Verzenio
Brand Information
| Brand name | Verzenio |
| Active ingredient | Abemaciclib |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Verzenio.
Summary CMI
VERZENIO™
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
▼ This medicine is new or being used differently. Please report side effects. See the full CMI for further details.
1. Why am I using VERZENIO?
VERZENIO contains the active ingredient abemaciclib. VERZENIO is used to treat patients with certain types of breast cancer which have not spread to other parts of the body or spread beyond the original tumour and/or to other organs.
For more information, see Section 1. Why am I using VERZENIO? in the full CMI.
2. What should I know before I use VERZENIO?
Do not use if you have ever had an allergic reaction to VERZENIO or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use VERZENIO? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with VERZENIO and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use VERZENIO?
- Swallow tablet whole with a full glass of water
- Do not chew, crush or split the tablets before swallowing. VERZENIO may be taken with or without food.
More instructions can be found in Section 4. How do I use VERZENIO? in the full CMI.
5. What should I know while using VERZENIO?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using VERZENIO? in the full CMI.
6. Are there any side effects?
Very common: diarrhoea, abdominal pain, infections, nausea, feeling tired, vomiting, constipation, indigestion or upset stomach, decreased appetite, abnormalities in liver blood tests, hair loss, alteration in taste, headache, dizziness, swelling in upper and lower limbs (peripheral oedema), cough, shortness of breath, dry mouth, inflammation of the mouth and lips (stomatitis) rash, itching, dry skin, nail disorders such as breaking or splitting of nails, watering eyes, muscular weakness, flu-like symptoms, fever, decreased weight.
Serious side effects: fever or chills, blood clots in the veins or cough, difficulty breathing or chest discomfort (lung inflammation). For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
▼ This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems.
VERZENIO™
Active ingredient: Abemaciclib
Consumer Medicine Information (CMI)
This leaflet provides important information about using VERZENIO. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using VERZENIO.
Where to find information in this leaflet:
1. Why am I using VERZENIO?
2. What should I know before I use VERZENIO?
3. What if I am taking other medicines?
4. How do I use VERZENIO?
5. What should I know while using VERZENIO?
6. Are there any side effects?
7. Product details
1. Why am I using VERZENIO?
VERZENIO contains the active ingredient abemaciclib. VERZENIO belongs to a group of medicine called antineoplastic drugs, an anticancer drug.
VERZENIO is used to treat patients with certain types of breast cancer (hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2-) which have not spread to other parts of the body (early breast cancer) or spread beyond the original tumour and/or to other organs (metastatic breast cancer).
2. What should I know before I use VERZENIO?
Warnings
Do not use VERZENIO if:
- you are allergic to abemaciclib, or any of the ingredients listed at the end of this leaflet.
- always check the ingredients to make sure you can use this medicine.
- you have an intolerance to some sugars. VERZENIO contains lactose (a type of sugar found in milk and dairy products).
Check with your doctor if you:
- have any other medical conditions. Severe liver problems; blood clots in the veins or arteries, infections, inflammation and infection of the lung
- take any medicines for any other condition.
- pregnant or breast feeding.
- a family history of blood clots.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not take this medicine if you are pregnant. It may affect your developing baby if you take it during pregnancy.
You should use effective birth control (contraception) during treatment with VERZENIO and for 3 weeks after the last dose of VERZENIO.
Do not breast-feed if you are taking this medicine and for at least 3 weeks after the last dose of VERZENIO.
It is not known if VERZENIO is excreted into breast milk and if the breast-fed infant is at risk of harm. Talk to your doctor about breast-feeding during or after treatment with VERZENIO.
VERZENIO may decrease fertility in men.
Child or adolescent
- Do not give this medicine to a child or adolescent under the age of 18 years. Safety and effectiveness in children younger than 18 years have not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with VERZENIO and affect how it works, or VERZENIO may affect how other medicines work.
These can either increase or decrease the amount of abemaciclib in the blood.
Medicines that may increase the effect of VERZENIO include:
- Clarithromycin (antibiotic used to treat bacterial infections).
- Ketoconazole, itraconazole, voriconazole, posaconazole (used to treat fungal infections).
- Diltiazem (used to treat chest pain (angina) and high blood pressure).
- Verapamil (used to treat chest pain (angina), high blood pressure and heart rhythm problems).
- Lopinavir/ritonavir (used to treat HIV/AIDS).
Do not take grapefruit products while on this therapy as it may increase the side effects of VERZENIO.
Medicines that may reduce the effect of VERZENIO include:
- Carbamazepine, phenytoin (used to treat epilepsy to prevent convulsions, fits).
- Rifampicin (used to treat tuberculosis (TB))
- St. John's wort (a herbal product used to treat mild depression and anxiety).
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect VERZENIO.
4. How do I use VERZENIO?
How much to take / use
- Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
- When given together with endocrine therapy to treat your breast cancer, the recommended dose of VERZENIO is 150 mg orally, twice daily.
- Swallow the tablet whole with a full glass of water. Do not chew, crush or split the tablets before swallowing. VERZENIO may be taken with or without food. Avoid grapefruit products.
- Women treated with the combination of VERZENIO plus endocrine therapy should be in a postmenopausal state prior to therapy.
When to take / use VERZENIO
- Take your medicine at about the same time each day.
- Taking it at the same time each day will have the best effect. It ensures that there is enough VERZENIO in your body all the time. It will also help you remember when to take it.
- For early breast cancer your doctor will advise you to take VERZENIO for adjuvant treatment for up to 2 years unless advised otherwise.
- For metastatic breast cancer treatment should be continued until disease progression or unacceptable toxicity. Consult your doctor regarding adverse events.
If you forget to use VERZENIO
VERZENIO should be used regularly at the same time each day. If you miss (vomit) a dose, take your next dose at its scheduled time.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you use too much VERZENIO
If you think that you have used too much VERZENIO, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using VERZENIO?
Things you should do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking VERZENIO.
Call your doctor straight away if you:
- become pregnant.
If you are about to have any blood tests, tell your doctor that you are taking this medicine. It may interfere with the results of some tests.
Remind any doctor, dentist or pharmacist you visit that you are using VERZENIO.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
- have diarrhoea
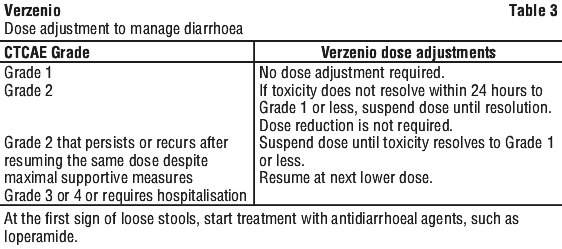
At the first sign of diarrhoea, tell your doctor or pharmacist. Your healthcare provider may tell you to temporarily stop taking VERZENIO, stop your treatment, or decrease your dose. Diarrhoea may cause you to develop dehydration or an infection.
If you have any loose stools, right away tell your healthcare provider, start taking an antidiarrheal medicine (such as loperamide), and drink more fluids.
Things you should not do
- Do not take VERZENIO to treat any other complaints unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Do not stop taking your medicine or lower the dosage without checking with your doctor.
- If necessary, your doctor will gradually reduce the amount you take each day before stopping the medicine completely.
- Do not take grapefruit products while on this therapy as it may increase the side effects of VERZENIO.
Blood tests
- Your doctor will perform a blood test before and during treatment to check whether VERZENIO affects the number of white cells in your blood or the concentration in your blood of enzymes that come from your liver. VERZENIO may reduce the number of white blood cells and produce abnormalities in liver blood tests.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how VERZENIO affects you.
This medicine may cause fatigue and dizziness in some people. If you experience any symptoms affecting your ability to concentrate and react, do not drive or use machines until the effect goes away.
Drinking alcohol
Tell your doctor if you drink alcohol.
Be careful when drinking alcohol while you are taking this medicine as this medicine may cause fatigue and dizziness in some people.
Looking after your medicine
- Keep your tablets in the pack until it is time to take them.
- If you take the tablets out of the pack they may not keep well.
