Voltaren Tablets and Suppositories
Brand Information
| Brand name | Voltaren Tablets and Suppositories |
| Active ingredient | Diclofenac sodium |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Voltaren Tablets and Suppositories.
Summary CMI
Voltaren® Tablets & Suppositories
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Voltaren?
Voltaren contains the active ingredient Diclofenac sodium. Voltaren tablets are used to treat certain joint conditions such as osteoarthritis, Rheumatoid arthritis, period cramps or pain, other painful conditions associated with swelling such as back pain, muscle sprains. Voltaren Suppositories are used to relieve pain after surgery in children and are also used to treat certain joint conditions such as osteoarthritis, Rheumatoid arthritis.
For more information, see Section 1. Why am I using Voltaren? in the full CMI.
2. What should I know before I use Voltaren?
Do not use if you have ever had an allergic reaction to Voltaren or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Voltaren? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Voltaren and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Voltaren?
- Your doctor will advise you the appropriate dose.
- Follow the instructions provided and use Voltaren until your doctor tells you to stop.
More instructions can be found in Section 4. How do I use Voltaren? in the full CMI.
5. What should I know while using Voltaren?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Voltaren? in the full CMI.
6. Are there any side effects?
Common side effects include headache, feeling sick, vomiting, dizziness, indigestion, loss of appetite, problems with vision.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Voltaren® Tablets & Suppositories
Active ingredient: Diclofenac sodium
Consumer Medicine Information (CMI)
This leaflet provides important information about using Voltaren. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Voltaren.
Where to find information in this leaflet:
1. Why am I using Voltaren?
2. What should I know before I use Voltaren?
3. What if I am taking other medicines?
4. How do I use Voltaren?
5. What should I know while using Voltaren?
6. Are there any side effects?
7. Product details
1. Why am I using Voltaren?
Voltaren contains the active ingredient Diclofenac sodium. It belongs to class of drugs called Nonsteroidal anti-inflammatory drugs (NSAID's). It helps to relieve both pain and swelling.
Voltaren Tablets are used to treat:
- certain conditions such as osteoarthritis, joint condition having symptoms such as joint pain, swelling, joint stiffness. Rheumatoid arthritis, joint disease characterized by joint swelling, redness and warmth, joint pain, fever
- other painful conditions where swelling is a problem such as back pain, muscle sprains.
- period cramps
Voltaren Suppositories are used to:
- treat certain joint diseases such as Osteoarthritis and Rheumatoid arthritis.
- relieve pain after surgery in children.
2. What should I know before I use Voltaren?
Warnings
Do not use Voltaren Tablets:
- If you are allergic to Diclofenac sodium, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine. Symptoms of allergic reaction may include shortness of breath, difficulty breathing, swelling of the face, lips, tongue or other parts of the body, rash, hives.
- If you have stomach or intestine ulcer, symptoms include heartburn, feeling sick, vomiting, dull or burning stomach pain, bleeding from the stomach or intestine (symptoms such as blood in your stools or black stools), or any other symptoms such as severe pain in tummy, fever, feeling sick, vomiting.
- In last three months of pregnancy.
- If you have liver or kidney problems.
- If you suffer from any heart diseases or if you have a history of heart bypass surgery.
- In children less than 12 months of age.
- If you are allergic to other medicines containing diclofenac (e.g. Voltfast powder), aspirin or any other NSAIDs (allergic reaction symptoms may include hives, difficulty breathing, swelling of the face, lips, tongue or other parts of the body).
- If you have ever developed a skin rash or skin peeling, blistering and/or mouth sore after taking Voltaren or other pain medications.
Do not use Voltaren Suppositories if:
- You suffer from Proctitis (sore back passage, sometimes with bleeding and discharge)
Check with your doctor if you:
- have heart problems, any history of heart attack, uncontrolled high blood pressure, problems with heart functions
- suffer from Leg pain while walking, muscle pain or cramps in the arms or legs or calf.
- have high levels of cholesterol in your blood.
- suffer from diabetes
- smoke or if you have history of smoking.
- have any stomach problems
- Suffer from loose and urgent bowel movements, blood in stool, stomach pain, feeling sick ulcerative colitis, or stomach cramps, constipation, weight loss.
- have bleeding disorders or other blood disorders
- have any skin problems, skin rash or any other sign of allergic reactions.
- have asthma (disease of airways), symptoms of hay fever such as runny nose, watery, itchy, red eyes or any long-standing infections of the respiratory tract that cause difficulty in breathing.
- suffer from symptoms such as feeling sick, vomiting, pain in stomach, diarrhea or if you have a rare genetic condition called glucose-galactose malabsorption, symptoms include severe diarrhea, which can cause dehydration and weight loss
- have symptoms such as severe abdominal pain, diarrhea or constipation, tiredness.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Do not use Voltaren in women who are trying to conceive.
If you are undergoing any treatment for infertility, check with your doctor. Your doctor will discuss with you the risks and benefits involved.
Do not use Voltaren during pregnancy. Do not use in the first 6 months of pregnancy, it may increase the risk of miscarriage. Do not use it during the last 3 months of pregnancy as it may affect your baby, may cause complications during pregnancy or may delay labour and birth. Do not use it if you are likely to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Do not use Voltaren if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Voltaren and affect how it works.
Tell your doctor if you take any of the following medicines:
- digoxin, drug used for treatment of heart problems.
- drugs used to increase the amount of urine e.g. diuretics (Furosemide)
- medicines used to treat high blood pressure, heart problems e.g. beta-blockers (such as Propranolol), angiotensin converting enzyme (ACE) inhibitors (such as Ramipril), angiotensin II receptor antagonists (such as Valsartan)
- drugs used to prevent blood clotting e.g. warfarin
- medicines used in treatment of depression e.g. Lithium or selective serotonin-reuptake inhibitors (SSRIs)
- medicines used to treat diabetes e.g. metformin
- methotrexate, a drug used to treat arthritis and some cancers
- medicines used in organ transplants e.g. cyclosporin and tacrolimus
- any other anti-inflammatory medicines, e.g. salicylates, ibuprofen or aspirin
- medicine used to treat urinary tract infections e.g. trimethoprim
- drugs used to treat infections e.g. quinolone antibacterials
- steroids, glucocorticoids, prednisone (medicines used to provide relief from swelling, arthritis)
- antibiotic medicine used to treat bacterial infections e.g. CYP2C9 inducers (such as rifampicin)
- phenytoin, a medicine used to treat seizures.
