Xylocaine Special Adhesive Ointment
Brand Information
| Brand name | Xylocaine Special Adhesive Ointment |
| Active ingredient | Lidocaine (lignocaine) |
| Schedule | S2 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Xylocaine Special Adhesive Ointment.
Summary CMI
XYLOCAINE SPECIAL ADHESIVE ointment
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using XYLOCAINE SPECIAL ADHESIVE?
XYLOCAINE SPECIAL ADHESIVE contains the active ingredient lidocaine. XYLOCAINE SPECIAL ADHESIVE is used in dentistry to prevent pain during scaling or injection and when fitting dentures. It is also used to relieve pain during the removal of baby teeth.
For more information, see Section 1. Why am I using XYLOCAINE SPECIAL ADHESIVE? in the full CMI.
2. What should I know before I use XYLOCAINE SPECIAL ADHESIVE?
Do not use if you have ever had an allergic reaction to lidocaine, other local anaesthetics or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, are pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use XYLOCAINE SPECIAL ADHESIVE? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with XYLOCAINE SPECIAL ADHESIVE and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use XYLOCAINE SPECIAL ADHESIVE?
- Your doctor, pharmacist or dentist will tell you how much ointment you should use. The dose depends on the size of the area and should be applied in a thin layer for adequate control of symptoms.
- More instructions can be found in Section 4. How do I use XYLOCAINE SPECIAL ADHESIVE? in the full CMI.
5. What should I know while using XYLOCAINE SPECIAL ADHESIVE?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using XYLOCAINE SPECIAL ADHESIVE? in the full CMI.
6. Are there any side effects?
Tell your doctor, pharmacist or dentist as soon as possible if you do not feel well while you are using XYLOCAINE SPECIAL ADHESIVE.
Some of the common side effects include skin rash or irritation, light headedness, dizziness, nervousness, blurred vision and drowsiness.
Some of the serious side effects include wheezing or difficulty breathing, chest pain, severe rash or itching and increased sweating.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
XYLOCAINE SPECIAL ADHESIVE ointment
Active ingredient: lidocaine
Consumer Medicine Information (CMI)
This leaflet provides important information about using XYLOCAINE SPECIAL ADHESIVE.
You should speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using XYLOCAINE SPECIAL ADHESIVE.
Where to find information in this leaflet:
1. Why am I using XYLOCAINE SPECIAL ADHESIVE?
2. What should I know before I use XYLOCAINE SPECIAL ADHESIVE?
3. What if I am taking other medicines?
4. How do I use XYLOCAINE SPECIAL ADHESIVE?
5. What should I know while using XYLOCAINE SPECIAL ADHESIVE?
6. Are there any side effects?
7. Product details
1. Why am I using XYLOCAINE SPECIAL ADHESIVE?
XYLOCAINE SPECIAL ADHESIVE contains the active ingredient lidocaine. It belongs to a group of medicines called local anaesthetics. It works by making nerves unable to pass messages to the brain.
XYLOCAINE SPECIAL ADHESIVE is used in dentistry to prevent pain during scaling or injection and when fitting dentures or orthodontic appliances. It is also used to relieve pain during the removal of baby teeth.
Ask your doctor or dentist if you have any questions about why XYLOCAINE SPECIAL ADHESIVE has been prescribed to you.
They may have prescribed it for another purpose.
This medicine is available without a doctor's prescription. It can be purchased at a pharmacy.
XYLOCAINE SPECIAL ADHESIVE is not addictive.
2. What should I know before I use XYLOCAINE SPECIAL ADHESIVE?
Warnings
Do not use XYLOCAINE SPECIAL ADHESIVE if:
- you are allergic to lidocaine, other local anaesthestics of the amide type or any of the ingredients listed at the end of this leaflet including other local anaesthetics e.g. bupivacaine or any other substances.
- Some symptoms of an allergic reaction may include a skin rash, hayfever, difficulty breathing or feeling faint. - the expiry date printed on the pack has passed
- the packaging is torn or shows signs of tampering.
