Vaxigrip
Brand Information
| Brand name | Vaxigrip |
| Active ingredient | Influenza virus vaccine, split virion, trivalent (inactivated) |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Vaxigrip.
Summary CMI
Vaxigrip®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Vaxigrip?
Vaxigrip is a vaccine. This vaccine helps to protect you or your child against influenza (flu). Vaxigrip is used to prevent flu in persons of 6 months of age and older. If you are pregnant, one dose of vaccine given to you during pregnancy may protect your baby from birth to less than 6 months of age.
For more information, see Section 1. Why am I using Vaxigrip? in the full CMI.
2. What should I know before I am given Vaxigrip?
Do not use if you have ever had an allergic reaction to Vaxigrip or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am given Vaxigrip? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Vaxigrip and affect how it works. Tell your doctor, nurse or pharmacist if you are taking, have recently taken or might take any other vaccines or medicines, including medicines obtained without a prescription.
For more information, see Section 3. What if I am taking other medicines? in the full CMI.
4. How is Vaxigrip given?
Vaxigrip is given by your doctor, nurse or pharmacist.
More instructions can be found in Section 4. How is Vaxigrip given? in the full CMI.
5. What should I know about after being given Vaxigrip?
| Things you should do | Call your doctor straight away if: you notice signs of allergic reaction may include difficulty breathing, shortness of breath, swelling of the face, lips, throat or tongue, rapid heart rate and weak pulse, cold, clammy skin, palpitations, dizziness, weakness, fainting, rash or itching. |
| Looking after your medicine | Vaxigrip is usually stored in the surgery or clinic, or at the pharmacy. However, if you need to store Vaxigrip:
|
For more information, see Section 5. What should I know about after being given Vaxigrip? in the full CMI.
6. Are there any side effects?
Serious side effects can include severe allergic reactions, pain situated on the nerve route, fits, neurological disorders that may result in stiff neck, confusion, numbness, pain and weakness of the limbs, loss of balance, loss of reflexes, paralysis of part or all of the body and blood vessel inflammation which may result in skin rashes and in very rare cases temporary kidney problems. See your doctor immediately if you notice this.
Common side effects include pain, tenderness, redness, swelling, bruising and hardness at the injection site, headache, muscle aches, feeling unwell, fever, and shivering. In children, other common side effects include irritability, abnormal crying, drowsiness, appetite loss and vomiting. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Vaxigrip®
Active ingredient(s): Influenza virus haemagglutinin
Consumer Medicine Information (CMI)
This leaflet provides important information about using Vaxigrip. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Vaxigrip.
This vaccine can be given to adults and children so you may be reading this leaflet for you or for your child.
Where to find information in this leaflet:
1. Why am I using Vaxigrip?
2. What should I know before I am given Vaxigrip?
3. What if I am taking other medicines?
4. How is Vaxigrip given?
5. What should I know about after being given Vaxigrip?
6. Are there any side effects?
7. Product details
1. Why am I using Vaxigrip?
Vaxigrip contains the active ingredient influenza virus haemagglutinin.
Vaxigrip is a vaccine for persons 6 months of age and older. This vaccine helps to protect you or your child against influenza (flu). If you are pregnant, one dose of vaccine given to you during pregnancy may protect your baby from birth to less than 6 months of age.
When a person is given the vaccine, the immune system (the body's natural defence system) will produce its own protection against the influenza virus. None of the ingredients in the vaccine can cause flu.
Flu is a disease that can spread rapidly and is caused by different types of strains that can change every year. This is why you need to be vaccinated every year. The greatest risk of catching flu is during the cold months between June and September. If you were not vaccinated in the autumn, it is still sensible to be vaccinated up until the spring since you run the risk of catching flu until then. Your doctor will be able to recommend the best time to be vaccinated.
As with all vaccines, Vaxigrip may not fully protect all persons who are vaccinated.
2. What should I know before I am given Vaxigrip?
Warnings
Do not use Vaxigrip:
- if you are allergic to the active ingredients or any of the ingredients listed at the end of this leaflet. Symptoms of allergic reaction may include difficulty breathing, shortness of breath, swelling of the face, lips, throat or tongue, low blood pressure, rapid heart rate and weak pulse, cold, clammy skin, palpitations, dizziness, weakness, fainting, rash or itching. If you are not sure if you are allergic, talk to your doctor, nurse or pharmacist before you receive Vaxigrip.
Always check the ingredients to make sure you can receive this vaccine.
Tell your doctor, nurse or pharmacist:
- If you have an acute illness with or without high temperature.
- If you have or have had an immune response problem because the immune response to the vaccine may be diminished.
- If you have a bleeding problem or bruise easily.
- If you have ever fainted from an injection. Fainting, sometimes with falling, can occur during, following, or even before, any injection with a needle.
- If you have or have had Guillain-Barré syndrome (severe muscle weakness) after getting a flu vaccine.
- If you have a known allergy to egg protein.
After vaccination, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. Your doctor will discuss the possible risks and benefits of having Vaxigrip during pregnancy or breastfeeding.
Vaxigrip can be used in all stages of pregnancy.
Vaxigrip may be used during breast-feeding.
Due to the known adverse consequences of influenza infection in pregnant women, health authorities recommend vaccination for pregnant women.
Your doctor should make sure the benefits of vaccination outweigh the risks when recommending Vaxigrip.
3. What if I am taking other medicines?
Some medicines may interfere with Vaxigrip and affect how it works. Tell your doctor, nurse or pharmacist if you are taking, have recently taken or might take any other vaccines or medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Your doctor will advise you if Vaxigrip is to be given with another vaccine.
Check with your doctor, nurse or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Vaxigrip.
4. How is Vaxigrip given?
How much is given
Vaxigrip is given by your doctor, nurse or pharmacist as a 0.5 mL injection in the muscle in the upper arm (preferably). For infants, the injection is normally given into the muscle of the thigh.
Children less than 9 years old, who have not been vaccinated against influenza before require a second injection a month later. Doses of influenza vaccine for infants and young children are decided by your doctor based on the official national recommendations.
5. What should I know about after being given Vaxigrip?
Things you should do
Call your doctor straight away if:
You notice signs of allergic reaction may include difficulty breathing, shortness of breath, swelling of the face, lips, throat or tongue, cold, clammy skin, palpitations, dizziness, weakness, fainting, rash or itching.
Driving or using machines
Do not drive or use machines if you are feeling unwell after vaccination. Wait until any effects of the vaccine have worn off before you drive or use machines.
Looking after your medicine
Vaxigrip is usually stored in the doctor's surgery or clinic, or at the pharmacy. However, if you need to store Vaxigrip:
- keep it where young children cannot reach it.
- keep Vaxigrip in the original pack until it is time for it to be given.
- keep it in the refrigerator, store at 2°C to 8°C. Do not freeze Vaxigrip.
Do not use Vaxigrip after the expiry date which is stated on the carton after EXP.
Do not use Vaxigrip if the packaging is torn or shows signs of tampering.