- medicine used to treat gout e.g. sulfinpyrazone
- voriconazole, a drug used to treat fungal infections.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Voltaren.
4. How do I use Voltaren?
How much to take
- Your doctor will advise you the appropriate dose.
- Follow the instructions provided and use Voltaren until your doctor tells you to stop.
For arthritis or other painful conditions
- Initial dose is 75 to 150 mg daily.
- After the early stages of treatment, it is usually possible to reduce the dose to 75mg to 100mg each day.
- A maximum daily dose of 150 mg should not be exceeded.
For menstrual cramps
- The usual starting dose of Voltaren tablets is 50mg to 100mg each day, usually taken during each period as soon as cramps begin and continued for a few days until the pain goes away.
- If necessary, the dose can be raised over several menstrual periods to a maximum of 200mg each day.
For post-operative pain in children
- Your doctor will advise you the appropriate dose of Voltaren suppositories for your child depending on your child's weight.
- Ask your doctor or pharmacist if you do not understand the dose.
- Voltaren 100 mg suppositories are not suitable for children and adolescents.
- Voltaren suppositories should not be used in children under 12 months of age.
When to take Voltaren
Voltaren Tablets:
- Take Voltaren tablets before meals or on an empty stomach. If they upset your stomach, you can take them with food or immediately after food.
Voltaren suppositories:
- Use the suppositories at bedtime to relieve pain during the night and early morning stiffness; or to relieve pain after an operation.
How to take Voltaren
Voltaren tablets:
- Swallow the tablets whole with liquid, preferably before meals. Do not divide or chew the tablets.
How to use Voltaren suppositories:
- If possible, go to the toilet and empty your bowels before using the suppository.
- Then follow the below steps:
- Clean your hands thoroughly with soap and water.
- If the suppository feels soft, cool it before removing the wrapper by placing it in the fridge or holding it under cold water for a few minutes.
- Put on a disposable glove, if desired.
- Remove the entire wrapper from the suppository.
- Briefly dip the suppository in cool water to moisten.
- Lie on your side and raise your knees to your chest.
- Push the suppository, blunt end first, gently into your rectum (back passage). Do not break the suppository.
- Remain lying down for a few minutes so that the suppository dissolves.
- Wash your hands again thoroughly.
- Avoid emptying your bowels for at least one hour after using the suppository.
- If you are not sure how to use a suppository, ask your pharmacist or doctor.
- Do not take suppositories by mouth.
Do not use Voltaren suppositories for more than 3 days in children.
If you forget to take/use Voltaren
Voltaren should be used as prescribed by your doctor. If you miss your dose at the usual time, take it as soon as you remember, and then go back to taking it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you use too much Voltaren
If you think that you have used too much Voltaren, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Voltaren?
Things you should do
- Tell your doctor immediately if you or your child is going to have any surgery.
Call your doctor straight away if you:
- notice any symptoms such as chest pain, shortness of breath, weakness, slurring of speech
- develop any signs of allergic reaction.
- become pregnant or intend to become pregnant or if you are breastfeeding.
- have symptoms like feeling sick, tiredness, lethargy, itching, yellowing of your skin or eyes pain in right upper part of stomach.
- suffer from any infections, symptoms such as fever, chills, sore throat
- have heart diseases, breathing problems.
Remind any doctor, dentist, pharmacist or specialist you visit that you are using Voltaren.
Things you should not do
- Do not give Voltaren to anyone else, even if they seem to have the same condition as you.
- Do not use Voltaren to treat any other complaints unless your doctor tells you to.
- Do not use Voltaren suppositories for more than 3 days in children.
laboratory tests
- Your doctor may do a routine blood checkup during treatment with Voltaren.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Voltaren affects you.
Voltaren may cause vision problems, dizziness, spinning sensation (vertigo), sleepiness or other central nervous system problems in some people.
If you have the above symptoms, do not drive a vehicle or operate machines.
Drinking alcohol
Tell your doctor if you drink alcohol.
Looking after your medicine
- Tablets: store below 30°C.
- Suppositories: store below 25°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
General problems:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
General problems:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Voltaren contains
| Active ingredient (main ingredient) | Voltaren Tablets are enteric coated and contain either 25 mg or 50 mg of diclofenac sodium. Voltaren Suppositories: contain either 12.5 mg, 25 mg, 50 mg or 100 mg diclofenac sodium |
| Other ingredients (inactive ingredients) | Tablets: colloidal anhydrous silica, microcrystalline cellulose, lactose monohydrate, magnesium stearate, maize starch, povidone, hypromellose, iron oxide yellow, titanium dioxide, sodium starch glycollate, purified talc, macrogol 8000, PEG-40 hydrogenated castor oil, acrylates copolymer, iron oxide red (50 mg tablet only), Wacker Silicone Fluid Emulsion E 2 (proprietary ingredient) Suppositories: triglyceride base. |
| Potential allergens | Tablets: lactose monohydrate |
Do not take this medicine if you are allergic to any of these ingredients.
What Voltaren looks like
Voltaren Tablets are Enteric coated tablets. They are available in the following strengths:
25 mg: They are yellow, round, slightly biconvex tablets with bevelled edges. Debossed with BZ on one side and CG on reverse side. They come in PVC/PE/PVDC/Al blister packs of 20 (not marketed) and 50; HDPE bottle packs of 20 (not marketed) and 50.
50 mg: They are light brown, round, slightly convex, coated tablets. Imprinted with CG on one side and GT on the other. They come in PVC/PE/PVDC/Al or PVC/Al blister packs of 10 (samples), and 50; HDPE bottle packs of 16, 20, and 50.
Voltaren Suppositories are available in the following strengths:
12.5 mg, 25 mg and 50 mg: They are white to yellowish torpedo-shaped with smooth surfaces. They come in PVC/PE blister packs of 10.