- Keep your tablets in a cool dry place where the temperature stays below 30°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sill.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
Your doctor may lower your dose or stop treatment temporarily to try to reduce certain side effects while you are taking VERZENIO.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Stomach-related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
General symptoms:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Other side effects you may not be aware of:
- low red blood cell counts (anaemia) which can make you feel lethargic and very tired
- low white blood cell counts which can make you more susceptible to developing infections
- low levels of platelets in blood (thrombocytopenia) which can lead to an increased risk of bleeding or bruising
- low levels of potassium (hypokalaemia) which can lead to feeling tired, leg cramps and weakness
- increased creatinine which can lead to tiredness, swelling and muscle cramps.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
These side effects may not be fully attributable to VERZENIO alone but may be caused by your condition or by other medicines that you may be taking at the same time.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What VERZENIO contains
| Active ingredient (main ingredient) | abemaciclib |
| Other ingredients (inactive ingredients) | croscarmellose sodium lactose monohydrate microcrystalline cellulose silicon dioxide sodium stearyl fumarate polyvinyl alcohol (E1203) titanium dioxide (E171) macrogol 3350 (E1521) purified talc (E553b) iron oxide yellow (E172) [50 mg and 150 mg tablets only] iron oxide red (E172) [50 mg tablets only] |
Do not take this medicine if you are allergic to any of these ingredients.
This medicine does not contain sucrose, gluten, tartrazine or any other azo dyes. Contains lactose.
What VERZENIO looks like
VERZENIO is a modified oval immediate-release film coated tablet with “Lilly” debossed on one side and tablet strength in mg debossed on the other.
50 mg tablets are beige in colour (Aust R 304765)
100 mg tablets are white in colour (Aust R 304767)
150 mg tablets are yellow in colour (Aust R 304766)
Who distributes VERZENIO
Eli Lilly Australia Pty Ltd
Level 9, 60 Margaret Street
Sydney NSW 2000
AUSTRALIA
™ = Trademark
If you have any questions about VERZENIO, contact Eli Lilly at 1800 454 559 (Australia) or your healthcare professional for assistance.
This leaflet was prepared in July 2025
vA08
Brand Information
| Brand name | Verzenio |
| Active ingredient | Abemaciclib |
| Schedule | S4 |
▼ This medicinal product is subject to additional monitoring in Australia. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse events at www.tga.gov.au/reporting-problems.
MIMS Revision Date: 01 September 2025
1 Name of Medicine
Abemaciclib.
2 Qualitative and Quantitative Composition
Abemaciclib (Verzenio) 50 mg film-coated tablets. Each film-coated tablet contains 50 mg abemaciclib.
Excipients with known effect. Each film-coated tablet contains 14 mg of lactose (monohydrate).
Abemaciclib (Verzenio) 100 mg film-coated tablets. Each film-coated tablet contains 100 mg abemaciclib.
Excipients with known effect. Each film-coated tablet contains 28 mg of lactose (monohydrate).
Abemaciclib (Verzenio) 150 mg film-coated tablets. Each film-coated tablet contains 150 mg abemaciclib.
Excipients with known effect. Each film-coated tablet contains 42 mg of lactose (monohydrate).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Verzenio 50 mg film-coated tablets. Beige, modified oval tablet debossed with "Lilly" on one side and "50" on the other.
Verzenio 100 mg film-coated tablets. White, modified oval tablet debossed with "Lilly" on one side and "100" on the other.
Verzenio 150 mg film-coated tablets. Yellow, modified oval tablet debossed with "Lilly" on one side and "150" on the other.
4 Clinical Particulars
4.1 Therapeutic Indications
Early breast cancer. Verzenio in combination with endocrine therapy is indicated for the adjuvant treatment of patients with hormone receptor (HR) positive, human epidermal growth factor receptor 2 (HER2) negative, node-positive early breast cancer at high risk of recurrence.
In pre- or peri-menopausal women, endocrine therapy should be combined with a luteinising hormone-releasing hormone (LHRH) agonist.
Advanced or metastatic breast cancer. Verzenio is indicated for the treatment of hormone receptor (HR) positive, human epidermal growth factor receptor 2 (HER2) negative, locally advanced or metastatic breast cancer either in combination with an aromatase inhibitor or fulvestrant as initial endocrine-based therapy, or following prior endocrine therapy.
In pre- or peri-menopausal women, the endocrine therapy should be combined with a luteinising hormone-releasing hormone (LHRH) agonist.
4.2 Dose and Method of Administration
Verzenio therapy should be initiated and supervised by physicians experienced in the use of anti-cancer therapies.
The recommended dose of Verzenio is 150 mg orally, twice daily in combination with endocrine therapy at its recommended dose.
Verzenio may be taken with or without food.
Early breast cancer. Treatment with Verzenio plus endocrine therapy should be combined with LHRH agonist for pre-menopausal women.
Verzenio should be taken continuously for two years, or until disease recurrence or unacceptable toxicity occurs whichever comes first.
Advanced or metastatic breast cancer. Women treated with the combination of Verzenio plus endocrine therapy should be in a postmenopausal state prior to therapy.
It is recommended that treatment be continued until disease progression or unacceptable toxicity.
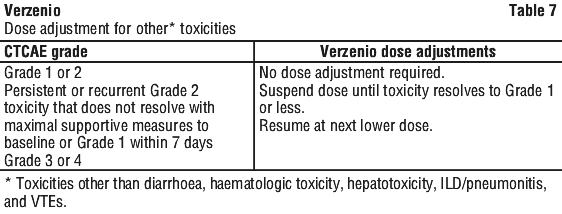
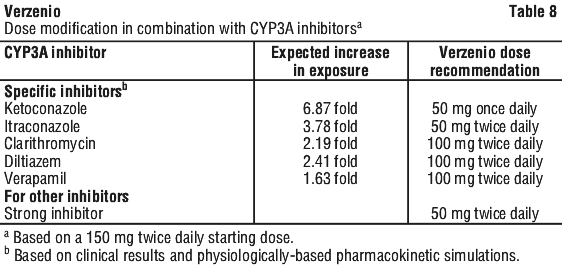
Dose adjustment for managing adverse effects. Management of some adverse reactions may require dose interruption and/or dose reduction. If dose reduction is necessary, decrease the dose by 50 mg at a time (see Table 1). Discontinue Verzenio for patients unable to tolerate 50 mg twice daily. See Tables 2-7.





Avoid foods that inhibit CYP3A activity, such as grapefruit or grapefruit juice. If a CYP3A inhibitor is discontinued, increase the Verzenio dose (after 3-5 half-lives of the inhibitor) to the dose that was used before starting the inhibitor (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
CYP3A inducers. Avoid concomitant use of CYP3A inducers. Consider alternative agents without CYP3A induction (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Dosing in severe hepatic impairment. Decrease the dosing frequency to once daily (see Section 5.2 Pharmacokinetic Properties).
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed (see Section 6.1 List of Excipients).
4.4 Special Warnings and Precautions for Use
Neutropenia. Grade ≥ 3 neutropenia was reported in patients receiving abemaciclib in breast cancer studies. Monitor complete blood counts prior to starting abemaciclib therapy, every 2 weeks for the first 2 months, monthly for the next 2 months, and as clinically indicated. Fatal events of neutropenic sepsis occurred in < 1% of patients with metastatic breast cancer. Patients should be instructed to report any episode of fever to their healthcare provider. Dose modification is recommended for patients who develop grade 3 or 4 neutropenia (see Section 4.2 Dose and Method of Administration).
Infections/infestations. Infections were reported in patients receiving abemaciclib plus endocrine therapy at a higher rate than in patients treated with endocrine therapy. Lung infection was reported in patients receiving abemaciclib without concurrent neutropenia. Fatal events occurred in < 1% of patients with metastatic breast cancer. Patients should be monitored for signs and symptoms of infection and treated as medically appropriate.
Venous thromboembolism. Verzenio has not been studied in patients with early breast cancer who had a history of venous thromboembolism. In early stage breast cancer, venous thromboembolisms of any grade were reported in 2.5% of patients treated with adjuvant abemaciclib plus endocrine therapy and 0.6% with endocrine therapy alone. Careful consideration should be given to the choice of endocrine therapy and known risks associated with VTEs, such as prior history of venous thromboembolic events.