Getting rid of any unwanted medicine
Medicines including vaccines should not be disposed of via wastewater or household waste. Ask your doctor, nurse or pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor, nurse or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor, nurse or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
What Vaxigrip contains
| Active ingredient (main ingredient) | Influenza virus haemagglutinin of the following strains
|
| Other ingredients (inactive ingredients) | Sodium chloride, potassium chloride, dibasic sodium phosphate dihydrate, monobasic potassium phosphate, water for injections, and traces of ovalbumin (egg protein), neomycin, octoxinol-9 and formaldehyde. |
| Potential allergens | Vaxigrip may contain less than 0.05 microgram ovalbumin (egg protein) per dose. |
Do not take this vaccine if you are allergic to any of these ingredients.
Vaxigrip syringes are not made with natural rubber latex.
What Vaxigrip looks like
Vaxigrip suspension for injection is clear and slightly opalescent in colour.
Vaxigrip is available in packs of 1 or 10 single dose (0.5 mL) pre-filled syringes with one separate needle or no needle provided per syringe.
AUST R 465534 – prefilled syringe needle free *
*Not all pack sizes may be marketed.
Who distributes Vaxigrip
Distributed by:
sanofi-aventis australia pty ltd
12-24 Talavera Road
Macquarie Park NSW 2113
Freecall: 1800 818 806
Email: medinfo.australia@sanofi.com
This leaflet was prepared in December 2025.
vaxi-ccdsv14-cmiv2-03dec25
Brand Information
| Brand name | Vaxigrip |
| Active ingredient | Influenza virus vaccine, split virion, trivalent (inactivated) |
| Schedule | S4 |
MIMS Revision Date: 01 March 2026
1 Name of Medicine
Inactivated trivalent influenza vaccine, split virion (Influenza virus haemagglutinin).
2 Qualitative and Quantitative Composition
Vaxigrip is formulated to contain the following three influenza strains*. See Table 1.

For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Suspension for injection in pre-filled syringe.
The vaccine, after shaking gently, is a colourless opalescent liquid.
4 Clinical Particulars
4.1 Therapeutic Indications
Vaxigrip is indicated for the prevention of influenza disease caused by the two influenza A virus subtypes and the one influenza B virus type contained in the vaccine for:
active immunisation of adults, including pregnant women, and children from 6 months of age and older;
passive protection of infant(s) from birth to less than 6 months of age following vaccination of pregnant women (see Section 4.2 Dose and Method of Administration; Section 4.4 Special Warnings and Precautions for Use; Section 4.6 Fertility, Pregnancy and Lactation; Section 5.1 Pharmacodynamic Properties, Clinical trials).
Vaxigrip should be given in accordance with the national recommendation as per the current Immunisation Handbook.
4.2 Dose and Method of Administration
Given the antigenic variation in circulating influenza viruses and the duration of immunity provided by the vaccine, it is recommended to perform vaccination against influenza every year at the beginning of the risk period.
Individuals from 9 years of age. A single 0.5 mL injection.
Children from 6 months to 8 years of age. If the child has not previously been vaccinated. Two 0.5 mL injections at least one month apart.
If the child has been previously vaccinated. A single 0.5 mL injection.
Infants younger than 6 months of age. No data are available regarding the safety and efficacy of Vaxigrip administration (active immunisation) in infants younger than 6 months of age.
Regarding passive protection, a single 0.5 mL injection given to pregnant women may protect infants from birth to less than 6 months of age; however not all these infants will be protected (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Method of administration. The vaccine should be given by intramuscular or deep subcutaneous injection.
The preferred site of administration is into the deltoid muscle in adults and children ≥ 12 months of age. The preferred site for infants (6 months to < 12 months of age) is the anterolateral aspect of the thigh. The vaccine should be administered into healthy well developed muscle and should not be injected into the gluteal region where there may be a risk of local neural, vascular and tissue injury.
Shake before use to distribute uniformly the suspension before administration.
Parenteral drug products should be inspected visually for particulate matter and/or discolouration prior to administration whenever solution and container permit. If either of these conditions exists, the vaccine should not be administered.
The syringe is for single use only in one patient and must not be reused. Discard any remaining unused contents.
4.3 Contraindications
Vaxigrip should not be given to individuals with a history of severe allergic reaction to any component of the vaccine (see Section 2 Qualitative and Quantitative Composition; Section 6.1 List of Excipients). Vaxigrip should not be given to individuals with a history of severe allergic reaction after previous administration of Vaxigrip or a vaccine containing the same components.
See Section 4.4 Special Warnings and Precautions for Use for influenza vaccination for individuals with a known egg allergy.
Vaccination should be postponed in case of moderate or severe febrile or acute disease.
4.4 Special Warnings and Precautions for Use
Vaxigrip should under no circumstances be administered intravenously.
Hypersensitivity. Prior to any vaccine injection, all known precautions should be taken to prevent hypersensitivity reactions. This includes a review of the individual's prior vaccination history with respect to possible hypersensitivity to the vaccine or similar vaccines. Adrenaline (epinephrine) injection (1:1000) and other appropriate agents used for the control of immediate allergic reactions must be available to treat unexpected reactions (e.g. anaphylaxis).
Individuals with egg allergy, including a history of anaphylaxis, can be safely vaccinated with influenza vaccines. Refer to the current Immunisation Handbook for guidance on the use of influenza vaccines in individuals with egg allergy.
Vaxigrip may contain traces of formaldehyde and octoxinol 9 which are used during vaccine production, caution should be exercised when the vaccine is administered to individuals with hypersensitivity to either one of these products.
As each dose may contain undetectable traces of neomycin, which is used during vaccine production, caution should be exercised when the vaccine is administered to individuals with hypersensitivity to this antibiotic (and other antibiotics of the same class).
Neurological disorders. Patients with a history of Guillain-Barré syndrome (GBS) with an onset related in time to influenza vaccination may be at increased risk of again developing GBS, but whether vaccination specifically might increase the risk for recurrence is unknown. Because patients with a history of GBS have an increased likelihood of again developing the syndrome, the chance of them coincidentally developing the syndrome following influenza vaccination may be higher than in individuals with no history of GBS. If GBS has occurred within 6 weeks following previous influenza vaccination, the decision to give Vaxigrip should be based on careful consideration of the potential benefits and risks.
Immunosuppressive treatments or conditions. If Vaxigirip is administered to immunocompromised individuals, whether due to genetic defect, immunodeficiency disease, or immunosuppressive therapy, they may have a reduced immune response to vaccination. For current recommendation, refer to the current Immunisation Handbook.
Protection. As with any vaccine, vaccination with Vaxigrip may not protect 100% of recipients.
Regarding passive protection, not all infants younger than 6 months of age born to women vaccinated during pregnancy will be protected (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Influenza virus is remarkably unpredictable in that significant antigenic changes may occur from time to time. It is known that influenza vaccines, as now constituted, are not effective against all possible strains of influenza virus. Protection is limited to those strains of virus from which the vaccine is prepared or to closely related strains.
