Wegovy FlexTouch
Brand Information
| Brand name | Wegovy FlexTouch |
| Active ingredient | Semaglutide |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Wegovy FlexTouch.
Summary CMI
Wegovy® FlexTouch®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
▼ This medicine is new or being used differently. Please report side effects. See the full CMI for further details.
1. Why am I using Wegovy®?
Wegovy® contains the active ingredient semaglutide. Wegovy® is used for weight loss and weight maintenance in addition to diet and physical activity in adults and adolescents ages 12 years and above, and to reduce the risk of a heart disease related event if you are an adult with a history of heart disease and have a BMI ≥27 kg/m2. It also has provisional approval for the treatment of Metabolic Dysfunction Associated Steatohepatitis (MASH) in adults with moderate to advanced liver scarring (fibrosis) without cirrhosis. For more information, see Section 1. Why am I using Wegovy®? in the full CMI.
2. What should I know before I use Wegovy®?
Do not use if you have ever had an allergic reaction to semaglutide or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Wegovy®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Wegovy® and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Wegovy®?
- The recommended dose is 2.4 mg once weekly.
- When you first start using Wegovy®, the starting dose is 0.25 mg once weekly.
- Your doctor will instruct you to gradually increase your dose every 4 weeks until you reach the recommended dose of 2.4 mg once weekly.
- Wegovy® is given as an injection under the skin (subcutaneous injection). Do not inject it into a vein or muscle.
More instructions can be found in Section 4. How do I use Wegovy®? in the full CMI.
5. What should I know while using Wegovy®?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Wegovy®? in the full CMI.
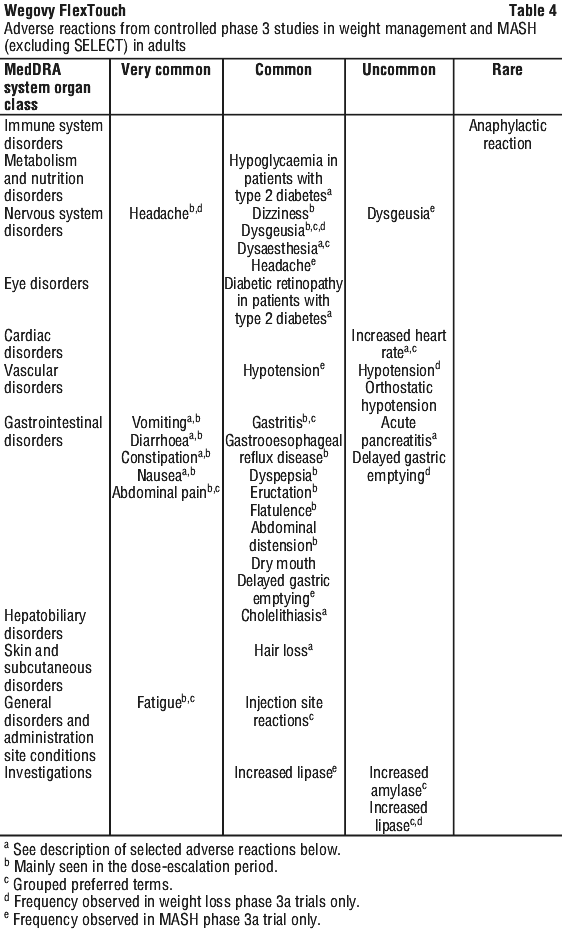
6. Are there any side effects?
During treatment with Wegovy®, you may feel sick (nausea), or be sick (vomiting), or have diarrhoea. These side effects can cause dehydration (loss of fluids). It is important that you drink enough fluids to prevent dehydration. Inflamed pancreas (acute pancreatitis) which could cause severe pain in the stomach and back which does not go away, allergic reaction (skin rashes over a large part of the body, shortness of breath, wheezing, swelling of the face, lips or tongue, fast pulse, sweating) and bowel obstruction are very serious side effects for which you may need urgent medical attention. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
▼ This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems.
Wegovy® FlexTouch®
Active ingredient(s): semaglutide
0.25 mg, 0.5 mg, 1 mg, 1.7 mg and 2.4 mg, solution for injection pre-filled pen
This medicine has provisional approval in Australia for the treatment of MASH in adults with moderate to advanced liver scarring (fibrosis) without cirrhosis. The decision to approve this indication has been made on the basis of promising results from preliminary data. More evidence is required to be submitted when available to fully confirm the benefit and safety of the medicine for this use.
Consumer Medicine Information (CMI)
This leaflet provides important information about using Wegovy® FlexTouch®. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Wegovy® FlexTouch®.
Keep this leaflet. You may need to read it again.
This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
Where to find information in this leaflet
1. Why am I using Wegovy®?
2. What should I know before I use Wegovy®?
3. What if I am taking other medicines?
4. How do I use Wegovy®?
5. What should I know while using Wegovy®?
6. Are there any side effects?
7. Product details
1. Why am I using Wegovy®?
Wegovy® contains the active ingredient semaglutide. Wegovy® is similar to a natural hormone called glucagon-like peptide-1 (GLP-1) that is released from the intestine after a meal. Wegovy® works by acting on receptors in the brain that control your appetite, causing you to feel fuller and less hungry and experience less craving for food. This will help you eat less food and reduce your body weight. In the liver, Wegovy® works by slowing down or stopping further liver damage caused by fat build-up and inflammation, and by supporting a reduction of liver scarring.
Wegovy® is used for weight loss and weight maintenance in addition to diet and physical activity in adults, who have
- a BMI of 30 kg/m² or greater (obesity), or
- a BMI of 27 kg/m² and less than 30 kg/m² (overweight) and weight-related health problems.
Wegovy® is used to reduce the risk of a heart disease related event in adults with a history heart disease who do not have Type 1 or Type 2 diabetes and have a BMI of 27 kg/m² or greater.
BMI (Body Mass Index) is a measure of your weight in relation to your height.
Wegovy® is used together with diet and physical activity for weight management in adolescents aged 12 years and above, who have
- obesity
and
- body weight >60 kg.
As an adolescent patient, you should only continue using Wegovy if you have lost at least 5% of your BMI after 12 weeks on the 2.4 mg dose or maximum tolerated dose (see Section 4. How do I use Wegovy®?). Consult your doctor before you continue.
Wegovy® also has provisional approval for the treatment of Metabolic Dysfunction Associated Steatohepatitis (MASH) in adults with moderate to advanced liver scarring (fibrosis) without cirrhosis. MASH is a condition where fat builds up in the liver creating inflammation, liver damage and scar tissue.
Wegovy® is an injection that is used once a week.
2. What should I know before I use Wegovy®?
Warnings
Do not use Wegovy® if:
- You are allergic to semaglutide, or any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- redness, swelling, rash and itching at the injection site
- rash, itching or hives on the skin
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body. - Always check the ingredients to make sure you can use this medicine.
- Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
- It is after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering.
- It has expired or is damaged, return it to your pharmacist for disposal. The expiry date refers to the last day of that month.
The use of Wegovy is not recommended if you:
- use other products for weight loss
- have type 1 diabetes
- have severe heart failure
There is little experience with Wegovy in patients:
- of 85 years and older
- with severe stomach or gut problem which results in delayed stomach emptying (called gastroparesis), or if you have an inflammatory bowel disease.
- MASH and BMI<25 kg/m² (or BMI<23 kg/m² for Asian population).
Please consult your doctor if one of the above applies to you.
Check with your doctor if you:
- have any other medical conditions
- have or have had depression, suicidal thoughts or any other major mental illness
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
This medicine should not be used during pregnancy, and for at least two months before a planned pregnancy because Wegovy® may harm your unborn child. Therefore, it is recommended to use contraception while using this medicine. If you wish to become pregnant, you should stop using this medicine at least two months in advance. If you become or are pregnant, think you may be pregnant or are planning to have a baby when using this medicine, talk to your doctor straight away, as your treatment will need to be stopped.
Do not use this medicine if you are breast-feeding, as it is unknown if it passes into breast milk.
Food or liquid getting into lungs during anaesthesia
- Some patients taking medicines like Wegovy® have had problems with food or liquid from their stomach getting into their lungs while under general anaesthesia or deep sedation.
- Tell your healthcare professional that you are taking Wegovy® before you have a procedure that requires general anaesthesia or deep sedation.
Effects on the digestive system
During treatment with Wegovy®, you may feel sick (nausea), or be sick (vomiting), or have diarrhoea. These side effects can cause dehydration (loss of fluids). It is important that you drink enough fluids to prevent dehydration. This is especially important if you have kidney problems. Talk to your doctor if you have any questions or concerns.
Inflammation of the pancreas
If you have severe and on-going pain in the stomach area (see Section 6. Are there any side effects?) – see a doctor straight away as this could be a sign of an inflamed pancreas (acute pancreatitis).
People with diabetes
Do not use Wegovy® as a substitute for insulin.
Low Blood Sugar (Hypoglycaemia)
Taking a sulfonylurea (such as gliclazide or glimepiride) or an insulin with Wegovy® might increase the risk of getting low blood sugar levels (hypoglycaemia). Please see section 6 for the warning signs of low blood sugar levels.
Your doctor may ask you to test your blood sugar levels. This will help your doctor decide if the dose of the sulfonylurea or insulin needs to be changed to reduce the risk of low blood sugar.
Diabetic eye disease (retinopathy)
Fast improvements in blood sugar control may lead to a temporary worsening of diabetic eye disease. If you have diabetic eye disease and experience eye problems while taking this medicine, talk to your doctor.
Children and adolescents
This medicine is not recommended in children and adolescents with overweight or obesity under 12 years as the safety and efficacy in this age group have not been established.
The safety and efficacy of Wegovy® in children and adolescents with MASH have not been studied and Wegovy® is not recommended for use in these populations.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
In particular, tell your doctor, pharmacist or nurse if you are using medicines containing the following:
- Warfarin or other similar medicines taken by mouth to reduce blood clotting (oral anti-coagulants). When you start treatment with e.g., warfarin or similar medicines, frequent blood testing to determine the ability of your blood to clot may be required.
People with diabetes
Your doctor may adjust the dose of your diabetes medicines to prevent you from getting low blood sugar.
Do not mix Wegovy® up with other medicines that you inject (e.g., insulins).
Do not use Wegovy® in combination with other medicines that contain GLP-1 receptor agonists (such as liraglutide, dulaglutide, exenatide or lixisenatide).
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Wegovy®.
4. How do I use Wegovy®?
How much to use
- Always use this medicine exactly as your doctor has told you. Check with your doctor, pharmacist or nurse if you are not sure.
Adults
- The recommended dose is 2.4 mg once weekly.
- Your treatment will start at a low dose which will be gradually increased over 16 weeks of treatment.
- When you first start using Wegovy®, the starting dose is 0.25 mg once weekly.
- Your doctor will instruct you to gradually increase your dose every 4 weeks until you reach the recommended dose of 2.4 mg once weekly.
- Once you reach the recommended dose of 2.4 mg, do not increase this dose further.
- You will be told to follow the table below.
- In case you are feeling very bothered by sickness (nausea) or by being sick (vomiting) talk with your doctor about adjusting your dose.
| Dose escalation | Weekly dose |
| Week 1-4 | 0.25 mg |
| Week 5-8 | 0.5 mg |
| Week 9-12 | 1 mg |
| Week 13-16 | 1.7 mg |
| From week 17 | 2.4 mg |
- Your doctor will assess your treatment on a regular basis.
Adolescents (above 12 years of age)
- For adolescents, the same dose escalation schedule as for adults should be applied (see above). The dose should be increased until 2.4 mg (maintenance dose) or maximum tolerated dose has been reached. Weekly doses higher than 2.4 mg are not recommended.
- As an adolescent patient, you should regularly review your treatment goals with your doctor.
Follow the instructions provided and use Wegovy® until your doctor tells you to stop.
When to use Wegovy®
- You should use this medicine once a week and if possible, on the same day each week
- You can give yourself the injection at any time of the day – regardless of meals.
- If necessary, you can change the day of your weekly injection of this medicine as long as it has been at least 3 days since your last injection. After selecting a new dosing day, continue with once a week dosing.
How Wegovy® is given
- Wegovy® is given as an injection under the skin (subcutaneous injection). Do not inject it into a vein or muscle.
- The best places to give the injection are the fronts of your upper arm, upper legs or stomach.
- Before you use the pen for the first time, ask your doctor, pharmacist or nurse how to use it.
Follow the detailed instructions on how to inject Wegovy® FlexTouch® in the instructions for use supplied with the product. These instructions are also available via the following hyperlink
medsinfo.com.au/media/noiwegof
If you forget to use Wegovy®
If you forgot to inject a dose and:
- it is 5 days or less since you should have used Wegovy®, use it as soon as you remember. Then inject your next dose as usual on your scheduled day.
- it is more than 5 days since you should have used Wegovy®, skip the missed dose. Then inject your next dose as usual on your next scheduled day. Do not take a double dose to make up for a forgotten dose.
If you use too much Wegovy®
Talk to your doctor straight away. You may get side effects such as feeling sick (nausea), being sick (vomiting) or have diarrhoea, which may cause dehydration (loss of fluids).
If you think that you have used too much Wegovy®, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26 in Australia or 0800 764766 in New Zealand), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Wegovy®?
Things you should do
- Tell your doctor or pharmacist if you are travelling.
- Ask them for a letter explaining why you are taking injecting devices with you. Each country you visit will need to see this letter, so you should take several copies.
- You may not be able to get Wegovy® FlexTouch® in the country you are visiting.
- Remind any doctor, dentist or pharmacist you visit that you are using Wegovy®.
Things you should not do
- Do not stop using this medicine without talking to your doctor.
- Do not use this medicine if you think it has been frozen or exposed to excessive heat.
- Do not use this medicine to treat any other complaints unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Do not share your pen with anyone else.
- Do not use this medicine after the expiry date which is stated on the pen label and carton after ‘EXP’. The expiry date refers to the last day of that month.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Wegovy® affects you.
Wegovy® is unlikely to affect your ability to drive and use machines. Some patients may feel dizzy when taking Wegovy® mainly during the first 4 months of treatment (see section 6). If you feel dizzy be extra careful while driving or using machines. If you need any further information, talk to your doctor, pharmacist or nurse.
People with diabetes
If you use this medicine in combination with a sulfonylurea or insulin, low blood sugar (hypoglycaemia) may occur which may reduce your ability to concentrate. Avoid driving or using machines if you get any signs of low blood sugar. See section 2 for information on increased risk of low blood sugar and section 6 for the warning signs of low blood sugar.
Looking after your medicine
- Store in a refrigerator (2°C – 8°C). Do not freeze. Keep away from the cooling element.
- Wegovy® can be taken out of the refrigerator for up to 6 weeks at a temperature below 30°C.
- Always store the pen in the original carton in order to protect from light.
- Do not use this medicine if you notice that the solution is not clear and colourless.
Keep it where young children cannot reach it.
When to discard your medicine
Discard the Wegovy® FlexTouch® pen you are using after 6 weeks even if there is still some medicine left in it.
After having injected the 4 doses, there might still be solution left in the pen despite having administered correctly. Any solution left is insufficient for a dose and the pen should be disposed of.
Dispose of used needles safely into a yellow plastic sharps container.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date. The expiry date refers to the last day of that month.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Very common: may affect more than 1 in 10 people:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
*Frequency based on weight management trials.
**Frequency based on MASH trials
Hypoglycaemia
The warning signs of low blood sugar may come on suddenly. They can include: cold sweat, cool pale skin, headache, fast heartbeat, feeling sick (nausea) or very hungry, changes in vision, feeling sleepy or weak, feeling nervous, anxious or confused, difficulty concentrating or shaking.
