine), hydroxyurea and pegylated interferons/ribavirin. Therefore, these medicines should only be considered for concomitant use with valganciclovir if the potential benefits outweigh the potential risks.
Zidovudine. Both zidovudine and valganciclovir have the potential to cause neutropenia and anaemia. A pharmacodynamic interaction may occur during concomitant administration of these drugs and some patients may not tolerate concomitant therapy at full dosage.
Didanosine. Didanosine plasma concentrations were found to be consistently raised when given with ganciclovir (both IV and oral). At ganciclovir oral doses of 3 g/day and 6 g/day, an increase in the AUC of didanosine ranging from 84% to 124% has been observed, and likewise at IV doses of 5 mg/kg/day and 10 mg/kg/day, and increase in the AUC of didanosine ranging from 38% to 67% has been observed confirming a pharmacokinetic interaction during the administration of these drugs. There was no significant effect on ganciclovir concentrations. Patients should be closely monitored for didanosine toxicity (e.g. pancreatitis).
Probenecid. Probenecid given with oral ganciclovir resulted in statistically significantly decreased renal clearance of ganciclovir (20%) leading to statistically significantly increased exposure (40%). These changes were consistent with a mechanism of interaction involving competition for renal tubular excretion. Therefore, patients taking probenecid and valganciclovir concomitantly should be closely monitored for ganciclovir toxicity.
Paediatric population. Interaction studies have only been performed in adults.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. In animal studies ganciclovir was found to impair fertility. In a clinical study, renal transplant patients receiving valganciclovir for CMV prophylaxis for up to 200 days were compared to an untreated control group. Spermatogenesis was inhibited during treatment with valganciclovir. At follow-up, approximately six months after treatment discontinuation, the mean sperm density in treated patients was comparable to that observed in the untreated control group. In valganciclovir treated patients, all patients with normal sperm density (n=7) and 8/13 patients with low sperm density at baseline, had normal density approximately six months after treatment cessation. In the control group, all patients with normal sperm density (n=6) and 2/4 patients with low sperm density at baseline, had normal density at the end of follow-up. Due to limitations of this study, these results are not sufficient to establish recovery of spermatogenesis. Reproductive toxicity studies have not been conducted with valganciclovir. Valganciclovir is rapidly and extensively converted to ganciclovir and therefore is expected to have similar reproductive toxicity effects as ganciclovir. Animal data indicate that the administration of ganciclovir causes inhibition of spermatogenesis and subsequent infertility. These effects were reversible at lower doses and irreversible at higher doses. It is considered probable that in humans, valganciclovir at the recommended doses may cause temporary or permanent inhibition of spermatogenesis.
Ganciclovir caused decreased mating behaviour, decreased fertility, and an increased incidence of embryolethality in female mice following IV doses of 90 mg/kg/day (approximately 2.1 times the mean drug exposure to ganciclovir in humans following the maximum recommended dose of valganciclovir, 900 mg twice daily, based on AUC comparisons).
Ganciclovir caused decreased fertility in male mice and hypospermatogenesis in mice and dogs following daily oral or IV administration of doses ranging from 0.2 to 10 mg/kg/day. Systemic drug exposure (AUC) at the lowest dose showing toxicity in each species ranged from 0.02 to 0.1 times the AUC of ganciclovir in humans following the maximum recommended dose of valganciclovir. Valganciclovir caused similar effects on spermatogenesis in mice, rats and dogs. It is considered likely that valganciclovir could cause inhibition of human spermatogenesis.
Prior to initiation of ganciclovir treatment, patients should be advised of the potential risks to the foetus and to use contraceptive measures.
Use in pregnancy. (Category D)
The safety of valganciclovir for use in pregnant women has not been established however, ganciclovir readily diffuses across the human placenta. Valganciclovir should be used during pregnancy only if the potential benefit justifies the potential risk to the foetus.
Valganciclovir may be teratogenic or embryotoxic at dose levels recommended for human use. In addition, the effect on the future fertility of boys is unknown. There are no adequate and well-controlled studies in pregnant women.
Contraception in males and females. Women of childbearing potential should be advised to use effective contraception during and for at least 30 days after treatment with valganciclovir because of the mutagenic and teratogenic potential of ganciclovir. Sexually active men are recommended to use condoms during, and for at least 90 days after cessation of treatment with valganciclovir, unless it is certain that the female partner is not at risk of pregnancy.
The safe use of valganciclovir during labour has not been established.
Valganciclovir is expected to have reproductive toxicity effects similar to ganciclovir. In animal studies ganciclovir was associated with reproductive toxicity and teratogenicity. Ganciclovir has been shown to be embryotoxic in rabbits and mice following IV administration, and teratogenic in rabbits. Foetal resorptions were present in at least 85% of rabbits at 60 mg/kg/day IV and mice at 108 mg/kg/day (2.7 times the mean drug exposure to ganciclovir in humans following the maximum recommended dose of valganciclovir, 900 mg twice daily, based on AUC comparisons). Effects observed in rabbits included: foetal growth retardation, embryolethality, teratogenicity and/or maternal toxicity. Teratogenic changes included cleft palate, anophthalmia/microphthalmia, aplastic organs (kidney and pancreas), hydrocephaly and brachygnathia. In mice, effects observed were foetal toxicity and embryolethality.
Daily intravenous doses of 90 mg/kg administered to female mice prior to mating, during gestation, and during lactation caused hypoplasia of the testes and seminal vesicles in the month-old male offspring, as well as pathologic changes in the nonglandular region of the stomach. The drug exposure in mice as estimated by the AUC was approximately 2.1 times the human AUC.
Use in lactation. Peri- and postnatal development has not been studied with valganciclovir or with ganciclovir but the possibility of ganciclovir being excreted in the breast milk and causing serious adverse reactions in the nursing infant cannot be discounted. Human data are not available but animal data indicates that ganciclovir is excreted in the milk of lactating rats. Therefore, valganciclovir should not be given to breastfeeding mothers or breastfeeding should be discontinued. The minimum time interval before breastfeeding can safely be resumed after the last dose of valganciclovir is unknown.
4.7 Effects on Ability to Drive and Use Machines
Adverse reactions such as seizures, dizziness and confusion have been reported with the use of valganciclovir and/or ganciclovir. If they occur, such effects may affect tasks requiring alertness including the patient's ability to drive and operate machinery.