Bleeding disorders. As with other vaccines administered intramuscularly, the vaccine should be administered with caution to individuals with thrombocytopenia or a bleeding disorder since bleeding may occur following an intramuscular administration to these individuals.
Syncope. Syncope (fainting) can occur following, or even before, any vaccination as a psychogenic response to the needle injection. Procedures should be in place to prevent injury from fainting and manage syncopal reactions.
Use in the elderly. Annual influenza vaccination is recommended for individuals 65 years of age and over.
Paediatric use. Infants younger than 6 months of age. No data are available regarding the safety and efficacy of Vaxigrip administration (active immunisation) in infants younger than 6 months of age. Regarding passive protection, one 0.5 mL dose given to pregnant women may protect infants from birth to less than 6 months of age; however not all these infants will be protected (see Section 4.2 Dose and Method of Administration; Section 5.1 Pharmacodynamic Properties, Clinical trials).
Effects on laboratory tests. Interference of Vaxigrip with laboratory and/or diagnostic tests has not been studied.
Following influenza vaccination, false positive results in serology tests using the ELISA method to detect antibodies against HIV1, Hepatitis C and especially HTLV1 have been observed. The Western Blot technique can be used to disprove these results. The transient false positive reactions could be due to IgM response by the vaccine.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Clinical data showing that Vaxigrip can be administered concomitantly with other vaccines are available for the following vaccines: 23-valent pneumococcal polysaccharide vaccine in elderly, dTpa-IPV (diphtheria-tetanus-acellular pertussis-inactivated poliovirus vaccine) in adults aged ≥ 60 years, and zoster vaccine in adults aged 50 and older.
Vaxigrip can be given at the same time as other vaccines.
Separate injection sites and separate syringes should be used in case of concomitant administration.
Individuals deficient in producing antibodies due to immunosuppressive therapy may have a reduced immune response to vaccination.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There are no fertility data available in humans. One animal study with Vaxigrip Tetra (Quadrivalent Influenza Vaccine (QIV)) did not indicate harmful effects on female fertility in rabbits.
Use in pregnancy. (Category A)
Animal reproductive studies have not been conducted with Vaxigrip. One development and reproductive study conducted in rabbits with Vaxigrip Tetra (QIV) can be extrapolated to Vaxigrip: this study did not indicate direct or indirect harmful effects with respect to pregnancy, embryo-fetal development or early post-natal development.
Data from studies involving large numbers of women (> 80,000) vaccinated during pregnancy with inactivated influenza vaccines do not indicate any adverse fetal and maternal outcomes attributable to the vaccine.
Data from four clinical studies conducted with Vaxigrip administered to pregnant women during the second and third trimesters (more than 5,000 exposed pregnancies and more than 5,000 live births, followed up to approximately 6 months postpartum) did not indicate any adverse fetal, newborn, infant, or maternal outcomes attributable to the vaccine.
In three of these clinical studies conducted in South Africa and Nepal, there were no significant differences between the Vaxigrip and placebo groups with regards to fetal, newborn, infant, or maternal outcomes (including miscarriage, stillbirth, premature birth, low birth weight).
In the fourth study conducted in Mali, there were no significant differences between the Vaxigrip and control vaccine (quadrivalent meningococcal conjugate vaccine) groups with regards to prematurity rate, stillbirth rate, or rate of low birth weight/small for gestational age.
Data from a clinical study conducted in Finland with Vaxigrip and Vaxigrip Tetra (QIV) administered in pregnant women during the second or third trimester (116 exposed pregnancies and 119 live births for Vaxigrip, 230 exposed pregnancies and 231 live births for Vaxigrip Tetra (QIV)) did not indicate any adverse fetal or maternal outcomes attributable to the vaccine.
Vaxigrip can be administered in all stages of pregnancy based on the safety data from clinical studies and post-marketing experience. Larger datasets on safety of inactivated influenza vaccines are available for the second and third trimesters than for the first trimester. Data from worldwide use of inactivated influenza vaccines, including Vaxigrip and Vaxigrip Tetra (QIV), do not indicate any adverse fetal and maternal outcomes attributable to the vaccine. Because of the known adverse consequences of influenza infection in pregnant women, health authorities recommend vaccination of pregnant women.
Use in lactation. There are no data on the effect of the vaccine in breastfed newborns/infants of women vaccinated with Vaxigrip during breastfeeding period. Based on experience with inactivated influenza vaccines, Vaxigrip may be used during breastfeeding.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed.
4.8 Adverse Effects (Undesirable Effects)
Within each system organ class, the adverse events are ranked under headings of frequency, using the following convention: very common ≥ 1/10 (≥ 10%); common ≥ 1/100 and < 1/10 (≥ 1% and < 10%); uncommon ≥ 1/1000 and < 1/100 (≥ 0.1% and < 1%); rare ≥ 1/10,000 and < 1/1000 (≥ 0.01% and < 0.1%); very rare < 1/10,000 (< 0.01%); not known: cannot be estimated from available data.
Clinical trial data. The safety profile of Vaxigrip is based on integrated safety analysis from clinical studies conducted with Vaxigrip and Vaxigrip Tetra (QIV). A total number of 17,917 individuals received an intramuscular or a subcutaneous injection of Vaxigrip or Vaxigrip Tetra (QIV).
Most reactions usually occurred within the first 3 days following vaccination and resolved spontaneously within 1 to 3 days after onset. The intensity of most of these reactions was mild to moderate.
The most frequently reported adverse reaction after vaccination was injection site pain in all populations including children from 6 to 35 months of age.
In the subpopulation of children less than 24 months of age, irritability (34.9%) was the most frequently reported adverse reaction.
In the subpopulation of children from 24 to 35 months of age, malaise (26.8%) was the most frequently reported adverse reaction.
