Versatis Dermal Patch
Brand Information
| Brand name | Versatis Dermal Patch |
| Active ingredient | Lidocaine (lignocaine) |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Versatis Dermal Patch.
Summary CMI
VERSATIS® dermal patch
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using VERSATIS® dermal patch?
VERSATIS® dermal patch contains the active ingredient lidocaine.
VERSATIS® dermal patch is used to treat a painful skin condition called postherpetic neuralgia.
For more information, see Section 1. Why am I using VERSATIS® dermal patch? in the full CMI.
2. What should I know before I use VERSATIS® dermal patch?
Do not use if you have ever had an allergic reaction to lidocaine, methyl hydroxybenzoate, propyl hydroxybenzoate or any of the ingredients listed at the end of the CMI. Do not use if you have an allergy to other medicines similar to lidocaine such as bupivacaine, etidocaine, mepivacaine or prilocaine.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use VERSATIS® dermal patch? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with VERSATIS® dermal patch and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use VERSATIS® dermal patch?
- The daily dose is between one and three patches depending on the size of the painful area of your skin.
- VERSATIS® dermal patches should be worn for no more than 12 hours per day.
More instructions can be found in Section 4. How do I use VERSATIS® dermal patch? in the full CMI.
5. What should I know while using VERSATIS® dermal patch?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using VERSATIS® dermal patch? in the full CMI.
6. Are there any side effects?
VERSATIS® dermal patch may cause the following skin conditions at or around the patch application site: redness, rash, itching burning, dermatitis, small blisters.
Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of the following of these serious side effects: Allergic reaction (symptoms may include shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue or other parts of the body, rash, itching or hives on the skin); open wound; skin injury; skin wound.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
VERSATIS® dermal patch
Active ingredient: lidocaine
Consumer Medicine Information (CMI)
This leaflet provides important information about using VERSATIS® dermal patch. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using VERSATIS® dermal patch.
Where to find information in this leaflet:
1. Why am I using VERSATIS® dermal patch?
2. What should I know before I use VERSATIS® dermal patch?
3. What if I am taking other medicines?
4. How do I use VERSATIS® dermal patch?
5. What should I know while using VERSATIS® dermal patch?
6. Are there any side effects?
7. Product details
1. Why am I using VERSATIS® dermal patch?
VERSATIS® dermal patch contains the active ingredient lidocaine. Lidocaine is a local anaesthetic, which works by reducing the pain in your skin.
VERSATIS® dermal patch is used to treat a painful skin condition called postherpetic neuralgia. This is the pain which persists after Herpes Zoster (Shingles) infection. It is generally characterised by localised symptoms such as burning, shooting or stabbing pain.
2. What should I know before I use VERSATIS® dermal patch?
Warnings
Do not use VERSATIS® dermal patch:
- if you are allergic to lidocaine, methyl hydroxybenzoate, propyl hydroxybenzoate or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - if you are allergic to any other medicines similar to lidocaine such as bupivacaine, etidocaine, mepivacaine or prilocaine.
- on injured skin or on open wounds.
- on the eyes or mouth.
- on unhealed shingles blisters.
Check with your doctor if you:
- severe liver disease.
- severe kidney disease.
- severe heart problems.
- have any other medical conditions.
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. If you become pregnant while taking VERSATIS® dermal patch, tell your doctor.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Children
Do not give VERSATIS® dermal patch to children.
Safety and effectiveness in children younger than 18 years has not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with VERSATIS® dermal patch and affect how it works. These include:
- some drugs used to slow the heart, including tocainide and mexiletine
- other local anaesthetics such as bupivacaine, etidocaine, mepivacaine or prilocaine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect VERSATIS® dermal patch.
4. How do I use VERSATIS® dermal patch?
How much VERSATIS® dermal patch to use
- The daily dose is between one and three patches depending on the size of the painful area of your skin.
- VERSATIS® dermal patch may be cut into smaller pieces to fit the affected area.
- You should not use more than 3 patches at the same time.
- VERSATIS® dermal patches should be worn for no more than 12 hours per day. Applying the patches for a longer time or using more than 3 patches per day could cause serious reactions.