4.8 Adverse Effects (Undesirable Effects)
This product is not indicated for paediatric patients. Information has been included for prescriber information only.
Clinical trials. Experience with valganciclovir. Valganciclovir, a prodrug of ganciclovir, is rapidly converted to ganciclovir after oral administration. The undesirable effects known to be associated with ganciclovir usage can therefore be expected to occur with valganciclovir. All of the adverse events observed in clinical studies with valganciclovir have been previously observed with ganciclovir.
Adult patients. Treatment of CMV retinitis in AIDS. The safety profiles of valganciclovir and IV ganciclovir during 28 days of randomised study phase (21 days induction dose and 7 days maintenance dose) in 158 patients were comparable. The most frequently reported events were diarrhoea, neutropenia and pyrexia. More patients reported diarrhoea, oral candidiasis, headache and fatigue in the oral valganciclovir arm, and nausea and injection site-related events in the IV ganciclovir arm (see Table 2).
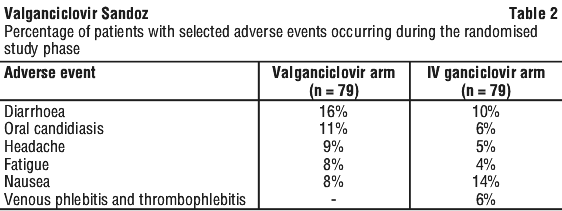
The most frequently reported adverse reactions regardless of seriousness, that were considered related (remotely, possibly or probably) to valganciclovir by the investigator were neutropenia, anaemia, diarrhoea and nausea.
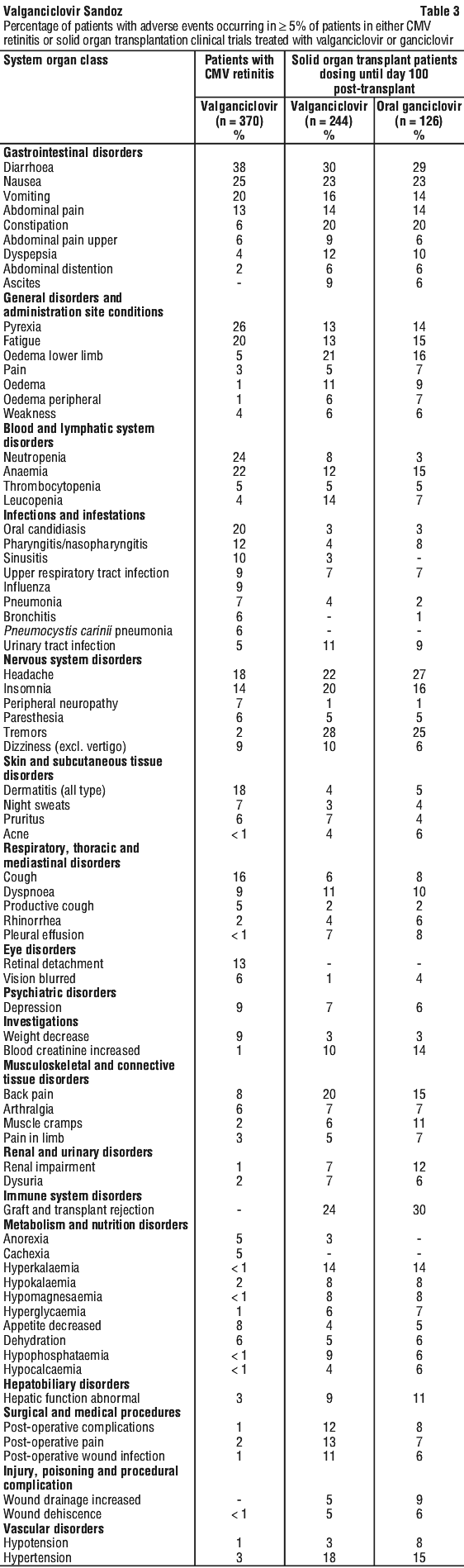
Prevention of CMV disease in transplantation. Table 3 shows the adverse events (up to 28 days after study treatment) regardless of seriousness and drug relationship with an incidence of ≥ 5% from a clinical trial, where solid organ transplant patients received valganciclovir or oral ganciclovir starting within 10 days post-transplantation until day 100 post-transplant.
The most frequently reported adverse reactions, regardless of seriousness, that were considered related (remotely, possibly or probably) to valganciclovir by the investigator in solid organ transplant patients treated until day 100 post-transplant were leukopenia, diarrhoea, nausea and neutropenia and were leukopenia, neutropenia, anaemia and diarrhoea in kidney transplant patients treated until day 200 post-transplant.
Blood and lymphatic system disorders. Pancytopenia, bone marrow depression, aplastic anaemia, febrile neutropenia.
Renal and urinary disorders. Decreased renal creatinine clearance.
Infections and infestations. Local and systemic infections and sepsis.
Bleeding complications. Potentially life-threatening bleeding associated with thrombocytopenia.
Nervous system disorders. Convulsion, psychotic disorder, hallucinations, confusion, agitation.
General disorder and administration site conditions. Valganciclovir hypersensitivity.
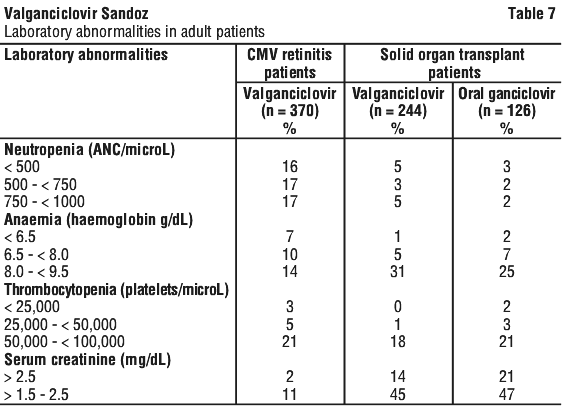
Severe neutropenia (ANC < 500/microL) is seen more frequently in CMV retinitis patients (16%) undergoing treatment with valganciclovir than in solid organ transplant patients receiving valganciclovir (5%) or oral ganciclovir (3%) until day 100 post-transplant or valganciclovir (10%) until day 200 post-transplant. There was a greater increase in serum creatinine seen in solid organ transplant patients treated until day 100 or day 200 post-transplant with both valganciclovir and oral ganciclovir when compared to CMV retinitis patients. Impaired renal function is a feature common to solid organ transplantation patients.