Your doctor will tell you how to treat low blood sugar and what to do if you notice these warning signs.
Low blood sugar is more likely to happen if you also take a sulfonylurea or insulin. Your doctor may reduce your dose of these medicines before you start using this medicine.
Serious side effects
| Serious side effects | What to do |
| Complications of diabetic eye disease (diabetic retinopathy). If you have diabetes you should inform your doctor if you experience eye problems, such as changes in vision, during treatment with this medicine. Common: may affect up to 1 in 10 people Inflamed pancreas (acute pancreatitis). Signs of inflamed pancreas may include severe and long-lasting pain in your stomach, with or without vomiting. You may feel the pain from your abdomen to your back. You should see your doctor immediately if you experience such symptoms. Uncommon: may affect up to 1 in 100 people Severe allergic reactions (anaphylactic reactions). You should seek immediate medical help and inform your doctor straight away if you get symptoms such as breathing problems, swelling of face and throat, wheezing, fast heartbeat, pale and cold skin, feeling dizzy or weak. Rare: may affect up to 1 in 1,000 people Bowel obstruction A severe form of constipation with additional symptoms such as stomach ache, bloating, vomiting etc. Not known: frequency cannot be estimated from the available data. | Call your doctor straight away, or go straight to the Accident and Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. If you are in New Zealand, you can report side effects at pophealth.my.site.com/carmreportnz/s/. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Wegovy® FlexTouch® contains
| Active ingredient (main ingredient) | semaglutide |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | Nil |
Wegovy® FlexTouch® contains sodium
This medicine contains less than 1 mmol sodium (23 mg) per dose, i.e. essentially ‘sodium-free’.
Do not take this medicine if you are allergic to any of these ingredients.
What Wegovy® FlexTouch® looks like
Wegovy® FlexTouch® is a clear and colourless solution for injection in a pre-filled disposable pen.
Each pen contains four doses. The pack size of each strength of Wegovy® FlexTouch® contains 1 pre-filled pen and 4 disposable NovoFine® Plus® needles.
Wegovy® FlexTouch® 0.25 mg solution for injection (Aust R 424390)
One mL of solution contains 0.68 mg of semaglutide. One pre-filled pen contains 1.0 mg semaglutide in 1.5 mL solution.
Wegovy® FlexTouch® 0.5 mg solution for injection (Aust R 424391)
One mL of solution contains 1.34 mg of semaglutide. One pre-filled pen contains 2.0 mg semaglutide in 1.5 mL solution.
Wegovy® FlexTouch® 1 mg solution for injection (Aust R 424392)
One mL of solution contains 1.34 mg of semaglutide. One pre-filled pen contains 4.0 mg semaglutide in 3 mL solution.
Wegovy® FlexTouch® 1.7 mg solution for injection (Aust R 424393)
One mL of solution contains 2.27 mg of semaglutide. One pre-filled pen contains 6.8 mg semaglutide in 3 mL solution.
Wegovy® FlexTouch® 2.4 mg solution for injection (Aust R 424394)
One mL of solution contains 3.2 mg of semaglutide. One pre-filled pen contains 9.6 mg of semaglutide in 3 mL solution.
Who distributes Wegovy®
Wegovy® is supplied in Australia by:
Novo Nordisk Pharmaceuticals Pty. Ltd.
Level 10
118 Mount Street
North Sydney NSW 2060
Wegovy® is supplied in New Zealand by:
Novo Nordisk Pharmaceuticals Ltd.
PO Box 51-268
Pakuranga
Auckland
New Zealand.
Wegovy®, FlexTouch® and NovoFine® are registered trademarks of Novo Nordisk A/S.
© 2025
Novo Nordisk A/S
Further information
For further information call Novo Nordisk Medical Information on 1800 668 626 (Australia) or 0800 733 737 (New Zealand).
wegovycare® is a free support program. To find out more go to:
The Patient Support Program is not authorised or approved by the Australian regulator of medicines, the TGA.
Always check the following websites to ensure you are reading the most recent version of the Wegovy® FlexTouch® medicine information:
www.ebs.tga.gov.au (AU)
www.medsafe.govt.nz/medicines/infosearch.asp (NZ)
This leaflet was prepared in March 2026
VV-LAB-114916 v8.0
Brand Information
| Brand name | Wegovy FlexTouch |
| Active ingredient | Semaglutide |
| Schedule | S4 |
▼ This medicinal product is subject to additional monitoring in Australia. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse events at www.tga.gov.au/reporting-problems.
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Semaglutide (rys).
2 Qualitative and Quantitative Composition
Single-dose pen. 0.25 mg dose pen. One mL of solution contains 0.5 mg of semaglutide. One pre-filled pen contains 0.25 mg semaglutide in 0.5 mL.
0.5 mg dose pen. One mL of solution contains 1 mg of semaglutide. One pre-filled pen contains 0.5 mg semaglutide in 0.5 mL.
1 mg dose pen. One mL of solution contains 2 mg of semaglutide. One pre-filled pen contains 1.0 mg semaglutide in 0.5 mL.
1.7 mg dose pen. One mL of solution contains 2.27 mg of semaglutide. One pre-filled pen contains 1.7 mg semaglutide in 0.75 mL.
2.4 mg dose pen. One mL of solution contains 3.2 mg of semaglutide. One pre-filled pen contains 2.4 mg of semaglutide in 0.75 mL.
FlexTouch pen. Each pen contains four doses.
0.25 mg dose pen. One mL of solution contains 0.68 mg of semaglutide. One pre-filled pen contains 1.0 mg semaglutide in 1.5 mL solution.
0.5 mg dose pen. One mL of solution contains 1.34 mg of semaglutide. One pre-filled pen contains 2.0 mg semaglutide in 1.5 mL solution.
1 mg dose pen. One mL of solution contains 1.34 mg of semaglutide. One pre-filled pen contains 4.0 mg semaglutide in 3 mL solution.
1.7 mg dose pen. One mL of solution contains 2.27 mg of semaglutide. One pre-filled pen contains 6.8 mg semaglutide in 3 mL solution.
2.4 mg dose pen. One mL of solution contains 3.2 mg of semaglutide. One pre-filled pen contains 9.6 mg semaglutide in 3 mL solution.
Semaglutide is a human glucagon-like peptide-1 (GLP-1) analogue produced by recombinant DNA technology in a Saccharomyces cerevisiae strain followed by purification.
Pre-filled syringe. 0.25 mg dose syringe. One mL of solution contains 0.5 mg of semaglutide. One pre-filled syringe contains 0.25 mg semaglutide in 0.5 mL.
0.5 mg dose syringe. One mL of solution contains 1 mg of semaglutide. One pre-filled syringe contains 0.5 mg semaglutide in 0.5 mL.
1 mg dose syringe. One mL of solution contains 2 mg of semaglutide. One pre-filled syringe contains 1.0 mg semaglutide in 0.5 mL.
1.7 mg dose syringe. One mL of solution contains 2.27 mg of semaglutide. One pre-filled syringe contains 1.7 mg semaglutide in 0.75 mL.
2.4 mg dose syringe. One mL of solution contains 3.2 mg of semaglutide. One pre-filled syringe contains 2.4 mg of semaglutide in 0.75 mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection in pre-filled pen and pre-filled syringe.
Clear and colourless isotonic solution; pH = 7.4.
4 Clinical Particulars
4.1 Therapeutic Indications
Weight management. Adults. Wegovy is indicated as an adjunct to a reduced-energy diet and increased physical activity for chronic weight management (including weight loss and weight maintenance) in adults with an initial Body Mass Index (BMI) of ≥ 30 kg/m2 (obesity), or ≥ 27 kg/m2 to < 30 kg/m2 (overweight) in the presence of at least one weight-related comorbidity (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Adolescents. Wegovy is indicated as an adjunct to a reduced-calorie diet and increased physical activity for weight management in adolescents ages 12 years and above with initial: obesity* and body weight above 60 kg.
Treatment with Wegovy should be re-evaluated and discontinued if adolescent patients have not reduced their BMI by at least 5% after 12 weeks on the 2.4 mg or maximum tolerated dose.
*Obesity (BMI ≥ 95th percentile) as defined on sex and age-specific BMI growth charts (CDC.gov) (see Table 1).
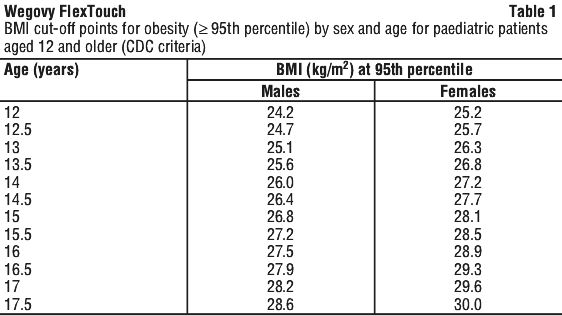
Metabolic dysfunction-associated steatohepatitis (MASH). Wegovy is indicated for the treatment of non-cirrhotic metabolic dysfunction-associated steatohepatitis (MASH) in adults with moderate to advanced liver fibrosis (consistent with stages F2 to F3 fibrosis). This indication was approved via the provisional approval pathway based on resolution of steatohepatitis and improvement in liver fibrosis. Continued approval of this indication depends on verification and description of clinical benefit in confirmatory trials.
4.2 Dose and Method of Administration
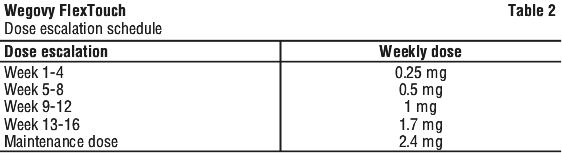
The recommended maintenance dose is 2.4 mg once weekly.
The maintenance dose of 2.4 mg once weekly is reached by starting with a weekly dose of 0.25 mg. To reduce the likelihood of gastrointestinal symptoms, the dose should be escalated over a 16-week period to a maintenance dose of 2.4 mg once weekly (see Table 2).
In case of significant gastrointestinal symptoms, consider delaying dose escalation until symptoms have improved. If patients do not tolerate the 2.4 mg dose, the dose can be decreased to a 1.7 mg weekly dose for maintenance. Patients should re-escalate the dose to 2.4 mg weekly, if tolerated. Weekly doses higher than 2.4 mg are not recommended.
Wegovy is to be injected subcutaneously in the abdomen, in the thigh or in the upper arm. The injection site can be changed without dose adjustment. Wegovy should not be administered intravenously or intramuscularly. The day of weekly administration can be changed if necessary, as long as the time between two doses is at least 3 days (> 72 hours). After selecting a new dosing day, once-weekly dosing should be continued.
Patients should be advised to read the instruction for use included in the package leaflet carefully before administering Wegovy.
Single-dose pen. The single-dose pen is for single use in one patient only. The single-dose pen should be discarded after use. When administering semaglutide, the pen should be pressed firmly against the skin until the yellow bar has stopped moving. The injection takes about 5-10 seconds.
FlexTouch pen. The Wegovy FlexTouch pen is for use by one patient only. It contains 4 doses. A new needle should be attached for each use. The needle should be discarded after each use.
Pre-filled syringe. When using the pre-filled syringe, abdominal injections must be at least 5 centimetres from the belly button and upper arm injections must be in the back of the arm and only with help from someone else.
Missed dose. If a dose is missed, it should be administered as soon as possible and within 5 days after the missed dose. If more than 5 days have passed, the missed dose should be skipped, and the next dose should be administered on the regularly scheduled day of the week. In each case, patients can then resume their regular once weekly dosing schedule. If more doses are missed, reducing the starting dose for re-initiation should be considered.
Special populations. Patients with type 2 diabetes. Wegovy should not be used in combination with other GLP-1 receptor agonist products.
When initiating Wegovy, consider reducing the dose of concomitantly administered insulin or insulin secretagogues (such as sulfonylureas) to reduce the risk of hypoglycaemia.
Elderly (≥ 65 years old). No dose adjustment is required based on age. Therapeutic experience in patients ≥ 85 years of age is limited (see Section 5.2 Pharmacokinetic Properties).
Gender. No dose adjustment is required based on gender.
Race and ethnicity. No dose adjustment is required based on race and ethnicity.
Patients with renal impairment. No dose adjustment is required for patients with renal impairment. Experience with the use of semaglutide in patients with severe (CrCl < 30 mL/min) renal impairment is limited. Semaglutide is not recommended for use in patients with end-stage renal disease (see Section 5.2 Pharmacokinetic Properties).
Patients with hepatic impairment (Child-Pugh A, B or C). No dose adjustment is required for patients with hepatic impairment (see Section 5.2 Pharmacokinetic Properties). Experience with the use of semaglutide in patients with severe hepatic impairment (Child-Pugh C) is limited. Caution should be exercised when treating these patients with semaglutide. In a trial in adult patients with MASH and compensated liver cirrhosis (F4c), no safety signals were identified (n=47 on active treatment).
Children and adolescents: weight management. Safety and efficacy of Wegovy in children below 12 years have not been studied.
For adolescents aged 12 to 17 years, the same dose escalation schedule as for adults should be applied (see Table 2). The dose should be increased until the maintenance dose of 2.4 mg weekly or a lower maximum tolerated dose has been reached. Weekly doses higher than 2.4 mg are not recommended.
It is recommended to regularly review the goals of treatment with Wegovy, particularly once the target weight (e.g. BMI < 85th percentile) has been achieved.
Children and adolescents: metabolic dysfunction-associated steatohepatitis (MASH). The safety and efficacy of Wegovy in children and adolescents with MASH below 18 years have not been established. No data are available.
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in Section 6.1 List of Excipients.
4.4 Special Warnings and Precautions for Use
Aspiration in association with general anaesthesia or deep sedation. Cases of pulmonary aspiration have been reported in patients receiving GLP-1 RAs undergoing general anaesthesia (GA) or deep sedation despite reported adherence to preoperative fasting recommendations. Therefore, the increased risk of residual gastric content because of delayed gastric emptying should be considered prior to performing procedures with GA or deep sedation.
Gastrointestinal effects and dehydration. Use of GLP-1 receptor agonists may be associated with gastrointestinal adverse reactions. This should be considered when treating patients with impaired renal function as nausea, vomiting, and diarrhoea, may cause dehydration which could cause a deterioration of renal function. There have been postmarketing reports of acute kidney injury and worsening of chronic renal failure, which have in some cases required haemodialysis, in patients treated with semaglutide.
Acute pancreatitis. Acute pancreatitis, including fatal and non-fatal haemorrhagic or necrotising pancreatitis, has been observed with the use of GLP-1 receptor agonists, including semaglutide. Patients should be informed of the characteristic symptoms of acute pancreatitis. If pancreatitis is suspected, Wegovy should be discontinued; if confirmed, Wegovy should not be restarted. Caution should be exercised in patients with a history of pancreatitis.
The clinical significance of elevations in lipase or amylase with Wegovy is unknown in the absence of other signs and symptoms of pancreatitis.
Psychiatric disorders. Suicidal behaviour and ideation have been reported with GLP-1 receptor agonists. Monitor patients for the emergence or worsening of depression, suicidal thoughts or behaviours, and/or any unusual changes in mood or behaviour. Consider the benefits and risks for individual patients prior to initiating or continuing therapy in patients with suicidal thoughts or behaviours or have a history of suicidal attempts.