- The patches should be removed after 12 hours of use, so that you have a 12 hour period with no patch in place.
- Usually, you will feel some pain relief on the first day you use the patch, but it may take up to 2 – 4 weeks until the full pain-relief effect of VERSATIS® dermal patch is seen.
If after that time you still have a lot of pain, talk to your doctor because the benefits of the treatment must be weighed against potential risks.
Your doctor will check how well VERSATIS® dermal patch is working at regular intervals. - Follow the instructions provided and use VERSATIS® dermal patch until your doctor tells you to stop.
How long to use VERSATIS® dermal patch for
Continue using it for as long as the doctor tells you.
How to use VERSATIS® dermal patch
Before you apply VERSATIS® dermal patch to the affected area:
- Cut any hairs on the affected skin using scissors. Do not shave hairs off.
- Ensure the skin is clean and dry.
- Allow your skin to cool off after a bath or shower.
- Creams and lotions may be used on the affected skin during the period when you are not wearing the patch.
VERSATIS® dermal patch should only be used on the areas of skin after the shingles rash has healed. It should not be used on or near the mouth.
Avoid contact of VERSATIS® dermal patch with the eye. If contact occurs, immediately wash the eye with water or saline and protect it until sensation returns.
Avoid the use of external heat sources, such as heating pads or electric blankets, as the effect of this has not been studied. Local heat may alter the absorption of lidocaine from the patch.
How to apply the patch:
Step 1: Open the sachet and remove one or more patches.


- Tear open or cut the sachet along the dotted line.
- When using scissors, be careful not to damage the patches.
- Take out one or more patches depending on the size of the painful area on your skin.
Step 2: Close the sachet.

- Close the sachet tightly after use.
- The patch contains water and will dry out if the sachet is not closed properly.
Step 3: Cut the patch, if necessary.

- If required, cut the patch to the required size to fit the painful area of skin before removing the liner.
Step 4: Remove the liner.
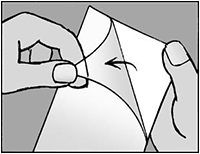
- Remove the transparent liner from the patch.
- Try not to touch the sticky part of the patch.
Step 5: Apply the patch and press it firmly onto the skin.
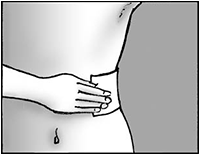
- Apply up to three patches to the painful area of skin.
- Press the patch onto your skin.
- Press for at least 10 seconds to make sure the patch sticks firmly.
- Make sure that all of it sticks to your skin, including the edges.
Leave the patch on for 12 hours only.
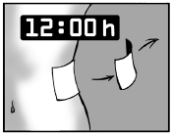
It is important that VERSATIS® dermal patch is in contact with your skin for only 12 hours.
For example, if you have more pain at night you might want to apply the patch at 7pm in the evening and remove it at 7am in the morning.
If you have more pain during the day than at night you might want to apply the patch at 7am in the morning and remove it at 7pm in the evening.
Bathing, showering and swimming
If at all possible, contact with water should be avoided whilst wearing VERSATIS® dermal patch. Bathing, showering or swimming can be done in the time period when you are not wearing the patch. If you have had a recent bath or shower, you should wait until your skin cools before applying the patch.
If the patch comes off
Very rarely the patch might fall off or come unstuck. If it does, try sticking it back on the same area. If it does not stay on, remove it and put a new patch on the same area.
How to remove VERSATIS® dermal patch
When changing the patch, remove the old patch slowly. If it does not come off easily, you can soak it in warm water for a few minutes before removing it.
Used patches still contain active ingredient, which may be harmful to others. Fold the used patch in half, with the sticky sides together and throw them away so that they are out of the reach of children.
If you forget to remove the patch after 12 hours:
As soon as you remember, remove the old patch. A new patch can be used again after 12 hours.
If you forget to use VERSATIS® dermal patch
VERSATIS® dermal patch should be used regularly at the same time each day.
If you miss your dose at the usual time and it is almost time for you to apply the next patch dose, skip the one you missed and apply the next patch dose when you are meant to.