Check with your doctor if you:
- have or have had any of these medical conditions:
- open wounds or infection in the mouth
- epilepsy
- heart, liver or kidney problems.
It may not be safe for you to use XYLOCAINE SPECIAL ADHESIVE if you have any of these conditions.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not use XYLOCAINE SPECIAL ADHESIVE if you are pregnant or breastfeeding unless your doctor or dentist says so. Ask them about the risks and benefits involved.
Lidocaine has been used widely during pregnancy and there have been no reports of any ill effects on the baby. Your baby can take in very small amounts of this medicine from breast milk if you are breastfeeding. It is unlikely that the amount available to the baby will do any harm.
3. What if I am taking other medicines?
Tell your doctor or dentist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and XYLOCAINE SPECIAL ADHESIVE may interfere with each other. These include:
- medicines to treat irregular heartbeats.
- cimetidine
- phenytoin and other anti-epileptic drugs such as phenobarbitone, primidone and carbamazepine.
Your doctor or dentist can tell you what to do if you are taking any of these medicines.
4. How do I use XYLOCAINE SPECIAL ADHESIVE?
How much to use
Your doctor, pharmacist or dentist will tell you how much ointment you should use. The dose depends on the size of the area, your age and medical condition and should be applied in a thin layer for adequate control of symptoms.
Do not exceed the recommended dose or use this medicine for long periods except on the advice or your health professional.
ADULTS: the maximum single dose is 2.5 g. This is roughly equivalent to squeezing a 7.5 cm length of ointment from the tube. No more than 8.5-10 g of ointment should be used in any 24 hour period.
CHILDREN (< 12 YEARS): Do not use more than 0.05 g ointment/kg bodyweight as a single dose. No more than three doses should be used in any 24 hour period.
If you use too much XYLOCAINE SPECIAL ADHESIVE
If you use too much XYLOCAINE SPECIAL ADHESIVE or you use it too often, you may experience side effects such as feeling nervous, dizzy and develop blurred vision and tremor.
If you think that you have used excessive XYLOCAINE SPECIAL ADHESIVE, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
If you accidentally swallow XYLOCAINE SPECIAL ADHESIVE it is unlikely to cause any problems due to the small amount of lidocaine it contains. However, you may have a numb tongue and throat. This will wear off without treatment.
Do not eat or drink anything until your tongue feels normal again.
5. What should I know while using XYLOCAINE SPECIAL ADHESIVE?
Things you should do
See your doctor or dentist if pain and discomfort continue.
Things you should not do
- Do not eat or drink anything for at least 1 hour after using XYLOCAINE SPECIAL ADHESIVE in the mouth or throat area.
You may swallow your food down the wrong way or burn or bite your mouth. - Do not use XYLOCAINE SPECIAL ADHESIVE on open wounds or infected areas.
- Do not get XYLOCAINE SPECIAL ADHESIVE in your eyes.
If any of the ointment does go in your eye, rinse immediately with lots of water for at least 15 minutes and call your doctor.
Driving or using machines
Be careful driving or operating machinery until you know how XYLOCAINE SPECIAL ADHESIVE affects you.
It may have a very mild effect on alertness and co-ordination. You may become drowsy, and your reflexes may be slow.
Looking after your medicine
Keep your XYLOCAINE SPECIAL ADHESIVE ointment in a cool place where the temperature stays below 25°C.
Do not store it or any other medicine in the bathroom or near a sink.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
Do not leave it in the car on hot days.
When to discard your medicine
Ask your pharmacist what to do with any ointment you have left over.
6. Are there any side effects?
Tell your doctor, pharmacist or dentist as soon as possible if you do not feel well while you are using XYLOCAINE SPECIAL ADHESIVE.
XYLOCAINE SPECIAL ADHESIVE will help relieve pain and discomfort in most people, but it may have unwanted side effects in a few people.
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your health professional if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor, pharmacist or dentist if you have any of these mild side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. You may need medical attention. Serious side effects are rare. |
Tell your health professional if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available without a doctor's prescription. It can be purchased at a pharmacy.