Populations not studied. The safety and efficacy of Wegovy have not been investigated in patients:
treated with other products for weight management;
with type 1 diabetes;
with congestive heart failure New York Heart Association (NYHA) class IV.
The use of semaglutide is not recommended in these patients.
There is limited experience with Wegovy in patients:
aged 85 years or more;
with inflammatory bowel disease;
with diabetic gastroparesis;
with MASH and BMI < 25 kg/m2 (or BMI < 23 kg/m2 for Asian population).
Use with caution in these patients.
Patients with type 2 diabetes. Wegovy must not be used as a substitute for insulin in patients with diabetes.
Semaglutide should not be used in combination with other GLP-1 receptor agonist products. It has not been evaluated and an increased risk of adverse reactions related to overdose is considered likely.
Hypoglycaemia in patients with overweight or obesity and type 2 diabetes. Insulin and sulfonylurea are known to cause hypoglycaemia. Patients treated with Wegovy in combination with a sulfonylurea or insulin may have an increased risk of hypoglycaemia. The risk of hypoglycaemia can be lowered by reducing the dose of sulfonylurea or insulin when initiating treatment with a GLP-1 receptor agonists.
The addition of Wegovy in patients treated with insulin has not been evaluated.
Diabetic retinopathy in patients with overweight or obesity and type 2 diabetes. Rapid improvement in glucose control has been associated with a temporary worsening of diabetic retinopathy. Long-term glycaemic control decreases the risk of diabetic retinopathy. Patients with a history of diabetic retinopathy should be monitored for worsening and treated according to local clinical guidelines. There is no experience with Wegovy in patients with type 2 diabetes with uncontrolled or potentially unstable diabetic retinopathy. In these patients, treatment with Wegovy is not recommended.
Use in the elderly. See Section 5.2 Pharmacokinetic Properties.
Paediatric use. Safety and efficacy of Wegovy in children with overweight or obesity below 12 years have not been studied. Semaglutide has not been studied in paediatric patients with MASH.
Use in renal impairment. Experience with the use of semaglutide in patients with severe (CrCl < 30 mL/min) renal impairment is limited. Semaglutide is not recommended for use in patients with end-stage renal disease (see Section 5.1 Pharmacodynamic Properties; Section 5.2 Pharmacokinetic Properties).
Use in hepatic impairment. Experience with the use of semaglutide in patients with severe hepatic impairment is limited. Caution should be exercised when treating these patients with semaglutide.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
In vitro studies have shown very low potential for semaglutide to inhibit or induce CYP enzymes, and to inhibit drug transporters.
The delay of gastric emptying with semaglutide may influence the absorption of concomitantly administered oral medicinal products; therefore, semaglutide should be used with caution in patients receiving oral medicinal products that require rapid gastrointestinal absorption. No clinically relevant effect on the rate of gastric emptying was observed with semaglutide 2.4 mg.
In clinical pharmacology trials assessing the effect of semaglutide 1.0 mg on the absorption of coadministered oral medications at steady state no clinically relevant drug-drug interactions with semaglutide (Figure 1) was observed based on the evaluated medications. Therefore, no dose adjustment is required when co-administered with semaglutide.
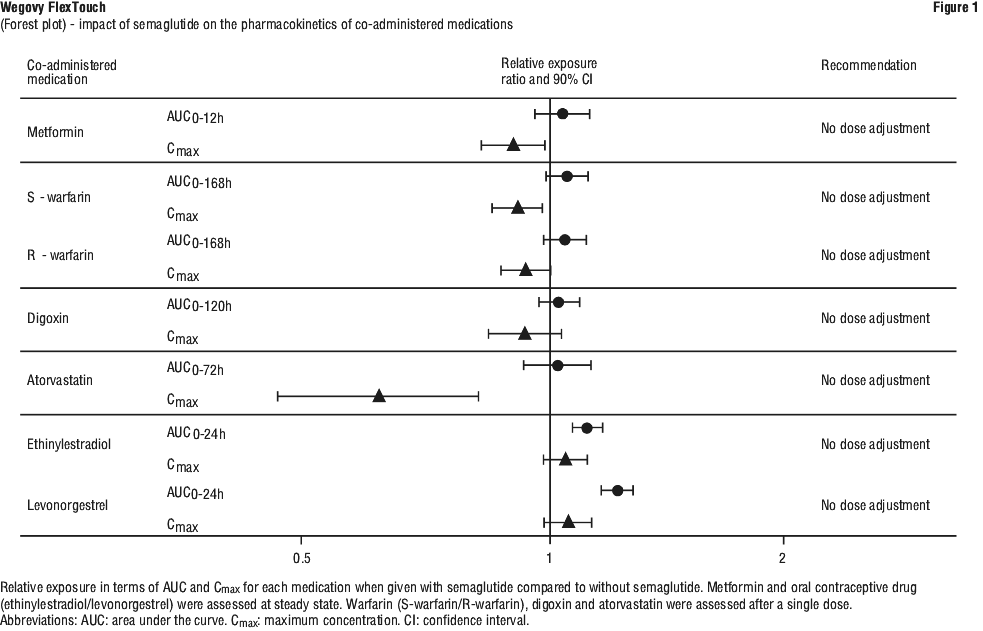
Atorvastatin. Semaglutide did not change the overall exposure of atorvastatin following a single dose administration of atorvastatin (40 mg). Atorvastatin Cmax was decreased by 38%. This was assessed not to be clinically relevant.
Digoxin. Semaglutide did not change the overall exposure or Cmax of digoxin following a single dose of digoxin (0.5 mg).
Metformin. Semaglutide did not change the overall exposure or Cmax of metformin following dosing of 500 mg twice daily over 3.5 days.
Warfarin and other coumarin derivatives. Semaglutide did not change overall exposure or Cmax of R and S-warfarin following a single dose of warfarin (25 mg), and the pharmacodynamic effects of warfarin as measured by the international normalised ratio (INR) were not affected in a clinically relevant manner. However, cases of decreased INR have been reported during concomitant use of acenocoumarol and semaglutide. Upon initiation of semaglutide treatment in patients on warfarin or other coumarin derivatives, frequent monitoring of INR is recommended.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. The effect of semaglutide on fertility in humans is unknown. Semaglutide did not affect male fertility in rats at daily subcutaneous (s.c.) doses of 828 microgram/kg, resulting in exposures approximately 4.5 times the clinical AUC. In female rats, an increase in oestrous length and a small reduction in number of ovulations were observed at doses associated with maternal body weight loss (≥ 30 microgram/kg/day SC, resulting in subclinical exposures).
Use in pregnancy. (Category D)
Semaglutide should not be used during pregnancy. Women of childbearing potential are recommended to use contraception when treated with semaglutide. If a patient wishes to become pregnant, or pregnancy occurs, semaglutide should be discontinued. Semaglutide should be discontinued at least 2 months before a planned pregnancy due to the long half-life (see Section 5.2 Pharmacokinetic Properties).
Studies in animals have shown reproductive toxicity when semaglutide was administered during organogenesis. In pregnant rats, embryofetal toxicity (lethality, impaired growth and an increased incidence of fetal abnormalities) was observed at subclinical plasma exposures. Mechanistic studies suggest a direct GLP-1 receptor mediated role of semaglutide on some of the effects in rats (species specific). In pregnant rabbits, pharmacologically mediated reductions in maternal body weight gain and food consumption were observed at all dose levels. Early pregnancy losses and increased incidences of minor visceral (kidney, liver) and skeletal (sternebra) fetal abnormalities were observed at ≥ 0.0025 mg/kg/day, at clinically relevant exposures. In pregnant cynomolgus monkeys, pharmacologically mediated, marked initial maternal body weight loss and reductions in body weight gain and food consumption coincided with the occurrence of sporadic abnormalities (vertebra, sternebra, ribs) and with an increase in early pregnancy losses at ≥ 0.075 mg/kg twice weekly (> 1.4-fold clinical exposure at 2.4 mg/week). Exposures at the NOAEL in all species were subclinical and a direct effect of semaglutide on the fetus cannot be excluded.
Use in lactation. In lactating rats, semaglutide was excreted in milk. A risk to a breast-fed child cannot be excluded. Semaglutide should not be used during breast-feeding.
4.7 Effects on Ability to Drive and Use Machines
Wegovy has no or negligible influence on the ability to drive or use machines. However, dizziness can be experienced mainly during the dose escalation period. Driving or use of machines should be done cautiously if dizziness occurs.
Effect on the ability to drive and use machines for patients with overweight or obesity and type 2 diabetes. If Wegovy is used in combination with a sulfonylurea or insulin, patients should be advised to take precautions to avoid hypoglycaemia while driving and using machines.
4.8 Adverse Effects (Undesirable Effects)
Summary of safety profile. Weight management. In 4 phase 3a trials, 2,650 adult patients were exposed to Wegovy. The duration of the trials were 68 weeks. Similar to other GLP-1 receptor agonists, the most frequently reported adverse reactions were gastrointestinal disorders including nausea, diarrhoea, constipation and vomiting.
MASH. In an ongoing 240-week phase 3 trial (ESSENCE), 800 adult patients were exposed to Wegovy at the time of interim analysis. Median duration of exposure was 95 weeks. Similar to other GLP-1 receptor agonists, the most frequently reported adverse reactions were gastrointestinal disorders including nausea, diarrhoea, constipation and vomiting.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Tabulated list of adverse events. See Table 3.
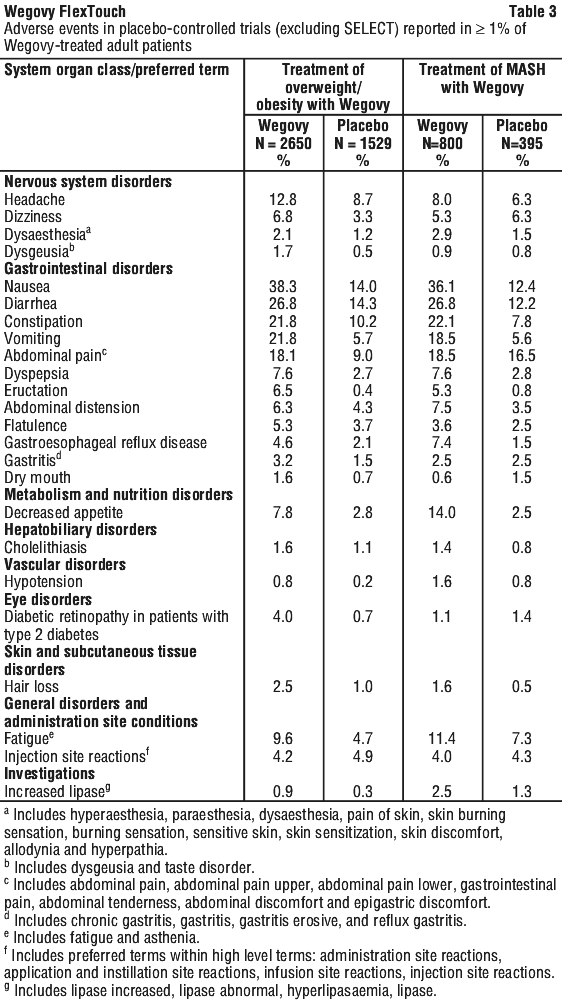
Adverse reactions associated with Wegovy are listed by body system and frequency. Frequency categories are defined as: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000).
Other events that occurred at a higher incidence among Wegovy-treated adult patients included dyspepsia, abdominal pain, abdominal distension, eructation, flatulence, gastroesophageal reflux disease, gastritis, haemorrhoids, and hiccups.
Patients with moderate renal impairment (eGFR ≥ 30 mL/min/1.73 m2) may experience more gastrointestinal effects when treated with semaglutide.
The gastrointestinal events led to permanent treatment discontinuation in 4.3% of patients.
Acute pancreatitis. The frequency of adjudication-confirmed acute pancreatitis reported in phase 3a clinical trials was 0.2% for Wegovy and < 0.1% for placebo, respectively.
In SELECT, the cardiovascular outcome trial, the frequency of acute pancreatitis confirmed by adjudication was 0.2% for Wegovy and 0.3% for placebo.
Acute gallstone disease/cholelithiasis. Cholelithiasis was reported in 1.6% and led to cholecystitis in 0.6% of patients treated with Wegovy. Cholelithiasis and cholecystitis were reported in 1.1% and 0.3%, respectively, of patients treated with placebo.
Hair loss. Hair loss was reported in 2.5% of patients treated with Wegovy and in 1.0% of patients treated with placebo. The events were mainly of mild severity and most patients recovered while on continued treatment. Hair loss was reported more frequently in patients with a greater weight loss (≥ 20%).
Increased heart rate. In the phase 3a trials, a mean increase of 3 beats per minute (bpm) from a baseline mean of 72 bpm was observed in patients treated with Wegovy. The proportions of patients with a maximum increase from baseline ≥ 20 bpm at any timepoint during the on-treatment period were 26.0% in the Wegovy group vs 15.6% in the placebo group.
Immunogenicity. Consistent with the potentially immunogenic properties of medicinal products containing proteins or peptides, patients may develop antibodies following treatment with semaglutide. The proportion of patients testing positive for anti-semaglutide antibodies at any time post-baseline was low (2.9%) and no patients had anti-semaglutide neutralising antibodies or anti-semaglutide antibodies with endogenous GLP-1 neutralising effect at end-of-trial.
Dysaesthesia. Events related to a clinical picture of altered skin sensation such as dysaesthesia, paraesthesia, hyperaesthesia, burning sensation, allodynia and sensitive skin were reported in 2.1% of patients treated with Wegovy injection and 1.2% of patients treated with placebo. The events were mild to moderate in severity and most patients recovered while on continued treatment.
Fractures. In SELECT, the cardiovascular outcomes trial in adults, more fractures of the hip and pelvis were reported on semaglutide than on placebo in female patients: 1.0% (24/2448) vs. 0.2% (5/2424), and in patients aged 75 years and older: 2.4% (17/703) vs. 0.6% (4/663), respectively.
Urolithiasis. In SELECT, the cardiovascular outcomes trial, 1.2% of semaglutide-treated patients and 0.8% of patients receiving placebo reported urolithiasis, including serious reactions that were reported more frequently among patients receiving semaglutide (0.6%) than placebo (0.4%).
Patients with type 2 diabetes. Hypoglycaemia in patients with overweight or obesity, MASH and type 2 diabetes. In STEP 2, clinically significant hypoglycaemia was observed in 6.2% (0.1 events/patient year) of subjects treated with Wegovy compared with 2.5% (0.03 events/patient year) of subjects treated with placebo. One episode (0.2% of subjects, 0.002 events/patient year) was reported as severe. The risk of hypoglycaemia was increased when Wegovy was used with a sulfonylurea.
In ESSENCE, severe hypoglycaemia was reported in 2.2% of patients (0.015 events/patient year) treated with Wegovy compared to 0.5% of patients (0.003 event /patient year) treated with placebo.
Diabetic retinopathy in patients with overweight or obesity and type 2 diabetes. Few episodes of diabetic retinopathy (4.0% vs 2.7% of patients treated with Wegovy vs placebo, respectively) were observed in STEP 2.
Long-term glycaemic control decreases the risk of diabetic retinopathy. A 2-year clinical trial investigated semaglutide 0.5 mg and 1 mg vs. placebo in 3,297 patients with type 2 diabetes, with high cardiovascular risk, long duration of diabetes and poorly controlled blood glucose. In this trial, adjudicated events of diabetic retinopathy complications occurred in more patients treated with semaglutide (3.0%) compared to placebo (1.8%). This was observed in insulin-treated patients with known diabetic retinopathy. The treatment difference appeared early and persisted throughout the trial.