Otherwise, apply a new patch as soon as you remember, and then go back to your normal schedule.
Do not take a double dose to make up for the dose you missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you use too much VERSATIS® dermal patch
If you think that you have used too much VERSATIS® dermal patch, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs or symptoms of discomfort or poisoning.
Signs or symptoms of an overdose may include:
- dizziness
- vomiting
- drowsiness
- taste disturbances or loss of taste
- seizures
- dilated pupils
- a low heartbeat
- changes in the way the heart beats (faster, slower, irregular);
- shock.
5. What should I know while using VERSATIS® dermal patch?
Things you should do
- Before starting any new medicine, tell your doctor or pharmacist that you are using VERSATIS® dermal patch.
- If irritation or burning sensation occurs whilst you are using the patch, it should be removed. The area of irritation should remain patch-free until the irritation stops.
- Keep all your doctor's appointments so that your progress can be checked.
Call your doctor straight away if you:
- Become pregnant while taking VERSATIS® dermal patch.
Remind any doctor, dentist, pharmacist, surgeons or anesthetists you visit that you are using VERSATIS® dermal patch.
Things you should not do
- Do not stop using this medicine or change the dose without checking with your doctor.
- Do not use VERSATIS® dermal patch to treat any other conditions unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Never re-use a patch.
Driving or using machines
This medicine is not expected to affect your ability to drive a car or operate machinery.
Looking after your medicine
- Keep your medicine in a cool dry place where the temperature stays below 25°C.
- Do not refrigerate or freeze.
- After first opening: Keep the sachet tightly closed.
- After opening the sachet, the patches should be used within 14 days.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children and pets cannot reach it.
Even a used VERSATIS® dermal patch contains a large amount of lidocaine. A small child or pet could suffer serious adverse effects from chewing or swallowing a new or used patch.
Store and dispose of patches out of the reach of children, pets and others.
When to discard your medicine (as relevant)
Do not use VERSATIS® dermal patch if the packaging is torn or shows signs of tampering. If the package has become damaged, the dermal patches may dry out and become less sticky.
Do not use VERSATIS® dermal patch if the expiry date (Exp.) printed on the pack has passed.
If it has expired or is damaged, return it to your pharmacist for disposal.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Skin conditions at or around the patch application site which may include:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
There is no evidence this medicine is addictive.
What VERSATIS® dermal patch contains
| Active ingredient (main ingredient) | lidocaine. |
| Other ingredients (inactive ingredients) | Self-Adhesive Layer:
|
| Potential allergens |
|
Do not take this medicine if you are allergic to any of these ingredients.
What VERSATIS® dermal patch looks like
VERSATIS® lidocaine 5% w/w dermal patch (AUST R 175178) is 14 cm long and 10 cm wide. It is white and made of fleece fabric marked with “lidocaine 5%”. The patches are packed in re-sealable sachets, each containing 5 patches.
Registered pack sizes are cartons containing 5, 10, 20, 25 or 30 patches packed in 1, 2, 4, 5 or 6 sachets respectively. Not all pack sizes may be marketed.
Who distributes VERSATIS® dermal patch
Sponsor:
Seqirus Pty Ltd
ABN: 26 160 735 035
63 Poplar Road
Parkville VIC 3052
Australia
Telephone: 1800 642 865
www.cslseqirus.com.au
Distributor:
Seqirus (Australia) Pty Ltd
Melbourne, Australia
Manufactured and licensed by:
Teikoku Pharma USA, Inc.
Registered trademark of
Grünenthal GmbH used by Seqirus
as authorised user.
This leaflet was prepared in December 2025.
Brand Information
| Brand name | Versatis Dermal Patch |
| Active ingredient | Lidocaine (lignocaine) |
| Schedule | S4 |
MIMS Revision Date: 01 January 2026
1 Name of Medicine
Lidocaine.
2 Qualitative and Quantitative Composition
Each dermal patch (10 cm x 14 cm) contains 700 mg (5% w/w) lidocaine (50 mg lidocaine per gram adhesive base).