100 mg: They are yellowish white "torpedo shaped" suppository, rounded one end, flat at other, being 30 mm long 12 mm diameter at widest part tapering to 9 mm small end. They come in PVC/PE blister packs of 5 (not currently marketed), 20.
Australian Registration Numbers:
25mg tablet blister AUST R 166496
50mg tablet blister AUST R 66880
12.5mg suppository AUST R 96797
25mg suppository AUST R 96810
50mg suppository AUST R 96811
100mg suppository AUST R 37582
Who distributes Voltaren
Voltaren is supplied in Australia by:
NOVARTIS Pharmaceuticals Australia Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone: 1800 671 203
®= Registered Trademark
This leaflet was prepared in November 2025.
Internal document code:
vlt171125c based on PI vlt171125i
Brand Information
| Brand name | Voltaren Tablets and Suppositories |
| Active ingredient | Diclofenac sodium |
| Schedule | S4 |
MIMS Revision Date: 01 January 2026
1 Name of Medicine
Active ingredient: Diclofenac sodium.
2 Qualitative and Quantitative Composition
Voltaren Tablets are enteric coated and contain either 25 mg or 50 mg of diclofenac sodium.
List of excipients with known effect. Lactose monohydrate.
For the full list of excipients, see Section 6.1 List of Excipients.
Voltaren Suppositories contain either 12.5 mg, 25 mg, 50 mg or 100 mg diclofenac sodium in a triglyceride base.
3 Pharmaceutical Form
Enteric coated tablets. 25 mg. Yellow, round, slightly biconvex tablets with bevelled edges. Debossed with BZ on one side and CG on reverse side.
50 mg. Light brown, round, slightly convex, coated tablets. Imprinted with CG on one side and GT on the other.
Suppositories. 12.5 mg, 25 mg and 50 mg. White to yellowish torpedo-shaped with smooth surfaces.
100 mg. Yellowish white "torpedo shaped" suppository, rounded one end, flat at other, being 30 mm long 12 mm diameter at widest part tapering to 9 mm small end.
4 Clinical Particulars
4.1 Therapeutic Indications
Tablets. Inflammatory and degenerative forms of rheumatism: rheumatoid arthritis and osteoarthritis.
Relief of acute or chronic pain states in which there is an inflammatory component.
Symptomatic treatment of primary dysmenorrhoea.
Suppositories. Inflammatory and degenerative forms of rheumatism: rheumatoid arthritis and osteoarthritis.
Short term (up to three days) treatment of post-operative pain in children.
4.2 Dose and Method of Administration
Pregnancy. See Section 4.3 Contraindications; Section 4.6 Fertility, Pregnancy and Lactation.
After assessing the risk/ benefit ratio in each individual patient, the lowest effective dose for the shortest possible duration should be used. Adverse effects may be minimized by using the lowest effective dose for the shortest duration necessary to control symptoms (see Section 4.4 Special Warnings and Precautions for Use). Patients on long-term treatment should be reviewed regularly with regards to efficacy, risk factors and ongoing need for treatment.
The tablets should be swallowed whole with liquid, preferably before meals, and must not be divided or chewed.
Dose. Adults. Initial dosage is 75 to 150 mg daily.
For long-term therapy, 75 to 100 mg daily is usually sufficient.
The daily dosage should generally be prescribed in 2 or 3 fractional doses. To suppress nocturnal pain and morning stiffness, treatment with tablets during the day can be supplemented by the administration of a suppository at bedtime (up to a maximum daily dose of 150 mg).
In primary dysmenorrhoea the daily dosage, which should be individually adapted, is generally 50 to 150 mg. Initially, a dose of 50 to 100 mg should be given and, if necessary, raised in the course of several menstrual cycles up to a maximum of 200 mg/day. Treatment should be started upon appearance of the first symptoms and, depending on the symptomatology, continued for a few days.
Post-operative analgesia in children. A first dose of 1-2 mg/kg followed by 1 mg/kg three times daily for a maximum of three days total therapy. The maximum daily dose is 3 mg/kg. Voltaren suppositories should not be used in children under 12 months of age.
A maximum daily dose of 150 mg should not be exceeded.
Voltaren 100 mg suppositories are not suitable for children and adolescents.
There is some evidence that post-operative analgesia is improved if the first dose of diclofenac is given one hour prior to surgery, however this may increase the risk of intra-operative bleeding (see Section 4.4 Special Warnings and Precautions for Use).
Method of administration. Tablets. The tablets should be swallowed whole with liquid, preferably before meals, and must not be divided or chewed.
Suppositories. The suppositories should be inserted well into the rectum. It is recommended to insert the suppositories after passing stools. The suppositories are not to be taken by mouth.
4.3 Contraindications
Tablets and suppositories. Gastric or duodenal ulcer, gastrointestinal bleeding or perforation.
Patients who are hypersensitive to the active ingredient, diclofenac, or any of the excipients contained in the tablets or suppositories.
Third trimester of pregnancy (see Section 4.6 Fertility, Pregnancy and Lactation).
Patients with severe hepatic impairment (see Section 4.4 Special Warnings and Precautions for Use).
Renal failure (see Section 4.4 Special Warnings and Precautions for Use).
Severe cardiac failure (see Section 4.4 Special Warnings and Precautions for Use).
Treatment of perioperative pain in setting of coronary artery bypass surgery (CABG).
Patients in whom diclofenac, aspirin or other NSAIDs induce asthma, angioedema, urticaria or other allergic-type reactions, because severe, rarely fatal, anaphylactic type reactions to diclofenac have been reported in such patients.
Suppositories. Proctitis.
4.4 Special Warnings and Precautions for Use
Cardiovascular thrombotic events. Observational studies have indicated that non-selective NSAIDs may be associated with an increased risk of serious cardiovascular events, including myocardial infarction and stroke, which may increase with dose or duration of use. Patients with cardiovascular disease, history of atherosclerotic cardiovascular disease or cardiovascular risk factors may also be at greater risk (see Section 4.2 Dose and Method of Administration).
Treatment with Voltaren is generally not recommended in patients with established cardiovascular disease (congestive heart failure, established ischemic heart disease, peripheral arterial disease) or uncontrolled hypertension. If needed, patients with established cardiovascular disease, uncontrolled hypertension, or significant risk factors for cardiovascular disease (e.g. hypertension, hyperlipidemia, diabetes mellitus and smoking) should be treated with Voltaren only after careful consideration and only at doses ≤ 100 mg daily when treatment continues for more than 4 weeks.