In STEP 2, retinal disorders were reported by 6.9% of patients treated with Wegovy, 6.2% of patients treated with semaglutide 1 mg, and 4.2% of patients treated with placebo. The majority of events were reported as diabetic retinopathy (4.0%, 2.7%, and 2.7%, respectively) and non-proliferative retinopathy (0.7%, 0%, and 0%, respectively).
Paediatric population. In a clinical trial conducted in adolescents of 12 years to below 18 years with obesity or overweight with at least one weight-related comorbidity, 133 patients were exposed to Wegovy. The trial duration was 68 weeks.
Overall, the frequency, type and severity of adverse reactions in the adolescents were comparable to that observed in the adult population. Cholelithiasis was reported in 3.8% of patients treated with Wegovy.
Semaglutide did not appear to affect growth or pubertal development during the trial period.
To date, there are no long-term (beyond 68 weeks) clinical trial data on safety or efficacy in adolescents.
Wegovy has not been studied in children and adolescents below 18 years with MASH.
Cardiovascular outcomes trial. In SELECT, the cardiovascular outcomes trial, the adverse event profile of systematically collected adverse events was similar to that seen in the phase 3a trials (see Section 5.1 Pharmacodynamic Properties, Clinical trials). Additional adverse events included urolithiasis and hip fractures. For non-systematically collected adverse events, no definite conclusions can be made.
Trial in adults with MASH. In ESSENCE, in adults with MASH, the adverse reaction profile was similar to that seen in the weight management phase 3a trials (described in Section 5.1 Pharmacodynamic Properties, Clinical trials). Due to the low number of patients with 'lean-MASH' (MASH with BMI < 25 kg/m2 or BMI< 23 kg/m2 for Asian population) included in the ESSENCE trial, safety data for this sub-group is limited.
Post-market adverse effects. Gastrointestinal disorders. Intestinal obstruction*.
*Grouped term covering PTs intestinal obstruction, ileus, small intestinal obstruction.
4.9 Overdose
Overdose with semaglutide may be associated with gastrointestinal disorders which could lead to dehydration. In the event of overdose the patient should be observed for clinical signs and appropriate supportive treatment initiated. A prolonged period of observation and treatment for these symptoms may be necessary, taking into account the long half-life of Wegovy of approximately 1 week (see Section 5.2 Pharmacokinetic Properties).
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Semaglutide is a GLP-1 analogue with 94% sequence homology to human GLP-1. Semaglutide acts as a GLP-1 receptor agonist that binds to and activates the GLP-1 receptor, the target for native GLP-1.
GLP-1 is a physiological regulator and has multiple actions in glucose and appetite regulation. The glucose and appetite effects are specifically mediated via GLP-1 receptors in the pancreas and the brain.
Clinical studies show that semaglutide reduces energy intake, increases feelings of satiety, fullness and control of eating, and reduces feelings of hunger, and frequency and intensity of cravings.
Animal studies show that semaglutide works in the brain through the GLP-1 receptor. Semaglutide has direct effects on areas in the brain involved in homeostatic regulation of food intake in the hypothalamus and the brainstem. Semaglutide affects the hedonic reward system through direct and indirect effects in brain areas including the septum, thalamus and amygdala. Semaglutide has shown an effect to change food intake in animals away from more rewarding high fat, sweet items.
Semaglutide orchestrates the homeostatic and hedonic contributions with executive function to regulate caloric intake, appetite, reward and food choice.
In addition, in clinical studies semaglutide have shown to reduce blood glucose through a mechanism where it stimulates insulin secretion and lowers glucagon secretion, both in a glucose-dependent manner. The mechanism of blood glucose lowering also involves a minor delay in gastric emptying in the early postprandial phase. During hypoglycaemia, semaglutide diminishes insulin secretion and does not impair glucagon secretion.
The mechanism of action of semaglutide for cardiovascular risk reduction has not been established.
MASH pathogenesis is characterised by accumulation of fat in the liver leading to lipotoxicity, cellular damage and upregulation of proinflammatory cytokines, activating the immune system and pathways leading to hepatic collagen deposition (fibrosis).
The liver-specific mechanism of action of semaglutide is multifactorial and thought to be mediated through improvement in metabolic factors, including weight loss, improved glucose and lipid metabolism, and reduced inflammation. Semaglutide affects gene pathways of both inflammation and fibrosis, consequently positively changing the proteomic pattern of an individual with MASH. Moreover, semaglutide lowers liver fat deposition.
Pharmacodynamic effects. Weight loss distribution. In a sub-study in STEP 1 (N = 140), body composition was measured using dual energy X-ray absorptiometry (DEXA). The results of the DEXA assessment showed that treatment with Wegovy was accompanied by greater reduction in fat mass than in lean body mass leading to an improvement in body composition compared to placebo after 68 weeks. Furthermore, this reduction in total fat mass was accompanied by a reduction in visceral fat. In the semaglutide group, there was a mean (SD) decrease in total fat mass proportion of 3.9%-points (5.4%-points), an increase in lean body mass proportion of 3.4% (5.1%) and a decrease in regional visceral fat mass proportion of 2.2%-points (4.4%). These results suggest that most of the total weight loss was attributable to a reduction in fat tissue, including visceral fat.
Appetite regulation, energy intake and food choice. Wegovy reduces appetite by increasing feelings of fullness and satiety, while lowering hunger and prospective food consumption. In a phase 1 trial, energy intake during an ad libitum meal was 35% lower with Wegovy compared to placebo after 20 weeks of dosing. This was supported by improved control of eating, less food cravings (for dairy and savoury foods), less desire for sweet food and a relatively lower preference for high fat food.
Cardiac electrophysiology (QTc). The effect of semaglutide on cardiac repolarization was tested in a thorough QTc trial. Semaglutide did not prolong QTc intervals at doses up to 1.5 mg at steady state.
The semaglutide exposure for subjects with overweight or obesity treated with Wegovy is comparable to the exposure evaluated in the semaglutide QTc study in healthy volunteers.
MASH disease activity. Liver steatosis*. A higher proportion of patients with MASH and F2 or F3 achieved an improvement in histology-assessed liver steatosis with semaglutide (70.0%) compared to placebo (42.3%) at week 72 in ESSENCE. Additionally, in ESSENCE semaglutide reduced liver steatosis assessed by TE using Controlled Attenuation Parameter (CAP) (dB/m) (43.4) compared to placebo (13.1) at 72 weeks.
Hepatocyte ballooning and lobular inflammation*. A higher proportion of patients with MASH and F2 or F3 achieved an improvement in histology-assessed hepatocyte ballooning with semaglutide (58.3%) compared to placebo (40.1%) at 72 weeks in ESSENCE. Additionally, in ESSENCE 36.0% of patients treated with semaglutide had an improvement in histology-assessed lobular inflammation compared to 20.6% in the placebo group at week 72.
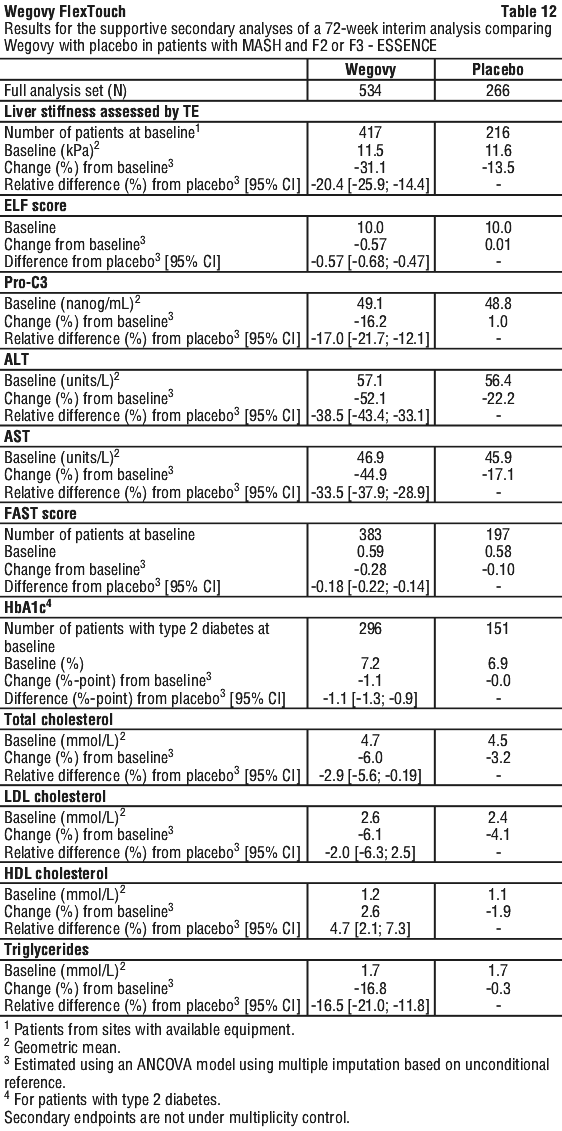
Plasma liver enzymes (ALT, AST) and FAST score*. Semaglutide reduced the levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST)in ESSENCE and in Trial NN9931-4296. Moreover, treatment with semaglutide resulted in a reduction in FAST score in ESSENCE.
Liver fibrosis*. Semaglutide decreased liver stiffness assessed by transient elastography (TE) and the levels of the pro-peptide of type III collagen biomarker (Pro-C3) in ESSENCE. Semaglutide also reduced the Enhanced Liver Fibrosis (ELF) score in ESSENCE and in Trial NN9931-4296.
*Secondary endpoints not under multiplicity control.
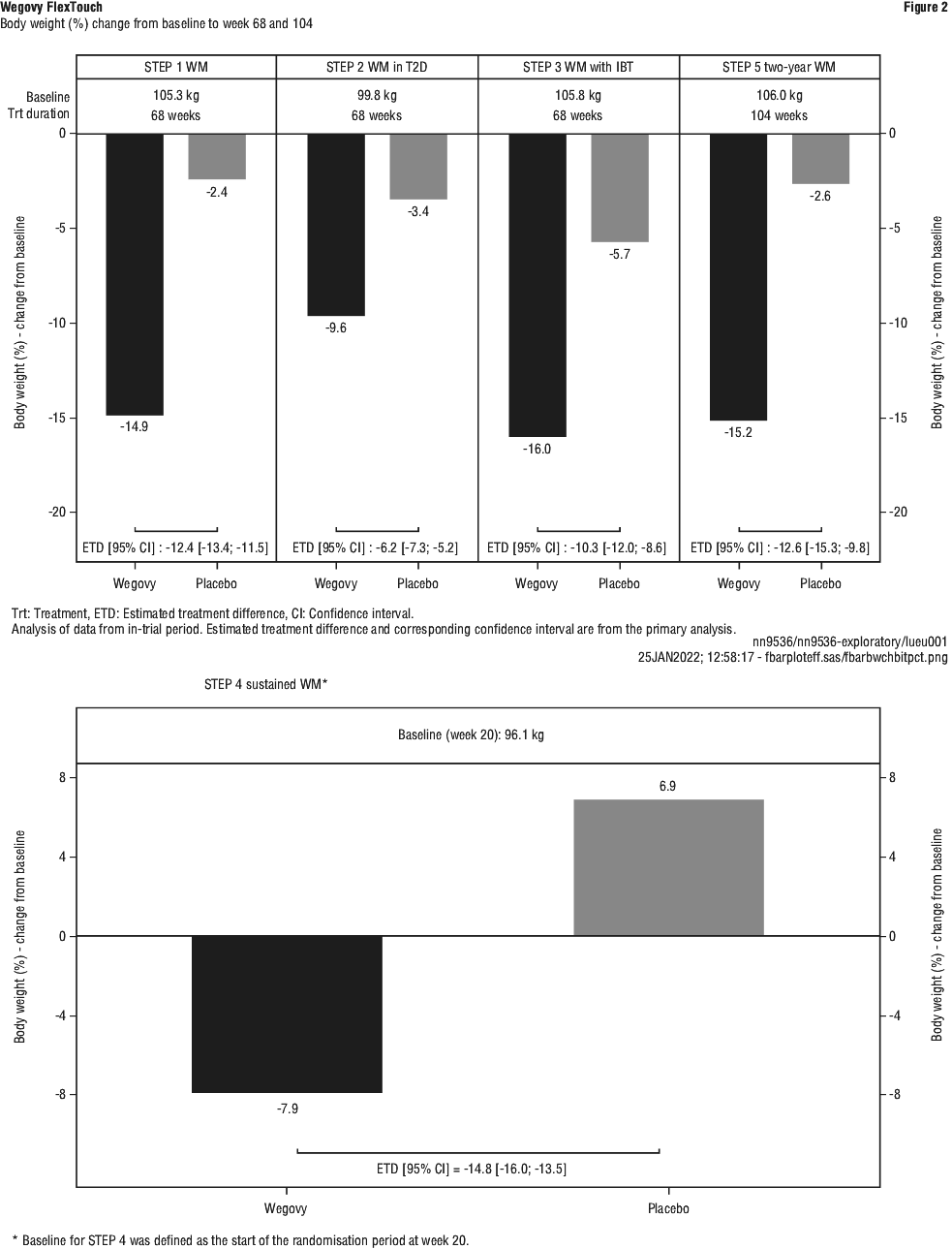
Clinical trials. Clinical programme for weight management (STEP). The efficacy and safety of Wegovy for weight management in combination with a reduced calorie intake and increased physical activity were evaluated in four 68-week double-blinded randomised placebo-controlled phase 3a trials (STEP 1-4). A total of 4684 patients (2652 randomised to treatment with Wegovy) were included in these trials. Furthermore, the two-year efficacy and safety of Wegovy compared to placebo were evaluated in a double-blinded randomised placebo-controlled phase 3b trial (STEP 5) including 304 patients (152 on treatment with Wegovy).
Treatment with Wegovy demonstrated superior, clinically meaningful, and sustained weight loss compared with placebo in patients with obesity (BMI ≥ 30 kg/m2) or overweight (BMI ≥ 27 kg/m2 to < 30 kg/m2) and at least one weight-related comorbidity (Figure 2). Furthermore, across the trials, a higher proportion of patients achieved ≥ 5%, ≥ 10% and ≥ 15% weight loss with Wegovy compared with placebo.
Treatment with Wegovy also showed statistically significant improvements in waist circumference and systolic blood pressure compared to placebo. Wegovy also showed statistically significant improvements in physical functioning compared to placebo, except for STEP 3 where the improvement was not statistically significant.
Wegovy demonstrated efficacy in weight loss regardless of age, sex, race, ethnicity, baseline body weight, BMI, presence of type 2 diabetes and level of renal function.