In metastatic breast cancer venous thromboembolic events were reported in 5.3% of patients treated with abemaciclib plus fulvestrant or aromatase inhibitors, compared to 0.8% of patients treated with placebo plus fulvestrant or aromatase inhibitors. Patients should be monitored for signs and symptoms of deep vein thrombosis and pulmonary embolism and treated as medically appropriate. Abemaciclib dose modification and suspension may be required (see Section 4.2 Dose and Method of Administration).
Arterial thromboembolic events. Serious arterial thromboembolic events (ATEs), including ischemic stroke and myocardial infarction, have been reported more frequently in the abemaciclib treatment arms in metastatic breast cancer studies when administered in combination with endocrine therapies.
In metastatic breast cancer, ATEs of any grade have been reported in 4.4% of patients treated with abemaciclib plus endocrine therapy, compared to 2.4% of patients treated with placebo plus endocrine therapy. A causal association has not been established. The benefits and risks of continuing abemaciclib in patients who experience a severe ATE should be considered.
Increased ALT/AST. Grade ≥ 3 increased ALT/AST was reported in patients receiving abemaciclib in breast cancer studies. Monitor ALT/AST prior to the start of abemaciclib therapy, every 2 weeks for the first 2 months, monthly for the next 2 months, and as clinically indicated. Based on the level of ALT/AST elevations, dose modification may be required (see Section 4.2 Dose and Method of Administration).
Interstitial lung disease (ILD)/pneumonitis. Severe, life threatening or fatal interstitial lung disease (ILD) and/or pneumonitis can occur in patients treated with Verzenio and other CDK4/6 inhibitors. In Verzenio treated patients in early breast cancer (monarchE), 3.2% of patients experienced ILD/pneumonitis of any grade: 0.4% were Grade 3 or 4 and there was one fatality (< 0.1%). In Verzenio treated patients in metastatic breast cancer (MONARCH 1, MONARCH 2, MONARCH 3), 3.3% of Verzenio-treated patients had ILD/pneumonitis of any grade, 0.6% had grade 3 or 4, and 0.4% had fatal outcomes. Additional cases of ILD/pneumonitis have been observed in the post-marketing setting, with fatalities reported.
Monitor patients for pulmonary symptoms indicative of ILD/pneumonitis. Symptoms may include hypoxia, cough, dyspnoea, or interstitial infiltrates on radiologic exams. Infectious, neoplastic and other causes for such symptoms should be excluded by means of appropriate investigations.
Dose interruptions or dose reduction is recommended for patients who develop persistent or recurrent grade 2 ILD/pneumonitis. Permanently discontinue Verzenio in all patients with grade 3 or 4 ILD or pneumonitis. (See Section 4.2 Dose and Method of Administration for dose modification).
Diarrhoea. Diarrhoea is the most common adverse reaction. Across clinical studies, median time to onset of the first diarrhoea event was approximately 6 to 8 days, and median duration of diarrhoea was 7 to 12 days (grade 2) and 5 to 8 days (grade 3). Diarrhoea can be associated with dehydration. Patients should start treatment with antidiarrhoeal agents such as loperamide at the first sign of loose stools, increase oral fluids and notify their healthcare provider. Dose modification is recommended for patients who develop ≥ grade 2 diarrhoea (see Section 4.2 Dose and Method of Administration).
Use in hepatic impairment. Abemaciclib is metabolised in the liver. In subjects with severe hepatic impairment, total abemaciclib unbound exposure increased 2.69-fold, and the abemaciclib half-life increased from 24 to 55 hours. Reduce the abemaciclib dosing frequency to once daily in patients with severe hepatic impairment (see Section 4.2 Dose and Method of Administration).
Use in renal impairment. Abemaciclib and its metabolites are not significantly cleared renally. Dose adjustment is not necessary in patients with mild or moderate renal impairment. There are no data in patients with severe renal impairment, end stage renal disease, or in patients on dialysis.
Use in the elderly. Age had no effect on the exposure of abemaciclib in a population pharmacokinetic analysis in patients with cancer (135 males and 859 females; age range 24-91 years; and body weight range 36-175 kg).
Paediatric use. The safety and efficacy of abemaciclib in children aged less than 18 years have not been established. No data are available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Effects of other medicinal products on the pharmacokinetics of abemaciclib. Abemaciclib is primarily metabolised by CYP3A4.
CYP3A4 inhibitors. Co-administration of abemaciclib with CYP3A4 inhibitors can increase plasma concentrations of abemaciclib. In patients with advanced and/or metastatic cancer, co-administration of the CYP3A4 inhibitor clarithromycin resulted in a 3.4-fold increase in the plasma exposure of abemaciclib and a 2.5-fold increase in the combined unbound potency adjusted plasma exposure of abemaciclib and its active metabolites.
Use of strong CYP3A4 inhibitors together with abemaciclib should be avoided. If strong CYP3A4 inhibitors need to be co-administered, the dose of abemaciclib should be reduced (see Section 4.2 Dose and Method of Administration), followed by careful monitoring of toxicity. Examples of strong CYP3A4 inhibitors include, but not limited to: clarithromycin, itraconazole, ketoconazole, lopinavir/ritonavir, posaconazole or voriconazole. Avoid grapefruit or grapefruit juice.
No dose adjustment is necessary for patients treated with moderate or weak CYP3A4 inhibitors. There should, however, be close monitoring for signs of toxicity.
CYP3A4 inducers. Co-administration of abemaciclib with the strong CYP3A4 inducer rifampicin decreased the plasma concentration of abemaciclib by 95% and unbound potency adjusted plasma concentration of abemaciclib plus its active metabolites by 77% based on AUC0-∞. Concomitant use of strong CYP3A4 inducers (including, but not limited to: carbamazepine, phenytoin, rifampicin and St. John's wort) should be avoided due to the risk of decreased efficacy of abemaciclib.
Effects of abemaciclib on the pharmacokinetics of other medicinal products. Medicinal products that are substrates of transporters. Abemaciclib and its major active metabolites inhibit the renal transporters organic cation transporter 2 (OCT2), multidrug and extrusion toxin protein (MATE1), and MATE2-K. In vivo interactions of abemaciclib with clinically relevant substrates of these transporters, such as dofetilide or creatinine, may occur (see Section 4.8 Adverse Effects (Undesirable Effects)). In a clinical drug interaction study with metformin (substrate of OCT2, MATE1 and 2) co-administered with 400 mg abemaciclib, a small but not clinically relevant increase (37%) in metformin plasma exposure was observed. This was found to be due to reduced renal secretion with unaffected glomerular filtration.
In healthy subjects, co-administration of abemaciclib and the P-glycoprotein (P-gp) substrate loperamide resulted in an increase in loperamide plasma exposure of 9% based on AUC0-∞ and 35% based on Cmax. This was not considered to be clinically relevant. However, based on the in vitro inhibition of P-gp and breast cancer resistance protein (BCRP) observed with abemaciclib, in vivo interactions of abemaciclib with narrow therapeutic index substrates of these transporters, such as digoxin or dabigatran etexilate, may occur.
In a clinical study in patients with breast cancer, there was no clinically-relevant pharmacokinetic drug interaction between abemaciclib and anastrozole, fulvestrant, exemestane, letrozole or tamoxifen. It is currently unknown whether abemaciclib may reduce the effectiveness of systemically acting hormonal contraceptives, and therefore patients using systemically acting hormonal contraceptives are advised to add a barrier method.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Effects on fertility and early embryonic development were assessed in rats. While no effects on male fertility were seen in rats with abemaciclib up to 10 mg/kg/day (approximately 4 times higher than clinical exposure based on AUC), effects to the male reproductive organs in mice, rats and dogs (see below) indicate that abemaciclib may impair fertility in males. No effects on female fertility and early embryonic development were observed in rats at up to 20 mg/kg/day (approximately 4.5 times the clinical exposure based on AUC).
In toxicity studies in mice (150 mg/kg/day), rats (≥ 0.3 mg/kg/day) and dogs (≥ 0.3 mg/kg/day), abemaciclib-related findings in the testis, epididymis, prostate, and seminal vesicle included decreased organ weights, intratubular cellular debris, hypospermia, tubular dilatation, atrophy, and/or degeneration/necrosis. Exposures at these doses in rats and dogs are less than the clinical exposure (based on AUC) and 11 times higher in mice than clinical exposure. In female mice, decreased number of corpora lutea were observed at ≥ 30 mg/kg/day, which is approximately 7.9 times higher than clinical exposure based.
