Zyrtec
Brand Information
| Brand name | Zyrtec |
| Active ingredient | Cetirizine hydrochloride |
| Schedule | S2 |
No current CMI was found.
Brand Information
| Brand name | Zyrtec |
| Active ingredient | Cetirizine hydrochloride |
| Schedule | S2 |
MIMS Revision Date: 01 August 2019
1 Name of Medicine
Cetirizine hydrochloride.
2 Qualitative and Quantitative Composition
Zyrtec tablets. White, film-coated, oblong tablets, scored on one face and embossed Y-Y, containing 10 mg cetirizine hydrochloride. Zyrtec tablets also contain microcrystalline cellulose, lactose, colloidal anhydrous silica, magnesium stearate, and Opadry white Y-1-7000.
Zyrtec oral liquid. Clear, colourless, banana flavoured liquid containing 1 mg/mL cetirizine hydrochloride. Zyrtec oral liquid also contains sorbitol, glycerol, propylene glycol, saccharin sodium, methyl hydroxybenzoate, propyl hydroxybenzoate, banana flavour, sodium acetate, acetic acid and purified water.
Zyrtec oral drops. Clear, colourless liquid with a slightly bitter-sweet taste which contains 10 mg/mL (2.5 mg/5 drops) cetirizine hydrochloride. Zyrtec oral drops also contains glycerol, propylene glycol, saccharin sodium, methyl hydroxybenzoate and propyl hydroxybenzoate, sodium acetate, acetic acid and purified water.
3 Pharmaceutical Form
Zyrtec is formulated as white, film-coated, scored 10 mg tablets, oral liquid 1 mg/mL and oral drops 10 mg/mL.
4 Clinical Particulars
4.1 Therapeutic Indications
Seasonal allergic rhinitis. Cetirizine is indicated for the relief of symptoms associated with seasonal allergic rhinitis (hay fever) in adults and children aged 1-12 years. Symptoms treated effectively include sneezing, rhinorrhoea, postnasal discharge, nasal pruritus, ocular pruritus and tearing and redness of the eyes.
Perennial allergic rhinitis. Cetirizine is indicated for the relief of symptoms associated with perennial allergic rhinitis in adults and children aged 1-12 years. Symptoms treated effectively include sneezing, rhinorrhoea, postnasal discharge, nasal pruritus, ocular pruritus and tearing.
Chronic idiopathic urticaria. Cetirizine is indicated for the treatment of the uncomplicated skin manifestations of chronic idiopathic urticaria in adults and children aged 1-12 years. It significantly reduces the occurrence, severity and duration of hives and markedly reduces pruritus. As with other antihistamines, patients should be advised to seek medical advice about the possibility that their urticaria is associated with ingestion of certain foods.
4.2 Dose and Method of Administration
Zyrtec tablets. The use of Zyrtec tablets is not recommended in children less than 6 years since this formulation does not allow for appropriate dose adaptation.
Children 6-12 years. The recommended dose is 5 mg (half a tablet) twice daily with or without food.
Adults and children over 12 years. The recommended dose is 10 mg (one tablet) once a day, with or without food. The time of administration may be varied to suit individual patient needs.
Zyrtec oral liquid. The use of Zyrtec Oral Liquid is not recommended in children aged less than 2 years as the amount of sorbitol (1.6 g/5 mL) in the formulation may cause diarrhoea in children under 2 years.
Children 2-12 years. The recommended dose is to be taken twice daily with or without food and should be calculated on the basis of body weight according to the following scale. See Table 1.
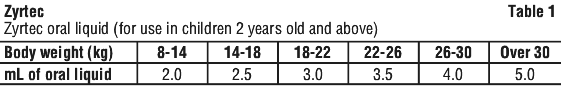
Zyrtec oral drops. The oral drops should not be used in children aged less than 1 year.
Children from 1 year of age and over. The recommended dose is to be taken twice daily with or without food and should be calculated on the basis of body weight according to the following scale. See Table 2.
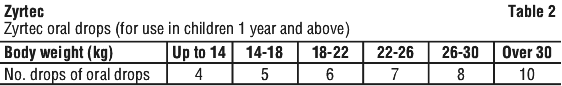
4.3 Contraindications
1. In patients with a known hypersensitivity to any of the ingredients of the Zyrtec formulations (see Section 6.1 List of Excipients).
2. In patients with a known hypersensitivity to hydroxyzine (the parent compound of cetirizine) or to any piperazine derivatives.
3. In patients with severe renal impairment (less than 10 mL/min creatinine clearance).
Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose/ galactose malabsorption should not take Zyrtec film-coated tablets.
Patients with rare hereditary problems of fructose intolerance should not take Zyrtec 1 mg/mL oral liquid.
4.4 Special Warnings and Precautions for Use
Concomitant treatment. Although it has been shown that cetirizine at the recommended dose of 10 mg does not potentiate the effect of alcohol, in sensitive patients, the simultaneous administration of cetirizine and alcohol or other CNS depressants may have effects on the CNS. Therefore, precaution is recommended if alcohol is taken concomitantly.