Excipients with known effect. Hydroxybenzoates.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Versatis is a white hydrogel patch containing adhesive material, which is applied to a non-woven polyethylene terephthalate backing embossed with 'Lidocaine 5%' and covered with a polyethylene terephthalate film release liner.
4 Clinical Particulars
4.1 Therapeutic Indications
Versatis is indicated for the symptomatic relief of neuropathic pain associated with previous herpes zoster infection (PHN).
4.2 Dose and Method of Administration
Adults and elderly patients. The painful area should be covered with Versatis once daily for up to 12 hours within a 24 hour period. Only the number of patches that are needed for an effective treatment should be used. When needed, the patches may be cut into smaller sizes with scissors prior to removal of the release liner. In total, not more than three patches should be used at the same time.
Versatis must be applied to intact, dry, nonirritated skin (after healing of the shingles).
Each patch must be worn no longer than 12 hours. The subsequent patch free interval must be at least 12 hours.
The patch must be applied to the skin immediately after removal from the sachet and following removal of the release liner from the gel surface. Hairs in the affected area must be cut off with a pair of scissors (not shaved).
Treatment outcome should be re-evaluated after 2-4 weeks. If there has been no response to Versatis after this period or if any relieving effect can solely be related to the skin protective properties of Versatis, treatment must be discontinued as potential risks may outweigh benefits in this context (see Section 4.4 Special Warnings and Precautions for Use; Section 5.1 Pharmacodynamic Properties). Treatment should be reassessed at regular intervals to decide whether the number of patches needed to cover the painful area can be reduced, or if the patch free period can be extended.
Use for patients under the age of 18 is not recommended because of the lack of data in this group.
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients. Versatis is also contraindicated in patients with known hypersensitivity to other local anaesthetics of the amide type, e.g. bupivacaine, etidocaine, mepivacaine and prilocaine.
Versatis must not be applied to inflamed or injured skin, such as active herpes zoster lesions, atopic dermatitis or wounds.
4.4 Special Warnings and Precautions for Use
Versatis should not be applied to mucous membranes. Eye contact with the patch should be avoided.
Versatis contains propylene glycol which may cause skin irritation. It also contains methyl hydroxybenzoate and propyl hydroxybenzoate which may cause allergic reactions (possibly delayed).
Excessive dosing. Excessive dosing by applying Versatis to larger areas or for longer than the recommended wearing time could result in increased absorption of lidocaine and higher blood concentrations, which may lead to systemic adverse drug reactions (see Section 4.8 Adverse Effects (Undesirable Effects)). The blood concentration of lidocaine is determined by the rate of systemic absorption and elimination. Longer duration of application, application of more than the recommended number of patches, smaller patients or impaired elimination may all contribute to increasing the blood concentration of lidocaine. Lidocaine toxicity could be expected at lidocaine blood concentrations above 5 microgram/mL. With recommended dosing of Versatis, the average peak blood concentration is about 0.13 microgram/mL, but individual concentrations up to 0.28 microgram/mL have been observed in clinical trials.
Use in hepatic impairment. Versatis should be used with caution in patients with severe hepatic impairment. The excretion of lidocaine and its metabolites may be delayed in patients with severe hepatic impairment. Higher systemic concentrations of lidocaine may occur.
Use in renal impairment. Versatis should be used with caution in patients with severe renal impairment. The excretion of lidocaine and its metabolites may be delayed in patients with severe renal impairment. Higher systemic concentrations of lidocaine may occur.
Use in cardiac impairment. Versatis should be used with caution in patients with severe cardiac impairment. The excretion of lidocaine and its metabolites may be delayed in patients with severe cardiac impairment. Higher systemic concentrations of lidocaine may occur.
Use in the elderly. Generally, no dosage adjustment is necessary for elderly patients (see Section 4.2 Dose and Method of Administration).
Paediatric use. Use for patients under the age of 18 is not recommended because of the lack of data in this group.
Effects on laboratory tests. Laboratory findings did not give any indication for safety-relevant issues during clinical trials.
External heat sources. Placement of external heat sources, such as heating pads or electric blankets, over Versatis is not recommended as this has not been evaluated and may increase plasma lidocaine levels.