Patients who could become pregnant should use highly effective contraception during treatment and for 3 weeks after the last dose of abemaciclib.
Use in pregnancy. (Category D)
There are no data on the use of Verzenio in human pregnancy. Based on findings in animals, and its mechanism of action, abemaciclib can cause fetal harm when administered to a pregnant patient. When pregnant rats were treated during the period of organogenesis (dose of ≥ 4 mg/kg/day; approximately equal to the human clinical exposure based on AUC), reduced fetal weights were observed in the absence of maternal toxicity, accompanied by an increased incidence of cardiovascular and skeletal malformations and variations (absent innominate artery and aortic arch, malpositioned subclavian artery, unossified sternebra, bipartite ossification of thoracic centrum, and rudimentary or nodulated ribs).
Verzenio is not recommended during pregnancy. Highly effective contraception is recommended.
Use in lactation. There are no data on the presence of abemaciclib in human milk, effects of abemaciclib on the breastfed child, or effects of abemaciclib on milk production. Due to the potential for serious adverse reactions in a child, patients should not breastfeed during treatment with abemaciclib, or for three weeks after the last dose of abemaciclib.
4.7 Effects on Ability to Drive and Use Machines
The effects of Verzenio on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Summary of the safety profile. The most commonly occurring adverse reactions are diarrhoea, infections, neutropenia, leukopenia, anaemia, fatigue, nausea, vomiting, decreased appetite and alopecia.
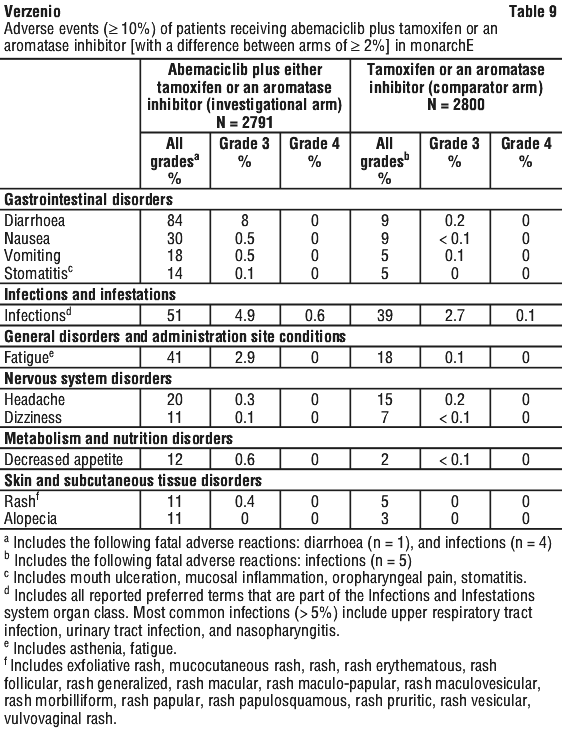
Safety in early breast cancer studies. monarchE - abemaciclib plus either tamoxifen or an aromatase inhibitor in patients with early breast cancer. The safety of Verzenio in early breast cancer was evaluated in monarchE, a study in which 2791 adult patients received Verzenio plus endocrine therapy (either tamoxifen or an aromatase inhibitor) and 2800 received endocrine therapy alone (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
The most frequently (≥ 5%) reported Grade ≥ 3 adverse reactions were neutropenia, leukopenia, diarrhoea and lymphopenia.
Fatal adverse reactions occurred in 0.8% of patients who received Verzenio plus endocrine therapy including: cardiac failure (0.1%), cardiac arrest, myocardial infarction, ventricular fibrillation, cerebral haemorrhage, cerebrovascular accident, pneumonitis, hypoxia, diarrhoea and mesenteric artery thrombosis (0.03% each).
Verzenio treatment discontinuation due to an adverse reaction was reported in 19% of patients receiving Verzenio, plus endocrine therapy. The most common adverse reactions leading to Verzenio discontinuations were diarrhoea (5%), fatigue (2%), and neutropenia (0.9%). Of the patients receiving endocrine therapy, 1% permanently discontinued due to an adverse reaction.
Dose interruption of Verzenio due to an adverse reaction occurred in 62% of patients receiving Verzenio plus endocrine therapy. Adverse reactions leading to Verzenio dose interruptions in ≥ 3% of patients were diarrhoea (20%), neutropenia (16%), leukopenia (7%), and fatigue (5%).
Dose reductions of Verzenio due to adverse reaction occurred in 44% of patients receiving Verzenio plus endocrine therapy. Adverse reactions leading to Verzenio dose reductions were diarrhoea (17%), neutropenia (8%), fatigue (5%) and leukopenia (4%).
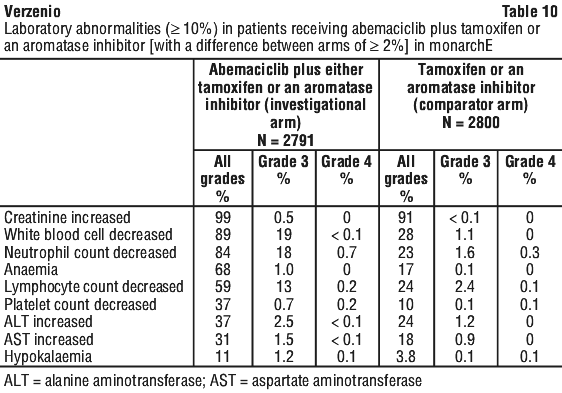
The most common adverse events and laboratory abnormalities with abemaciclib treatment in monarchE are summarised in Table 9 and Table 10, respectively.
Pruritus - 9%.
Dyspepsia - 8%.
Nail disorder - 6% (includes nail bed disorder, nail bed inflammation, nail discoloration, nail disorder, nail dystrophy, nail pigmentation, nail ridging, nail toxicity, onychalgia, onychoclasis, onycholysis, onychomadesis).
Lacrimation increased - 6%.
Dysgeusia - 5%.
Interstitial lung disease (ILD)/pneumonitis - 3% (includes pneumonitis, radiation pneumonitis, interstitial lung disease, pulmonary fibrosis, organising pneumonia, radiation fibrosis - lung, lung opacity, sarcoidosis).
Venous thromboembolic events (VTEs) - 3% (includes catheter site thrombosis, cerebral venous thrombosis, deep vein thrombosis, device related thrombosis, embolism, hepatic vein thrombosis, jugular vein occlusion, jugular vein thrombosis, ovarian vein thrombosis, portal vein thrombosis, pulmonary embolism, subclavian vein thrombosis, venous thrombosis limb).
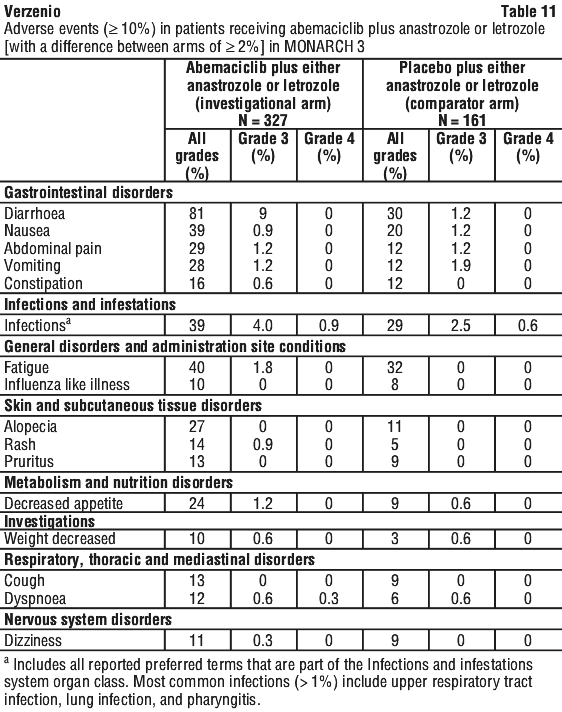
MONARCH 3 - abemaciclib plus aromatase inhibitor in postmenopausal women with HR-positive, HER2-negative locoregionally recurrent or metastatic breast cancer with no prior systemic therapy in this disease setting. In MONARCH 3, 488 women were randomly assigned to receive 150 mg of Verzenio or placebo orally twice daily, plus physician's choice of anastrozole or letrozole once daily (see Section 5.1 Pharmacodynamic Properties, Clinical trials). Median duration of treatment was 15 months for the Verzenio arm and approximately 14 months for the placebo arm.