As the cardiovascular risks of diclofenac may increase with dose and duration of exposure, the lowest effective daily dose should be used for the shortest duration possible. The patient's need for symptomatic relief and response to therapy should be re-evaluated periodically, especially when treatment continues for more than 4 weeks.
Physicians and patients should remain alert for the signs and symptoms of serious arteriothrombotic events (e.g. chest pain, shortness of breath, weakness, slurring of speech), which can occur without warnings. Patients should be informed about signs and/or symptoms of serious cardiovascular toxicity and be instructed to see a physician immediately in case of such an event.
There is no consistent evidence that the concurrent use of aspirin mitigates the possible increased risk of serious cardiovascular thrombotic events associated with NSAID use.
Hypertension. NSAIDs may lead to the onset of new hypertension or worsening of pre-existing hypertension and patients taking anti-hypertensives with NSAIDs may have an impaired anti-hypertensive response. Caution is advised when prescribing NSAIDs to patients with hypertension. Blood pressure should be monitored closely during initiation of NSAID treatment and at regular intervals thereafter.
Heart failure. Fluid retention and oedema have been observed in some patients taking NSAIDs, including diclofenac, therefore, caution is advised in patients with fluid retention or heart failure.
Gastrointestinal effects. Close medical surveillance is imperative and particular caution should be exercised when prescribing NSAIDs, including diclofenac, in patients with symptoms indicative of gastrointestinal disorders (GI) or with a history suggestive of gastrointestinal ulceration, bleeding or perforation (see Section 4.8 Adverse Effects (Undesirable Effects)).
Upper GI ulcers, gross bleeding or perforation caused by NSAIDs, including diclofenac, occur in approximately 1% of patients treated for 3-6 months and in about 2-4% of patients treated for one year. The risk of GI bleeding is higher with increasing NSAID doses, with increasing duration of use and in patients with a history of ulcer, particularly if complicated with haemorrhage or perforation, and in the elderly.
Gastric or duodenal ulceration, perforation or gastrointestinal bleeding, which can be fatal, have been reported in patients receiving Voltaren. Studies to date have not identified any subset of patients who are not at risk of developing these problems.
Caution is advised in patients with risk factors for gastrointestinal events who may be at greater risk of developing serious gastrointestinal events, e.g. the elderly, those with a history of serious gastrointestinal events, smoking and alcoholism.
The concurrent use of aspirin and NSAIDs, including diclofenac, also increases the risk of serious gastrointestinal adverse events.
To reduce the risk of GI toxicity in patients with a history of ulcer, particularly if complicated with haemorrhage or perforation, and in the elderly, the treatment should be initiated and maintained at the lowest effective dose. Gastrointestinal bleeding, ulceration and perforation in general have more serious consequences in the elderly. They can occur at any time during treatment with or without warning symptoms or a previous history. In instances where gastrointestinal bleeding or ulcerations occur in patients receiving Voltaren, the drug should be withdrawn immediately. Physicians should warn patients about the signs and symptoms of serious gastrointestinal toxicity and what steps to take if they occur.
Combination therapy with protective agents (e.g. proton pump inhibitors or misoprostol) should be considered for these patients, and also for patients requiring concomitant use of medicinal products containing low-dose acetylsalicylic acid (ASA)/ aspirin or other medicinal products likely to increase gastrointestinal risk.
Patients with a history of GI toxicity, particularly the elderly, should report any unusual abdominal symptoms (especially GI bleeding). Caution is recommended in patients receiving concomitant medications which could increase the risk of ulceration or bleeding, such as systemic corticosteroids, anticoagulants, anti-platelet agents or selective serotonin-reuptake inhibitors (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Close medical surveillance and caution should also be exercised in patients with ulcerative colitis, or with Crohn's disease, as well as in patients suffering from pre-existing dyshaemopoiesis or disorders of blood coagulation, as their condition may be exacerbated (see Section 4.8 Adverse Effects (Undesirable Effects)).
NSAIDs, including diclofenac, may be associated with increased risk of gastrointestinal anastomotic leak. Close medical surveillance and caution are recommended when using Voltaren after gastrointestinal surgery.
Serious skin reactions. Serious skin reactions, some of them fatal, including exfoliative dermatitis, Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia with systemic symptoms (DRESS) (see Drug reaction with eosinophilia with systemic symptoms (DRESS)) and generalised bullous fixed drug eruption, have been reported very rarely in association with the use of NSAIDs, including Voltaren (see Section 4.8 Adverse Effects (Undesirable Effects)). These serious adverse events are idiosyncratic and are independent of dose or duration of use. Patients appear to be at highest risk of these reactions early in the course of therapy, the onset of the reaction occurring in the majority of cases within the first month of treatment. Patients should be advised of the signs and symptoms of serious skin reactions and to consult their doctor at the first appearance of skin rash, mucosal lesion or any other sign of hypersensitivity, and Voltaren should be discontinued.
Drug reaction with eosinophilia with systemic symptoms (DRESS). DRESS has been reported in patients taking NSAIDs. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, haematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue the NSAID and evaluate the patient immediately.
Pre-existing asthma. In patients with asthma, seasonal allergic rhinitis, swelling of the nasal mucosa (i.e. nasal polyps), chronic obstructive pulmonary diseases or chronic infections of the respiratory tract (especially if linked to allergic rhinitis-like symptoms), reactions to NSAIDs such as asthma exacerbations (so-called intolerance to analgesics/ analgesics-asthma), Quincke's oedema or urticaria, are more frequent than in other patients. Therefore, special precaution is recommended in such patients. This is applicable as well for patients who are allergic to other substances, e.g. with skin reactions, pruritus or urticaria.
Infection. Like other NSAIDs, Voltaren may mask the usual signs and symptoms of infection due to its pharmacodynamic properties.
Hypersensitivity. As with NSAIDs, allergic reactions, including anaphylactic/ anaphylactoid reactions, have been reported with diclofenac. These reactions can occur without earlier exposure to the drug.