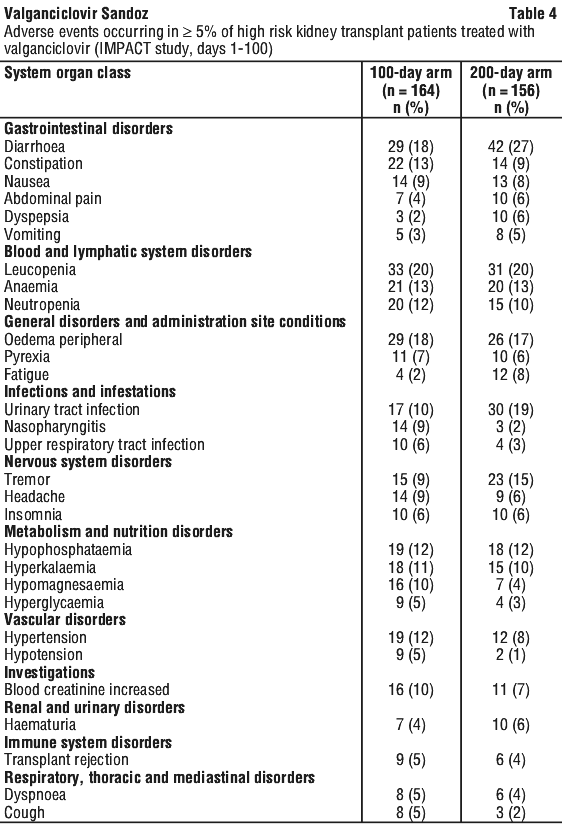
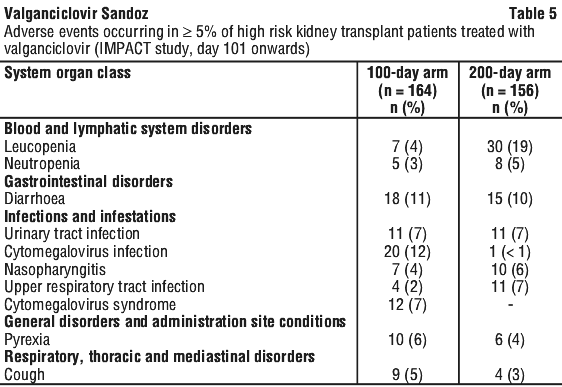
The overall safety profile of valganciclovir did not change with the extension of prophylaxis up to 200 days in high risk kidney transplant patients. Leucopenia was reported with a slightly higher incidence in the 200 days arm while the incidence of neutropenia, anaemia and thrombocytopenia were similar in both arms. The incidence of adverse events in this patient population from the IMPACT study is shown in Table 4 and Table 5.
Table 4 shows adverse events occurring in the first 100 days of the study when all patients were receiving valganciclovir prophylaxis. While, Table 5 shows adverse events occurring after day 100 of the study when only patients in the 200 days arm were receiving valganciclovir (patients in the 100 day arm were receiving placebo).
Gastrointestinal disorders. Cholangitis, dysphagia, eructation, oesophagitis, faecal incontinence, flatulence, gastritis, gastrointestinal disorder, gastrointestinal haemorrhage, mouth ulceration, pancreatitis, tongue disorder.
General disorders and administration site conditions. Asthenia, bacterial, fungal and viral infections, haemorrhage, malaise, mucous membrane disorder, photosensitivity reaction, rigors, sepsis, taste disturbance, decreased libido.
Hepatobiliary disorders. Hepatitis, jaundice.
Skin and subcutaneous tissue disorders. Acne, alopecia, exfoliative dermatitis, dry skin, increased sweating, urticaria.
Nervous system disorders. Abnormal dreams, amnesia, anxiety, ataxia, coma, dry mouth, emotional disturbance, hyperkinetic syndrome, hypertonia, myoclonic jerks, nervousness, somnolence, tremor.
Psychiatric disorder. Abnormal thinking.
Musculoskeletal and connective tissue disorders. Musculoskeletal pain, myasthenic syndrome.
Renal and urinary disorders. Haematuria present, impotence, renal failure, urinary frequency.
Metabolic and nutritional disorders. Increased blood alkaline phosphatase, increased blood creatine phosphokinase, decreased blood glucose, increased blood lactic dehydrogenase, decreased blood magnesium, diabetes mellitus, oedema, abnormal hepatic function, hypocalcaemia, hypokalaemia, hypoproteinaemia.
Eye disorders. Amblyopia, blindness, eye haemorrhage, eye pain, glaucoma, abnormal vision, vitreous disorder.
Ear and labyrinth disorders. Earache, deafness, tinnitus.
Blood and lymphatic system disorders. Eosinophilia, leucocytosis, lymphadenopathy, splenomegaly.
Cardiac disorders. Arrhythmia (including ventricular arrhythmia), deep thrombophlebitis, hypertension, hypotension, migraine, phlebitis, tachycardia, vasodilatation.
Respiratory, thoracic and mediastinal disorders. Pleural effusion, sinus congestion.
Paediatric patients. Valganciclovir has been studied in 179 paediatric solid organ transplant patients who are at risk of developing CMV disease (aged 3 weeks to 16 years) and in 133 neonates with symptomatic congenital CMV disease (aged 2 to 31 days), with duration of ganciclovir exposure ranging from 2 to 200 days (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
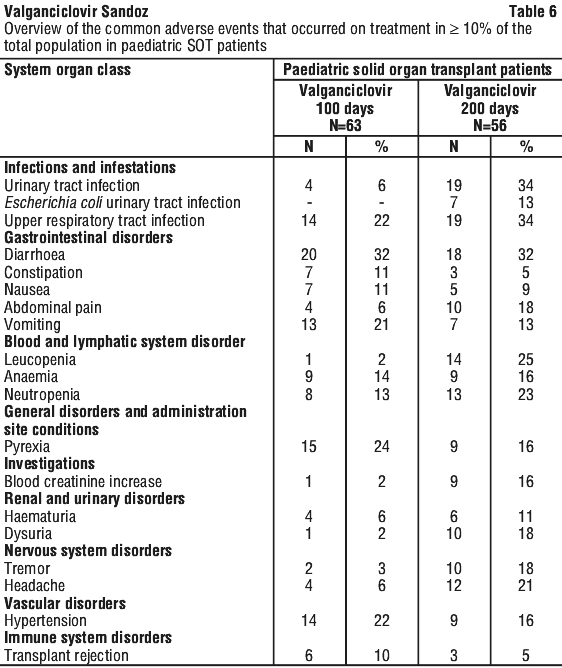
Prevention of CMV disease in solid organ transplant (SOT). Table 6 shows the adverse events (up to 28 days after completion of study treatment) regardless of seriousness and relationship with an incidence of ≥ 10 % from two clinical trials where solid organ transplant paediatric patients received valganciclovir starting within 10 days post-transplantation until day 100 post-transplant and where kidney transplant paediatric patients received valganciclovir once daily starting within 10 days post transplantation until day 200 post-transplant.