The most frequently reported (≥ 5%) Grade 3 or 4 adverse reactions were neutropenia, diarrhoea, leukopenia, increased ALT, and anaemia.
Deaths during treatment or during the 30-day follow up, regardless of causality, were reported in 11 cases (3%) of Verzenio plus an aromatase inhibitor treated patients versus 3 cases (2%) of placebo plus an aromatase inhibitor treated patients. Causes of death for patients receiving Verzenio plus an aromatase inhibitor included: 3 (0.9%) patient deaths due to underlying disease, 3 (0.9%) due to lung infection, 3 (0.9%) due to VTE, 1 (0.3%) due to pneumonitis, and 1 (0.3%) due to cerebral infarction.
Permanent treatment discontinuation due to an adverse reaction was reported in 13% of patients receiving Verzenio plus an aromatase inhibitor and in 3% of patients receiving placebo plus an aromatase inhibitor. Adverse reactions leading to permanent discontinuation for patients receiving Verzenio plus an aromatase inhibitor were diarrhoea (2%), ALT increased (2%), infection (1%), venous thromboembolic events (VTE) (1%), neutropenia (0.9%), renal impairment (0.9%), AST increased (0.6%), dyspnoea (0.6%), pulmonary fibrosis (0.6%) and anaemia, rash, weight decreased and thrombocytopenia (each 0.3%).
Dose interruption of Verzenio due to an adverse reaction occurred in 56% of patients receiving Verzenio plus anastrozole or letrozole. Adverse reactions leading to Verzenio dose interruptions in ≥ 5% of patients were neutropenia (16%) and diarrhoea (15%).
Dose reductions due to an adverse reaction occurred in 43% of patients receiving Verzenio plus anastrozole or letrozole. Adverse reactions leading to dose reductions in ≥ 5% of patients were diarrhoea and neutropenia. Verzenio dose reductions due to diarrhoea of any grade occurred in 13% of patients receiving Verzenio plus an aromatase inhibitor compared to 2% of patients receiving placebo plus an aromatase inhibitor. Verzenio dose reductions due to neutropenia of any grade occurred in 11% of patients receiving Verzenio plus an aromatase inhibitor compared to 0.6% of patients receiving placebo plus an aromatase inhibitor.
The most common adverse events are shown in Table 11. The most common laboratory abnormalities in patients receiving Verzenio in MONARCH 3 were similar to those in monarchE, in the early breast cancer setting (see Table 10).
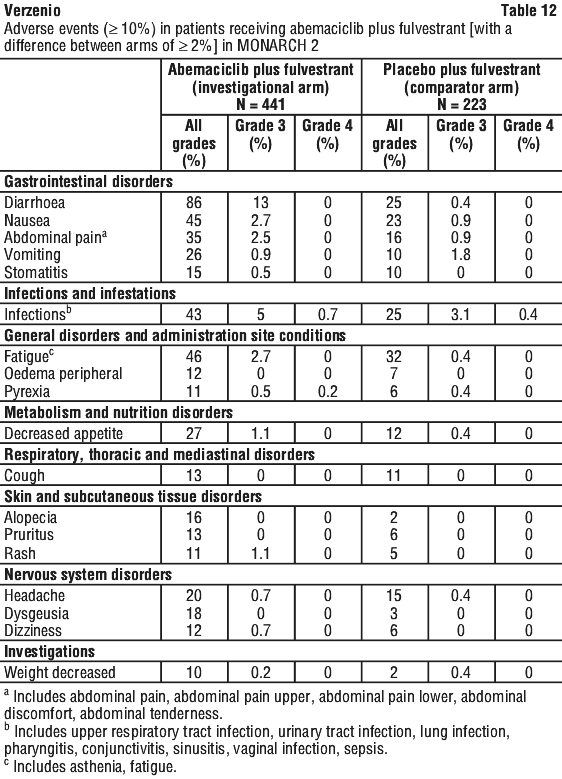
In MONARCH 2, deaths during treatment or during the 30-day follow up period, regardless of causality, were reported for 18 patients (4%) receiving Verzenio plus fulvestrant and 10 patients (5%) receiving placebo plus fulvestrant. Causes of death for patients receiving Verzenio plus fulvestrant included: 7 (2%) patient deaths due to underlying disease, 4 (0.9%) due to sepsis, 2 (0.5%) due to pneumonitis, 2 (0.5%) due to hepatotoxicity, and one (0.2%) due to cerebral infarction.
The most common adverse events in MONARCH 2 are summarised in Table 12. The most common laboratory abnormalities in patients receiving Verzenio in MONARCH 2 were similar to those in monarchE, in the early breast cancer setting (see Table 10).
Description of selected adverse reactions. Neutropenia. In the monarchE study, neutropenia was reported in 46% of patients. Grade ≥ 3 decrease in neutrophil counts (based on laboratory findings) was reported in 19% of patients receiving abemaciclib in combination with endocrine therapy. The median time to onset of Grade ≥ 3 neutropenia was 30 days, and median time to resolution was 16 days. Febrile neutropenia was reported in 0.3% patients. Neutropenia was also very common in MONARCH 2 and MONARCH 3 (45%) and a grade 3 or 4 decrease in neutrophil counts (based on laboratory findings) was reported in 28.2% of patients receiving abemaciclib in combination with aromatase inhibitors or fulvestrant. The median times to onset of grade 3 or 4 neutropenia were 29 and 33 days, and median times to resolution were 11 and 15 days. Febrile neutropenia was reported in 0.9% of patients. Dose modification is recommended for patients who develop grade 3 or 4 neutropenia [see Section 4.2 Dose and Method of Administration].
Diarrhoea. Diarrhoea was the most commonly reported adverse reaction (see Table 9, Table 11 and Table 12). Incidence was greatest during the first month of abemaciclib treatment. The median time to onset of a first diarrhoea event of any grade was 6 to 8 days across studies. The median duration of Grade 2 diarrhoea was 6 to 11 days across studies and of Grade 3 diarrhoea was 5 to 8 days across studies. Most diarrhoea events recovered or resolved with supportive treatment such as loperamide and/or dose adjustment [see Section 4.2 Dose and Method of Administration]. In MONARCH 3, 19% of patients with diarrhoea required a dose omission and 13% required a dose reduction. The median time to the first dose reduction due to diarrhoea in MONARCH 3 was 38 days.
Increased aminotransferases ((ALT/AST). Dose modification is recommended for patients who develop grade 3 or 4 ALT or AST increase [see Section 4.2 Dose and Method of Administration].
In the monarchE study, ALT and AST elevations were very common in patients receiving abemaciclib in combination with endocrine therapy (see Table 10). Grade 3-4 ALT and AST elevations (based on laboratory findings) were reported in 2.6% and 1.6% of patients, respectively. The median time to onset of Grade ≥ 3 ALT elevation was 118 days, and median time to resolution was 15 days. The median time to onset of Grade ≥ 3 AST elevation was 91 days, and median time to resolution was 11 days.
ALT and AST elevations were also very common in the MONARCH 2 and MONARCH 3 studies amongst patients receiving abemaciclib in combination with aromatase inhibitors or fulvestrant. Grade 3 or 4 ALT or AST elevations (based on laboratory findings) were reported in 6.1% and 4.2% of patients, respectively. The median time to onset of Grade 3-4 ALT elevation was 57 days in MONARCH 2 and 61 days in MONARCH 3, and median time to resolution was 14 days in both studies. The median time to onset of Grade 3-4 AST elevation was 71 days in MONARCH 3 and 185 days in MONARCH 2, and median time to resolution was 13 days (MONARCH 2) and 15 days (MONARCH 3).