Accidental exposure. Even a used Versatis patch contains a large amount of lidocaine (at least 665 mg). The potential exists for a small child or pet to suffer serious adverse effects from chewing or ingesting a new or used Versatis patch, although the risk with this formulation has not been evaluated. It is important for patients to store and dispose of Versatis out of the reach of children, pets and others.
Lidocaine metabolite toxicity. The lidocaine metabolite, 2,6-xylidine, has been shown to be genotoxic and carcinogenic in rats and a secondary metabolite has been shown to be mutagenic (see Section 5.3 Preclinical Safety Data). The clinical significance of these findings is unknown. Consequently long-term treatment with Versatis is only justified if there is a therapeutic benefit for the patient (see Section 4.2 Dose and Method of Administration).
4.5 Interactions with Other Medicines and Other Forms of Interactions
No interaction studies have been performed. No clinically relevant interactions have been observed in clinical studies with Versatis.
Antiarrhythmic drugs. Although normally the absorption of lidocaine from the skin is low, Versatis must be used with caution in patients receiving class I antiarrhythmic medicinal products (e.g. tocainide, mexiletine) since the cardiac effects (including arrhythmia) and CNS effects (including convulsions, CNS depression) may be additive and potentially synergistic.
Local anaesthetic agents. When Versatis is used concomitantly with other products containing local anaesthetic agents, the amount absorbed from all formulations must be considered.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No clinical data regarding fertility are available for lidocaine. Animal studies have not shown effects on female fertility.
Use in pregnancy. (Category A)
Lidocaine crosses the placenta. However, there are no adequate data from the use of lidocaine in pregnant women. Animal studies are incomplete with respect to effects on pregnancy, embryo fetal development, parturition or postnatal development.
Lidocaine had no effect on general reproductive performance in rats at plasma concentrations up to 130-fold of those observed in patients. No adverse effects were seen in an embryofetal/ teratogenicity study in rats at plasma concentrations more than 200-fold of that observed in patients.
The potential risk for humans is unknown. Therefore, Versatis should not be used during pregnancy unless clearly necessary.
Use in lactation. Lidocaine is excreted in milk. However, there are no studies of Versatis in breastfeeding women. Since the metabolism of lidocaine is relatively rapid and almost completely in the liver, only very low levels of lidocaine are expected to be excreted into milk.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed. An effect on the ability to drive and use machines is unlikely because systemic absorption is minimal (see Section 5.2 Pharmacokinetic Properties).
4.8 Adverse Effects (Undesirable Effects)
Systemic adverse drug reactions following the appropriate use of Versatis are unlikely since the systemic concentration of lidocaine is very low (see Section 5.2 Pharmacokinetic Properties). Systemic adverse drug reactions to lidocaine are similar in nature to those observed with other amide local anaesthetic agents (see Section 4.9 Overdose).
Adverse events identified in clinical trials. Adverse events occurring in at least 1.5% of patients treated for up to 6 months are displayed in Table 1. The adverse events listed may be associated with the underlying disease and concomitant medications.
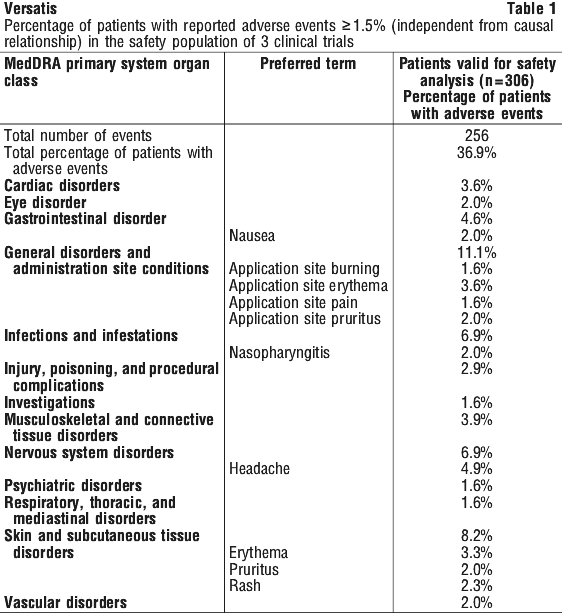
Approximately 16% of patients can be expected to experience adverse drug reactions. These are localised reactions due to the nature of the medicinal product.