Lactose intolerance. Voltaren tablets contain lactose and therefore are not recommended for patients with rare hereditary problems of galactose intolerance, severe lactase deficiency or glucose-galactose malabsorption.
Perioperative bleeding. Pre-operative administration of Voltaren may increase the risk of post-operative bleeding. The safety of Voltaren suppositories in children has not been established in major operations or in procedures where minor bleeding could pose a critical safety risk (e.g. patients undergoing intracranial surgery or receiving spinal anaesthesia). The use of Voltaren suppositories in children for such procedures is not recommended.
Since Voltaren may temporarily inhibit platelet aggregation, children undergoing minor procedures such as tonsillectomy, myringotomy, circumcision, orchidopexy and strabismus surgery, should be carefully monitored.
Use in hepatic impairment. Close medical surveillance is required when prescribing Voltaren to patients with impaired hepatic function, as their condition may be exacerbated (see Section 4.3 Contraindications).
As with other NSAIDs, including diclofenac, elevations of one or more liver enzymes may occur during Voltaren therapy. These laboratory abnormalities may progress, remain unchanged, or revert to normal despite continued therapy. Borderline elevations (i.e. 1.2 to 3 times the upper limit of normal (ULN)), or greater elevations of transaminases occurred in about 15% of Voltaren-treated patients. In clinical trials, meaningful elevations (i.e. more than 3 times the ULN) of AST and/or ALT occurred in about 4% of patients treated for several months, including marked elevations (i.e. more than 8 times the ULN) in about 1% of patients. Transaminase elevations were seen more frequently in patients with osteoarthritis than in those with rheumatoid arthritis (see Section 4.8 Adverse Effects (Undesirable Effects)).
Transaminase elevations were reversible on cessation of therapy, and even among patients with marked elevations, signs and symptoms of liver disease occurred only in isolated cases. Most patients with borderline elevations did not have therapy interrupted, and transaminase elevations in most of these cases disappeared or did not progress. There were no identifying features to distinguish those patients who developed marked elevations from those who did not.
In addition to the enzyme elevations seen in clinical trials, rare cases of severe hepatic reactions, including jaundice and fatal fulminant hepatitis, have been reported.
Severe hepatotoxicity may develop without prodromal symptoms, so transaminases should be measured periodically in patients receiving long-term therapy with Voltaren. The optimum times for making the measurements are not known. In most patients who have developed marked transaminase elevations, abnormal tests occurred during the first 2 months of therapy with Voltaren. Based on this experience the first transaminase measurement should be made no later than 8 weeks after the start of Voltaren treatment. As with other NSAIDs, including diclofenac, if abnormal liver tests persist or worsen, if clinical signs and/or symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g. eosinophilia, rash, etc.), Voltaren should be discontinued.
To minimise the possibility of hepatic injury becoming severe between transaminase measurements, physicians should inform patients of the warning signs and symptoms of hepatotoxicity (e.g. nausea, fatigue, lethargy, pruritus, jaundice, abdominal tenderness in the right upper quadrant and "flu-like" symptoms) and the appropriate action to take should these signs and symptoms appear.
Caution should be exercised when using Voltaren in patients with hepatic porphyria, since Voltaren may trigger an attack.
Use in renal impairment. As a class, NSAIDs have been associated with renal papillary necrosis and other pathology during long-term administration in animals.
Fluid retention and oedema have been reported in association with NSAID therapy, including diclofenac. Owing to the importance of prostaglandins for maintaining renal blood flow, particular caution is called for in patients with impaired cardiac or renal function, history of hypertension, in the elderly, in patients being treated with diuretics or medicinal products that can significantly impact renal function, and in those with extracellular volume depletion from any cause, e.g. in the peri- or post-operative phase of major surgical operations (see Section 4.3 Contraindications). Monitoring of renal function as a precautionary measure is, therefore, recommended when using Voltaren in such cases. Discontinuation of therapy is typically followed by recovery to the pre-treatment state.
Combination use of ACE inhibitors or angiotensin receptor antagonists, anti-inflammatory drugs and thiazide diuretics. The use of an ACE inhibiting drug (ACE-inhibitors or angiotensin receptor antagonist), an anti-inflammatory drug (NSAID or COX-2 inhibitor) and a thiazide diuretic at the same time increases the risk of renal impairment. This includes use in fixed combination products containing more than one class of drug. Combined use of these medications should be accompanied by increased monitoring of serum creatinine, particularly at the institution of the combination. The combination of drugs from these three classes should be used with caution particularly in elderly patients or those with pre-existing renal impairment.
Use in the elderly. In elderly patients, who are generally more prone to side effects, particular caution should be exercised. It is recommended that the lowest effective dosage be used in elderly patients or those with a low body weight.
Paediatric use. The safety and efficacy of Voltaren suppositories in children under 12 months has not been established. Therefore, the use of Voltaren suppositories for peri-operative pain, is not recommended in this population.
Effects on laboratory tests. Haematological effects. Like other NSAIDs, Voltaren may temporarily inhibit platelet aggregation. Patients with haemostatic disorders should be carefully monitored.
During prolonged treatment with Voltaren, a slight reduction in haemoglobin has been noted in some patients. On rare occasions, blood dyscrasias have been reported. Periodic blood counts are, therefore, recommended.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The following interactions include those observed with Voltaren tablets/ suppositories and/or other pharmaceutical forms of diclofenac.
Lithium/ digoxin. When given together with preparations containing lithium or digoxin, diclofenac may raise their plasma concentrations and these concentrations should be monitored during treatment with Voltaren.
Diuretics and antihypertensive agents. Like other NSAIDs, concomitant use of diclofenac with diuretics or antihypertensive agents (e.g. beta-blockers, angiotensin converting enzyme (ACE) inhibitors) may cause a decrease in their antihypertensive effect. Therefore, the combination should be administered with caution and patients, especially the elderly, should have their blood pressure periodically monitored. When NSAIDs, including diclofenac, are combined with diuretics, ACE inhibitors or angiotensin II receptor antagonists, the risk of worsening of renal function, including possible acute renal failure (which is usually reversible), may be increased in some patients, especially when renal function is compromised (e.g. dehydrated or elderly patients). Patients should be adequately hydrated and monitoring of renal function is recommended after initiation of concomitant therapy and periodically thereafter (see Section 4.4 Special Warnings and Precautions for Use).