What XYLOCAINE SPECIAL ADHESIVE contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Do not use this medicine if you are allergic to any of these ingredients.
What XYLOCAINE SPECIAL ADHESIVE looks like
XYLOCAINE SPECIAL ADHESIVE is a white to slightly yellow flavoured ointment. Available in a 15 g tube.
Australian Registration number: AUST R 12006
Who distributes XYLOCAINE SPECIAL ADHESIVE
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australia
This leaflet was revised in July 2025.
Brand Information
| Brand name | Xylocaine Special Adhesive Ointment |
| Active ingredient | Lidocaine (lignocaine) |
| Schedule | S2 |
MIMS Revision Date: 01 December 2024
1 Name of Medicine
Lidocaine.
2 Qualitative and Quantitative Composition
Each gram of Xylocaine Special Adhesive contains lidocaine base 100 mg as the active ingredient.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Xylocaine Special Adhesive is formulated to adhere to moist oral mucosa and provide topical anaesthesia. It is a white to slightly yellow ointment.
4 Clinical Particulars
4.1 Therapeutic Indications
Surface anaesthesia of the gums prior to injection, before scaling and in conjunction with the fitting of new dentures or orthodontic appliances.
Temporary relief of pain associated with removal of deciduous teeth.
4.2 Dose and Method of Administration
As with any local anaesthetic, reactions and complications are best averted by employing the minimal effective dosage. Debilitated or elderly patients and children should be given doses commensurate with their age and physical condition.
The dose of topical lidocaine should be taken into consideration in estimating the total dose of lidocaine if parenteral lidocaine is to be administered concomitantly.
The following dosage recommendations should be regarded as a guide. The clinician's experience and knowledge of the patient's physical status are of importance in calculating the required dose.
Adults. The maximum single recommended dose of Xylocaine Special Adhesive is 2.5 g, containing 250 mg of lidocaine base (approximately equivalent to 300 mg lidocaine hydrochloride). This is roughly equivalent to squeezing a 7.5 cm length of Special Adhesive from the tube. Not more than 8.5-10 g of the Special Adhesive should be administered in any 24 hour period.
Children. In children less than 12 years of age, the maximum single recommended dose should not exceed 0.05 g ointment/kg bodyweight (corresponding to lidocaine 5 mg/kg bodyweight). Not more than three doses should be administered in a 24 hour period.
The Special Adhesive should be applied in a thin layer for adequate control of symptoms.
In dentistry, apply to previously dried oral mucosa, allow at least 3-5 minutes for anaesthesia to become effective. When inserting new dentures, apply to all denture surfaces contacting mucosa.
4.3 Contraindications
Known history of hypersensitivity to lidocaine or other local anaesthetics of the amide type or to other components of the ointment.
4.4 Special Warnings and Precautions for Use
Warning. Excessive dosage, or short intervals between doses, can result in high levels of lidocaine or its metabolites and serious adverse effects. Patients should be instructed to strictly adhere to the recommended dosage and administration guidelines. The management of serious adverse reactions may require the use of resuscitative equipment, oxygen and other resuscitative drugs.
Patients should not exceed the recommended dose or use Xylocaine Special Adhesive for prolonged periods except on the advice of their physician or dentist.
The lowest dose that results in effective anaesthesia should be used to avoid high plasma levels and serious adverse effects. Tolerance to elevated blood levels varies with the status of the patient.
Dosage reduction. Debilitated, elderly and/or acutely ill patients and children should be given reduced doses commensurate with their age and physical status.
Excessive absorption. Absorption from mucous membranes is relatively high, especially in the bronchial tree. This should be taken into consideration when the Special Adhesive is used in children for treatment of large areas. Because of the possibility of significant systemic absorption, Xylocaine Special Adhesive should be used with caution in patients with traumatised mucosa and/or sepsis in the region of the proposed application.
If the dose or site of administration is likely to result in high blood levels, lidocaine, in common with other local anaesthetics, should be used with caution in patients with epilepsy, impaired cardiac conduction, bradycardia, impaired hepatic function and in severe shock.