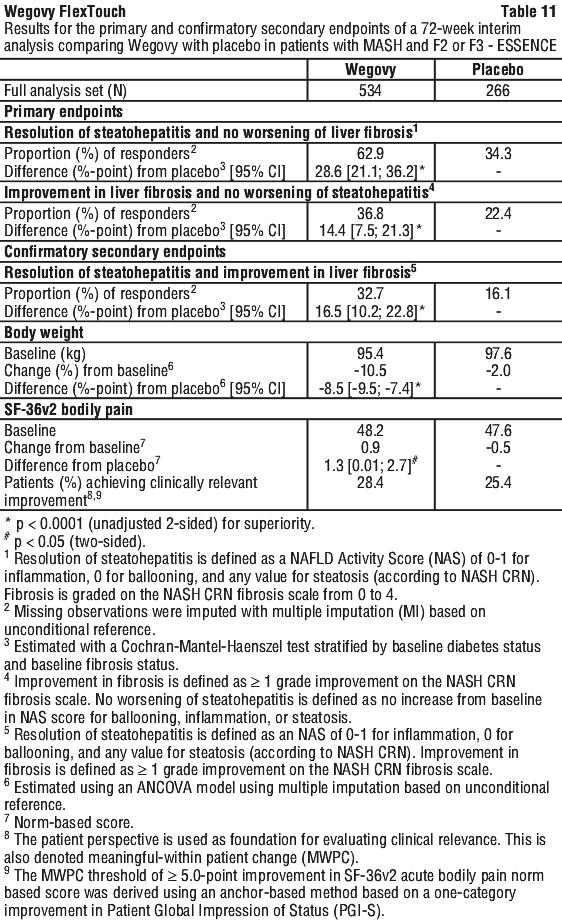
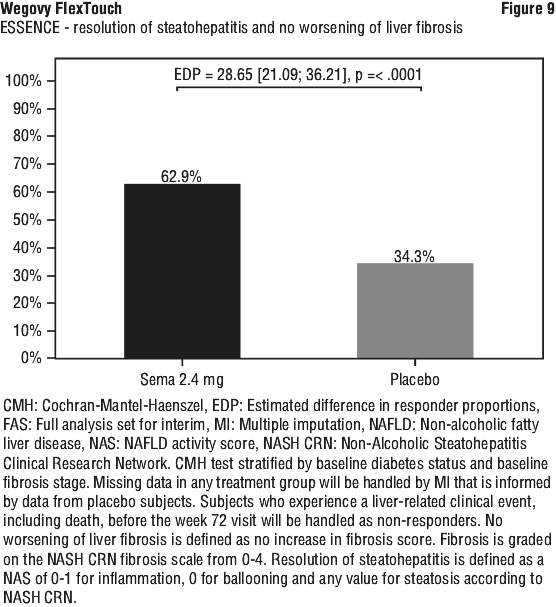
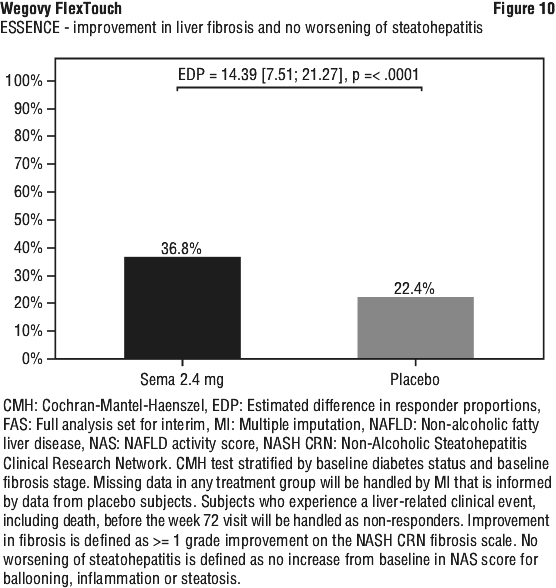
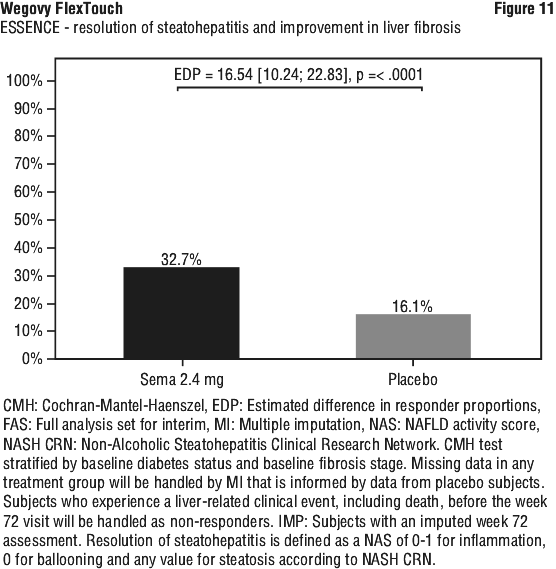
Clinical programme for MASH (ESSENCE and NN9931-4296). The efficacy and safety of Wegovy for MASH have been evaluated in one phase 3 trial (ESSENCE, semaglutide s.c. 2.4 mg once weekly) in adult patients with MASH and fibrosis stage 2 (F2) or 3 (F3) and in one phase 2 trial (NN9931-4296, semaglutide s.c. 0.1, 0.2, or 0.4 mg once daily) in adult patients with MASH and F1 to F3. Treatment with Wegovy in ESSENCE demonstrated superior and clinically meaningful differences relative to placebo in the proportion of patients achieving resolution of steatohepatitis with no worsening of liver fibrosis, improvement in liver fibrosis with no worsening of steatohepatitis, as well as in the proportion of patients achieving resolution of steatohepatitis with improvement in liver fibrosis.
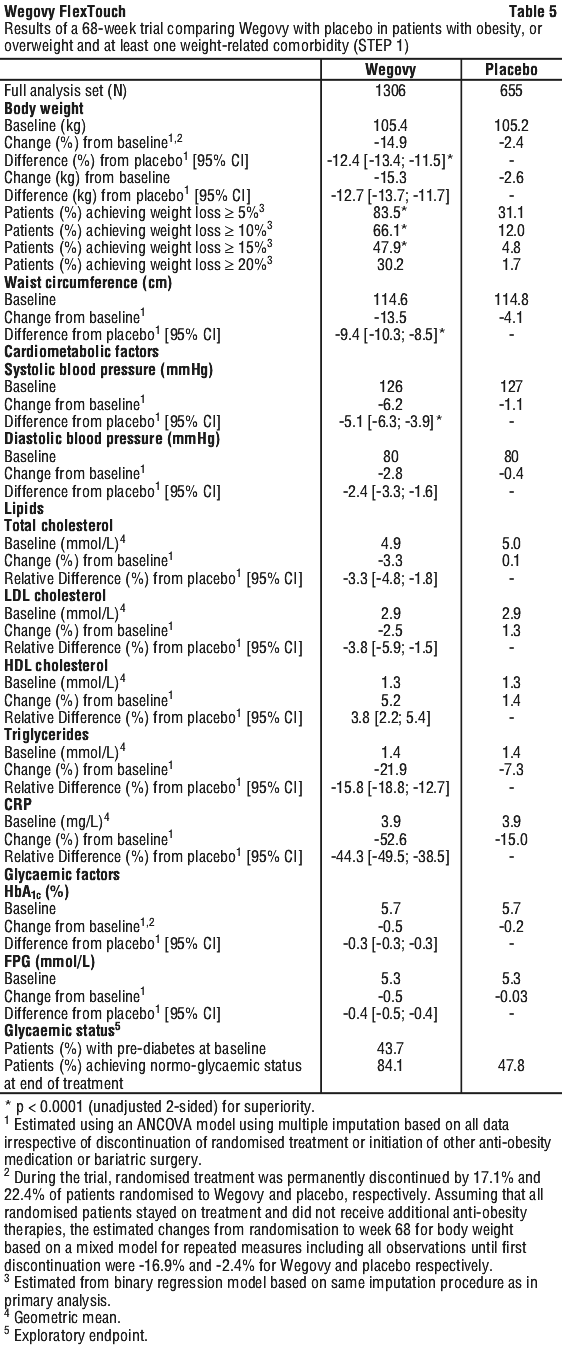
Weight management. STEP 1: weight management. In a 68-week double-blind trial, 1961 patients with obesity (BMI ≥ 30 kg/m2), or with overweight (BMI ≥ 27 to < 30 kg/m2) and at least one weight-related comorbidity were randomised to Wegovy or placebo. All patients received counselling with regards to diet (500 kcal/2092 kJ deficit per day) relative to the estimated total energy expenditure (TEE) (calculated once at randomisation), and physical activity (150 min of physical activity per week was encouraged).
At baseline, patients had a mean BMI of 37.9 kg/m2 and a mean body weight of 105.3 kg. The mean age of patients was 46 years. There were 75.1% Caucasian/White, 5.7% Black/African American, and 13.3% Asian. A total of 12.0% were Hispanic or Latino. The majority of patients had at least one weight-related comorbidity. These included, but were not limited to, pre-diabetes (43.7%), dyslipidaemia (37.0%), hypertension (36.0%), knee or hip osteoarthritis (15.9%), obstructive sleep apnoea (11.7%), asthma/chronic obstructive pulmonary disease (COPD) (11.6%), liver disease (metabolic dysfunction-associated steatotic liver disease (MASLD or MASH)) (8.6%) and polycystic ovary syndrome (PCOS) (6.6%).
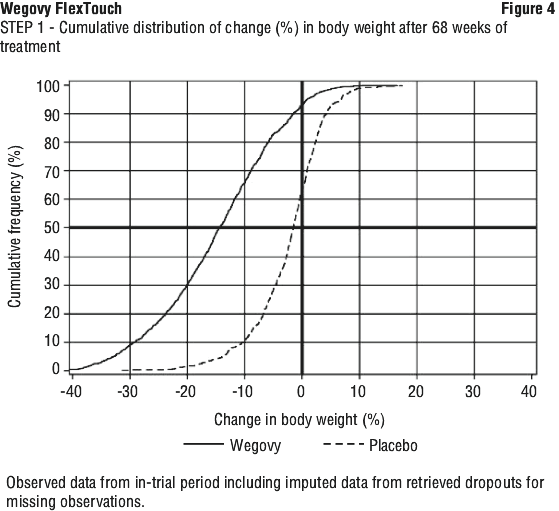
Weight loss with Wegovy occurred early and continued throughout the trial. At end of treatment (week 68), the weight loss with Wegovy was superior and clinically meaningful compared with placebo (see Table 5 and Figure 3). Furthermore, a higher proportion of patients achieved ≥ 5%, ≥ 10%, ≥ 15% and ≥ 20% weight loss with Wegovy compared with placebo (see Table 5 and Figure 4). Among patients with pre-diabetes at baseline, 84.1% and 47.8% achieved a normo-glycaemic status at end of treatment with Wegovy and placebo, respectively.
Following the 68-week trial, a 52-week off-treatment extension was conducted including 327 patients who had completed the main trial period on the maintenance dose of Wegovy or placebo.
The trial extension consisted of four clinic visits and did not include structured lifestyle intervention. In the off-treatment period from week 68 to week 120, mean body weight increased in both treatment groups. However, for patients that had been treated with semaglutide for the main trial period the weight remained 5.6% below baseline compared to 0.1% for the placebo group.
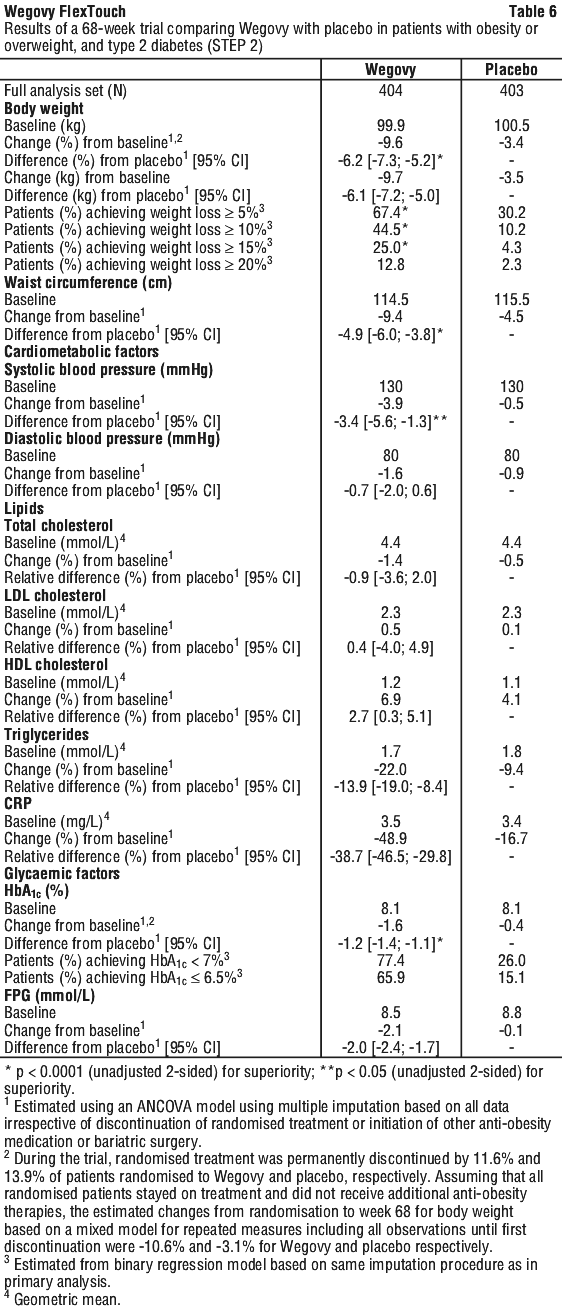
At baseline, patients had a mean BMI of 35.7 kg/m2, a mean body weight of 99.8 kg and a mean HbA1c of 8.1%. The mean age was 55 years. There were 62.1% Caucasian/White, 8.3% Black/African American, and 26.2% Asian. A total of 12.8% were Hispanic or Latino. The majority of patients had at least two weight-related comorbidities. Besides type 2 diabetes these included, but were not limited to, hypertension (69.8%), dyslipidaemia (68.0%), liver disease (MASLD or MASH) (22.6%), knee or hip osteoarthritis (19.6%), obstructive sleep apnoea (15.1%), asthma/COPD (8.4%) and PCOS (4.1%).
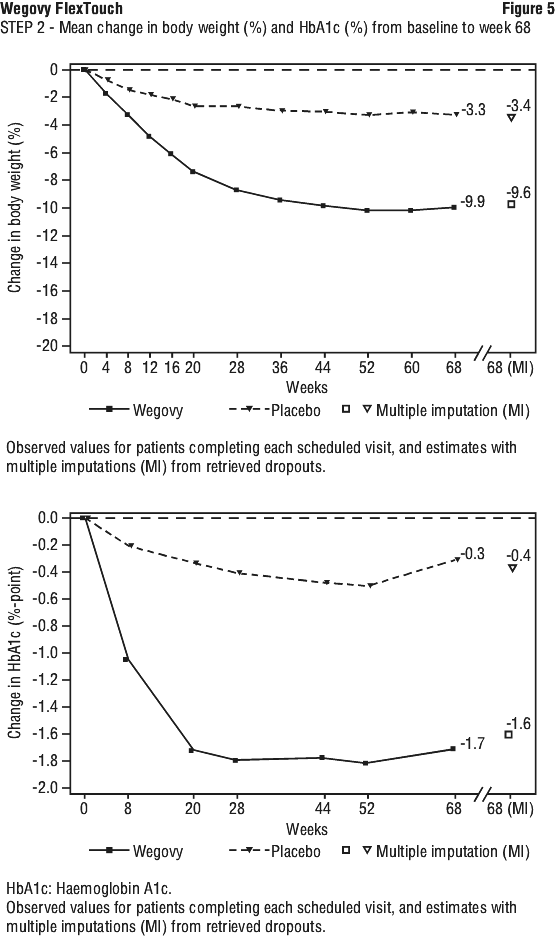
Treatment with Wegovy for 68 weeks resulted in superior and clinically meaningful reductions in body weight and in HbA1c compared to placebo (see Table 6 and Figure 5).