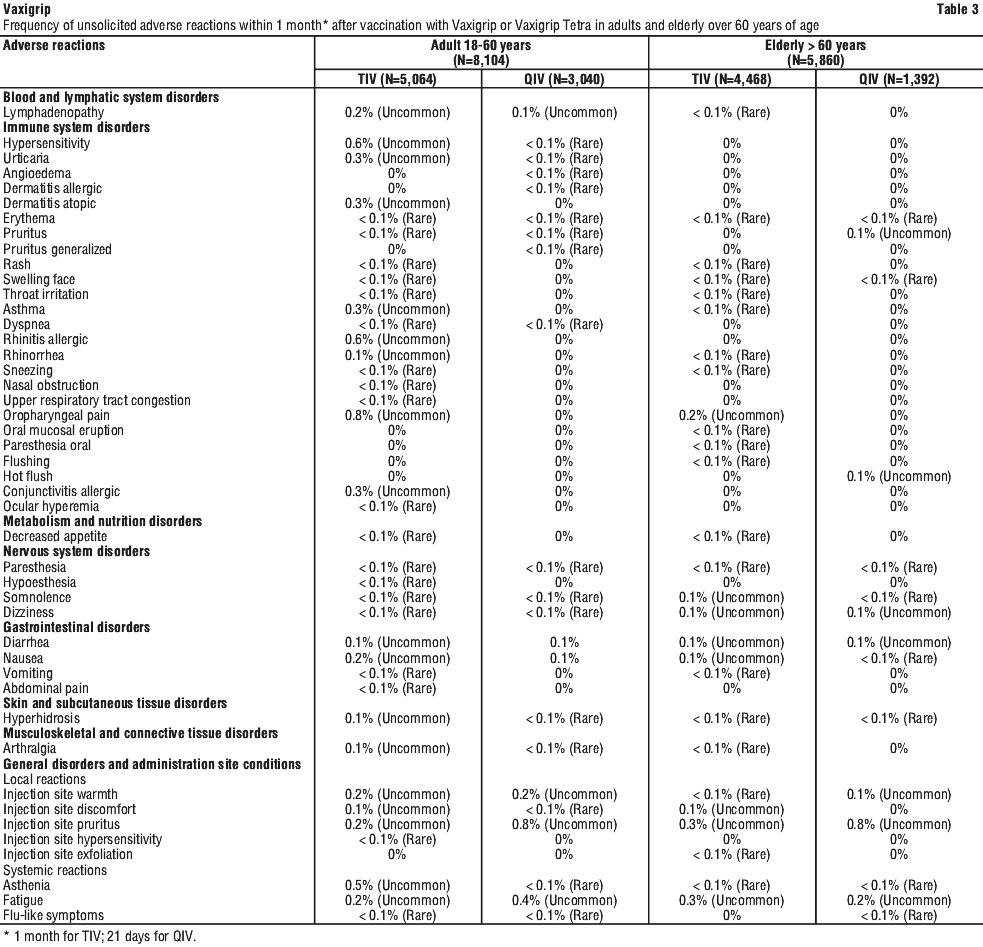
Adults and the elderly. A total of 8,104 adults from 18 to 60 years and 5,860 adults over 60 years of age received one 0.5 mL dose of Vaxigrip or of Vaxigrip Tetra (QIV) during clinical studies.
In adults, the most frequently reported adverse reactions after vaccination were injection site pain (52.8%), headache (27.8%), myalgia (23.0%), and malaise (19.2%).
In the elderly, the most frequently reported adverse reactions after vaccination were injection site pain (25.8%), headache (15.6%), and myalgia (13.9%).
Overall, adverse reactions were generally less frequent in the elderly than in adults and children. See Table 2 and Table 3.

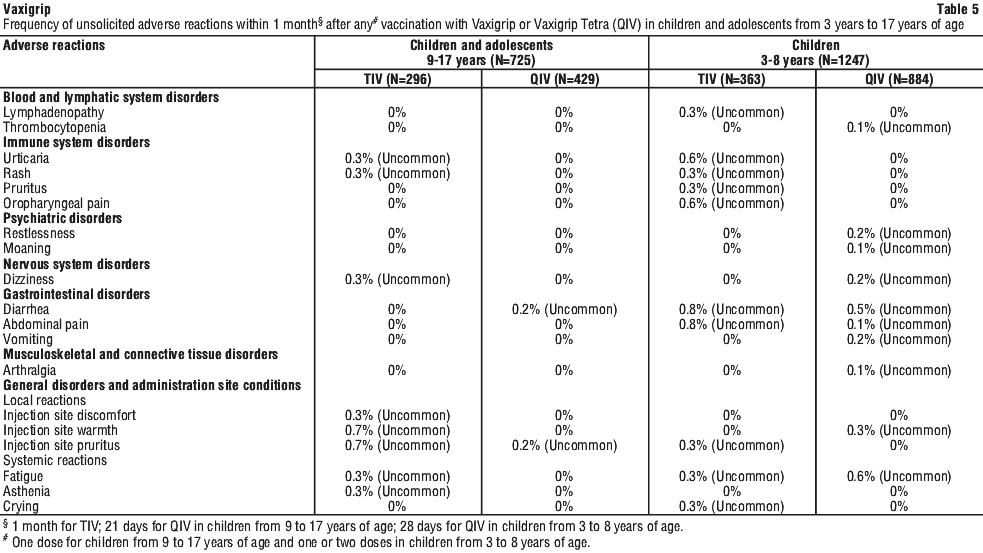
In children/adolescents from 9 to 17 years of age, the most frequently reported adverse reactions after vaccination were injection site pain (65.3%), myalgia (29.1%), headache (28.6%), malaise (20.3%), shivering (13.0%), injection site erythema (11.7%) and injection site swelling (11.4%).
In children from 3 to 8 years of age, the most frequently reported adverse reactions after any vaccination were injection site pain (59.1%), malaise (30.7%), injection site erythema (30.3%), myalgia (28.5%), headache (25.7%), injection site swelling (22.1%), injection site induration (17.6%), and shivering (11.2%).
The safety profile was similar after the first and the second injections.
Table 4 and Table 5 summarise the frequencies of solicited and unsolicited adverse reactions that were recorded after any vaccination in children and adolescents from 3 years to 17 years of age.

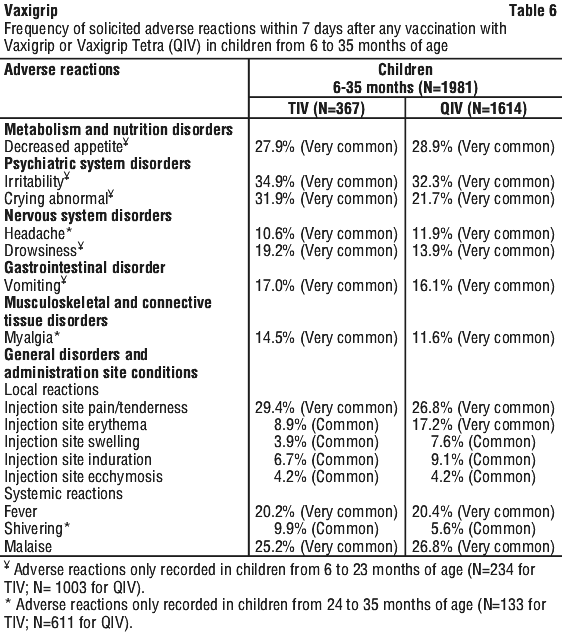
In children from 6 to 35 months of age, the most frequently reported adverse reactions after any vaccination were injection site pain/tenderness (29.4%), fever (20.4%) and injection site erythema (17.2%).
In the subpopulation of children from 6 to 23 months of age, the most frequently reported adverse reactions after any vaccination were irritability (34.9%), crying abnormal (31.9%), appetite lost (28.9%), drowsiness (19.2%) and vomiting (17.0%).
In the subpopulation of children from 24 to 35 months of age, the most frequently reported adverse reactions after any vaccination were malaise (26.8%), myalgia (14.5%), and headache (11.9%).
The safety profile was similar after the first and the second injections with a trend of lower incidence of adverse reactions after the second injection compared to the first one.
Table 6 and Table 7 summarise the frequencies of solicited and unsolicited adverse reactions that were recorded after any vaccination in children from 6 to 35 months of age.