The overall safety profile was similar in paediatric patients as compared to adults. However, the rates of certain adverse events, such as, but not limited to, upper respiratory tract infection, pyrexia, abdominal pain and dysuria, that may be characteristic of the paediatric population, were reported in somewhat higher incidence in paediatrics than in adults.
Neutropenia was also reported with slightly higher incidence in the two paediatric studies as compared to adults but neutropenia and infectious adverse events were generally not correlated in the paediatric populations.
In the first study, the primary toxicity associated with ganciclovir was neutropenia with 9 out of 24 subjects (38%) developing Grade 3 or 4 neutropenia while on therapy. Only one subject had to permanently discontinue antiviral treatment, due to neutropenia. Most events were manageable with continuation of antiviral therapy in this study. Growth (head circumference, weight and height) of all neonates, who had growth measurements recorded, increased over time in this non-comparative study.
In the second study, the most frequent treatment-related AEs associated with oral valganciclovir were neutropenia, anaemia, liver function abnormality and diarrhoea, all seen more frequently in the placebo group. The only treatment-related SAEs were neutropenia and anaemia, both seen more frequently in the placebo arm. No statistically or clinically significant differences were observed in the rate of growth (average head circumference, weight and length) over time at each time point between the two treatment groups.
Laboratory abnormalities. Laboratory abnormalities reported in adult CMV retinitis and SOT patients receiving with valganciclovir tablets until day 100 post-transplant are listed in Table 7. The incidence of laboratory abnormalities was comparable with the extension of prophylaxis up to 200 days in high risk kidney transplant patients.
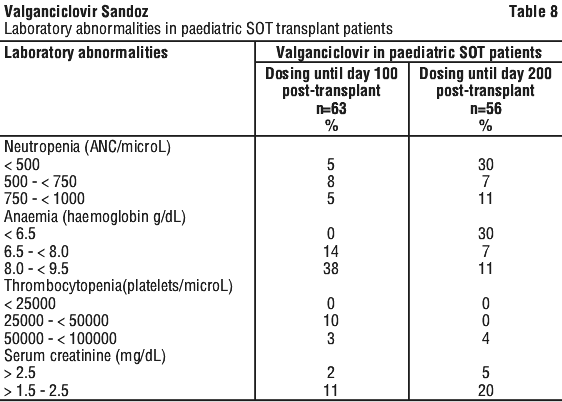
Laboratory abnormalities reported in paediatric SOT patients are listed in Table 8. The incidence of severe neutropenia (ANC < 500/microL) was higher in paediatric kidney patients treated until day 200 as compared to paediatric patients treated until day 100 and as compared to adults kidney transplant patients treated until day 100 or day 200.
Anaphylaxis.
Decreased fertility in males.
Agranulocytosis.
Granulocytopenia.
Adverse events that have been reported during the post-marketing period are consistent with those seen in clinical trials with valganciclovir and ganciclovir (see Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties, Clinical trials).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdose experience with valganciclovir tablets and intravenous valganciclovir. It is expected that an overdose of valganciclovir could also possibly result in increased renal toxicity (see Section 4.2 Dose and Method of Administration, Dosage adjustment, Renal impairment; Section 4.4 Special Warnings and Precautions for Use).
Overdose experience with intravenous ganciclovir. Reports of overdoses with intravenous ganciclovir, some with fatal outcomes, have been received from clinical trials and during post-marketing experience. In some of these cases, no adverse events were reported. The majority of patients experienced one or more of the following adverse events.
Haematological toxicity. Myelosuppression including pancytopenia, bone marrow depression, medullary aplasia, leucopenia, neutropenia, granulocytopenia.
Hepatotoxicity. Hepatitis, liver function disorder.
Renal toxicity. Worsening of haematuria in a patient with pre-existing renal impairment, acute renal failure, and elevated creatinine.
Gastrointestinal toxicity. Abdominal pain, diarrhoea, vomiting.
Neurotoxicity. Generalised tremor, convulsion.
Haemodialysis and hydration may be of benefit in reducing blood plasma levels in patients who receive an overdose of valganciclovir.
Treatment of overdose should consist of general supportive measures.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: antivirals for systemic use, nucleosides and nucleotides excl. reverse transcriptase inhibitors, ATC code: J05A B14.
Mechanism of action. Valganciclovir is an L-valyl ester salt (prodrug) of ganciclovir which, after oral administration, is rapidly converted to ganciclovir by intestinal and hepatic esterases. Ganciclovir is a synthetic analogue of 2'-deoxyguanosine, which inhibits replication of herpes viruses in vitro and in vivo. Sensitive human viruses include human cytomegalovirus (HCMV), herpes-simplex virus-1 and 2 (HSV-1 and HSV-2), human herpes virus type 6, 7 and 8 (HHV-6, HHV-7, HHV-8), Epstein-Barr virus (EBV), varicella-zoster virus (VZV) and hepatitis B virus.
In cytomegalovirus (CMV)-infected cells, ganciclovir is initially phosphorylated to ganciclovir monophosphate by the viral protein kinase, UL97. Further phosphorylation occurs by cellular kinases to produce ganciclovir triphosphate, which is then slowly metabolised intracellularly. This has been shown to occur in CMV-infected cells (half-life 18 hours) and HSV-infected cells (half-life between 6 and 24 hours) after removal of extracellular ganciclovir. As the phosphorylation is largely dependent on the viral kinase, phosphorylation of ganciclovir occurs preferentially in virus-infected cells.
