Zoladex 10.8 mg Implant SafeSystem
Brand Information
| Brand name | Zoladex 10.8 mg Implant SafeSystem |
| Active ingredient | Goserelin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Zoladex 10.8 mg Implant SafeSystem.
Summary CMI
ZOLADEX® 10.8 mg Implant SafeSystem®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ZOLADEX 10.8 mg?
ZOLADEX 10.8 mg contains the active ingredient goserelin acetate. ZOLADEX 10.8 mg is used to lower the amount of sex hormones in the body. ZOLADEX 10.8 mg is used to treat prostate cancer in some men.
For more information, see Section 1. Why am I using ZOLADEX 10.8 mg? in the full CMI.
2. What should I know before I use ZOLADEX 10.8 mg?
Do not use if you have ever had an allergic reaction to goserelin acetate or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions or take any other medicines.
For more information, see Section 2. What should I know before I use ZOLADEX 10.8 mg? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ZOLADEX 10.8 mg and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is ZOLADEX 10.8 mg given?
A ZOLADEX 10.8 mg implant will be injected into the skin of your stomach every 3 months.
More instructions can be found in Section 4. How is ZOLADEX 10.8 mg given? in the full CMI.
5. What should I know while using ZOLADEX 10.8 mg?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ZOLADEX 10.8 mg? in the full CMI.
6. Are there any side effects?
All medicines can have side effects. Sometimes they are serious, most of the time they are not. Tell your doctor immediately if you have trouble passing urine, feel weak in the arms or legs, feel numb in the arms or legs, have pain in the kidneys, have pain in the bones or backbone or have a tumour in your pituitary gland which causes severe headaches, sickness, loss of eyesight and unconsciousness. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ZOLADEX® 10.8 mg Implant SafeSystem®
Active ingredient: goserelin acetate
Consumer Medicine Information (CMI)
This leaflet provides important information about using ZOLADEX 10.8 mg. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ZOLADEX 10.8 mg.
Where to find information in this leaflet:
1. Why am I using ZOLADEX 10.8 mg?
2. What should I know before I use ZOLADEX 10.8 mg?
3. What if I am taking other medicines?
4. How is ZOLADEX 10.8 mg given?
5. What should I know while using ZOLADEX 10.8 mg?
6. Are there any side effects?
7. Product details
1. Why am I using ZOLADEX 10.8 mg?
ZOLADEX 10.8 mg contains the active ingredient goserelin acetate. ZOLADEX 10.8 mg is a member of the anti-hormonal group of medicines. This means that it affects the levels of various hormones (natural chemicals produced by the body). In men it reduces the level of testosterone.
ZOLADEX 10.8 mg is used to treat prostate cancer in some men. It is not a cure for prostate cancer.
2. What should I know before I use ZOLADEX 10.8 mg?
Warnings
Do not use ZOLADEX 10.8 mg if:
- you are allergic to goserelin acetate, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
Symptoms of an allergic reaction may include:
- rash
- hay fever
- difficulty breathing
- feeling faint
Check with your doctor if you:
- have any other medical conditions (have heart or blood vessel conditions, including heart rhythm problems (arrhythmia), or are being treated with medicines for these conditions). The risk of you having further heart rhythm problems may increase if you are taking ZOLADEX 10.8 mg.
- have any condition which affects the strength of your bones.
- you have had any problems passing urine or if you have had any lower back problems.
- you have diabetes.
- take any medicines for any other conditions
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
ZOLADEX 10.8 mg will not normally be used in females. There is not enough evidence to support its use in women.
It is also unlikely that you can father a child while using ZOLADEX 10.8 mg, but you must use your normal contraceptive method to make sure.
Children
Do not give ZOLADEX 10.8 mg to children.
There is no information on its use in children.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
ZOLADEX 10.8 mg might interfere with some medicines used to treat heart rhythm problems or might increase the risk of heart rhythm problems when used with some other drugs that can cause heart rhythm abnormalities.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ZOLADEX 10.8 mg.