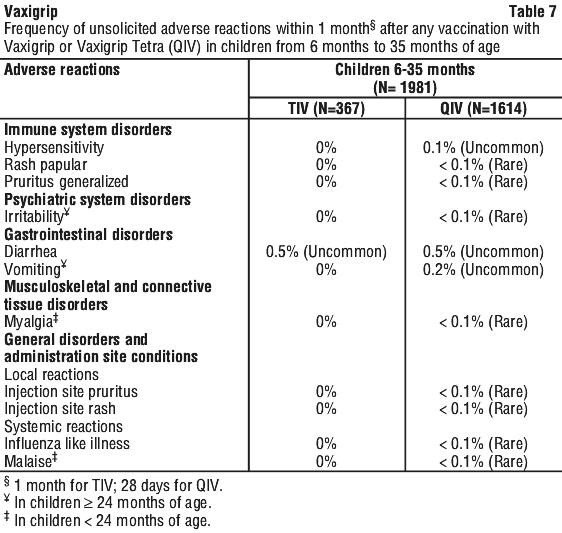
Pregnant women. In clinical studies conducted in pregnant women in South Africa and Mali with Vaxigrip (see Section 4.6 Fertility, Pregnancy and Lactation; Section 5.1 Pharmacodynamic Properties, Clinical trials), frequencies of local and systemic solicited reactions reported within 7 days following administration of Vaxigrip, were generally consistent with those reported for the adult population during clinical studies. In the study conducted in South Africa, local reactions were more frequent in the Vaxigrip group than in the placebo group in both HIV-negative and HIV-positive cohorts. There were no other significant differences in solicited reactions between Vaxigrip and placebo groups in both cohorts.
In one clinical study conducted in pregnant women in Finland with Vaxigrip and Vaxigrip Tetra (QIV) (see Section 4.6 Fertility, Pregnancy and Lactation; Section 5.1 Pharmacodynamic Properties, Immunogenicity of Vaxigrip), frequencies of local and systemic solicited reactions reported were consistent with those reported for the non-pregnant adult population during clinical studies conducted with Vaxigrip or Vaxigrip Tetra (QIV) even though higher for some adverse reactions (injection site pain, malaise, shivering, headache, myalgia).
Post marketing experience. Blood and lymphatic system disorders. Transient thrombocytopenia*, lymphadenopathy*.
Immune system disorders. Allergic reactions including anaphylactic reactions, angioedema, shock, dyspnoea, urticaria, pruritus, rash erythematous.
Nervous system disorders. Paraesthesia*, Guillain-Barré Syndrome (GBS), neuritis, neuralgia, convulsions, encephalomyelitis.
Vascular disorders. Vasculitis, such as Henoch-Schönlein purpura, with transient renal involvement in certain cases.
* These adverse events were reported during clinical trials only in some age groups (see Section 4.8 Adverse Effects (Undesirable Effects), Clinical trial data).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Cases of administration of more than the recommended dose (overdose) have been reported with Vaxigrip. When adverse reactions were reported, the information was consistent with the known safety profile of Vaxigrip described in Section 4.8 Adverse Effects (Undesirable Effects).
For information on the management of overdose, contact the Poisons Information Centre, on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Influenza vaccine, ATC code: J07BB02.
Mechanism of action. Vaxigrip provides active immunisation against three influenza virus strains (two A subtypes and one B type) contained in the vaccine.
Vaxigrip induces humoral antibodies against the haemagglutinins within 2 to 3 weeks. These antibodies neutralise influenza viruses.
Specific levels of haemagglutination-inhibition (HAI) antibody titre post-vaccination with inactivated influenza virus vaccines have not been correlated with protection from influenza illness but the HAI antibody titres have been used as a measure of vaccine activity. In some human challenge studies, HAI antibody titres of ≥ 1:40 have been associated with protection from influenza illness in up to 50% of subjects.
Since influenza viruses constantly evolve, the virus strains selected in the vaccine are reviewed annually by the WHO.
Annual influenza vaccination is recommended given the duration of immunity provided by the vaccine and because circulating strains of influenza virus change from year to year.
Clinical trials. Efficacy of Vaxigrip. Efficacy data for Vaxigrip are available in pregnant women and in infants less than 6 months of age born to vaccinated pregnant women (passive protection).
In children from 6 to 35 months of age and from 3 to 8 years of age (active immunisation) Vaxigrip efficacy is based on extrapolation of Vaxigrip Tetra (Quadrivalent Influenza Vaccine) efficacy.
No efficacy data are available for Vaxigrip in children and adolescents from 6 months to 17 years of age, in adults and in the elderly.
Children aged from 6 to 35 months (active immunisation). A randomised placebo controlled study was conducted in 4 regions (Africa, Asia, Latina America and Europe) over 4 influenza seasons, in more than 5400 children from 6 to 35 months of age who received two doses (0.5 mL) of Vaxigrip Tetra (QIV) (N=2722), or placebo (NaCl 0.9%, N=2717) 28 days apart to assess Vaxigrip Tetra (QIV) efficacy for the prevention of laboratory confirmed influenza illness caused by any strain A and/or B and caused by vaccine similar strains (as determined by sequencing).
Laboratory-confirmed influenza illness was defined as influenza like-illness (ILI) [occurrence of fever ≥ 38°C (that lasts at least 24 hours) concurrently with at least one of the following symptoms: cough, nasal congestion, rhinorrhoea, pharyngitis, otitis, vomiting, or diarrhoea] laboratory-confirmed by reverse transcriptase polymerase chain reaction (RT-PCR) and/or viral culture. See Table 8.
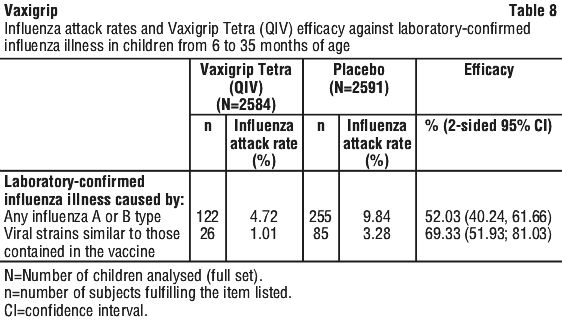
Severe laboratory-confirmed influenza illnesses were defined as ILI laboratory-confirmed by RT-PCR and/or viral culture with at least one of the following items:
fever > 39.5°C for subjects aged < 24 months or ≥ 39.0°C for subjects aged ≥ 24 months,
and/or at least one significant ILI symptom which prevents daily activity (cough, nasal congestion, rhinorrhoea, pharyngitis, otitis, vomiting, diarrhoea),
and/or one of the following events: acute otitis media, acute lower respiratory infection (pneumonia, bronchiolitis, bronchitis, croup), inpatient hospitalisation.
Children from 3 to 8 years of age (active immunisation). Based on immune responses observed in children 3 to 8 years of age, the efficacy of Vaxigrip Tetra (QIV) in this population is expected to be at least similar to the efficacy observed in children from 6 to 35 months (see Children from 6 to 35 months of age above and Immunogenicity of Vaxigrip Tetra below).