The most commonly reported adverse drug reactions were skin reactions (such as erythema, rash, application site pruritus, application site burning, application site dermatitis, application site erythema, application site vesicles, dermatitis, skin irritation, and pruritus).
Table 2 lists adverse drug reactions that have been reported in studies of postherpetic neuralgia patients receiving Versatis. They are listed by system organ class and frequency. Frequencies are defined as very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000), not known (cannot be estimated from the available data).

Postmarketing experience. Additionally, Table 3 presents adverse drug reactions have been observed in patients receiving Versatis under postmarketing conditions.

Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdose with Versatis is unlikely but it cannot be excluded that inappropriate use, such as use of a higher number of patches at the same time, with prolonged application period, or using Versatis on broken skin might result in higher than normal plasma concentrations. Possible signs of systemic toxicity will be similar in nature to those observed after administration of lidocaine as a local anaesthetic agent, and may include the following signs and symptoms: dizziness, vomiting, drowsiness, dysgeusia, seizures, mydriasis, bradycardia, arrhythmia, and shock.
In addition, known drug interactions related to systemic lidocaine concentrations with beta-blockers, CYP3A4 inhibitors (e.g. imidazole derivatives, macrolides) and antiarrhythmic agents might become relevant with overdose.
In case of suspected overdose Versatis should be removed and supportive measures taken as clinically needed. If there is any suspicion of lidocaine overdose, drug blood concentration should be checked. There is no antidote to lidocaine.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: local anaesthetics, amides.
Mechanism of action. Lidocaine is a local anaesthetic of the amide type which stabilises excitable membranes by inactivation of sodium channels. Its action on neuronal membranes prevents neuronal conduction. When applied topically in the form of Versatis, lidocaine produces a local analgesic effect.
Versatis has a dual mode of action: the pharmacological action of lidocaine diffusion and the mechanical action of the hydrogel patch that protects the hypersensitive area. The lidocaine contained in Versatis diffuses continuously into the skin, providing a local analgesic effect.
Clinical trials. Pain management in PHN is difficult. There is evidence of efficacy with Versatis in the symptomatic relief from the allodynic component of PHN in some cases.
There were two key controlled studies carried out to assess the efficacy of Versatis. A third study provides information on maintenance of effect.
Efficacy was demonstrated in a multicentre, enriched enrolment, double blind placebo controlled, multiple dose randomised withdrawal, parallel group study with Versatis and corresponding placebo patch. This study had an initial 8 week run-in, open label phase in which patients with PHN responding to treatment were randomised to a 2 week, double blind treatment period with either Versatis or placebo patch. PHN was defined as neuropathic pain persisting for at least 3 months after healing of a herpes zoster skin rash.
Patients aged 50 years and older, suffering from PHN and having an average pain intensity (during last week prior to screening and enrolment visit) of at least 4 on the 11 point numeric rating scale (NRS) applied Versatis for up to 8 weeks. Only those patients who reported regular use of Versatis during the last 4 weeks of the run-in phase, an average daily pain intensity of 7 or less on the 11 point NRS in the last week prior to randomisation, increase in pain intensity when Versatis was not worn, and an average pain relief of 'moderate' or better at randomisation (recall period of 1 week prior to the visit) were considered to be responders and were eligible for randomisation.
The primary endpoint was the time to exit during the double blind phase due to lack of efficacy (decrease of pain relief score by two or more categories on a 6 item pain relief scale on two consecutive application days in comparison to the average pain relief in last week on treatment with Versatis before randomisation in run-in phase).
Of the 265 patients, 137 (52%) entered the run-in phase and responded to Versatis. Of these, 71 were randomised to the double blind phase. About 40% of patients randomised to each treatment group were not regularly taking any concomitant medication for PHN pain at study entry. The median time to exit the double blind period was 13.5 days for patients randomised to Versatis (n = 36) and 9.0 days for those randomised to placebo patch (n = 35). This difference was not statistically significant. For the per protocol population, median time to exit was 14 days for Versatis (n = 17) and 6 days for the placebo patch (n = 17). This difference between groups was statistically significant.