Creatinine. Abemaciclib causes asymptomatic serum creatinine elevation by inhibiting renal tubular secretion transporters, but does not affect glomerular function as measured by iohexol clearance (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). In clinical studies, increases in serum creatinine occurred within the first month of abemaciclib dosing, remained elevated but stable through the treatment period, and were reversible upon treatment discontinuation. Creatinine elevations were not accompanied by changes in markers of renal function, such as blood urea nitrogen (BUN), cystatin C, or calculated glomerular filtration rate based on cystatin C.
Laboratory findings of serum creatinine elevation occurred in 99% of patients randomised to receive abemaciclib in the monarchE study, and 98% of those in the MONARCH 2 and MONARCH 3 studies. The incidence of Grade 3 or higher elevations was 0.5% and 2%, respectively. The incidence of laboratory findings of serum creatinine elevation amongst patients in the comparator arm was 91% in monarchE and 78% in MONARCH 2 and MONARCH 3.
Post-marketing experience. The following adverse drug reactions are based on post-marketing reports.
Respiratory, thoracic, and mediastinal disorders. Interstitial lung disease/pneumonitis: common (≥ 1.0% - < 10%).
Skin and subcutaneous tissue disorders. Erythema multiforme: rare (≥ 0.01% - < 0.1%).
4.9 Overdose
There is no known antidote for abemaciclib overdose. In case of overdose, use supportive therapy.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
In cancer patients, abemaciclib inhibits CDK4 and CDK6 as indicated by inhibition of phosphorylation of Rb and topoisomerase II alpha, which results in cell cycle inhibition upstream of the G1 restriction point at doses of 50 mg to 200 mg twice daily. MONARCH 2 and MONARCH 3 exposure-response analyses support the 150 mg twice daily starting dose in combination with endocrine therapy and support dose reductions as needed for tolerability to a dose as low as 50 mg twice daily. MONARCH 1 exposure-response analysis supports the 200 mg twice daily starting dose when used as a single agent. The effect of abemaciclib on the QTcF interval was evaluated in 144 patients with advanced cancer. No large change (that is, > 20 ms) in the QTcF interval was detected at the mean observed maximal steady state abemaciclib concentration following a therapeutic dosing schedule. In an exposure-response analysis in healthy subjects at the highest clinically relevant exposures, abemaciclib did not prolong the QTcF interval to any clinically relevant extent.
Mechanism of action. Abemaciclib is an inhibitor of cyclin D-dependent kinases 4 and 6 (CDK4 and CDK6) and was most active against cyclin D1/CDK4 in enzymatic assays. In breast cancer, cyclin D1/CDK4 has been shown to promote phosphorylation of the retinoblastoma protein (Rb), cell proliferation, and tumour growth. Abemaciclib prevents Rb phosphorylation, blocking progression from G1 into S phase of the cell cycle, leading to suppression of tumour growth in preclinical models following short duration target inhibition. In oestrogen receptor-positive breast cancer cell lines, sustained target inhibition by abemaciclib prevents rebound of Rb phosphorylation and cell cycle reentry, resulting in senescence and apoptosis. In breast cancer xenograft models, abemaciclib dosed daily without interruption at clinically relevant doses - as a single agent or in combination with antioestrogens - resulted in reduction of tumour size.
Clinical trials. Cardiac electrophysiology. The effect of abemaciclib on the QTcF interval was evaluated in 144 patients with advanced cancer. No large change (that is, > 20 ms) in the QTcF interval was detected at the mean observed maximal steady state abemaciclib concentration following a therapeutic dosing schedule.
In an exposure-response analysis in healthy subjects at exposures comparable to a 200 mg twice daily dose, abemaciclib did not prolong the QTcF interval to any clinically relevant extent.
Early breast cancer. Randomised phase 3 study monarchE: Verzenio in combination with endocrine therapy. The efficacy and safety of Verzenio in combination with adjuvant endocrine therapy was evaluated in monarchE a randomised, open-label, multicentre study in adult women and men with HR-positive, HER2-negative, node-positive, resected, early breast cancer (EBC) with clinical and pathological features consistent with a high risk of disease recurrence. To be enrolled, all patients had to have HR positive, HER2-negative EBC with tumour involvement in at least 1 axillary lymph node (pALN). Two cohorts of patients were enrolled. To be enrolled in cohort 1, patients needed to have either ≥ 4 pALN, or pALN 1-3 and either tumour grade 3 or tumour size ≥ 50 mm. To be enrolled in cohort 2, patients were required to have pALN 1-3 and Ki-67 index of ≥ 20% as measured in untreated breast tumour tissue, using a clinical trial assay at a central laboratory. The intent to treat (ITT) population included patients from both cohort 1 (n=5120) and cohort 2 (n=517). Patients with history of VTEs were excluded from this study. Randomisation to treatment was stratified by prior treatment (neoadjuvant chemotherapy versus adjuvant chemotherapy versus no chemotherapy); menopausal status (premenopausal versus postmenopausal); and region (North America/Europe versus Asia versus other). Men were stratified as post-menopausal.
A total of 5637 patients were randomised in a ratio 1:1 to receive 2 years of Verzenio 150 mg twice daily plus physician's choice of standard endocrine therapy or standard endocrine therapy alone; with or without LHRH agonist. After the end of the study treatment period, standard adjuvant endocrine therapy is to be continued for a duration of at least 5 years if deemed medically appropriate. Initial endocrine therapy received by patients included letrozole (38.2%), tamoxifen (31.4%), anastrozole (22.0%), or exemestane (8.1%). The use of LHRH analogues during the study was 21.7% and 22.4% in Verzenio plus endocrine therapy and endocrine therapy arms, respectively. In premenopausal women, (N=2451), aromatase inhibitor and tamoxifen was used in 41.2% and 58.2% patients respectively, and LHRH agonists was used in 74.6% patients on aromatase inhibitor and in 30.1% patients on tamoxifen.
Patient median age was 51 years (range, 22-89 years), 99% were women, 71% were White, and 24% were Asian. Forty-four percent of patients were premenopausal. Most patients received prior chemotherapy (37% neoadjuvant, 62% adjuvant) and prior radiotherapy (95%). Sixty percent of the patients had 4 or more positive lymph nodes with 20% having ≥ 10 positive lymph nodes, 38% had Grade 3 tumour, and 22% had pathological tumour size ≥ 50 mm. Most patients were progesterone receptor positive (87%) and 44% had high Ki-67 index as determined by the central clinical trial assay.
The primary end point was invasive disease-free survival (IDFS). IDFS was defined as the time from randomisation to the first occurrence of ipsilateral invasive breast tumour recurrence, regional invasive breast cancer recurrence, distant recurrence, contralateral invasive breast cancer, second primary non-breast invasive cancer, or death attributable to any cause. Secondary end point distant relapse free survival is defined as the time from randomisation to distant recurrence or death from any cause, whichever occurs first.
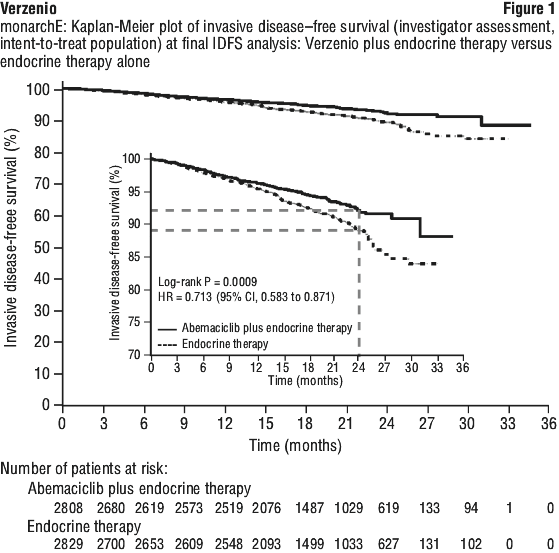
At the pre planned interim analysis (IA2) with a median follow-up time of 15.4 months, the primary objective of the study was met. A statistically significant improvement in IDFS was observed in patients who received Verzenio plus endocrine therapy versus endocrine therapy alone (HR = 0.747, 95% CI [0.598, 0.932], p = 0.0096). In addition, a clinically meaningful benefit in DRFS (HR = 0.717, 95% CI [0.559, 0.920], nominal p=0.00853) was observed with Verzenio plus endocrine therapy, reflecting a 28.3% reduction in the risk of distant recurrence or death. Consistent results were observed in patient subgroups including geographic region, prior chemotherapy, and menopausal status.