Pregnant women. In the randomised, controlled, phase IV, clinical studies conducted in Mali, Nepal, and South Africa, approximately 5,000 pregnant women received Vaxigrip and approximately 5,000 pregnant women received placebo or control vaccine (quadrivalent meningococcal conjugate vaccine) during the second or third trimester of pregnancy. Vaccine efficacy against laboratory-confirmed influenza in pregnant women was evaluated as a secondary endpoint in all three studies.
The studies conducted in Mali and South Africa demonstrated the efficacy of Vaxigrip for the prevention of influenza in pregnant women (during pregnancy and for approximately 6 months post-delivery), following vaccination during these trimesters of pregnancy.
In the study conducted in Nepal, the efficacy of Vaxigrip for the prevention of influenza in pregnant women following vaccination during these trimesters of pregnancy was not demonstrated. See Table 9.

Efficacy in infants younger than 6 months of age whose mothers received a single 0.5-mL dose of Vaxigrip during the second or third trimester has been demonstrated in clinical trials in Nepal, Mali and South Africa. Efficacy of Vaxigrip in infants younger than 6 months of age whose mothers were vaccinated during the first trimester has not been studied in these trials. Nevertheless, influenza vaccination during the first trimester should not be postponed (see Section 4.6 Fertility, Pregnancy and Lactation). See Table 10.

In the trial conducted in South Africa, vaccine efficacy was highest among infants 8 weeks of age or younger (85.8% [95% CI, 38.3 to 98.4]) and decreased over time; vaccine efficacy was 25.5% (95% CI, -67.9 to 67.8) for infants > 8 to 16 weeks of age and 30.4% (95% CI, -154.9 to 82.6) for infants > 16 to 24 weeks of age.
In the trial conducted in Mali, there is also a trend of higher efficacy of Vaxigrip in infants during the first 4 months after birth (70.2% [95% CI, 35.7 to 87.6]), with lower efficacy within the fifth month of surveillance (60.7% [95% CI, 33.8 to 77.5]) and a marked fall within the sixth month (37.3% [95% CI, 7.6 to 57.8]).
The prevention of influenza disease can only be expected if the infant(s) are exposed to strains included in the vaccine administered to the mother.
Immunogenicity of Vaxigrip. Clinical studies performed in adults from 18 to 60 years of age, in elderly over 60 years of age, in children from 3 to 8 years of age and from 6 to 35 months of age assessed the non-inferiority of Vaxigrip versus Vaxigrip Tetra (QIV) for HAI (hemagglutinin inhibition) Geometric Mean antibody Titre (GMT) at Day 21 (for adults) and at Day 28 (for children), HAI seroconversion rate (4-fold rise in reciprocal titre or change from undetectable [< 10] to a reciprocal titre of ≥ 40), and HAI GMTR (post-/pre-vaccination titres).
One clinical study performed in adults from 18 to 60 years of age and in children from 9 to 17 years of age described the immune response of Vaxigrip versus Vaxigrip Tetra (QIV) for HAI GMT at Day 21. Another clinical study performed in children from 9 to 17 years of age described only the immune response of Vaxigrip Tetra (QIV).
One clinical study performed in pregnant women described the immune response of Vaxigrip and Vaxigrip Tetra (QIV) for HAI GMT at Day 21, HAI seroconversion rate, and HAI GMTR after one dose administered during the second or third trimester of pregnancy. In this study, the transplacental transfer was evaluated using HAI GMTs of maternal blood, of cord blood and of ratio cord blood/maternal blood, at delivery.
Vaxigrip induced a significant immune response to the 3 influenza strains contained in the vaccine.
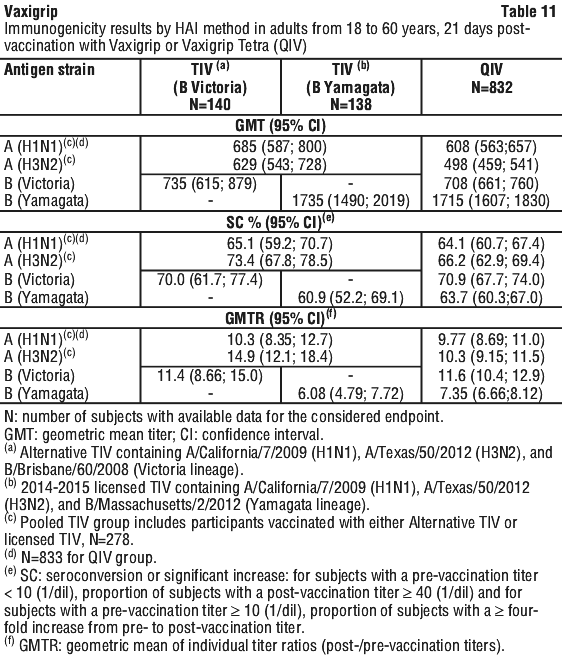
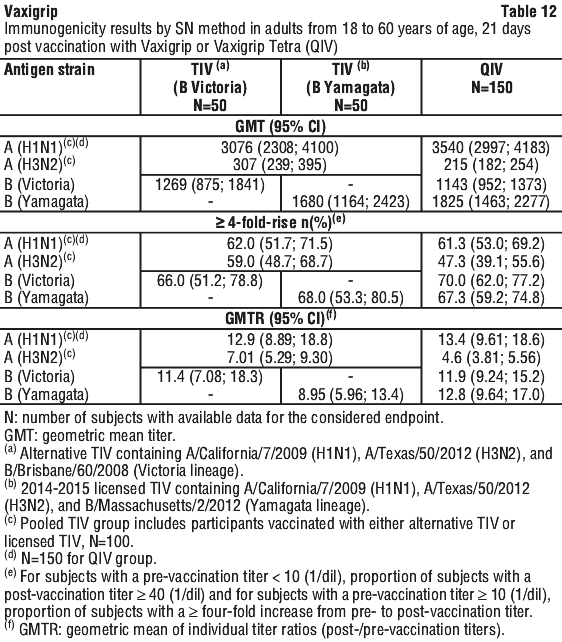
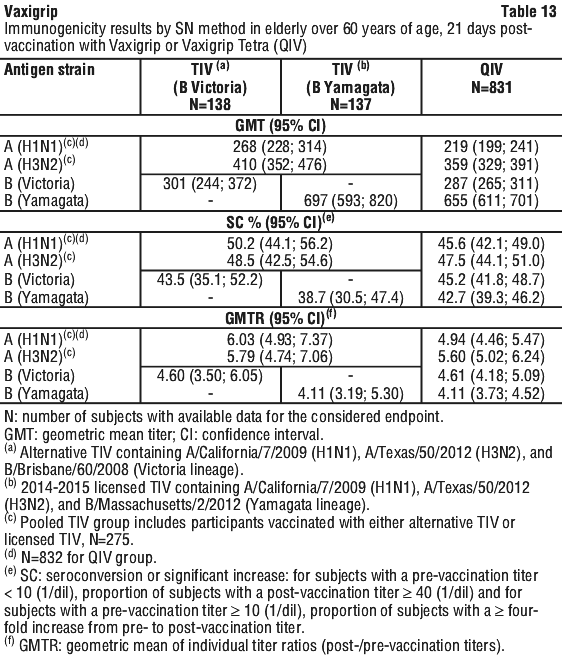
Adults and elderly. A randomised, active controlled non-inferiority study was conducted to assess the immunogenicity of Vaxigrip Tetra (QIV) compared to Vaxigrip. A total of 1114 adults from 18 to 60 years of age and 1111 elderly over 60 years of age were randomised to receive either one dose of Vaxigrip (one of two formulations), each containing a B strain that corresponds to one of the two B strains in Vaxigrip Tetra (QIV) (a B strain of the Yamagata lineage and a B strain of the Victoria lineage) or one dose of Vaxigrip Tetra (QIV).