Other NSAIDs and corticosteroids. The concomitant use of diclofenac with systemic NSAIDs, including cyclooxygenase-2 selective inhibitors, should be avoided due to the absence of any evidence demonstrating synergistic benefits and the potential for additive undesirable effects. Concomitant administration of diclofenac and other systemic NSAIDs or corticosteroids may increase the frequency of gastrointestinal undesirable effects. Concurrent treatment with aspirin lowers the plasma concentration, peak plasma levels and AUC values of diclofenac. The use of both drugs concurrently is not recommended.
Anticoagulants and anti-platelet agents. Caution is recommended since concomitant administration could increase the risk of bleeding (see Section 4.4 Special Warnings and Precautions for Use). The concurrent use of NSAIDs and warfarin has been associated with severe, sometimes fatal, haemorrhage. The exact mechanism of the interaction between NSAIDs and warfarin is unknown, but may involve enhanced bleeding from NSAID-induced gastrointestinal ulceration or an additive effect of anticoagulation by warfarin and inhibition of platelet function by NSAIDs. Diclofenac should be used with caution in combination with warfarin and such patients should be closely monitored.
Selective serotonin reuptake inhibitors (SSRIs). Concomitant administration of systemic NSAIDs, including diclofenac, and SSRIs may increase the risk of gastrointestinal bleeding (see Section 4.4 Special Warnings and Precautions for Use).
Antidiabetic agents. Diclofenac can be given together with oral antidiabetic agents without influencing their clinical effect. However, there are isolated reports of both hypoglycaemic and hyperglycaemic effects in the presence of diclofenac which necessitated changes in the dosage of the antidiabetic agents. For this reason, monitoring of the blood glucose level is recommended as a precautionary measure during concomitant therapy.
There have also been isolated reports of metabolic acidosis when diclofenac was co-administered with metformin, especially in patients with pre-existing renal impairment.
Methotrexate. Caution should be exercised when NSAIDs, including diclofenac, are administered less than 24 hours before or after treatment with methotrexate, since the blood concentration of methotrexate may rise and the toxicity of this substance be increased.
Cyclosporin and tacrolimus. Nephrotoxicity of cyclosporin may be enhanced through effects of NSAIDs, including diclofenac, on renal prostaglandins. Therefore, diclofenac should be given at doses lower than those that would be used in patients not receiving cyclosporin or tacrolimus.
Drugs known to cause hyperkalaemia. Concomitant treatment with potassium-sparing drugs (e.g. diuretics, cyclosporin, tacrolimus or trimethoprim) may be associated with increased serum potassium levels, which should therefore be monitored frequently (see Section 4.4 Special Warnings and Precautions for Use).
Glucocorticoids. The addition of glucocorticoids to NSAIDs, though sometimes necessary for therapeutic reasons, may aggravate gastrointestinal side effects.
Quinolone antibacterials. There have been isolated reports of convulsions which may have been due to concomitant use of quinolones and NSAIDs.
CYP2C9 inhibitors. Caution is recommended when co-prescribing diclofenac with potent CYP2C9 inhibitors (such as sulfinpyrazone and voriconazole), which could result in a significant increase in peak plasma concentrations and exposure to diclofenac due to inhibition of diclofenac metabolism. Concomitant administration of voriconazole with diclofenac may increase plasma diclofenac levels.
CYP2C9 inducers. Caution is recommended when co-prescribing diclofenac with CYP2C9 inducers (such as rifampicin), which could result in a significant decrease in plasma concentration and exposure to diclofenac.
Phenytoin. When using phenytoin concomitantly with diclofenac, monitoring of phenytoin plasma concentrations is recommended due to an expected increase in exposure to phenytoin.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. The use of Voltaren may impair female fertility and is not recommended in women attempting to conceive. In women who have difficulties conceiving or who are undergoing investigation of infertility, withdrawal of Voltaren should be considered.
Use in pregnancy. (Category C)
The use of diclofenac in pregnant women has not been studied and safety in pregnancy has not been established. Therefore, Voltaren should not be used in pregnant women during the first two trimesters or in women who are likely to become pregnant unless the potential benefit to the mother outweighs the risk to the foetus.
Data from epidemiological studies suggest an increased risk of miscarriage after the use of a prostaglandin synthesis inhibitor in early pregnancy.
NSAIDs inhibit prostaglandin synthesis and, when given during the latter part of pregnancy, may cause closure of the foetal ductus arteriosus, foetal renal impairment, inhibition of platelet aggregation, and delay labour and birth.
Use of Voltaren during the third trimester of pregnancy is contraindicated owing to the possibility of uterine inertia, premature closure of the ductus arteriosus and oligohydramnios and neonatal renal impairment (see Oligohydramnios and neonatal renal impairment).
Oligohydramnios and neonatal renal impairment. Use of NSAIDs from about 20 weeks gestation may cause foetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment.
These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. Oligohydramnios is often, but not always, reversible with treatment discontinuation.
Complications of prolonged oligohydramnios may, for example, include limb contractures and delayed lung maturation. In some post-marketing cases of impaired neonatal renal function, invasive procedures such as exchange transfusion or dialysis were required.
If, after careful consideration of alternative treatment options for pain management, NSAID treatment is necessary from about 20 weeks to the end of the second trimester, limit use to the lowest effective dose and shortest duration possible. Use of Voltaren during the third trimester of pregnancy is contraindicated (see Use in pregnancy). Consider ultrasound monitoring of amniotic fluid if treatment extends beyond 48 hours. Discontinue treatment with NSAIDs if oligohydramnios occurs.
Use in lactation. Following oral doses of 50 mg administered every 8 hours, the active substance, diclofenac, passes into the breast milk. As with other drugs that are excreted in milk, Voltaren is not recommended for use in nursing women.
4.7 Effects on Ability to Drive and Use Machines
Patients experiencing visual disturbances, dizziness, vertigo, somnolence or other central nervous disturbances while taking Voltaren should refrain from driving a vehicle or operating machines (see Section 4.8 Adverse Effects (Undesirable Effects)).