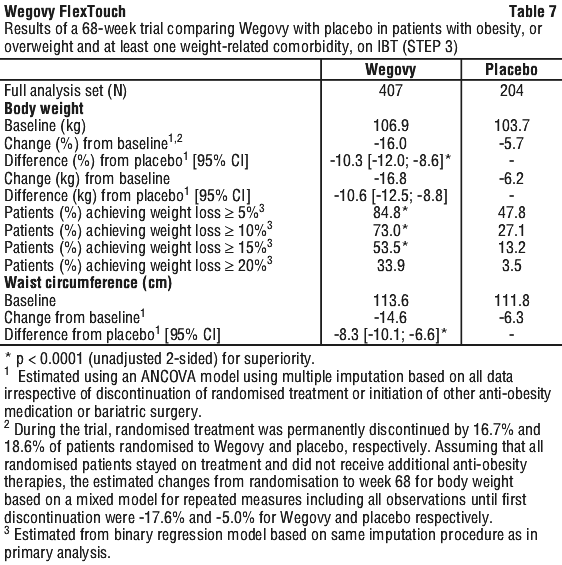
At baseline, patients had a mean BMI of 38.0 kg/m2 and a mean body weight of 105.8 kg. The mean age was 46 years. There were 76.1% Caucasian/White, 19.0% Black/African American, and 1.8% Asian. A total of 19.8% were Hispanic or Latino. The majority of patients had at least one weight-related comorbidity. These included, but were not limited to, pre-diabetes (49.8%), hypertension (34.7%), dyslipidaemia (34.7%), knee or hip osteoarthritis (18.7%), asthma/COPD (15.1%), obstructive sleep apnoea (12.6%), liver disease (MASLD or MASH) (6.1%) and PCOS (5.5%).
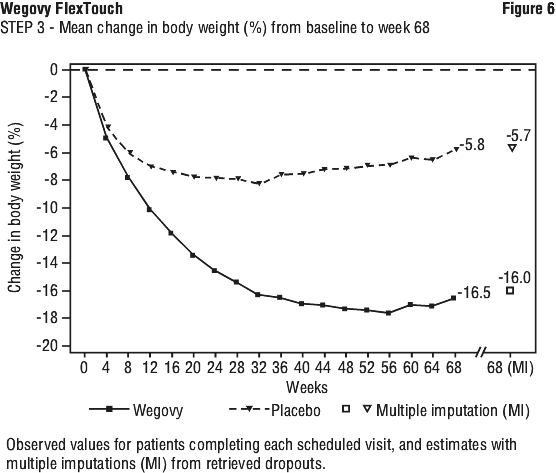
Treatment with Wegovy and IBT for 68 weeks resulted in superior and clinically meaningful reduction in body weight compared to placebo (see Table 7 and Figure 6). Among patients with pre-diabetes at baseline, 89.5% and 55.0% of patients achieved normo-glycaemic status at end of treatment with Wegovy and placebo, respectively.
At the beginning of the run-in period (week 0), patients had a mean BMI of 38.4 kg/m2, a mean body weight of 107.2 kg and a mean age of 46 years. At baseline (week 20), patients had a mean BMI of 34.4 kg/m2 and a mean body weight of 96.1 kg. There were 83.7% Caucasian/White, 13.0% Black/African American, and 2.4% Asian. A total of 7.8% were Hispanic or Latino. The majority of patients had at least one weight-related comorbidity. These included, but were not limited to, prediabetes (46.8%), hypertension (37.1%), dyslipidaemia (35.9%), knee or hip osteoarthritis (13.3%), obstructive sleep apnoea (11.7%), asthma/COPD (11.5%), liver disease (MASLD and MASH) (7.3%) and PCOS (3.9%).
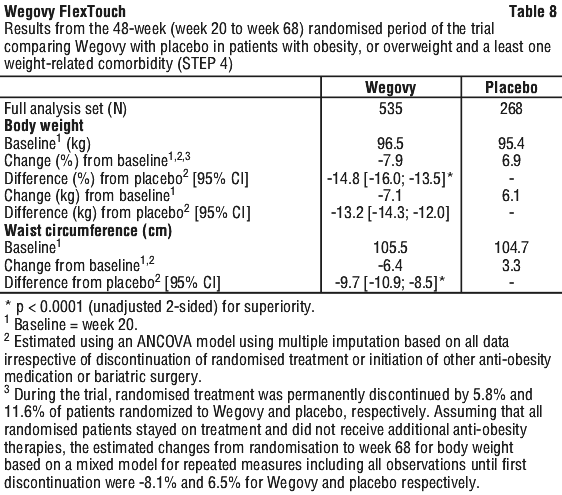
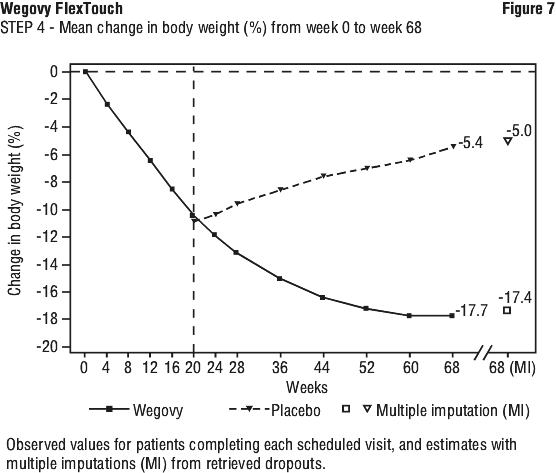
Patients who had reached the maintenance dose of Wegovy (2.4 mg) at week 20 (baseline) and continued treatment with Wegovy for 48 weeks continued losing weight and had a superior and clinically meaningful reduction in body weight compared to those switched to placebo (see Table 8 and Figure 7). On the other hand, in patients switching to placebo at week 20 (baseline), body weight increased steadily from week 20 to week 68. However, the observed mean body weight was lower at week 68 than at start of the run-in period (week 0) (see Figure 7). Patients treated with Wegovy from week 0 (run-in) to week 68 (end of treatment) achieved a mean change in body weight of 17.4%, with weight loss ≥ 5% achieved by 87.8%, ≥ 10% achieved by 78.0%, ≥ 15% achieved by 62.2% and ≥ 20% achieved by 38.6% of these patients.
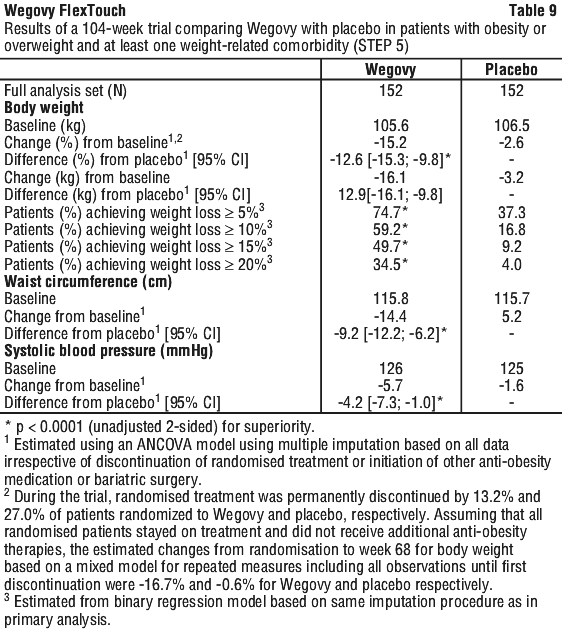
At baseline, patients had a mean BMI of 38.5 kg/m2, a mean body weight of 106.0 kg. The mean age was 47 years. There were 93.1% Caucasian/White, 3.9% Black/African American, and 0.7% Asian. A total of 12.8% were Hispanic or Latino. The majority of patients had at least one weight-related comorbidity. These included, but were not limited to, hypertension (38.8%), dyslipidaemia (35.2%), obstructive sleep apnoea (16.8%) and knee osteoarthritis (15.1%).
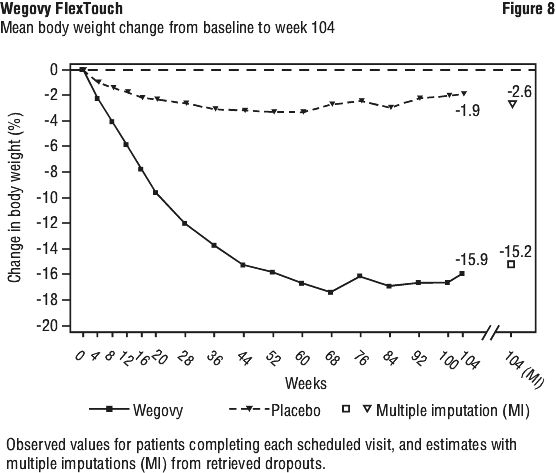
Treatment with Wegovy for 104 weeks resulted in a superior and clinically meaningful reduction in body weight compared to placebo (see Table 9 and Figure 8). Mean body weight decreased from baseline through to week 68 with Wegovy after which a plateau was reached. With placebo, mean body weight decreased less, and a plateau was reached after approximately 20 weeks of treatment. A higher proportion of patients achieved ≥ 5%, ≥ 10%, ≥ 15% and ≥ 20% weight loss with Wegovy compared with placebo. Among patients with pre-diabetes at baseline, 80% and 37% achieved a normo-glycaemic status at end of treatment with Wegovy and placebo, respectively.
At baseline, patients had a mean BMI of 37.5 kg/m2, a mean body weight of 104.5 kg. The mean age was 49 years. There were 73.7% Caucasian/White, 18.9% Black/African American, and 3.8% Asian. A total of 11.5% were Hispanic or Latino. The majority of patients had at least one weight-related comorbidity. These included, but were not limited to, dyslipidaemia (47.6%), hypertension (42.0%), knee osteoarthritis (18.3%) and obstructive sleep apnoea (18.0%).
Treatment with Wegovy once weekly for 68 weeks resulted in superior and clinically meaningful reduction in body weight compared to liraglutide. Mean body weight decreased from baseline through to week 68 with Wegovy, with liraglutide, mean body weight decreased less, and with placebo less yet. 37.4% of the patients treated with semaglutide lost ≥ 20%, compared to 7.0% treated with liraglutide. Table 10 shows the results of the confirmatory endpoints ≥ 10%, ≥ 15% and ≥ 20% weight loss.

There were 67.5% Caucasian/White, 0.6% Black/African American, and 27.0% Asian. A total of 18.3% were Hispanic or Latino. The mean BMI was 34.6 kg/m2, 6.6% had BMI < 25, 72.8% had BMI ≥ 30 and 55.9% had type 2 diabetes.
At week 72, Wegovy was superior to placebo in inducing resolution of steatohepatitis with no worsening of liver fibrosis and improvement in liver fibrosis with no worsening of steatohepatitis (primary endpoints). As for the confirmatory secondary endpoints, Wegovy was superior to placebo in inducing resolution of steatohepatitis with improvement in liver fibrosis. Treatment with Wegovy resulted in a greater and sustained weight loss. These treatment effects were clinically meaningful. Wegovy did not meet the pre-specified significance level for the confirmatory secondary endpoint SF-36 Bodily pain. See Table 11, Figure 9, Figure 10 and Figure 11.
Treatment with Wegovy also resulted in improvements in ALT, AST, liver stiffness assessed by TE, ELF score, Pro-C3 and FAST score compared with placebo at week 72 (see Table 12). These treatment effects were clinically meaningful. Wegovy increased the levels of high-density lipoprotein (HDL) cholesterol and decreased the levels of total cholesterol and triglycerides compared with placebo at week 72 (see Table 12).
Wegovy demonstrated efficacy in improving liver histology regardless of age, sex, race and ethnicity, as well as baseline fibrosis stage, hepatic function, BMI (≥ 25 kg/m2 or ≥ 23 kg/m2 for Asian populations), presence of type 2 diabetes and renal function.
Body weight. Across all weight management trials, treatment with Wegovy demonstrated clinically meaningful and sustained weight loss for up to 104 weeks compared to placebo in patients with obesity or overweight. In the weight management phase 3a trials, the reduction in body weight was shown to occur irrespective of the presence of gastrointestinal symptoms such as nausea, vomiting or diarrhoea. In ESSENCE, treatment with Wegovy resulted in superior, clinically meaningful and sustained weight loss compared with placebo in patients with MASH and F2 or F3.
Cardiovascular risk factors. Wegovy reduced waist circumference, systolic blood pressure and CRP, and improved lipid profile compared with placebo in the weight management phase 3a trials. Similar results were observed for waist circumference, systolic blood pressure and CRP in the ESSENCE trial after 72 weeks of treatment with semaglutide in patients with MASH and F2 or F3.
Lipids. Wegovy improved total cholesterol by -0.9% to -5.9%, HDL-cholesterol by 0.3% to 3.8% LDL cholesterol by 0.4% to -7.1%, and triglycerides by -17.8% to -15.8%, after 68 weeks of treatment in the weight management phase 3a trials.
Blood pressure. Wegovy reduced systolic and diastolic blood pressure by 3.4 mmHg to 5.1 mmHg and 0.7 mmHg to 2.4 mmHg respectively after 68 weeks of treatment in the weight management phase 3a trials.
Waist circumference. Wegovy reduced waist circumference by 4.9 cm to 9.4 cm after 68 weeks of treatment in the weight management phase 3a trials.
C-reactive protein (CRP). Wegovy reduced CRP (marker of systemic inflammation) by 38.7% to 47.6% after 68 weeks of treatment in the weight management phase 3a trials.
Cardiovascular outcomes. SELECT: cardiovascular outcome trial in patients with overweight or obesity. SELECT was a randomised, double-blind, placebo-controlled, event driven trial which included 17,604 patients with established cardiovascular disease (67.6% with prior myocardial infarction only, 17.8% with prior stroke only and 4.4% with peripheral arterial disease only; 8.2% with 2 or more prior CV events) and BMI ≥ 27 kg/m2. Patients with history of type 1 and type 2 diabetes were excluded. The median time in trial was 41.8 months. The study population consisted of 27.7% female and 72.3% male, with a mean age of 61.6 years, including 38.2% patients ≥ 65 years (n = 6,728) and 7.8% patients ≥ 75 years (n = 1,366). The mean BMI was 33.3 kg/m2 and mean body weight was 96.7 kg.
Patients were randomised to either semaglutide 2.4 mg (n = 8,803) or placebo (n = 8,801) in addition to standard-of-care. At baseline, 92.0% of patients were receiving cardiovascular medication (70.2% beta blockers, 45.0% angiotensin-converting enzyme (ACE) inhibitors, 29.5% angiotensin receptor blockers and 26.9% calcium-channel blockers), 90.1% lipid lowering agents (primarily statins 87.6%) and 86.2% anti-platelet agents.
At baseline most patients had cardiovascular related comorbidities including 66.5% with HbA1c ≥ 5.7% indicative of prediabetes, 24.3% with chronic heart failure, 81.8% with hypertension, 46.8% with inflammation (hsCRP ≥ 2 mg/L) as well as patients with mild (48.7%), moderate (10.4%) or severe (0.4%) renal impairment.