4. How is ZOLADEX 10.8 mg given?
How much you will be given
- A ZOLADEX 10.8 mg implant will be injected into the skin of your stomach every 3 months.
- The implant is a very small pellet that is given by a special needle and syringe known as SafeSystem. The injection will not hurt very much.
- The pellet is designed to slowly release the medicine into your body over 3 months.
- The ZOLADEX 10.8 mg SafeSystem incorporates a protective needle sleeve that automatically locks in place following administration of the implant to help prevent needle stick injury.
If you forget to be given ZOLADEX 10.8 mg
Your doctor will make an appointment for your next dose, so you will not forget your next dose.
If you use too much ZOLADEX 10.8 mg
The doctor or nurse giving you ZOLADEX 10.8 mg will be experienced in the use of it, so it is unlikely that you will be given an overdose.
5. What should I know while using ZOLADEX 10.8 mg?
Things you should do
Make sure to keep your appointment for being given ZOLADEX 10.8 mg every 3 months.
If you do not, your testosterone level will rise and the prostate cancer may grow more quickly.
Remind any doctor, dentist or pharmacist you visit that you are using ZOLADEX 10.8 mg.
Things you should not do
- Do not stop being given ZOLADEX 10.8 mg unless you have discussed it with your doctor first.
They will have explained why you need to be given ZOLADEX 10.8 mg and for how long. - Do not use if the packaging is torn or shows signs of tampering.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ZOLADEX 10.8 mg affects you.
Looking after your medicine
- Keep in a cool, dry place where the temperature stays below 25°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Serious side effects
| Serious side effects | What to do |
Tell your doctor immediately if any of these things happen while you are using ZOLADEX 10.8 mg:
These may mean that the cancer is growing.
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
You may experience the following side effects:
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these very common side effects and they worry you. |
ZOLADEX 10.8 mg lowers the amount of sex hormones in your body so your sex drive will probably be reduced.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ZOLADEX 10.8 mg contains
| Active ingredient (main ingredient) | goserelin (as the acetate) |
| Other ingredients (inactive ingredients) | polyglactin |
Do not take this medicine if you are allergic to any of these ingredients.
What ZOLADEX 10.8 mg looks like
ZOLADEX 10.8 mg (AUST R 55242) comes in a special syringe, called the SafeSystem, and a siliconised needle.
The small pellet containing ZOLADEX 10.8 mg is about the size of a grain of rice. It can be seen in the "window" half way up the syringe and is white or cream coloured.
Who distributes ZOLADEX 10.8 mg
AstraZeneca Pty Ltd
ABN 54 009 682 311
66 Talavera Road
MACQUARIE PARK NSW 2113
Telephone:- 1800 805 342
This leaflet was prepared in March 2026.
Zoladex, Zoladex SafeSystem and SafeSystem are trade marks of the AstraZeneca group of companies.
© AstraZeneca 2026
VV-RIM-06055551 v 4.0
Brand Information
| Brand name | Zoladex 10.8 mg Implant SafeSystem |
| Active ingredient | Goserelin |
| Schedule | S4 |
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Goserelin acetate.
2 Qualitative and Quantitative Composition
Each Zoladex SafeSystem Implant contains goserelin 10.8 mg (as goserelin acetate).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
A sterile white to cream coloured cylindrical sustained release implant in which goserelin acetate is dispersed in a polyglactin co-polymer biodegradable matrix.
The implant is supplied in a single dose syringe applicator. The SafeSystem incorporates a protective needle sleeve that automatically locks in place following administration of the implant to aid in the prevention of needle stick injury.
4 Clinical Particulars
4.1 Therapeutic Indications
Prostate cancer. Palliative treatment of metastatic (M+) or locally advanced prostate cancer where suitable for hormonal manipulation.
Adjuvant and neoadjuvant therapy in combination with radiotherapy for the management of locally advanced prostate cancer in men suitable for hormonal manipulation.
4.2 Dose and Method of Administration
Caution should be taken while inserting Zoladex into the anterior abdominal wall due to the proximity of underlying inferior epigastric artery and its branches. Do not penetrate into muscle or peritoneum.