4.8 Adverse Effects (Undesirable Effects)
Adverse reactions are ranked under heading of frequency, the most frequent first, using the following convention: common (≥ 1/100, < 1/10); uncommon (≥ 1/1,000, < 1/100); rare (≥ 1/10,000, < 1/1,000); very rare (< 1/10,000), including isolated reports.
The following undesirable effects include those reported with Voltaren tablets/ suppositories and/or other pharmaceutical forms of diclofenac, with either short-term or long-term use.
Blood and lymphatic system disorders. Very rare: Thrombocytopenia, leucopoenia, anaemia (including haemolytic and aplastic anaemia), agranulocytosis, positive Coombs' test.
Immune system disorders. Rare: Hypersensitivity, anaphylactic and anaphylactoid reactions (including hypotension and shock).
Very rare: Angioneurotic oedema (including face oedema).
Psychiatric disorders. Very rare: Disorientation, depression, insomnia, nightmare, irritability, psychotic disorder.
Nervous system disorders. Common: Headache, dizziness.
Rare: Somnolence.
Very rare: Paraesthesia, memory impairment, convulsion, anxiety, tremor, aseptic meningitis, taste disturbances, cerebrovascular accident, myoclonic encephalopathy (described in two patients).
Eye disorders. Very rare: Visual disturbance, blurred vision, diplopia.
Ear and labyrinth disorders. Common: Vertigo.
Very rare: Tinnitus, impaired hearing.
Cardiac disorders. Uncommon*: Myocardial infarction, cardiac failure, palpitations, chest pain.
Frequency unknown: Kounis syndrome.
Vascular disorders. Very rare: Hypertension, vasculitis.
Respiratory, thoracic and mediastinal disorders. Rare: Asthma (including dyspnoea).
Very rare: Pneumonitis.
Gastrointestinal disorders. Common: Nausea, vomiting, diarrhoea, dyspepsia, abdominal pain, flatulence, anorexia.
Rare: Gastritis, gastrointestinal haemorrhage, haematemesis, haemorrhagic diarrhoea, melaena, gastrointestinal ulcer (with or without bleeding or perforation), gastrointestinal stenosis, or perforation, which may lead to peritonitis, proctitis (Voltaren suppositories).
Very rare: Colitis (including haemorrhagic colitis, ischemic colitis and exacerbation of ulcerative colitis or Crohn's disease), constipation, stomatitis, glossitis, oesophageal disorder, diaphragm-like intestinal strictures, pancreatitis, haemorrhoids aggravated (Voltaren suppositories).
Hepatobiliary disorders. Common: Transaminases increased.
Rare: Hepatitis, jaundice, liver disorder.
Very rare: Fulminant hepatitis, hepatic necrosis, hepatic failure.
Pregnancy, puerperium and perinatal conditions. Unknown: Oligohydramnios, neonatal renal impairment.
Skin and subcutaneous tissue disorders. Common: Rashes or skin eruptions.
Rare: Urticaria.
Very rare: Bullous eruptions, eczema, erythema, erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis (Lyell's syndrome), exfoliative dermatitis, loss of hair, photosensitivity reaction, purpura, allergic purpura, pruritus.
Unknown: Drug reaction with eosinophilia with systemic symptoms (DRESS), fixed drug eruption, generalised bullous fixed drug eruption.
Renal and urinary disorders. Very rare: Acute kidney injury (acute renal failure), haematuria, proteinuria, nephrotic syndrome, interstitial nephritis, renal papillary necrosis.
General disorders and administration site conditions. Common: Application site irritation.
Rare: Oedema.
Very rare: Impotence (association with Voltaren intake is doubtful). Worsening of haemorrhoids has been reported with use of Voltaren suppositories. Toxic shock syndrome has been reported in patients administered NSAIDs postoperatively.
* The frequency reflects data from long-term treatment with a high dose (150 mg/day).
Description of selected adverse drug reactions. Arteriothrombotic events. Meta-analysis and pharmacoepidemiological data point towards an increased risk of arteriothrombotic events (for example myocardial infarction) associated with the use of diclofenac, particularly at a high dose (150 mg daily) and during long-term treatment (see Section 4.4 Special Warnings and Precautions for Use). A recent meta-analysis (CNT) estimates that, in comparison with placebo, allocation to diclofenac caused around 3 additional major vascular events per 1000 participants per year. This estimate reflects data from long-term treatment with high dose diclofenac (150 mg/day).
Visual effects. Visual disturbances such as visual impairment, blurred vision or diplopia appear to be NSAID class effects and are usually reversible on discontinuation. A likely mechanism for the visual disturbances is the inhibition of prostaglandin synthesis and other related compounds that alter the regulation of retinal blood flow resulting in potential changes in vision. If such symptoms occur during diclofenac treatment, an ophthalmological examination may be considered to exclude other causes.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Management of acute poisoning with NSAIDs, including diclofenac, consists essentially of supportive measures and symptomatic treatment. There is no typical clinical picture resulting from an overdosage of diclofenac. Overdosage can cause symptoms such as vomiting, gastrointestinal haemorrhage, diarrhoea, dizziness, tinnitus or convulsions. In the event of significant poisoning, acute renal failure and liver damage are possible.
Activated charcoal may reduce absorption of the medicine if given within one or two hours after ingestion. In patients who are not fully conscious or have impaired gag reflex, consideration should be given to administering activated charcoal via nasogastric tube, once the airway is protected.
Supportive and symptomatic treatment should be given for complications such as hypotension, renal failure, convulsions, gastrointestinal disorder, and respiratory depression. Haematological and biochemical parameters, and the presence or absence of blood in the stools, should be monitored.
Specific therapies such as forced diuresis, dialysis, or haemoperfusion, are probably of no help in eliminating NSAIDs, including diclofenac, because of their high protein-binding rate and extensive metabolism.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Diclofenac sodium, a non-steroidal compound, exhibits pronounced antirheumatic, anti-inflammatory, analgesic, and antipyretic properties.
As with other NSAIDs, its mode of action is not known; however, its ability to inhibit prostaglandin synthesis may be involved in the anti-inflammatory effect.