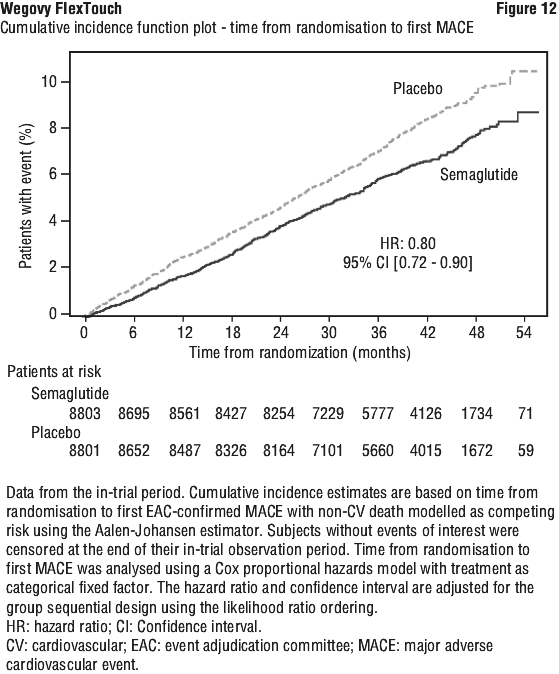
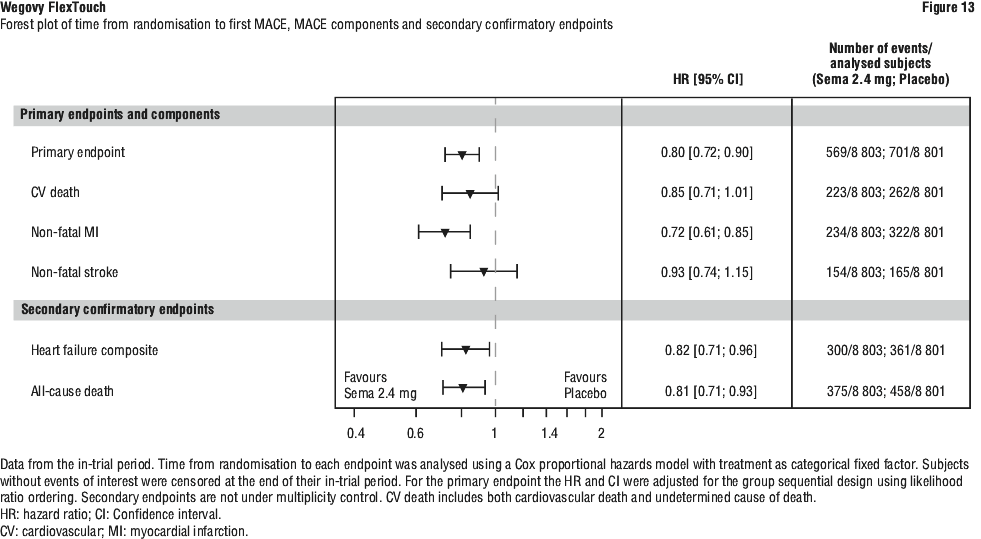
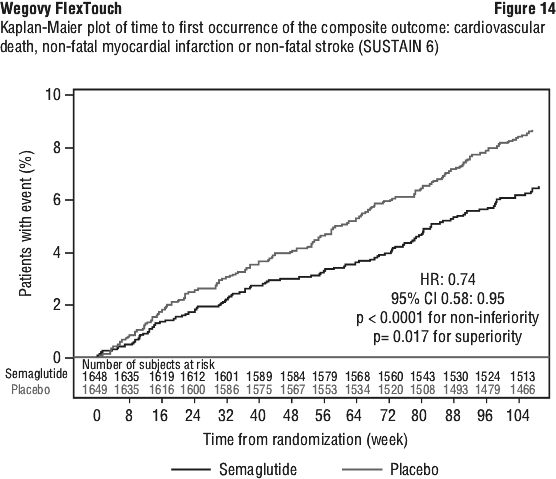
For the primary MACE endpoint, the superiority of semaglutide 2.4 mg to placebo was confirmed demonstrated with a hazard ratio 0.80 [0.72; 0.90] [95% CI], corresponding to a relative risk reduction in MACE of 20% (see Figure 12). The primary MACE endpoint results showed statistical significance for one out of the three components, namely non-fatal MI, but not for CV death or non fatal stroke (see Figure 13).
Each component contributed to the reduction of MACE (Figure 13). The cardiovascular risk reduction appeared largely independent of weight loss. The treatment effect was consistent across all subgroups defined by age, sex, race, ethnicity, region, prior CV disease (MI, stroke, PAD, or any combination thereof), BMI, chronic heart failure status, HbA1c (< 5.7% or ≥ 5.7%), and renal function (eGFR < 60 mL/min/1.73 m2 or ≥ 60 mL/min/1.73 m2).
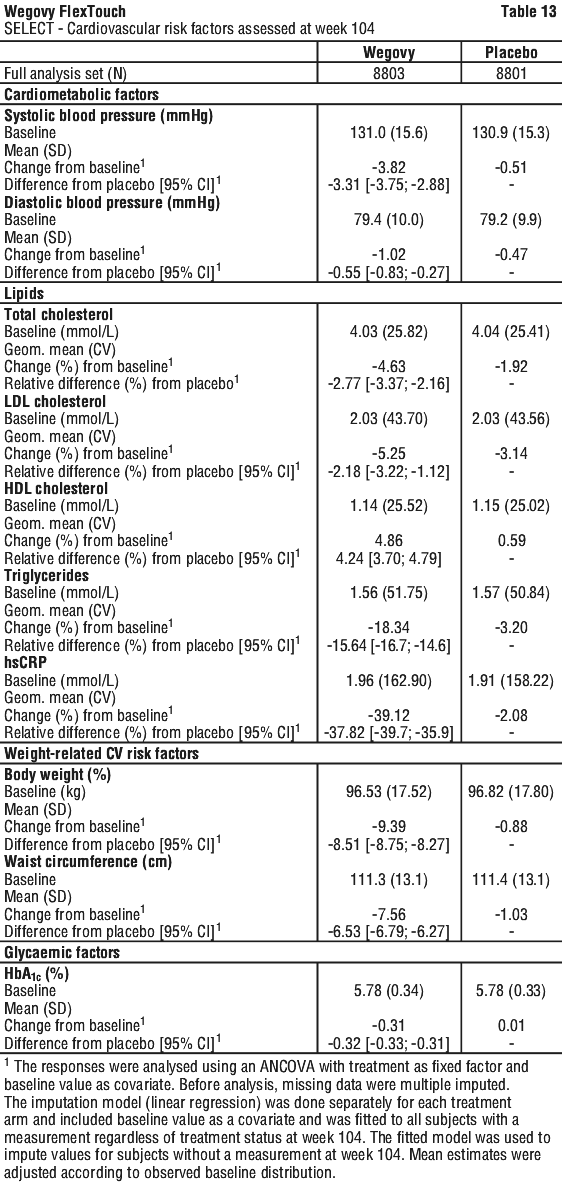
SUSTAIN 6: cardiovascular outcome trial in patients with type 2 diabetes unrelated to weight loss. In the SUSTAIN 6 trial for Ozempic (semaglutide) for a type 2 diabetes indication unrelated to weight loss, 3,297 patients with type 2 diabetes and at high risk of cardiovascular events were randomised to semaglutide s.c. 0.5 mg or 1 mg once-weekly or placebo in addition to standard-of-care. The treatment duration was 104 weeks. The mean age was 65 years and the mean BMI was 33 kg/m2.
The primary endpoint was the time from randomisation to first occurrence of a major adverse cardiovascular event (MACE): cardiovascular death, non-fatal myocardial infarction or non-fatal stroke. The secondary endpoint was time from randomisation to first occurrence of an expanded composite cardiovascular outcome, defined as MACE, revascularisation (coronary and peripheral), unstable angina requiring hospitalisation or hospitalisation for heart failure. The total number of the primary component MACE endpoint was 254, including 108 (6.6%) with semaglutide and 146 (8.9%) with placebo.
Treatment with semaglutide reduced the rate of MACE vs. placebo with a risk reduction of 26%, HR 0.74, [0.58, 0.95] [95% CI]. This was mainly driven by a significant (39%) decrease in the rate of nonfatal stroke and a non-significant (26%) decrease in non-fatal myocardial infarction with no difference in cardiovascular death (see Figure 14).
Wegovy improved glycaemic control through sustained reduction of HbA1c and fasting plasma glucose (FPG) levels. Reduction in HbA1c was 1.2% and reduction in FPG was 2.0 mmol/L in STEP 2.
In STEP 2, 78.5% of patients with type 2 diabetes treated with Wegovy achieved an HbA1c < 7% compared to 26.5% with placebo. A total of 67.5% of the patients treated with Wegovy achieved an HbA1c ≤ 6.5% compared to 15.5% with placebo.
In STEP 1, STEP 3 and STEP 5, among those patients with pre-diabetes at baseline, more Wegovy-treated patients had achieved normo-glycaemic status by the end of the trials (week 68 or week 104) compared to placebo-treated patients (STEP 1: 84.1% vs. 47.8%; STEP 3: 89.5% vs. 55.0%; STEP 5: 80% vs. 37%).
At baseline, patients had a mean BMI of 37.0 kg/m2 and a mean body weight of 107.5 kg. The mean age of patients was 15.4 years. There were 37.8% males, 79.1% Caucasian/White, 8.0% Black/African American and 2.0% Asian. A total of 10.9% were Hispanic or Latino.
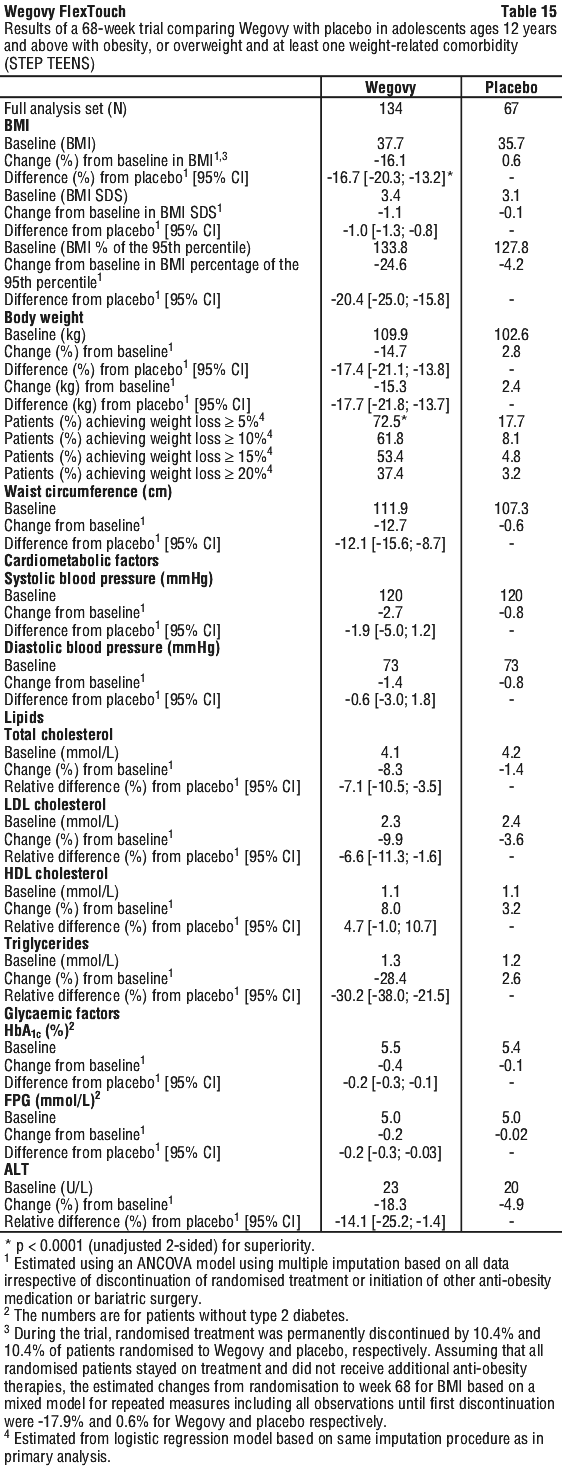
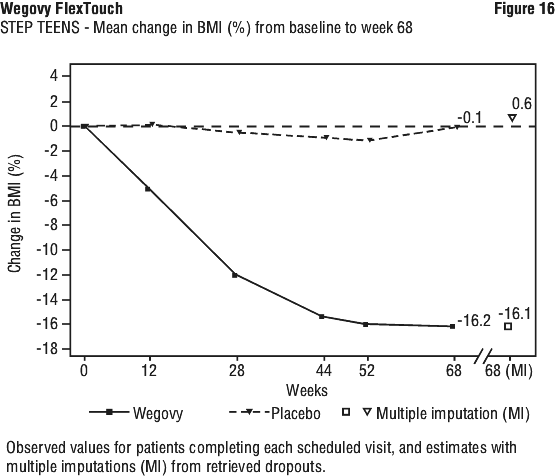
At end of treatment (week 68), the improvement in BMI with Wegovy was superior and clinically meaningful compared with placebo (see Table 15 and Figure 16). Furthermore, a higher proportion of patients achieved ≥ 5%, ≥ 10%, ≥ 15% and ≥ 20% weight loss with Wegovy compared with placebo (see Table 16). Also, improvement in weight category was seen for a larger proportion of patients with Wegovy (71.8%) compared to placebo (21.0%). Improvement was defined as changing from a higher to a lower obesity class, or to overweight or normal weight. At end of treatment, a higher proportion of patients with Wegovy (53.7%) changed at least 2 categories compared to placebo (3.8%).
Greater reductions in absolute BMI, BMI standard deviation score (SDS), BMI percentage of the 95th percentile (sex and age-specific growth charts) and body weight were observed with Wegovy compared to placebo at week 68.
Beneficial effects of Wegovy vs. placebo were also demonstrated in STEP 1 and 2 in all additional scores on IWQOL-Lite-CT (Physical, Psychosocial, and Total). In STEP 3, beneficial effects of Wegovy vs. placebo were demonstrated for the following scores in SF-36: General Health, Vitality, Social Functioning, Role-Emotional and Mental Component Summary.
5.2 Pharmacokinetic Properties
Compared to native GLP-1, semaglutide has a prolonged half-life of around 1 week making it suitable for once weekly s.c. administration. The principal mechanism of protraction is albumin binding, which results in decreased renal clearance and protection from metabolic degradation. Furthermore, semaglutide is stabilised against degradation by the DPP-4 enzyme.
Absorption. The average semaglutide steady state concentration following s.c. administration of Wegovy was approximately 75 nanomol/L in patients with either overweight (BMI ≥ 27 kg/m2 to < 30 kg/m2) or obesity (BMI ≥ 30 kg/m2) and approximately 80 nanomol/L in patients with MASH and F2 or F3 based on data from ESSENCE. The steady state exposure of semaglutide increased proportionally with doses up to 2.4 mg once weekly.
Similar exposure was achieved with s.c. administration of semaglutide in the abdomen, thigh, or upper arm. The absolute bioavailability of semaglutide was 89%.
Distribution. The mean volume of distribution of semaglutide following s.c. administration in patients with overweight or obesity is approximately 12.4 L and approximately 13.7 L in patients with MASH and F2 or F3. Semaglutide is extensively bound to plasma albumin (> 99%).
Metabolism. Semaglutide is metabolised through proteolytic cleavage of the peptide backbone and sequential beta-oxidation of the fatty acid side chain.
Excretion. Semaglutide has pharmacokinetic properties compatible with once-weekly administration, with an elimination half-life of approximately 1 week.
The primary excretion routes of semaglutide-related material are via the urine and faeces. Approximately 3% of the dose was excreted in the urine as intact semaglutide.
Clearance of semaglutide in patient with overweight (BMI ≥ 27 kg/m2 to < 30 kg/m2) or obesity (BMI ≥ 30 kg/m2) and in patients with MASH and F2 or F3 was approximately 0.05 L/h. With an elimination half-life of approximately 1 week, semaglutide can be present in the circulation for approximately 7 weeks after the last dose of 2.4 mg.