The Zoladex syringe cannot be used for aspiration. If the hypodermic needle penetrates a large vessel, blood will be seen instantly in the syringe chamber. If a vessel is penetrated, withdraw the needle and immediately control any resultant bleeding, monitoring the patient for signs or symptoms of abdominal haemorrhage. After ensuring the patient is haemodynamically stable, another Zoladex implant may be injected with a new syringe elsewhere.
Use extra care when administering Zoladex to patients with a low BMI and/or who are receiving full anticoagulation medication (see Section 4.4 Special Warnings and Precautions for Use).
Before injection, it should be ensured that the implant is visible in the window of the applicator. The plunger should not be withdrawn once the needle is in position. The plunger should be fully depressed to expel the implant into subcutaneous tissue, well away from point of entry and to activate the protective needle sleeve.
For correct administration of Zoladex, see instructions on the administration card.
Do not omit or delay injections, as serum testosterone levels may rise in males.
In the unlikely event of the need to surgically remove a Zoladex implant, it may be localised by ultrasound.
Adult men (including the elderly). One implant of Zoladex 10.8 mg injected subcutaneously into the anterior abdominal wall, every 3 months (see Section 5.1 Pharmacodynamic Properties). Adjuvant and/or neoadjuvant Zoladex therapy in combination with radiotherapy may include short-term use of an anti-androgen to prevent flare (see Section 5.1 Pharmacodynamic Properties, Clinical trials, Prostate cancer - adjuvant and neoadjuvant Zoladex therapy in combination with radiotherapy).
Adult women. Zoladex 10.8 mg Implant is not indicated for use in females.
Special patient populations. Renal impairment. No dosage adjustment is necessary for patients with renal impairment.
Hepatic impairment. No dosage adjustment is necessary for patients with hepatic impairment.
Paediatric use. Zoladex 10.8 mg Implant is not indicated for use in children.
4.3 Contraindications
Zoladex is contraindicated in patients with known hypersensitivity to luteinising hormone releasing hormone (LHRH), LHRH agonist analogues or any of the components of Zoladex.
4.4 Special Warnings and Precautions for Use
Injection site injury has been reported with Zoladex, including events of pain, haematoma, haemorrhage and vascular injury. Monitor affected patients for signs or symptoms of abdominal haemorrhage. In very rare cases, administration error resulted in vascular injury and haemorrhagic shock requiring blood transfusions and surgical intervention.
Extra care should be taken when administering Zoladex to patients with a low BMI and/or receiving full anticoagulation medications (see Section 4.2 Dose and Method of Administration).
Zoladex 10.8 mg is not indicated for use in females, since there is insufficient evidence of reliable suppression of serum oestradiol. For female patients requiring treatment with goserelin, refer to the prescribing information for Zoladex 3.6 mg.
Initially Zoladex, like other gonadotrophin releasing hormone (GnRH) agonists, transiently increases serum testosterone. Although not necessarily associated, there have been reports of temporary increase in bone pain in patients with advanced cancer and bony metastases. These events may last up to two weeks and may need to be managed symptomatically.
The use of Zoladex in patients with metastatic cancer who are at particular risk of developing ureteric obstruction or spinal cord compression should be considered carefully and the patients monitored closely during the first month of therapy. If spinal cord compression or renal impairment due to ureteric obstruction are present or develop, specific standard treatment of these complications should be instituted. Isolated cases of short-term worsening of these signs and symptoms have been reported during the initial four weeks of Zoladex therapy. Consideration should be given to anti-androgen therapy at the start of Zoladex therapy since this has been reported to prevent the possible sequelae of the initial rise in serum testosterone.
Serum testosterone concentrations may rise if an implant is omitted or delayed.
Zoladex causes loss of bone mineral density.