The immunogenicity of Vaxigrip and Vaxigrip Tetra (QIV) were assessed 21 days after injection by HAI method in all subjects (278 adults from 18 to 60 years of age for Vaxigrip, 832 adults from 18 to 60 years of age for Vaxigrip Tetra (QIV) and 275 adults over 60 years of age for Vaxigrip, 831 for Vaxigrip Tetra (QIV)) and by seroneutralisation (SN) method in subsets of subjects (100 adults from 18 to 60 years of age for Vaxigrip, 150 for Vaxigrip Tetra (QIV) and 98 adults over 60 years of age, 150 for Vaxigrip Tetra (QIV)).
Immunogenicity results in adults from 18 to 60 years of age and in elderly over 60 years of age for Vaxigrip are presented in Table 11, Table 12, Table 13 and Table 14 respectively.
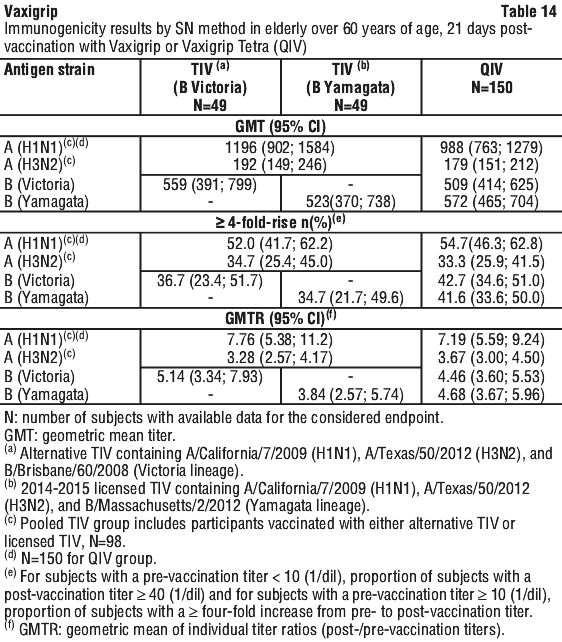
Pregnant women and transplacental transfer. In a randomised, controlled clinical study conducted in Finland, a total of 116 pregnant women received Vaxigrip and 230 pregnant women received Vaxigrip Tetra (QIV) during the second or third trimester of pregnancy (from 20 to 32 weeks of pregnancy).
Immunogenicity results by HAI method, in pregnant women 21 days after vaccination with Vaxigrip or Vaxigrip Tetra (QIV) are presented in Table 15.
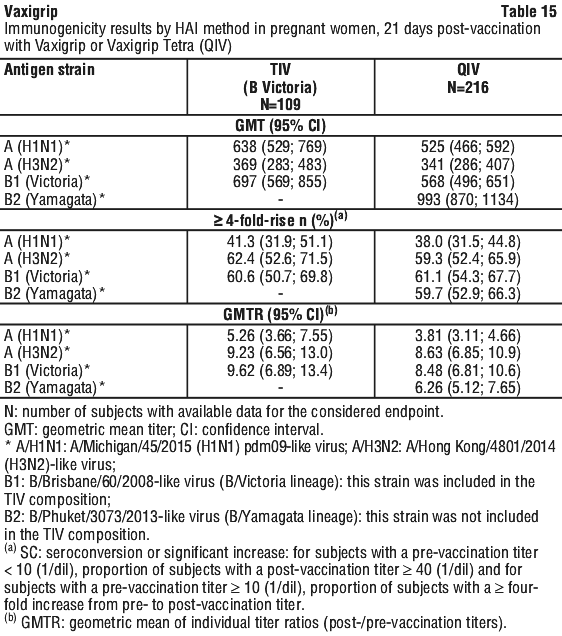
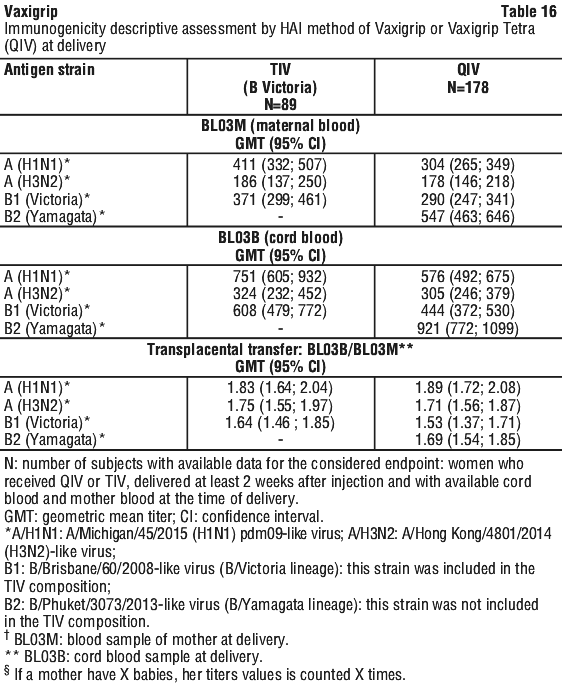
These data are consistent with the passive protection demonstrated in infants from birth to approximately 6 months of age following vaccination of women during the second or third trimester of pregnancy with Vaxigrip in studies conducted in Mali, Nepal, and South Africa.