Antiarrhythmic drugs class III. Patients treated with antiarrhythmic drugs class III (e.g. amiodarone) should be kept under close surveillance and ECG monitoring considered, since cardiac effects may be additive.
Eating and drinking. The use of topical anaesthetic agents in the oral cavity may interfere with swallowing and thus enhance the danger of aspiration of food or drink. For this reason, food or drink should not be ingested within 60 minutes of using local anaesthetics in the mouth or throat area. Numbness of the tongue or buccal mucosa may increase the danger of biting or heat trauma. Food, chewing gum or hot drinks should not be taken while the mouth or throat area is anaesthetised.
Malignant hyperthermia. Many drugs used during the conduct of anaesthesia are considered potential triggering agents for familial malignant hyperthermia. It has been shown that the use of amide local anaesthetics in malignant hypothermia patients is generally safe, but cases of malignant hyperthermia have occasionally been documented after use.
Sterile instruments. Xylocaine Special Adhesive is not intended for use with sterile instruments.
Use in hepatic impairment. See Section 4.4 Special Warnings and Precautions for Use, Excessive absorption.
Use in the elderly. See Section 4.2 Dose and Method of Administration.
Paediatric use. See Section 4.2 Dose and Method of Administration.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Antiarrhythmic drugs. Lidocaine should be used with caution in patients receiving other local anaesthetics or agents structurally related to local anaesthetics, e.g. antiarrhythmic drugs such as mexiletine, since the toxic effects are additive.
Specific interaction studies with lidocaine and antiarrhythmic drugs class III (e.g. amiodarone) have not been performed, but caution is advised.
Enzyme inducing drugs. Cimetidine has been shown to reduce clearance of IV administered lidocaine. Caution should be taken if administered concurrently with lidocaine.
Phenytoin and other antiepileptic drugs such as phenobarbitone, primidone and carbamazepine appear to enhance the metabolism of lidocaine but the significance of this effect is not known. Phenytoin and lidocaine have additive cardiac depressant effects.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Lidocaine crosses the placental barrier and may be taken up by foetal tissues. When used for surface anaesthesia, lidocaine blood levels after normal doses are low so little drug is available for placental transfer.
There are, however, no adequate and well-controlled studies in pregnant women. Reproduction studies have been performed in rats at doses of 500 mg/kg/day and have revealed no evidence of harm to the foetus caused by lidocaine.
It is reasonable to assume that a large number of pregnant women and women of child-bearing age have used lidocaine. No specific disturbances to the reproduction process have so far been reported.
Lidocaine is not contraindicated in labour and delivery.
Use in lactation. Lidocaine enters breast milk, but in such small quantities that there is generally no risk of affecting the child at therapeutic dose levels.
4.7 Effects on Ability to Drive and Use Machines
Depending on the dose, local anaesthetics may have a very mild effect on mental function and may temporarily impair locomotion and coordination. With the recommended doses of lidocaine ointment adverse effects on the CNS are unlikely.
Xylocaine Special Adhesive is probably porphyrinogenic and should only be prescribed to patients with acute porphyria on strong or urgent indications. Appropriate precautions should be taken for all porphyric patients.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Systemic adverse reactions are rare and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy or reduced tolerance on the part of the patient. Such reactions are systemic in nature and involve the central nervous system and/or the cardiovascular system.
Central nervous system. CNS reactions are excitatory and/or depressant and may be characterised by lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness and, possibly, respiratory arrest. The excitatory reactions may be brief or may not occur at all, in which case the first manifestations of toxicity may be drowsiness, progressing to unconsciousness and respiratory arrest.
Drowsiness following administration of lidocaine is usually an early sign of a high blood level of the drug and may occur as a result of rapid absorption.
Cardiovascular. Cardiovascular reactions are usually depressant and may be characterised by hypotension, myocardial depression, bradycardia and possibly cardiac arrest.
Allergic reactions. Allergic reactions may occur as a result of sensitivity either to the local anaesthetic agent or to other ingredients in the formulation. Allergic reactions as a result of sensitivity to lidocaine are rare (< 0.1%). The detection of sensitivity by skin testing is of doubtful value.