Clinical trials. In rheumatic diseases, the anti-inflammatory and analgesic properties of Voltaren elicit a clinical response characterised by relief from signs and symptoms such as pain at rest, pain on movement, morning stiffness, and swelling of the joints, as well as by an improvement in function.
In addition, clinical studies have revealed that in primary dysmenorrhoea, Voltaren is capable of relieving the pain and reducing the extent of bleeding. Low concentrations of diclofenac sodium inhibit the aggregation of platelets induced in vitro by collagen and by adenosine diphosphate. Diclofenac sodium in vitro does not suppress proteoglycan biosynthesis in canine cartilage at concentrations equivalent to the concentrations reached in humans. It is unknown whether or not diclofenac sodium affects the integrity of human osteoarthritic cartilage.
5.2 Pharmacokinetic Properties
Absorption. Diclofenac is completely absorbed from the enteric coated tablets after their passage through the stomach. Following ingestion of one tablet with or after a meal, its passage through the stomach is slower than when it is taken before a meal, but the amount of active substance absorbed remains the same. In fasting subjects, the mean peak plasma concentration of 1.5 microgram/mL (5 micromol/L) is attained on average 2 hours after ingestion of one tablet of 50 mg. Suppositories of 50 mg produce a corresponding mean peak plasma concentration of 1.2 microgram/mL (4 micromol/L). The plasma concentrations, as measured by the area under the time-concentration curve, are in linear relation to the size of the dose.
In a comparative bioavailability study of Voltaren 50 mg enteric coated tablets and Voltaren 100 mg suppositories, the rectal absorption of diclofenac was almost immediate (Tmax 0.62 hr) and the Cmax was lower but more sustained, suggesting slower absorption from this formulation compared with the tablet. The t1/2 was significantly longer for the suppository, however, the total AUC was not significantly different for the two formulations.
Distribution. Diclofenac becomes bound to serum proteins to the extent of 99.7%, chiefly to albumin (99.4%).
Metabolism. Following oral or rectal administration, about half the active substance is metabolised during its first passage through the liver ("first-pass" effect). The biotransformation of diclofenac involves partly glucuronidation of the intact molecule, but mainly single and multiple hydroxylation followed by glucuronidation.
Excretion. The total systemic clearance of diclofenac in plasma is 263 ± 56 mL/min (mean value ± SD). The terminal half-life in plasma is 1 to 2 hours.
After administration of diclofenac for 15 days in an oral dose of 25 mg three times daily, there was no evidence of drug accumulation in plasma.
In a study in 16 patients with rheumatoid arthritis and knee joint effusions it was found that diclofenac enters the synovial fluid, where maximum concentrations were measured 2 to 4 hours after oral administration. The apparent half-life for elimination from the synovial fluid was 3 to 6 hours. Only 4 to 6 hours after administration, therefore, concentrations of the active substance were already higher in the synovial fluid than they were in the plasma and remained higher for up to 12 hours. These results could possibly explain that the duration of clinical effect is longer than might be inferred from the short plasma half-life of Voltaren.
The biotransformation of diclofenac involves partly glucuronidation of the intact molecule, but mainly single and multiple hydroxylation followed by glucuronidation. About 60% of the administered dose is excreted in the urine in the form of metabolites from one of these two processes. Less than 1% is excreted as unchanged substance. The remainder of the dose is eliminated as metabolites through the bile in the faeces.
Special patient populations. No relevant age-dependent differences in the drug's absorption, metabolism or excretion have been observed.
In patients suffering from renal impairment, no accumulation of the unchanged active substance could be inferred from the single-dose kinetics when applying the usual dosage schedule. At a creatinine clearance of < 10 mL/min, the theoretical steady-state plasma levels of metabolites are about four times higher than in normal subjects. However, the metabolites appear to be satisfactorily cleared through the bile.
In the presence of impaired hepatic function (chronic hepatitis, non-decompensated cirrhosis), the kinetics and metabolism of diclofenac were the same as in patients without liver disease.
5.3 Preclinical Safety Data
Genotoxicity. Diclofenac showed no mutagenic effects in the studies conducted.
Carcinogenicity. Dietary administration of diclofenac to mice and rats at doses up to 0.5 mg/kg/day revealed no carcinogenic activity. However, the plasma concentration of diclofenac at this dose level was 20 to 100 times lower than that in humans. Administration of higher doses to rats and mice resulted in increased mortality due to gastrointestinal ulceration. Diclofenac showed no carcinogenic effects in the studies conducted.
6 Pharmaceutical Particulars
6.1 List of Excipients
Tablets. Colloidal anhydrous silica, microcrystalline cellulose, lactose monohydrate, magnesium stearate, maize starch, povidone, hypromellose, iron oxide yellow, titanium dioxide, sodium starch glycollate, purified talc, macrogol 8000, PEG-40 hydrogenated castor oil, acrylates copolymer, iron oxide red (50 mg tablet only), Wacker Silicone Fluid Emulsion E 2 (proprietary ingredient).
Suppositories. Triglyceride base.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Tablets. Store below 30°C. Keep out of reach of children.
Suppositories. Store below 25°C. Keep out of the reach of children.
6.5 Nature and Contents of Container
Tablets. 25 mg. PVC/PE/PVDC/Al blister packs of 20 (not marketed) and 50; HDPE bottle packs of 20 (not marketed) and 50.
50 mg. PVC/PE/PVDC/Al or PVC/Al blister packs of 10 (samples), and 50; HDPE bottle packs of 16, 20, and 50.
Suppositories. 12.5 mg, 25 mg and 50 mg. PVC/PE blister packs of 10.
100 mg. PVC/PE blister packs of 5 (not currently marketed), 20.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
The active ingredient of Voltaren is diclofenac sodium, a phenylacetic acid derivative, structurally similar to both the phenylalkanoic acid and the anthranilic acid series of compounds. Diclofenac sodium is an odourless, yellowish-white, crystalline powder sparingly soluble in water.
Chemical structure.
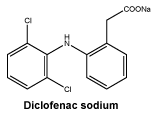
Molecular weight: 296.15.
Molecular formula: C14H11Cl2NO2.
CAS number. 15307-86-5.
7 Medicine Schedule (Poisons Standard)
Prescription only medicine (Schedule 4).
Date of First Approval
02 August 1991
Date of Revision
17 November 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.