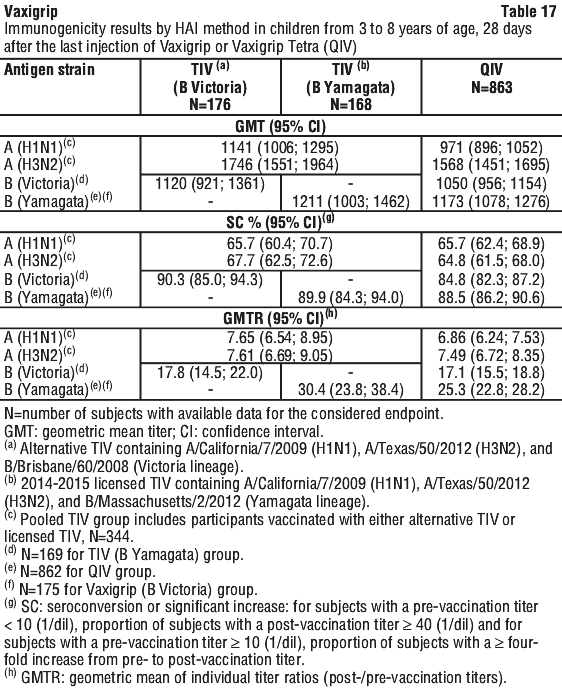
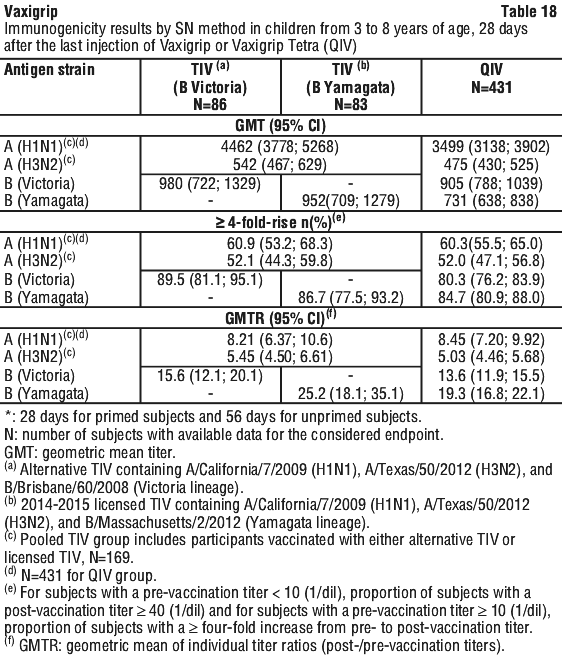
Children from 3 to 8 years of age. A randomised, active controlled study was conducted to assess the immunogenicity of Vaxigrip compared to Vaxigrip Tetra (QIV). A total of 1242 children 3 to 8 years of age were randomised to receive either one or two doses of Vaxigrip or of Vaxigrip Tetra (QIV) depending on their previous influenza vaccination history.
The immunogenicity of Vaxigrip and Vaxigrip Tetra (QIV) were assessed 28 days after receipt of the last injection of Vaxigrip Tetra by HAI method in all subjects and by SN method in subsets of subjects.
Children who received a one-or two-dose schedule of Vaxigrip or Vaxigrip Tetra (QIV) presented a similar immune response following the last dose of the respective schedule. See Table 17.
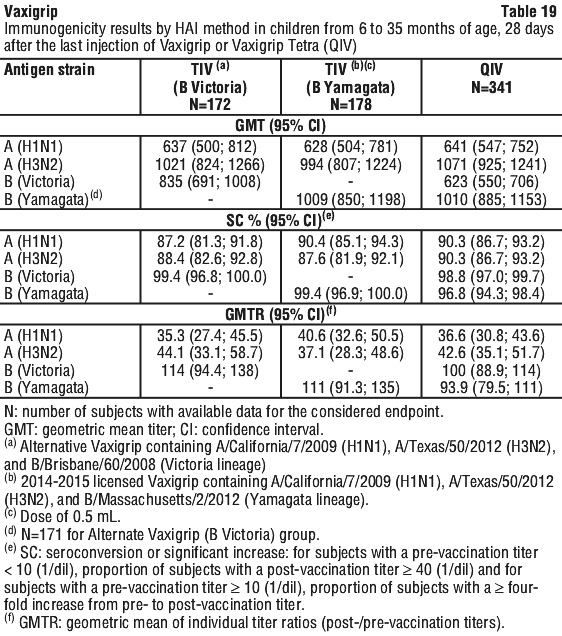
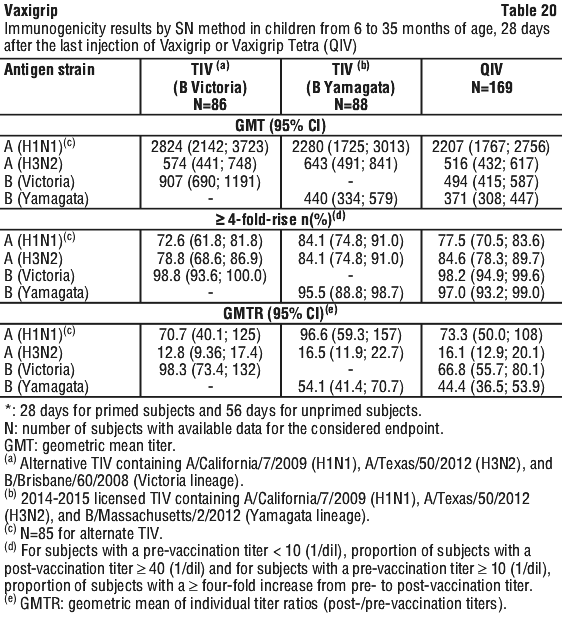
Children from 6 months to 35 months of age. In addition to the Vaxigrip Tetra (QIV) efficacy, the immunogenicity of two 0.5 mL of doses of Vaxigrip (N=369) compared to two 0.5 mL of doses of Vaxigrip Tetra QIV (N=341) was assessed 28 days after receipt of the last injection of Vaxigrip Tetra (QIV) by HAI method in children 6 to 35 months of age and by SN method in subsets of subjects. See Table 19.
5.2 Pharmacokinetic Properties
No pharmacokinetic studies have been performed.
5.3 Preclinical Safety Data
Genotoxicity. Vaxigrip has not been tested for genotoxic potential.
Carcinogenicity. Vaxigrip has not been tested for carcinogenic potential.
6 Pharmaceutical Particulars
6.1 List of Excipients
Buffer solution. Sodium chloride; potassium chloride; dibasic sodium phosphate dihydrate; monobasic potassium phosphate; water for injections.
No adjuvant and no preservative are added.
Vaxigrip may contain traces of ovalbumin (≤ 0.05 micrograms), neomycin (≤ 10.1 picograms), formaldehyde (≤ 30 micrograms) and octoxinol-9 (≤ 222.5 micrograms), which are used during the manufacturing process (see Section 4.4 Special Warnings and Precautions for Use).
6.2 Incompatibilities
This vaccine must not be mixed with other vaccines or medicinal products.
6.3 Shelf Life
Vaxigrip has a shelf life of 12 months when stored at 2°C to 8°C.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze). Discard if vaccine has been frozen. In the absence of photostability studies, this vaccine should be protected from light.
6.5 Nature and Contents of Container
0.5 mL of suspension in pre-filled syringe with or without separate needle - pack size of 1 or 10*.
Vaxigrip pre-filled syringe is not made with natural rubber latex.
*Not all pack sizes or presentations may be marketed.
6.6 Special Precautions for Disposal
After use, any remaining vaccine and container must be disposed of safely, preferably by heat inactivation or incineration, according to locally agreed procedures.
6.7 Physicochemical Properties
Chemical structure and CAS number. Not applicable/defined for vaccines.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
05 November 2025
Date of Revision
03 December 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.