Special populations. Based on a population pharmacokinetic analysis across the weight management trials, age, sex, race, and ethnicity, and renal impairment do not have a clinically meaningful effect on the pharmacokinetics of semaglutide. The exposure of semaglutide decreases with an increase in body weight. However, semaglutide 2.4 mg provide adequate systemic exposure over the body weight range of 54.4-245.6 kg evaluated in the clinical trials. In patients with overweight or obesity, the effects of intrinsic factors on the pharmacokinetics of semaglutide are shown in Figure 17. Similar results have been shown in patients with MASH (body weight range 42.7-206.0 kg). Fibrosis stage (F1 to F4c) did not impact semaglutide exposure in patients with MASH.
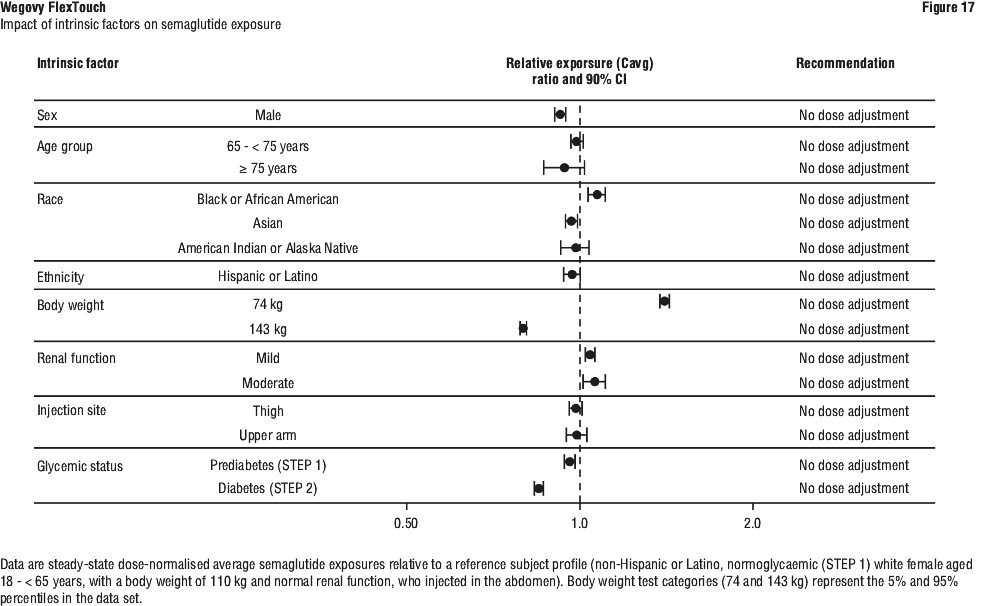
Sex. Gender had no effect on the pharmacokinetics of semaglutide.
Race. Race (White, Black or African-American, Asian) had no effect on the pharmacokinetics of semaglutide.
Ethnicity. Ethnicity (Hispanic or Latino) had no effect on the pharmacokinetics of semaglutide.
Body weight. Body weight had an effect on the exposure of semaglutide. Higher body weight was associated with lower exposure. The 2.4 mg weekly dose of semaglutide provided adequate systemic exposures over the body weight range of 54.4-245.6 kg evaluated for exposure response in the clinical trials for weight management and 42.7-206.0 kg for MASH.
Renal impairment. Renal impairment did not impact the pharmacokinetics of semaglutide in a clinically relevant manner. This was shown with a single dose of 0.5 mg semaglutide for patients with different degrees of renal impairment (mild, moderate, severe or subjects in dialysis) compared with subjects with normal renal function. This was also shown for subjects with overweight (BMI ≥ 27 kg/m2 to < 30 kg/m2) or obesity (BMI ≥ 30 kg/m2) and mild to moderate renal impairment based on data from phase 3a trials and for patients with MASH and mild to moderate renal impairment based on population PK analysis.
Hepatic impairment. Hepatic impairment did not have any impact on the exposure of semaglutide. The pharmacokinetics of semaglutide were evaluated in patients with different degrees of hepatic impairment (mild, moderate, severe) compared with subjects with normal hepatic function in a study with a single-dose of 0.5 mg semaglutide.
Liver fibrosis. The stage of liver fibrosis (F1 to F4c) did not have any effect on the exposure of semaglutide based on population PK analysis.
Paediatrics. Semaglutide has not been studied in children below 12 years of age with obesity or overweight. Subcutaneously administered semaglutide has not been studied in paediatric patients with MASH. Pharmacokinetic properties for semaglutide 2.4 mg were assessed in a clinical trial for adolescent patients with obesity or overweight and at least one weight-related comorbidity ages 12 to less than 18 years (124 patients, body weight 61.6-211.9 kg). The semaglutide exposure in adolescents was similar to that in adults with obesity or overweight.
5.3 Preclinical Safety Data
Genotoxicity. Semaglutide was not mutagenic in the bacterial reverse mutation assay, and was not clastogenic in vitro (cytogenetic assay in human lymphocytes), or in vivo (rat bone marrow micronucleus test).
Carcinogenicity. Non-lethal thyroid C-cell tumours observed in rodents are a class effect for GLP-1 receptor agonists. In 2-year carcinogenicity studies in rats and mice, semaglutide caused thyroid C-cell tumours at clinically relevant exposures (at ≥ 1.2x the clinical AUC in mice [based on the plasma AUC at the maximum recommended human dose of 2.4 mg/week] and subclinical exposures in rats; a no effect level was not established in either species). No other treatment-related tumours were observed. The rodent C-cell tumors are caused by a non-genotoxic, specific GLP-1 receptor mediated mechanism to which rodents are particularly sensitive. The relevance for humans is considered to be low, but cannot be excluded.
Juvenile toxicity. In juvenile rats, semaglutide caused delayed sexual maturation in both males and females. These delays had no impact upon fertility and reproductive capacity of either sex, or on the ability of the females to maintain pregnancy.
6 Pharmaceutical Particulars
6.1 List of Excipients
Single-dose pen and pre-filled syringe. Dibasic sodium phosphate dihydrate, sodium chloride, hydrochloric acid, sodium hydroxide, water for injections.
FlexTouch pen. Dibasic sodium phosphate dihydrate, propylene glycol, phenol, hydrochloric acid, sodium hydroxide, water for injections.
6.2 Incompatibilities
Substances added to Wegovy may cause degradation of semaglutide. Wegovy must not be mixed with other medicinal products, e.g. infusion fluids.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Single-dose pen. Store in a refrigerator (2°C to 8°C). Keep away from the cooling element.
May be stored unrefrigerated for up to 28 days at a temperature not above 30°C.
Always store the pen in the original carton in order to protect from light.
Do not freeze Wegovy and do not use Wegovy if it has been frozen.
Wegovy should be protected from excessive heat.
Wegovy should not be used if it does not appear clear and colourless.
The Wegovy pen is for single use in one patient only.
After use. Discard Wegovy single-dose pen after use.
FlexTouch pen. Do not freeze Wegovy FlexTouch and do not use Wegovy FlexTouch if it has been frozen.
Keep the pen cap on in order to protect from light.
Wegovy FlexTouch should be protected from excessive heat.
Wegovy FlexTouch should not be used if it does not appear clear and colourless.
The Wegovy FlexTouch pen is for use by one person only.
Before first use. Store in a refrigerator (2°C to 8°C). Keep away from the cooling element.
After first use. May be stored unrefrigerated for up to 42 days at a temperature below 30°C.
Always remove the injection needle after each injection and store the pen without a needle attached. This may prevent blocked needles, contamination, infection, leakage of solution and inaccurate dosing.
Pre-filled syringe. Store in a refrigerator (2°C - 8°C). Keep away from the cooling element.
May be stored unrefrigerated for up to 28 days at a temperature not above 30°C.
Discard the syringe if it has been out of the refrigerator for more than 28 days.
Always store the syringe in the original carton in order to protect from light.
Do not freeze Wegovy and do not use Wegovy if it has been frozen.
Wegovy should be protected from excessive heat.
After use. Discard Wegovy pre-filled syringe after use.
6.5 Nature and Contents of Container
Single-dose pen. The primary packaging contains a 1 mL glass syringe (Type I glass) with attached stainless-steel needle, rigid needle shield (Type II/polyisoprene) and a rubber plunger (Type I/chlorobutyl).
There are five variants of the pre-filled pen for Wegovy:
Wegovy 0.25 mg/dose is provided in a pre-filled, fixed dose, single use disposable pen, which contains the drug solution semaglutide 0.5 mg/mL, 0.5 mL in a 1 mL pre-filled syringe. The pen contains one dose. This pen is intended to be used for dose escalation.
Wegovy 0.5 mg/dose is provided in a pre-filled, fixed dose, single use disposable pen, which contains the drug solution semaglutide 1.0 mg/mL, 0.5 mL in a 1 mL pre-filled syringe. The pen contains one dose. This pen is intended to be used for dose escalation.
Wegovy 1 mg/dose is provided in a pre-filled, fixed dose, single use disposable pen, which contains the drug solution semaglutide 2.0 mg/mL, 0.5 mL in a 1 mL pre-filled syringe. The pen contains one dose. This pen is intended to be used for dose escalation.
Wegovy 1.7 mg/dose is provided in a pre-filled, fixed dose, single use disposable pen, which contains the drug solution semaglutide 2.27 mg/mL, 0.75 mL in a 1 mL pre-filled syringe. The pen contains one dose. This pen is intended to be used for dose escalation.
Wegovy 2.4 mg/dose is provided in a pre-filled, fixed dose, single use disposable pen, which contains the drug solution semaglutide 3.2 mg/mL, 0.75 mL in a 1 mL pre-filled syringe. The pen contains one dose. This pen is intended to be used for maintenance treatment.
Pack sizes of*:
Wegovy 0.25 mg/dose. 2 or 4 pre-filled pens.
Wegovy 0.5 mg/dose. 2 or 4 pre-filled pens.
Wegovy 1 mg/dose. 2 or 4 pre-filled pens.
Wegovy 1.7 mg/dose. 2 or 4 pre-filled pens.
Wegovy 2.4 mg/dose. 2 or 4 pre-filled pens.
*Not all pack sizes may be available.
FlexTouch pen. The primary packaging contains a 1.5 mL or 3 mL glass cartridge (Type I glass) closed at the one end with a rubber plunger (Type I/chlorobutyl) and at the other end with an aluminium cap with a rubber disc (Type I/bromobutyl/isoprene) inserted.
The cartridge is assembled into a pre-filled, multi-dose disposable pen made of polypropylene polyoxymethylene, polycarbonate and acrylonitrile butadiene styrene.
There are five strengths of the Wegovy FlexTouch pen:
Wegovy 0.25 mg/dose solution for injection in pre-filled pen delivers 4 doses of 0.25 mg. This pen is intended to be used for dose escalation. The pen contains 1.5 mL solution.
Wegovy 0.5 mg/dose solution for injection in pre-filled pen delivers 4 doses of 0.5 mg. This pen is intended to be used for dose escalation. The pen contains 1.5 mL solution.
Wegovy 1 mg/dose for injection in pre-filled pen delivers 4 doses of 1 mg. This pen is to be used for dose escalation. The pen contains 3 mL solution.
Wegovy 1.7 mg/dose for injection in pre-filled pen delivers 4 doses of 1.7 mg. This pen is to be used for dose escalation. The pen contains 3.0 mL solution.
Wegovy 2.4 mg/dose solution for injection in pre-filled pen delivers 4 doses of 2.4 mg. This pen is intended to be used for maintenance treatment. The pen contains 3.0 mL solution.
NovoFine Plus needles are included in the Wegovy FlexTouch package.
Wegovy can be administered with 30G, 31G, and 32G disposable needles up to a length of 8 mm.
Pack sizes of:
Wegovy FlexTouch 0.25 mg/dose. 1 pre-filled pen and 4 disposable NovoFine Plus needles.
Wegovy FlexTouch 0.5 mg/dose. 1 pre-filled pen and 4 disposable NovoFine Plus needles.
Wegovy FlexTouch 1 mg/dose. 1 pre-filled pen and 4 disposable NovoFine Plus needles.
Wegovy FlexTouch 1.7 mg/dose. 1 pre-filled pen and 4 disposable NovoFine Plus needles.
Wegovy FlexTouch 2.4 mg/dose. 1 pre-filled pen and 4 disposable NovoFine Plus needles.
Pre-filled syringe. The primary packaging contains a 1 mL glass syringe (type I glass) with attached stainless-steel needle, rigid needle shield (type II/polyisoprene - inner shield and polypropylene - outer shield) and a rubber plunger (type I/chlorobutyl). The pre-filled syringe is assembled with a polypropylene plunger rod and a polypropylene finger grip.
There are five variants of the pre-filled syringe for Wegovy:
Wegovy 0.25 mg/dose solution for injection in pre-filled syringe delivers 1 dose of 0.25 mg. This syringe is intended to be used for dose escalation. The syringe contains 0.5 mL solution.
Wegovy 0.5 mg/dose solution for injection in pre-filled syringe delivers 1 dose of 0.5 mg. This syringe is intended to be used for dose escalation. The syringe contains 0.5 mL solution.
Wegovy 1 mg/dose solution for injection in pre-filled syringe delivers 1 dose of 1 mg. This syringe is intended to be used for dose escalation. The syringe contains 0.5 mL solution.
Wegovy 1.7 mg/dose solution for injection in pre-filled syringe delivers 1 dose of 1.7 mg. This syringe is intended to be used for dose escalation. The syringe contains 0.75 mL solution.
Wegovy 2.4 mg/dose solution for injection in pre-filled syringe delivers 1 dose of 2.4 mg. This syringe is intended to be used for maintenance treatment. The syringe contains 0.75 mL solution.
Pack sizes of:
Wegovy 0.25 mg/dose. 4 pre-filled syringes.
Wegovy 0.5 mg/dose. 4 pre-filled syringes.
Wegovy 1 mg/dose. 4 pre-filled syringes.
Wegovy 1.7 mg/dose. 4 pre-filled syringes.
Wegovy 2.4 mg/dose. 4 pre-filled syringes.
6.6 Special Precautions for Disposal
Single-dose pen. The patient should be advised to discard the pre-filled pen after each injection in accordance with local requirements.
FlexTouch pen. The patient should be advised to discard the injection needle in accordance with local requirements after each injection and store the Wegovy FlexTouch pen without an injection needle attached.
After the final dose of Wegovy FlexTouch the pen should be discarded in accordance with local requirements.
After having injected the 4 doses, there might still be solution left in the pen despite having administered correctly. Any solution left is insufficient for a dose and the pen should be disposed of.
Pre-filled syringe. The patient should be advised to discard the pre-filled syringe after injection in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. Molecular formula: C187H291N45O59. Molecular weight: 4113.6 daltons.
Semaglutide is an Aib8, Arg34-GLP-1(7-37) analogue substituted on the ε-amino group of the lysine residue in position 26 with an (S)-22,40-dicarboxy-10,19,24-trioxo-3,6,12,15-tetraoxa-9,18,23-triazatetracontan-1-oyl side chain. The side chain consists of two 8-amino-3,6-dioxaoctanoic acid (ADO) spacers, one γ-glutamic acid (Glu) spacer, and a fatty diacid (1,18-octadecanedioic acid). Semaglutide is produced using recombinant DNA technology in yeast (Saccharomyces cerevisiae) and chemical modification.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
01 September 2022
Date of Revision
30 March 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.