Post hoc analyses of this study showed that the initial response was independent of the duration of pre-existing PHN. However, the notion that patients with longer duration of PHN (> 12 months) do benefit more from active treatment is supported by the finding that this group of patients was more likely to drop out due to lack of efficacy when switched to placebo during the double blind withdrawal part of this study.
A double blind, placebo controlled, crossover trial was conducted to test whether patients who had been using Versatis could distinguish between the lidocaine patch and a placebo patch. Half of the patients were randomised to receive Versatis as first treatment and half received the placebo patch as first treatment then vice versa in the second treatment phase. After up to 14 days in the first phase patients were directly switched to second treatment phase. The primary endpoint was 'time to exit' comparing treatment phases.
Patients were to withdraw from the treatment phase if their pain relief was 2 points lower than their usual response to Versatis on a 6 point verbal rating scale (worse, no pain relief, slight relief, moderate relief, a lot of relief, complete relief). The secondary endpoint was patient's preference between treatments. Of the 33 patients with PHN in this study, 32 of them received the investigational products and completed the study. In general, patients used up to 3 patches per application with only three exceptions (two patients used up to 4 and one patient 5 patches per application). The median times to exit in each treatment arm were > 14 days for Versatis and 3.8 days for placebo patch (across both treatment periods) p < 0.001. Three patients (9.4%) preferred placebo patch, 25 patients (78.1%) preferred Versatis, and 4 patients (12.5%) had no preference.
Maintenance of efficacy was assessed over up to 12 months in an open label, multicentre, multiple dose study conducted in 259 patients aged 50 years and older with PHN for > 3 m after herpes zoster (HZ) rash healing who participated in an earlier efficacy study, or had PHN with average pain intensity of ≥ 4 (0-11 scale) over last week prior to screening. At 12 months, 143 patients had completed the study. Pain relief was generally maintained.
5.2 Pharmacokinetic Properties
Absorption. The amount of lidocaine systemically absorbed from Versatis is directly related to both the duration of application and the surface area over which it is applied. When Versatis is used according to the maximum recommended dose (3 patches applied simultaneously for 12 h) about 3 ± 2% of the total applied lidocaine dose is systemically available and similar for single and multiple administrations. Maximum systemic concentrations of lidocaine were observed between 9 and 12 h after application of Versatis.
A population kinetics analysis of the clinical efficacy studies in patients suffering from postherpetic neuralgia (PHN) revealed a mean maximum concentration for lidocaine of 45 nanogram/mL after application of 3 patches simultaneously 12 h per day after repeated application for up to one year. This concentration is in accordance with the observation in pharmacokinetic studies in PHN patients (52 nanogram/mL) and in healthy volunteers (85 nanogram/mL and 125 nanogram/mL).
For lidocaine and its metabolites MEGX, GX, and 2,6-xylidine no tendency for accumulation was found; steady state concentrations were reached within the first four days.
The population kinetic analysis indicated that when increasing the number from 1 to 3 patches worn simultaneously, the systemic exposure increased less than proportionally to the number of used patches.
Distribution. After intravenous administration of lidocaine to healthy volunteers, the volume of distribution was found to be 1.3 ± 0.4 L/kg (mean ± S.D., n = 15). The lidocaine distribution volume showed no age dependency; however it is decreased in patients with congestive heart failure and increased in patients with liver disease. At plasma concentrations produced by application of Versatis, approximately 70% of lidocaine is bound to plasma proteins. Lidocaine crosses the placental and blood brain barriers presumably by passive diffusion.
Metabolism. Lidocaine is metabolised rapidly in the liver to a number of metabolites. The primary metabolic route for lidocaine is N-dealkylation to monoethylglycinexylidide (MEGX) and glycinexylidide (GX), both of which are less active than lidocaine and available in low concentrations. These are hydrolysed to 2,6-xylidine, which is converted to 4-hydroxy-2,6-xylidine and the N-hydroxylation product N-(2,6-dimethlyphenyl)-hydroxylamine (DMHA).