The virustatic activity of ganciclovir is due to inhibition of viral DNA synthesis by: (a) competitive inhibition of incorporation of deoxyguanosine-triphosphate into DNA by viral DNA polymerase, and (b) incorporation of ganciclovir triphosphate into viral DNA causing termination of, or very limited, further viral DNA elongation. Typical anti-viral IC50 against CMV in vitro is in the range 0.08 microM (0.02 microgram/mL) to 14.32 microM (3.58 microgram/mL).
Pharmacodynamic effect. Valganciclovir allows systemic exposure of ganciclovir comparable to that achieved with recommended doses of intravenous (IV) ganciclovir, which has been shown to be efficacious in the treatment of CMV.
The clinical antiviral effect of valganciclovir has been demonstrated in the treatment of AIDS patients with newly diagnosed CMV retinitis (study WV15376). CMV shedding was decreased in urine from 46% (32/69) of patients at study entry to 7% (4/55) of patients following four weeks of valganciclovir treatment.
Viral resistance.Viral resistance to ganciclovir can arise after chronic dosing with valganciclovir by selection of mutations in either the viral kinase gene (UL97) responsible for ganciclovir monophosphorylation or the viral polymerase gene (UL54). UL97 mutations arise earlier and more frequently than mutations in UL54. Virus containing mutations in the UL97 gene is resistant to ganciclovir alone, with M460V/I, H520Q, C592G, A594V, L595S, C603W being the most frequently reported ganciclovir resistance-associated substitutions. Mutations in the UL54 gene may show cross-resistance to other antivirals targeting the viral polymerase and vice versa. Amino acid substitutions in UL54 conferring cross-resistance to ganciclovir and cidofovir are generally located within the exonuclease domains and region V, however amino acid substitutions conferring cross-resistance to foscarnet are diverse, but concentrate at and between regions II (codon 696-742) and III (codon 805-845).
Adult patients. Treatment of CMV retinitis in AIDS. Genotypic analysis of CMV in polymorphonuclear leucocytes (PMNL) isolates from 148 patients enrolled in one clinical study has shown that 2.2% (3/137), 6.5% (8/123), 12.8% (13/101) and 15.3% (13/85) contain UL97 mutations after 3, 6, 12 and 18 months, respectively, of valganciclovir treatment (using the number of patients still on treatment at the assessment time as the denominator). Phenotypic resistance was not identified, but very few CMV culture isolates were available for analysis.
Prevention of CMV disease in transplantation. Resistance was studied by genotypic analysis of CMV in PMNL samples collected i) on day 100 (end of study drug prophylaxis) and ii) in cases of suspected CMV disease up to 6 months after transplantation. From the 245 patients randomised to receive valganciclovir, 198 day 100 samples were available for testing and no ganciclovir resistance mutations were observed. This compares with 2 ganciclovir resistance mutations detected in the 103 samples tested (1.9%) for patients in the oral ganciclovir comparator arm.
Of the 245 patients randomised to receive valganciclovir, samples from 50 patients with suspected CMV disease were tested and no resistance mutations were observed. Of the 127 patients randomised on the ganciclovir comparator arm, samples from the 29 patients with suspected CMV disease were tested, from which two resistance mutations were observed, giving an incidence of resistance of 6.9%.
Resistance was evaluated in a study that extended valganciclovir CMV prophylaxis from 100 days to 200 days post-transplant in adult kidney transplant patients at high risk for CMV disease (D+/R-) (see Section 5.1 Pharmacodynamic Properties, Clinical trials). Five subjects from the 100 day group and four subjects from the 200 day group meeting the resistance analysis criteria had known ganciclovir resistance-associated amino acid substitutions detected. In six subjects, the following resistance associated amino acid substitutions were detected within pUL97: 100 day group: A440V, M460V, C592G; 200 day group: M460V, C603W. In three subjects, the following resistance-associated amino acid substitutions were detected within pUL54: 100 day group: E315D, 200 day group: E315D, P522S. Overall, the detection of known ganciclovir resistance-associated amino acid substitutions was observed more frequently in patients during prophylaxis therapy than after the completion of prophylaxis therapy (during therapy: 5/12 [42%] versus after therapy: 4/58 [7%]). The possibility of viral resistance should be considered in patients who show poor clinical response or experience persistent viral excretion during therapy.
Clinical trials. Adult patients. Study WV15376: treatment of CMV retinitis in AIDS. In a randomised, controlled study, 160 patients with AIDS and newly diagnosed CMV retinitis were randomised to receive treatment with either valganciclovir tablets (900 mg twice daily for 21 days, then 900 mg daily for 7 days) or with IV ganciclovir solution (5 mg/kg twice daily for 21 days, then 5 mg/kg daily for 7 days). Participants in the two treatment arms were comparable with respect to age, sex, weight, height and race. The mean age in the valganciclovir treatment arm was 39.6 years compared with 37.7 years in the ganciclovir arm. There was a higher proportion of males in each treatment group; 90% in the valganciclovir arm and 91% in the ganciclovir arm. The median CD4+ T-cell count at screening was 20.0 cells/microL for patients on the valganciclovir arm, and 26.0 cells/microL for patients on the ganciclovir arm; and the median HIV viral load was 4.8 log10 copies/mL in the valganciclovir arm and 4.9 log10 copies/mL in the ganciclovir arm.
In the final analysis of CMV retinitis progression by week 4 based on masked assessment of fundus photographs, 146 of 160 patients were included (73 in the valganciclovir tablets group and 73 in the IV ganciclovir group). The proportion of patients with retinitis progression at week 4 was the same in both treatment groups: 0.099 for the valganciclovir treatment group and 0.1 for the ganciclovir treatment group. The difference in progression proportions (IV ganciclovir minus valganciclovir tablets) was 0.001, with a 95% confidence interval of -0.097 to 0.100.
After week 4, all patients in this study were allowed to continue to receive treatment with valganciclovir tablets given at the dosage of 900 mg once daily. The mean (median) time from randomisation to progression of CMV retinitis in the group receiving induction and maintenance treatment with valganciclovir tablets (n = 80) was 226 (160) days and in the group receiving induction treatment with IV ganciclovir and maintenance treatment with valganciclovir tablets (n = 80) was 219 (125) days.