The efficacy results for the final IDFS analysis are summarised in Table 13. At the final IDFS analysis, with a median follow-up of 19.1 months, a further analysis of IDFS and DRFS in the ITT population was performed (see Table 13).

The overall survival (OS) data were not mature at the time of the first OS interim analysis with a total of 186 (3.3%) deaths. Patients will continue to be followed for the final OS analysis. See Figure 1.
Patients were well matched for baseline demographics and prognostic characteristics between the abemaciclib and aromatase inhibitor arm (AI) and the placebo plus AI arm. The median age of patients enrolled was 63 years (range 32-88). Approximately 39% of patients had received chemotherapy and 44% had received antihormonal therapy in the (neo)adjuvant setting prior to their diagnosis of advanced breast cancer. The majority of patients (96%) had metastatic disease at baseline. Approximately 22% of patients had bone-only disease, and 53% patients had visceral metastases.
At the pre-planned interim analysis, the study met the primary endpoint demonstrating a statistically significant prolongation in PFS and a clinically meaningful treatment effect. Primary efficacy results are summarised in Table 14 and Figure 2.
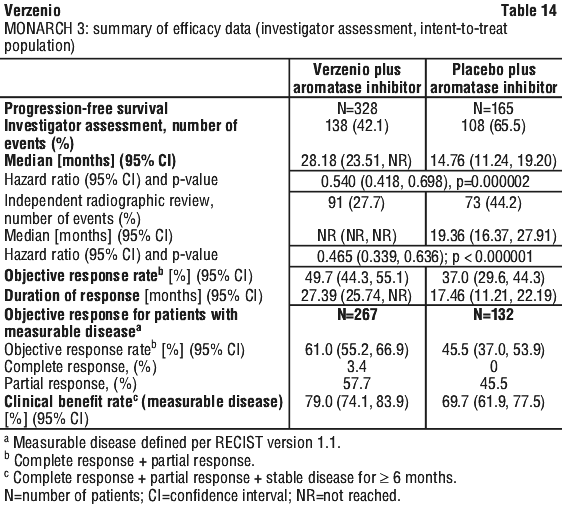
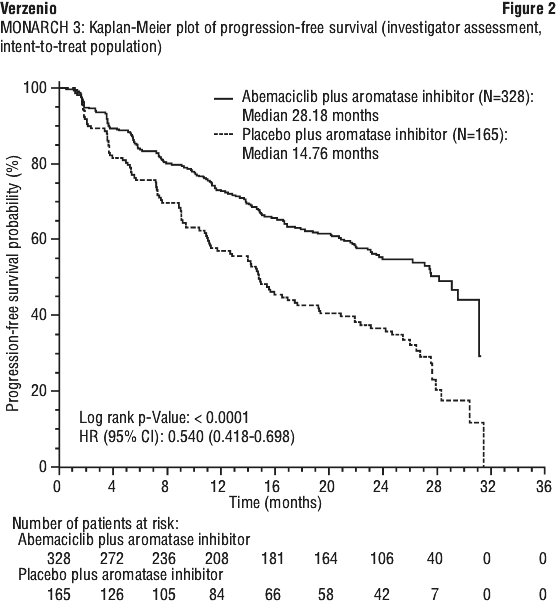
Overall survival was not mature at the final PFS analysis (93 events observed across the two arms). The HR was 1.057 (95% CI: 0.683, 1.633), p = 0.8017.
A series of prespecified subgroup PFS analyses showed consistent results across patient subgroups including age (< 65 or ≥ 65 years), disease site, disease setting (de novo metastatic vs recurrent metastatic vs locally advanced recurrent), presence of measurable disease, progesterone receptor status, and baseline ECOG performance status. A reduction in the risk of disease progression or death was observed in patients with visceral disease, (HR of 0.567 [95% CI: 0.407, 0.789]), median PFS 21.6 months versus 14.0 months; in patients with bone-only disease (HR 0.565, [95% CI: 0.306, 1.044]); and in patients with measurable disease (HR 0.517, [95% CI: 0.392, 0.681]).
Randomised phase 3 study MONARCH 2: Verzenio in combination with fulvestrant. The efficacy and safety of Verzenio was evaluated in MONARCH 2, a randomised, double blind, placebo controlled phase 3 study in women with HR positive, HER2 negative locally advanced or metastatic breast cancer. Patients were randomised in a 2:1 ratio to receive Verzenio 150 mg twice daily plus fulvestrant 500 mg at intervals of one month, with an additional 500 mg dose given two weeks after the initial dose, versus placebo plus fulvestrant alone according to the same schedule. The primary endpoint was investigator-assessed PFS evaluated according to RECIST 1.1; key secondary efficacy endpoints included objective response rate (ORR), clinical benefit rate (CBR) and overall survival (OS).
Patients were well matched for baseline demographics and prognostic characteristics between the abemaciclib plus fulvestrant arm and the placebo plus fulvestrant arm. The median age of patients enrolled was 60 years (range, 32-91 years). In each treatment arm the majority of patients were white and had not received chemotherapy or any prior endocrine therapy for metastatic disease. 17% of patients were pre/perimenopausal. Approximately 56% patients had visceral metastases. Approximately 25% patients had primary resistance to endocrine therapy as per ESMO International Consensus Guidelines for Advanced Breast Cancer, with the majority of patients having secondary resistance.
The study met the primary endpoint demonstrating a statistically significant prolongation in PFS and a clinically meaningful treatment effect. Primary efficacy results are summarised in Table 15 and Figure 3.
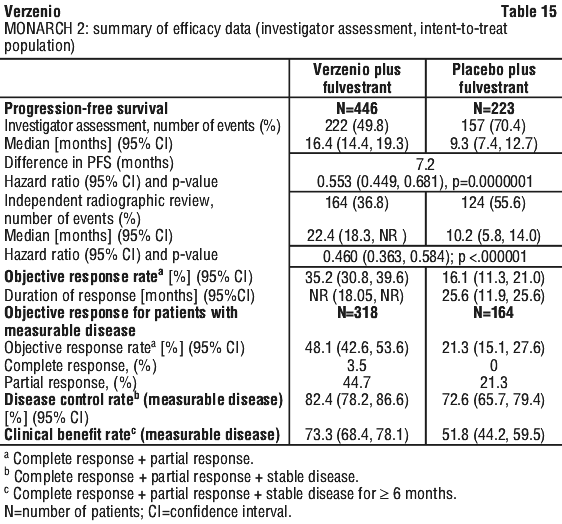
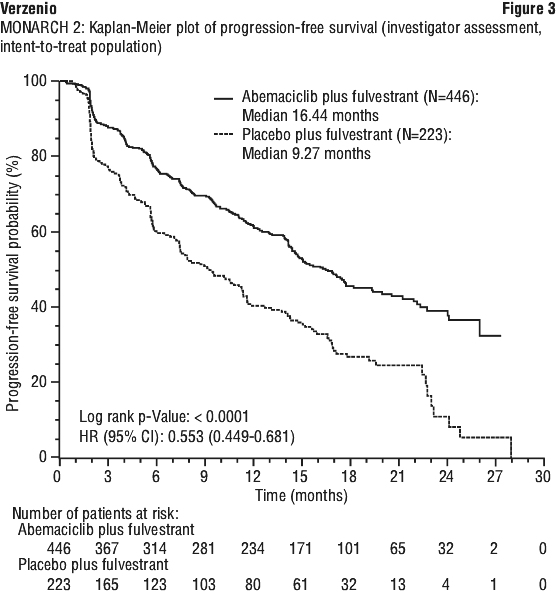
The addition of Verzenio to fulvestrant significantly delayed the time to post discontinuation chemotherapy, hazard ratio 0.651 (95% CI: 0.502, 0.845). The median time to chemotherapy for the abemaciclib arm was not yet reached and for the placebo plus fulvestrant arm was 26 months.