The extremely rare cases of allergy to local anaesthetic preparations have included bronchospasm, chest pain, dyspnoea, pruritus, rash, rhinitis, increased sweating, urticaria, sleepiness, dizziness, paraesthesia, oedema and, in the most severe instances, anaphylactic shock.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Management of local anaesthetic emergencies. The first consideration is prevention, best accomplished by careful and constant monitoring of cardiovascular and respiratory vital signs and the patient's state of consciousness after each local anaesthetic administration. At the first sign of change, oxygen should be administered.
Treatment. If convulsions occur then immediate attention is required for the maintenance of a patent airway and assisted or controlled ventilation with oxygen, via a positive airway pressure delivery system mask. Adequacy of the circulation should then be evaluated, bearing in mind that drugs used to treat convulsions depress the circulation when administered IV. Should convulsions persist despite adequate respiratory support and if the status of the circulation permits, appropriate anticonvulsant medication such as an ultra short acting barbiturate (e.g. thiopental) or a benzodiazepine (e.g. diazepam), may be administered IV. The clinician should be familiar with these anticonvulsant drugs prior to use of local anaesthetics.
Dialysis is of negligible value in the treatment of acute overdosage with lidocaine.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Lidocaine, the active ingredient of Xylocaine Special Adhesive, stabilises the neuronal membrane and prevents the initiation and conduction of nerve impulses, thereby effecting local anaesthetic action.
The onset of action of Xylocaine Special Adhesive occurs within 3-5 minutes on mucous membrane and the effect lasts for approximately 15-20 minutes. It is ineffective when applied to intact skin.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Lidocaine may be absorbed following topical administration to mucous membranes, its rate of absorption and amount of dose absorbed depending upon concentration and total dose administered, the specific site of application and duration of exposure. In general, the rate of absorption occurs most rapidly after intratracheal administration. Lidocaine is well absorbed from the gastrointestinal tract, but little intact drug appears in the circulation because of biotransformation in the liver.
Metabolism. Lidocaine is metabolised rapidly by the liver, and metabolites and unchanged drug are excreted by the kidney.
Excessive blood levels may cause changes in cardiac output, total peripheral resistance and mean arterial pressure. These changes may be attributable to a direct depressant effect of the anaesthetic agent on various components of the cardiovascular system.
Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage and conjugation. The pharmacological/ toxicological actions of the metabolites are similar to, but not less potent than, those of lidocaine. Approximately 90% of lidocaine is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2,6-dimethylaniline.
The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of free base 1 to 4 microgram/mL, 60 to 80% of lidocaine is protein bound. Binding is also dependent on the plasma concentrations of the alpha-1-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion.
Studies of lidocaine metabolism following IV bolus injection have shown that the elimination half-life is usually 1.5 to 2 hours. The half-life may be prolonged 2-fold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine kinetics, but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above free base 6.0 microgram/mL. In the rhesus monkey arterial blood levels of 18 to 21 microgram/mL have been shown to be the threshold for convulsive activity.
5.3 Preclinical Safety Data
Genotoxicity. Genotoxicity tests with lidocaine are inconclusive. In genotoxicity studies, a metabolite of lidocaine, 2,6-xylidine, showed evidence of activity in some tests but not in other tests.
Carcinogenicity. This metabolite of lidocaine, 2,6-xylidine, has been shown to have carcinogenic potential (nasal and subcutaneous tumours) in preclinical toxicological studies evaluating chronic exposure.
6 Pharmaceutical Particulars
6.1 List of Excipients
Carmellose sodium, macrogol 1500, macrogol 300, macrogol 400, propylene glycol, spearmint oil.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
15 g tube.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
The molecular formula of lidocaine is C14H22N2O. The molecular weight is 234.3.
Chemical structure.
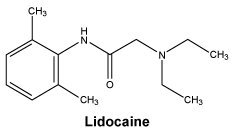
7 Medicine Schedule (Poisons Standard)
S2- Pharmacy Medicine.
Date of First Approval
13 August 1991
Date of Revision
18 October 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.