Satisfactory induction was achieved at week 4 in 47/61 (77%) patients given ganciclovir and 46/64 (72%) patients given valganciclovir. Satisfactory induction was defined as no progression, no increase in lesion activity and a reduction in retinitis border activity. Response was reassessed at 6 weeks when 39/62 (63%) patients given ganciclovir and 39/56 (70%) patients given valganciclovir maintained a satisfactory response to induction therapy. Three (8%) patients in each group had active retinitis at the week 6 assessment.
Study PV16000: prevention of CMV disease in solid organ transplantation. A double-blind, double-dummy clinical active comparator study has been conducted in 372 heart, liver and kidney transplant patients at high-risk for CMV disease (donor seropositive/ recipient seronegative [(D+/R-)]). The study was designed to test for non-inferiority between the 2 treatment arms. Patients were randomised (2 valganciclovir: 1 oral ganciclovir) to receive either valganciclovir (900 mg once daily) or oral ganciclovir (1000 mg three times daily) starting within 10 days of transplantation until day 100 post-transplant.
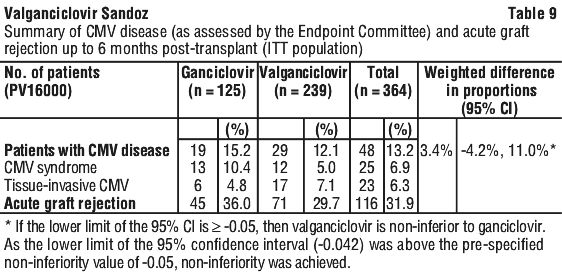
The primary analysis of the primary endpoint, the proportion of patients who developed CMV disease, including CMV syndrome and/or tissue invasive disease during the first 6 months post-transplant was 12.1% in the valganciclovir arm (n = 239) compared with 15.2% in the oral ganciclovir arm (n = 125) as assessed by a blinded Endpoint Committee. The study achieved its objective and it was concluded that valganciclovir was non-inferior to oral ganciclovir for the prevention of CMV disease in solid organ transplant patients.
The majority of cases of CMV disease occurred following cessation of prophylaxis (post-day 100) with cases in the valganciclovir arm occurring on average later than those in the oral ganciclovir arm. The incidence of acute rejection in the first 6 months was 29.7% in patients randomised to valganciclovir compared with 36.0% in the oral ganciclovir arm. For a summary of PV16000 see Table 9.
IMPACT study (study NT18435): prevention of CMV disease in kidney transplant patients. A double-blind, placebo controlled study has been conducted in 326 kidney transplant patients at high risk of CMV disease (D+/R-) to assess the efficacy and safety of extending valganciclovir CMV prophylaxis from 100 to 200 days post-transplant.
The inclusion criteria in this study required the patients to have adequate haematological (absolute neutrophil count > 1000 cells/microL, platelets > 25,000/microL, haemoglobin > 8 g/dL) and renal function (creatinine clearance > 15 mL/min and improving) in the immediate post-transplant period. The mean age of the patients who participated in this trial was about 48 years.
Patients were randomised (1:1) to receive valganciclovir tablets (900 mg once daily) within 10 days of transplantation until day 200 post-transplant or until day 100 post-transplant followed by 100 days placebo.
The proportion of patients who developed CMV disease during the first 12 months post-transplant is shown in Table 10.
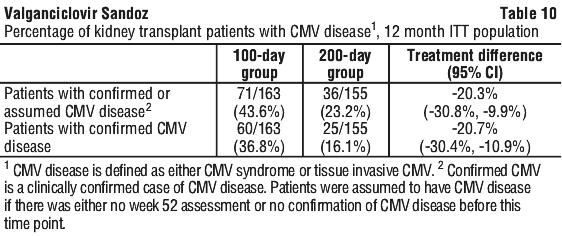
No clinical trials have been conducted in patients following haematological or lung transplants.
Paediatric population. Prevention of CMV disease in transplantation. Valganciclovir powder for oral solution has been studied in five open-label, multi-centre clinical trials in paediatric solid transplant (SOT) patients.
Three of these studies assessed only the pharmacokinetics and safety of oral valganciclovir in SOT patients aged from 4 weeks to 16 years of age who were requiring CMV prophylaxis (see Section 5.2 Pharmacokinetic Properties, Special populations). One study enrolled 20 liver transplant patients with a median age of 2 years (6 months to 16 years) who received a single daily dose of valganciclovir on 2 consecutive days. A second study enrolled 26 kidney patients with a median age of 12 years (1 to 16 years) who received multiple doses of valganciclovir on 2 consecutive days. The third study enrolled 14 heart transplant patients with a median age of 13 weeks (4 weeks to 125 days) who received a single daily dose of valganciclovir on 2 consecutive days. The other two studies assessed the development of CMV disease, as a measure of efficacy; following prophylaxis of valganciclovir for up to 100 days and 200 days post-transplant using the described paediatric dosing algorithm (see Section 4.2 Dose and Method of Administration).
One solid organ transplant study enrolled 63 kidney, liver or heart patients with a median age of 9 years (4 months to 16 years) who received multiple doses of valganciclovir for up to 100 days. There was no CMV event reported during the study that would fulfil the definition of CMV disease. CMV events were reported in 7 patients during the study of which 3 did not require adjustment to the study drug or were not treated and, therefore, were not considered clinically significant (see Section 4.8 Adverse Effects (Undesirable Effects); Section 5.2 Pharmacokinetic Properties). The second study in solid organ transplant enrolled 57 paediatric kidney patients with a median age of 12 years (1 to 16 years) who received daily doses of valganciclovir for up to 200 days. There was no CMV event reported during the study that would fulfill the definition of CMV disease. While 4 patients reported CMV events, one could not be confirmed by the central laboratory and of the 3 remaining events one did not require treatment and, therefore, was not considered clinically significant (see Section 4.8 Adverse Effects (Undesirable Effects)).
Congenital CMV. The efficacy and safety of ganciclovir and/or valganciclovir were studied in neonates and infants with congenital symptomatic CMV infection in two studies, with patients receiving up to 6 weeks or 6 months of treatment. The dose of valganciclovir that was determined in the first study and carried forward to the second study was twice daily doses of valganciclovir oral solution based on body weight using the following equation: Dose (mg) = 16 mg per kg of body weight.