A series of prespecified subgroup PFS analyses were performed based on prognostic factors and baseline characteristics to confirm consistency of the treatment effect. A reduction in the risk of disease progression or death in favour of the Verzenio plus fulvestrant arm was observed in all patient subgroups. Consistent results were observed across patient subgroups including age (< 65 or ≥ 65 years), race, geographic region, disease site, endocrine therapy resistance, presence of measurable disease, progesterone receptor status, and menopausal status. A reduction in the risk of disease progression or death was evident in patients with visceral disease, (HR of 0.481 [95% CI: 0.369, 0.627], median PFS 14.7 months versus 6.5 months); in patients with bone only disease (HR of 0.543 [95% CI: 0.355, 0.833]); patients with measurable disease (HR of 0.523 [95% CI: 0.412, 0.644]). In patients who were pre/perimenopausal, the hazard ratio was 0.415 (95% CI: 0.246, 0.698); in patients who were progesterone receptor negative, the HR was 0.509 [95% CI: 0.325, 0.797]).
In the population of 44 patients who presented de novo with locally advanced or metastatic disease, and had not received any prior endocrine therapy, the addition of Verzenio to fulvestrant reduced the risk of disease progression or death in this population by 54.6% (HR of 0.454 [95% CI: 0.179, 1.154]).
Overall survival (OS) analysis in the ITT population showed a statistically significant improvement in patients receiving Verzenio plus fulvestrant compared with those receiving placebo plus fulvestrant. The overall survival results are summarised in Table 16 and Figure 4.

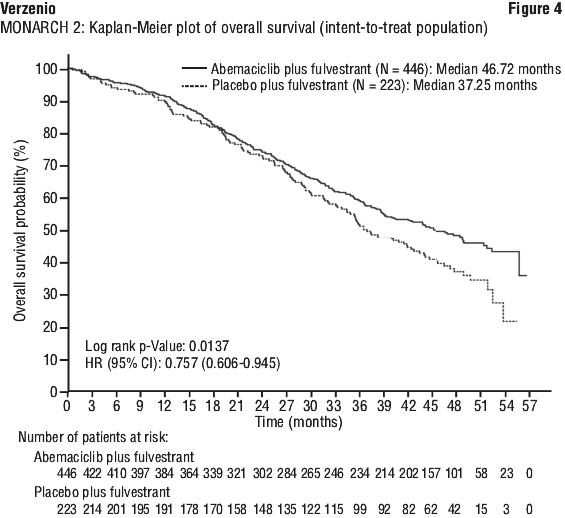
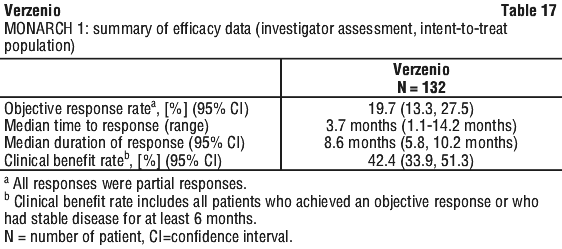
Phase 2 study MONARCH 1: Verzenio monotherapy. The efficacy and safety of Verzenio was evaluated in MONARCH 1, a single-arm, open-label trial in 132 women with HR positive, HER2 negative metastatic breast cancer who had failed prior endocrine therapies and had received one or 2 prior chemotherapy regimens in the metastatic setting. Patients received Verzenio 200 mg twice daily. The primary endpoint was objective response rate (ORR). Efficacy results for MONARCH 1 are summarised in Table 17.
Visceral crisis. There are no data on the efficacy and safety of abemaciclib in patients with visceral crisis.
5.2 Pharmacokinetic Properties
Absorption. Abemaciclib absorption is slow, with a median Tmax of 8.0 hours. The absolute bioavailability of abemaciclib is 45% (90% confidence interval: 40-51%). In the therapeutic dose range of 50-200 mg, the increase in plasma exposure (AUC) and Cmax is dose proportional. Steady state was achieved within 5 days following repeated twice daily dosing, and abemaciclib accumulated with a geometric mean accumulation ratio of 3.7 (58% CV) and 5.8 (65% CV) based on Cmax and AUC, respectively.
Distribution. Abemaciclib was highly bound to plasma proteins in humans (mean bound fraction was approximately 96-98%), and the binding was independent of concentration from 152 nanogram/mL to 5066 nanogram/mL. Abemaciclib binds to both human serum albumin and alpha-1-acid glycoprotein. The geometric mean systemic volume of distribution is approximately 747 L (68.6% CV). In patients with advanced cancer, concentrations of abemaciclib and its active metabolites M2 and M20 in cerebrospinal fluid are comparable to unbound plasma concentrations.
Metabolism. Hepatic metabolism is the main route of clearance for abemaciclib. Abemaciclib is metabolized to several metabolites primarily by cytochrome P450 (CYP) 3A, with formation of N-desethyl abemaciclib (M2) representing the major metabolism pathway. Additional metabolites include hydroxyabemaciclib (M20), hydroxy-N-desethylabemaciclib (M18), and an oxidative metabolite (M1). Metabolites N-desethylabemaciclib (M2) and hydroxyabemaciclib (M20) are active with similar potency as abemaciclib.
Excretion. The geometric mean hepatic clearance (CL) of abemaciclib was 21.8 L/h (39.8% CV), and the mean plasma elimination half-life for abemaciclib in patients was 24.8 hours (52.1% CV). After a single oral dose of [14C]-abemaciclib, approximately 81% of the dose was excreted in faeces and 3.4% excreted in urine. The majority of the dose eliminated in faeces was metabolites.
Use in hepatic impairment. Abemaciclib is metabolised in the liver. In subjects with severe hepatic impairment, total abemaciclib unbound exposure increased 2.69-fold, and the abemaciclib half-life increased from 24 to 55 hours. Reduce the abemaciclib dosing frequency to once daily in patients with severe hepatic impairment.
Use in renal impairment. Abemaciclib and its metabolites are not significantly cleared renally. Dose adjustment is not necessary in patients with mild or moderate renal impairment. There are no data in patients with severe renal impairment, end stage renal disease, or in patients on dialysis.
5.3 Preclinical Safety Data
Genotoxicity. Abemaciclib was not mutagenic in a bacterial reverse mutation (Ames) assay and did not induce structural chromosomal aberrations in the in vitro chromosome aberration assay with human lymphocytes, or the in vivo rat micronucleus test. Metabolites M2 and M20 were not mutagenic in the Ames assay did not induce structural chromosomal aberrations in Chinese hamster ovary cells in the in vitro chromosome aberration assay and were not clastogenic in the rat bone marrow micronucleus assay.
Carcinogenicity. In male rats treated for 95 weeks with abemaciclib at ≥ 1 mg/kg/day (less than or similar to clinical exposure based on AUC), abemaciclib caused interstitial (Leydig) cell hyperplasia and benign adenomas in testes. It is unknown if this effect will translate to humans. There were no neoplastic findings in mice or female rats.
6 Pharmaceutical Particulars
6.1 List of Excipients
Tablet cores. Croscarmellose sodium, lactose monohydrate, microcrystalline cellulose, silicon dioxide, sodium stearyl fumarate.
Film coating. Polyvinyl alcohol (E1203), titanium dioxide (E171), macrogol 3350 (E1521), purified talc (E553b), iron oxide yellow (E172) [50 mg and 150 mg tablets only], iron oxide red (E172) [50 mg tablets only].
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Verzenio is available as a modified oval immediate-release film-coated tablet with "Lilly" debossed on one side and tablet strength in mg debossed on the other.
The 50 mg tablets are beige in colour, 100 mg is white and 150 mg tablets are yellow.
Verzenio is supplied in PVC/PE/PCTFE blister packs sealed with aluminum foil lidding.
Pack sizes. 50 mg, 100 mg, 150 mg: 14 tablets*.
50 mg, 100 mg, 150 mg: 42 tablets*.
50 mg, 100 mg, 150 mg: 56 tablets*.
50 mg, 100 mg, 150 mg: 70 tablets*.
* Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. The molecular formula for abemaciclib is C27H32F2N8 and it has the following structural formula:
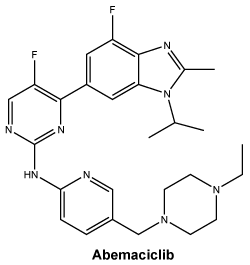
7 Medicine Schedule (Poisons Standard)
Schedule 4.
Date of First Approval
08 April 2019
Date of Revision
18 July 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.