The metabolite, 2,6-xylidine, has unknown pharmacological activity but has shown carcinogenic potential in rats (see Section 5.3 Preclinical Safety Data). A population kinetics analysis revealed a mean maximum concentration for 2,6-xylidine of 9 nanogram/mL after repeated daily applications for up to one year. This finding is confirmed by a phase I pharmacokinetic study.
Lidocaine may undergo metabolism in the skin.
Excretion. Lidocaine and its metabolites are excreted by the kidneys. More than 85% of the dose is found in the urine in the form of metabolites or active substance. Less than 10% of the lidocaine dose is excreted unchanged. The main metabolite in urine is a conjugate of 4-hydroxy-2,6-xylidine, accounting for about 70 to 80% of the dose excreted in the urine. 2,6-xylidine is excreted in the urine in man at a concentration of less than 1% of the dose. The elimination half-life of lidocaine after patch application in healthy volunteers is 7.6 hours. After multiple administration of Versatis in healthy subjects, the elimination half-lives of MEGX, GX and 2,6-xylidine are 6.4 h, 13 h, and 15 h, respectively. The systemic clearance of lidocaine is 0.635 ± 0.175 L/min.
The excretion of lidocaine and its metabolites may be delayed in cardiac, renal or hepatic insufficiency.
5.3 Preclinical Safety Data
Genotoxicity. Lidocaine itself has shown no evidence of genotoxicity when investigated in vitro or in vivo. Metabolites of lidocaine, 2,6-xylidine and N-(2,6-dimethylphenyl)-hydroxylamine (DMHA), have shown genotoxic activity in several assays.
Carcinogenicity. Carcinogenicity studies have not been performed with lidocaine. A two year dietary study in rats with the metabolite 2,6-xylidine noted inflammation and hyperplasia of the nasal olfactory epithelium and carcinomas and adenomas in the nasal cavity.
Tumorigenic changes were also found in the liver and subcutis. Estimated 2,6-xylidine exposure (plasma AUC) at the lowest tumorigenic dose was at least 60-fold clinical exposure at the maximum recommended human dose (MRHD) of Versatis dermal patch. Because the risk to humans is unclear, long-term treatment with high doses of lidocaine should be avoided.
6 Pharmaceutical Particulars
6.1 List of Excipients
Self adhesive layer. Glycerol, sorbitol solution (70%) (crystallising), carmellose sodium, propylene glycol, urea, heavy kaolin, tartaric acid, gelatin, polyvinyl alcohol, aluminium glycinate, disodium edetate, methyl hydroxybenzoate, propyl hydroxybenzoate, polyacrylic acid, sodium polyacrylate, purified water.
Backing fabric and release liner. Polyethylene terephthalate (PET).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Do not refrigerate or freeze.
After first opening: keep the sachet tightly closed.
Use opened sachets within 14 days.
6.5 Nature and Contents of Container
Versatis dermal patches are contained in a resealable sachet composed of paper/polyethylene/aluminium/ethylene meta-acrylic acid co-polymer containing 5 patches.
Each carton contains 5, 10, 20, 25 or 30 patches. Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
After use Versatis still contains active substance. After removal, the used patches should be folded in half, adhesive side inwards so that the self adhesive layer is not exposed, and the patch should be discarded.
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. Structural formula:
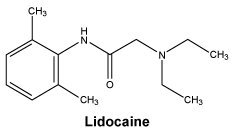
Molecular formula: C14H22N2O.
Molecular weight: 234.3.
CAS number. 137-58-6.
Lidocaine is a white or almost white crystalline powder, practically insoluble in water, very soluble in ethanol, and freely soluble in ether. The pKa is 7.9. The coefficient (log D) is defined as the ratio of the equilibrium concentrations of a single molecular species in a 1-octanol/aqueous buffered solution 2-phase system at pH 7.4. The value of log D for lidocaine in 1-octanol/water is 1.61.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
16 March 2012
Date of Revision
28 October 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.