Efficacy was evaluated using relevant endpoints such as hearing outcomes, neurodevelopmental outcomes and correlations of CMV blood viral load with ganciclovir plasma concentrations and hearing (see Section 4.8 Adverse Effects (Undesirable Effects)).
5.2 Pharmacokinetic Properties
The pharmacokinetic properties of valganciclovir have been evaluated in HIV- and CMV-seropositive patients, patients with AIDS and CMV retinitis and in solid organ transplant patients.
The parameters which control the exposure of ganciclovir from valganciclovir are the oral absorption of valganciclovir and the renal excretion of ganciclovir.
Absorption. Valganciclovir is a prodrug of ganciclovir, which is well absorbed from the gastrointestinal tract and rapidly metabolised in the intestinal wall and liver to ganciclovir. The absolute bioavailability of ganciclovir from valganciclovir is approximately 60%. Systemic exposure to valganciclovir is transient and low. AUC0-24h and Cmax values are approximately 1% and 3% of those of ganciclovir, respectively.
Dose proportionality with respect to ganciclovir AUC following administration of valganciclovir in the dose range 450 to 2625 mg was demonstrated only under fed conditions. When valganciclovir is given with food mean ganciclovir AUC0-24h increased by 24% to 56% depending on the dose. When valganciclovir was given with food at a dose of 875 mg, increases were seen in both mean ganciclovir AUC0-24h (approximately 30%) and mean ganciclovir Cmax values (approximately 14%). Therefore, it is recommended that valganciclovir be administered with food (see Section 4.2 Dose and Method of Administration).
For ganciclovir, average AUC0-24h has been shown to correlate with time to progression of CMV retinitis.
The bioavailability of ganciclovir from valganciclovir is comparable across all the patient populations studied (adult and paediatric). The systemic exposure of ganciclovir to heart, kidney and liver transplant recipients was similar after oral administration of valganciclovir according to the renal function dosing algorithm and paediatric dosing algorithm (see Section 4.2 Dose and Method of Administration).
Distribution. Due to the rapid conversion of valganciclovir to ganciclovir, protein binding of valganciclovir was not determined. Plasma protein binding of ganciclovir was 1% to 2%. The steady state volume of distribution of ganciclovir after IV administration was 0.680 ± 0.161 L/kg. For IV ganciclovir, the volume of distribution is correlated with body weight with values for the steady state volume of distribution ranging from 0.54−0.87 L/kg. Ganciclovir penetrates the cerebrospinal fluid.
Metabolism. Valganciclovir is rapidly hydrolysed to ganciclovir; no other metabolites have been detected. Ganciclovir itself is not metabolized to a significant extent.
Excretion. Following dosing with oral valganciclovir, the drug is rapidly hydrolysed to ganciclovir. Ganciclovir is eliminated from the systemic circulation by glomerular filtration and active tubular secretion.
The terminal half-life (t½) of ganciclovir following oral administration of valganciclovir to either healthy or HIV- and CMV-positive subjects was 4.18 ± 0.80 hours (n = 244), and that following administration of IV ganciclovir was 3.85 ± 0.74 hours (n = 87).
In patients undergoing haemodialysis, approximately half of the ganciclovir present at the start of a dialysis session is removed during dialysis. The mean intra-dialysis half-life and the mean inter-dialysis half-life was estimated to be 3.47 hours and 51.0 hours, respectively.
Pharmacokinetics in special populations. Patients with renal impairment. The pharmacokinetics of ganciclovir from a single oral dose of 900 mg valganciclovir were evaluated in 24 individuals with renal impairment without HIV and/or CMV infections. See Table 11.
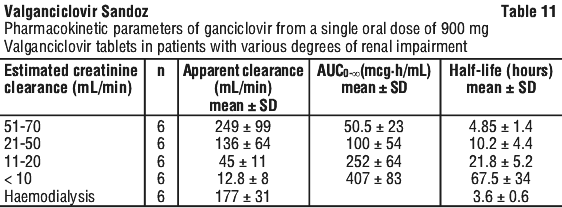
Patients undergoing haemodialysis. Ganciclovir is readily removable by haemodialysis. Data obtained during intermittent haemodialysis in patients dosed with valganciclovir showed estimated dialysis clearance as 138 mL/min ± 9.1% (N = 3) and intra-dialysis half-life estimated to 3.47 h (N = 6). 55% of ganciclovir was removed during a 3 hour dialysis session.
Haemodialysis reduces plasma concentrations of ganciclovir by about 42 to 55% following valganciclovir administration. Adult patients receiving haemodialysis (CrCl less than 10 mL/min) cannot use valganciclovir tablets because the daily dose of valganciclovir tablets required for these patients is less than 450 mg (see Section 4.2 Dose and Method of Administration).
Stable liver transplant patients. The pharmacokinetics of valganciclovir in stable liver transplant recipients were investigated in one open label 4-part cross-over study (n = 28). The absolute bioavailability of ganciclovir from valganciclovir following a single dose of 900 mg valganciclovir under fed conditions was approximately 60%. Ganciclovir AUC0-24h was comparable to that achieved by 5 mg/kg IV ganciclovir in liver transplant recipients.
Patients with hepatic impairment. No pharmacokinetic study has been conducted and no population PK data was collected in patients with hepatic impairment undergoing valganciclovir therapy (see Section 4.4 Special Warnings and Precautions for Use; Section 4.2 Dose and Method of Administration).
Elderly. No investigations on valganciclovir or ganciclovir pharmacokinetics in adults older than 65 years of age have been undertaken. However as valganciclovir is a pro-drug of ganciclovir and because ganciclovir is mainly renally excreted and since renal clearance decreases with age, a decrease in ganciclovir total body clearance and a prolongation of ganciclovir half-life can be anticipated in elderly (see Section 4.2 Dose and Method of Administration).
Paediatric patients. Prevention of CMV disease in transplantation. The pharmacokinetics of ganciclovir following the administration of valganciclovir were characterised using a population PK model based on data from four studies in paediatric solid organ transplant patients aged 3 weeks to 16 years. PK data were evaluable from 119 of the 123 patients enrolled. In these studies, patients received daily intravenous doses of ganciclovir to produce exposure equivalent to an adult 5 mg/kg intravenous dose (70 kg reference body weight) and/or received oral doses of valganciclovir to produce exposure equivalent to an adult 900 mg dose.