Hyperglycaemia and diabetes. Hyperglycaemia and an increased risk of developing diabetes have been reported in men receiving GnRH agonists. Hyperglycaemia may represent development of diabetes mellitus or worsening of glycaemic control in patients with diabetes. Monitor blood glucose and/or glycosylated haemoglobin (HbA1c) periodically in patients receiving a GnRH agonist and manage with current practice for the treatment of hyperglycaemia or diabetes.
Cardiovascular disease. An increased risk of developing myocardial infarction, sudden cardiac death and stroke has been reported in association with use of GnRH agonists in men. The risk appears low based on the reported odds ratios and should be evaluated carefully along with cardiovascular risk factors when determining a treatment for patients with prostate cancer. Patients receiving a GnRH agonist should be monitored for symptoms and signs suggestive of development of cardiovascular disease and managed according to current clinical practice.
QT/QTc interval prolongation. Androgen deprivation therapy may prolong QT/QTc interval. Prescribers should consider whether the benefits of androgen deprivation therapy outweigh the potential risks in patients with congenital long QT syndrome, congestive heart failure, frequent electrolyte abnormalities and in patients taking drugs known to prolong the QT interval. Electrolyte imbalances should be corrected. Consider periodic monitoring of electrocardiograms and electrolytes (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Use in the elderly. See Section 4.2 Dose and Method of Administration.
Paediatric use. Zoladex 10.8 mg is not indicated for use in children as safety and efficacy have not been established in this group of patients.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No data available.
Since androgen deprivation treatment may prolong the QT interval, the concomitant use of Zoladex with medicinal products known to prolong the QT interval or medicinal products able to induce Torsades de Pointes should be carefully evaluated (see Section 4.4 Special Warnings and Precautions for Use).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. The expected pharmacology of Zoladex is the suppression of gonad function to castrate levels. As a result there is profound impairment of fertility.
In rats this is expressed as:
Male. Decrease in weight and atrophic histological changes in the testes, epididymis, seminal vesicle and prostate gland with complete suppression of spermatogenesis.
Female. Suppression of ovarian function with decreased size and weight of the ovaries and secondary sex organs; arrest of follicular development at the antral stage and reduction in size and number of the corpora lutea.
Except for the testes, almost complete reversal of these effects in male and female rats was observed several weeks after dosing was stopped, however, fertility and general reproductive performance were reduced in those that became pregnant after goserelin was discontinued.
Based on histological examination, drug effects on reproductive organs seem to be completely reversible in male and female dogs when drug treatment was stopped after continuous administration for 1 year at doses equivalent to 214 microgram/kg/day (approximately 57 times the recommended monthly dose for a human based on AUC).
Use in pregnancy. Zoladex 10.8 mg is not indicated for use in females.
Use in lactation. Zoladex 10.8 mg is not indicated for use in females.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
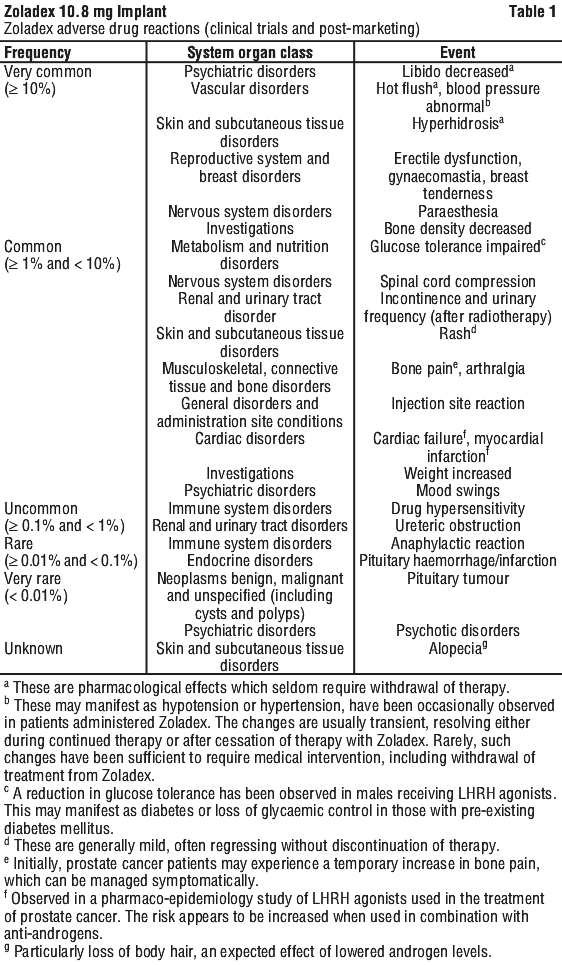
The following frequency categories for adverse drug reactions (ADRs) were calculated based on reports from Zoladex clinical trials and post-marketing sources. See Table 1.