The model indicated that clearance is influenced by body weight and creatinine clearance while the central and peripheral volumes of distribution were influenced by body weight (see Section 4.2 Dose and Method of Administration).
The mean ganciclovir Cmax, AUC and half-life by age and organ type in studies using the paediatric dosing algorithm are listed in Table 12 and are consistent with estimates obtained in adult solid organ transplant patients.
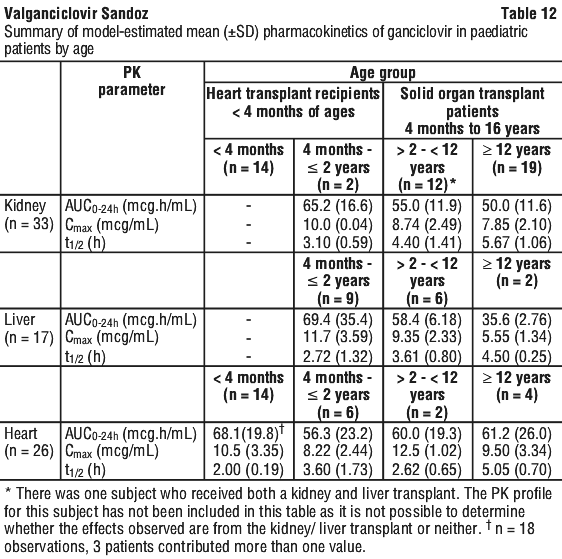
The mean ganciclovir AUC0-12hr after oral dose administration of valganciclovir was approximately 23.2 microgram.h/mL (equivalent to 46.4 microgram.h/mL in AUC0-24hr) in the first study. Similar exposure was also observed in the second study.
5.3 Preclinical Safety Data
Ganciclovir was genotoxic and carcinogenic in animal studies.
Genotoxicity. Valganciclovir increased mutations in mouse lymphoma cells and was clastogenic in the mouse micronucleus assay. Valganciclovir was not mutagenic in the Ames Salmonella assay.
Ganciclovir increased mutations in mouse lymphoma cells and DNA damage in human lymphocytes in vitro. Ganciclovir was clastogenic in the mouse micronucleus assay. Ganciclovir was not mutagenic in the Ames Salmonella assay.
Carcinogenicity. Ganciclovir was genotoxic and carcinogenic in animal studies. Valganciclovir should be considered a potential carcinogen in humans with the potential to cause cancers. No long-term carcinogenicity studies have been conducted with valganciclovir. However, upon oral administration, valganciclovir is rapidly and extensively converted to ganciclovir.
Toxicity in mice, dogs and rats was primarily characterised by testicular atrophy. Male infertility occurred at doses of 2 mg/kg/day and above which was consistent with the infertility and testicular atrophy seen in toxicity studies with doses between 2 and 10 mg/kg/day. In females, a more complex range of effects were induced which were characterised by embryo-foetal abnormalities and embryo-foetal losses in mice and rabbits and in multi-dose studies, by toxic and eventually carcinogenic changes to the reproductive system in mice.
Ganciclovir was carcinogenic in the mouse after oral doses of 20 mg/kg/day for 18 months and 1000 mg/kg/day for 15 months. All ganciclovir-induced tumours were of haematopoietic epithelial or vascular origin. Epithelial tumours involved a wide variety of tissues, including the female reproductive organs, pancreas, gastrointestinal tract and skin, as well as rodent specific glands (preputial, clitoral and Harderian). Vascular tumours were observed in females, mainly in the reproductive organs, but also in the mesenteric lymph nodes and liver. No carcinogenic effects occurred at 1 mg/kg/day. Based on data on plasma drug concentrations, exposure of humans to ganciclovir would be similar to or greater than the exposure of mice in the above study at 1000 mg/kg/day. This potential is likely to be markedly greater in children, as cell division occurs more rapidly in children.
6 Pharmaceutical Particulars
6.1 List of Excipients
The excipients are microcrystalline cellulose, crospovidone, stearic acid and povidone. The film coat applied to the tablets contains Opadry Pink 15B24005 which consists of hypromellose, titanium dioxide, macrogol 400, iron oxide red and polysorbate 80.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
For information on interactions with other medicines and other forms of interactions, see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Valganciclovir Sandoz tablets should be stored at below 25°C.
Protect from light.
6.5 Nature and Contents of Container
Valganciclovir Sandoz is supplied in blister packs (Al/Al) of 60 tablets.
6.6 Special Precautions for Disposal
Caution should be exercised in the handling of valganciclovir tablets. Valganciclovir tablets should not be broken or crushed. Since valganciclovir is considered a potential teratogen and carcinogen in humans and inhibits spermatogenesis, caution should be observed in handling valganciclovir tablets. Avoid direct contact of broken or crushed tablets with skin or mucous membranes. If such contact occurs, wash thoroughly with soap and water, and rinse eyes thoroughly with sterile water, or plain water if sterile water is unavailable.
The release of medicines into the environment should be minimised. Medicines should not be disposed of via wastewater and disposal through household waste should be avoided. Unused or expired medicine should be returned to a pharmacy for disposal.
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Valganciclovir hydrochloride (valganciclovir HCl) is the hydrochloride salt of the L-valyl ester of ganciclovir. Ganciclovir is a synthetic nucleoside analogue of guanine.
Valganciclovir HCl is a white to off white crystalline powder.
Valganciclovir HCl is a polar hydrophilic compound with a solubility of 70 mg/mL in water at 25°C at a pH of 7.0 and an n-octanol/water partition coefficient of 0.0095 at pH 7.0. The pKa for valganciclovir is 7.6.
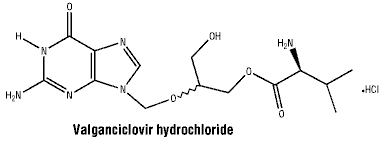
Chemical structure. The chemical name of valganciclovir hydrochloride is L-valine, 2-[(2-amino-1,6 dihydro-6-oxo-9H-purin-9-yl)methoxy]-3-hydroxypropyl ester, monohydrochloride. The molecular formula is C14H22N6O5.HCl (MW: 390.83) and its chemical structure is:
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
09 December 2016
Date of Revision
25 March 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.