4.9 Overdose
There is limited experience of overdosage in humans. In cases where Zoladex has unintentionally been readministered early or given at a higher dose, no clinically relevant adverse effects have been seen. Animal tests suggest that no effect other than the intended therapeutic effects on sex hormone concentrations and on the reproductive tract will be evident with higher doses of Zoladex. If overdosage occurs, this should be managed symptomatically.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Goserelin acetate is a GnRH agonist (also known as LHRH agonist).
Goserelin acetate is a potent synthetic decapeptide analogue of LHRH. When given acutely, goserelin acetate will release luteinising hormone (LH) from the pituitary gland. However, following chronic administration, goserelin acetate is a potent inhibitor of gonadotrophin production resulting in gonadal suppression and consequently sex organ regression.
In animals and humans, following an initial stimulation of pituitary LH secretion and a transient elevation in serum testosterone, chronic administration results in inhibition of gonadotrophin secretion. The result is a sustained suppression of pituitary LH occurring within approximately 3 weeks after initiation of therapy, and a reduction in serum testosterone levels in males to a range normally seen in surgically castrated men. This suppression is then maintained as long as therapy is continued. If in exceptional circumstances repeat dosing does not occur at 3 months, data indicate that castrate levels of testosterone are maintained for up to 16 weeks in the majority of patients.
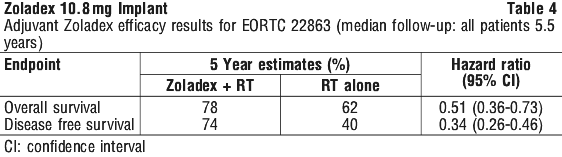
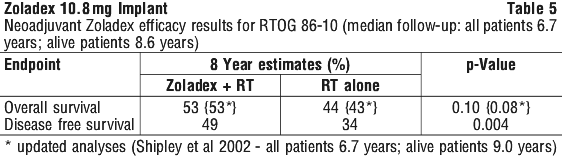
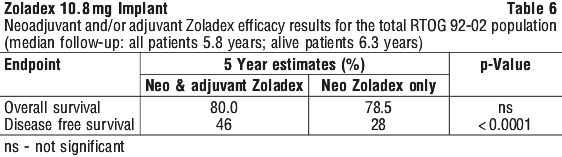
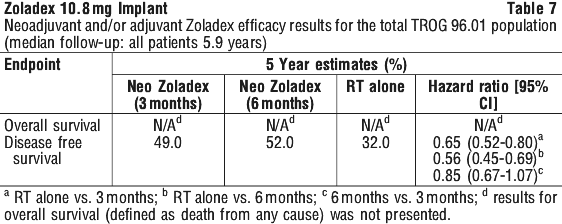
Clinical trials. Prostate cancer - adjuvant and neoadjuvant Zoladex therapy in combination with radiotherapy. Five phase III, open-labelled, randomised, controlled, multi-centred clinical trials have been conducted to evaluate the added value of adjuvant and/or neoadjuvant Zoladex therapy in combination with radiotherapy in patients with histologically proven prostate cancer. The majority of patients had locally advanced disease (T2 N+, T3 or T4, N0/Nx, M0). All studies have been performed by three independent collaborative oncology groups (European Organisation for Research and Treatment of Cancer [EORTC], the Radiation Therapy Oncology Group [RTOG]) and the Trans-Tasman Radiation Oncology Group [TROG]), and have reported results from median follow-up of more than 5 years. Table 2 summarises the study design, patient populations and median follow-up periods for these studies.


5.2 Pharmacokinetic Properties
Goserelin acetate has a serum elimination half-life of approximately 4.2 hours in subjects with normal renal function compared to 13 minutes for natural LHRH.
Although the half-life is increased in patients with impaired renal function, absolute clearance is still relatively rapid. The existence of a non-renal, presumably hepatic, clearance and the absence of an increased incidence of possible adverse reactions in such patients imply that no adjustment in the proposed dosage regimen is necessary in patients with renal impairment.
There is no significant change in pharmacokinetics in patients with hepatic failure.
The implant formulation of the drug is dispersed in a cylindrical rod of a biodegradable and biocompatible polyglactins and is released continuously when injected subcutaneously. The implant is supplied in a purpose-designed applicator with 14-gauge needle.
Administration of Zoladex 10.8 mg, in accordance with the dosage recommendations, ensures that exposure to goserelin is maintained with no clinically significant accumulation. The peak serum concentrations occur during the first 24 hours post administration. The mean serum concentration at 2 hours post administration is 8.6 ± 2.9 (SD) nanogram/mL with an inter-individual range of up to 11-fold.
Mean systemic clearance values for goserelin were about 100 to 200 mL/min with an inter-individual range of up to 6-fold.
Serum goserelin concentrations become low by end of the dosing interval; delaying or omitting scheduled doses should be avoided as it may lead to increased testosterone levels and loss of efficacy.
5.3 Preclinical Safety Data
Genotoxicity. Mutagenicity tests for gene mutations and chromosomal damage have provided no evidence for mutagenic effects.
Carcinogenicity. After subcutaneous implant injections once every 4 weeks for 1 year to male and female rats at doses equivalent to 4 times the recommended monthly dose for a human (based on AUC), an increased incidence of benign pituitary microadenomas was found.
This finding is similar to that previously noted in this species following surgical castration and appears to be a species specific response to castration. Any relevance to humans has not been established. No increase in pituitary adenomas was seen in mice receiving injections of goserelin every 3 weeks for 2 years at doses up to 2400 microgram/kg/day (approximately 18 to 37 times the recommended monthly dose for a human [based on Cmax]). An increased incidence of histiocytic sarcomas of the bone marrow of the vertebral column and femur were observed in male mice given 2400 microgram/kg/day but not in female mice, or rats of either sex. The relevance of these tumours to humans has not been established.
In mice, long-term repeated dosing with multiples of the human dose produced histological changes in some regions of the digestive system manifested by pancreatic islet cell hyperplasia and a benign proliferation condition in the pyloric region of the stomach, also reported as a spontaneous lesion in this species. The clinical relevance of these findings is unknown.
6 Pharmaceutical Particulars
6.1 List of Excipients
Zoladex 10.8 mg SafeSystem Implant contains the excipient polyglactin.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Zoladex 10.8 mg SafeSytem Implant is supplied as a sterile, biodegradable cylindrical implant containing the equivalent of 10.8 mg of goserelin base together with the inactive ingredient polyglactin and is presented as 1x (one) pre-filled syringe applicator for subcutaneous injection per carton.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
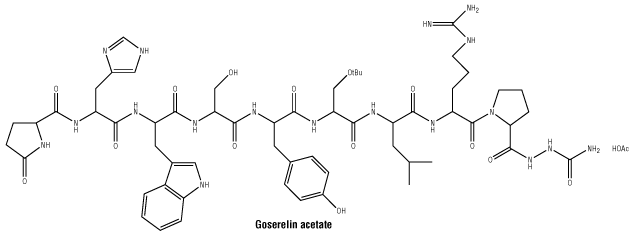
Molecular weight: 1269 (base).
CAS number. 65807-02-5 (goserelin base).
7 Medicine Schedule (Poisons Standard)
Prescription only medicine (Schedule 4).
Date of First Approval
22 May 1996
Date of Revision
06 March 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.