Mental alertness. Some patients may experience a degree of drowsiness with cetirizine. Studies using objective measurements have shown no effect of cetirizine on cognitive function, motor performance of sleep latency. However, in clinical trials, the occurrence of CNS effects has been observed in some individual patients and due caution should be exercised when driving a car or operating potentially dangerous machinery.
In sensitive patients, concurrent use with alcohol or other CNS depressants may cause additional reductions in alertness and impairment of performance.
Epilepsy. CNS stimulation may occur with antihistamines, especially in children. Therefore, caution is recommended when treating patients suffering from epilepsy or patients at risk of convulsions.
Renal impairment. Cetirizine clearance is reduced in patients with renal impairment. In patients with renal insufficiency, dosage should be reduced to half the usual recommended dose. Zyrtec is contraindicated in patients with severe renal impairment (less than 10 mL/min creatinine clearance) (see Section 4.3 Contraindications).
Skin and subcutaneous tissue. Severe skin reactions such as acute generalized exanthematous pustulosis (AGEP) have been reported very rarely with cetirizine-containing products. This acute pustular eruption may occur within the first 2 days of treatment, with fever, and numerous, small, mostly non-follicular pustules arising on a widespread oedematous erythema and mainly localized on the skin folds, trunk, and upper extremities. Patients should be carefully monitored. If signs and symptoms such as formation of small pustules occur, with or without pyrexia or erythema, then treatment with cetirizine should be discontinued and a physician should be consulted.
Urinary retention. Caution should be taken in patients with predisposing factors of urinary retention (e.g. spinal cord lesion, prostatic hyperplasia) as cetirizine may increase the risk of urinary retention.
Use in the elderly. There are no data to suggest that elderly who have normal renal function require a lower dose. However, as advancing age may be associated with declining renal function, dosage may need to be reduced in the elderly if creatinine clearance is reduced. Cetirizine is well tolerated by patients 65 years of age and over. Clearance of cetirizine is reduced in proportion to creatinine clearance. In patients whose creatinine clearance is reduced (i.e. those with moderate renal impairment), a dose of 5 mg/day is recommended.
Paediatric use. Zyrtec tablets is not recommended in children aged less than 6 years since this formulation does not allow for appropriate dose adaptation.
Zyrtec oral liquid is not recommended in children aged less than 2 years as the amount of sorbitol (1.6 g/5 mL) in the formulation may cause diarrhoea in children under 2 years.
Zyrtec oral drops should not be used in children aged less than 1 year.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Studies with cetirizine demonstrated that there were no clinically relevant adverse interactions with pseudoephedrine, antipyrine, cimetidine, ketoconazole, erythromycin, azithromycin, glipizide and diazepam. A small decrease in the clearance of cetirizine (16%) was observed in a multiple dose study with theophylline (400 mg once a day); while the disposition of theophylline was not altered by concomitant cetirizine administration. In a multiple dose study of ritonavir (600 mg twice daily) and cetirizine (10 mg daily), the extent of exposure to cetirizine was increased by about 40% while the disposition of ritonavir was slightly altered (-11%) further to concomitant cetirizine administration.
4.6 Fertility, Pregnancy and Lactation
Use in pregnancy. (Category B2)
Reproduction studies in mice, rats and rabbits failed to show evidence of teratogenicity using doses up to 96, 225, and up to 135 mg/kg/day, respectively. However, the short half-life of cetirizine in these species suggests that fetal exposure may have been inadequate. In mice, postnatal development was inhibited after 96 mg/kg/day. Clinical data for cetirizine or other compounds of the class are inadequate to establish safety in pregnancy. Until such data are available, cetirizine should be used in pregnancy only if the expected benefits clearly outweigh potential risks to mother and fetus.
Use in lactation. Studies in beagle dogs indicate that approximately 3% of the dose is excreted in milk. Cetirizine is excreted in human milk at concentrations representing 0.25 to 0.90 those measured in plasma, depending on sampling time after administration. Use of cetirizine in breastfeeding mothers is not recommended.
Effects on fertility. No data available.
4.7 Effects on Ability to Drive and Use Machines
Although studies using objective measurements have shown no effect of cetirizine on cognitive function, motor performance of sleep latency, due to its potential for sedation, caution should be used when driving a motor vehicle or operating machinery.
In sensitive patients, concurrent use with alcohol or other CNS depressants may cause additional reductions in alertness and impairment of performance.
4.8 Adverse Effects (Undesirable Effects)
The more commonly observed untoward reactions reported during cetirizine administration and not associated with an equivalent incidence among placebo treated patients are somnolence, dry mouth and fatigue.
Table 3 shows adverse events occurring with an incidence of greater than 1% after intake of cetirizine 5 to 20 mg cetirizine a day. It pools data obtained from American and European clinical studies (including open studies with access to rescue drug) in urticaria, perennial and seasonal rhinitis. The incidence of somnolence associated with Zyrtec was 14.3% (7.6% under placebo) and was predominantly mild to moderate in severity. After pooling the same studies in the three registered indications, sedation is reported more in the patients suffering from seasonal allergic rhinitis than in the patients suffering from perennial allergic rhinitis and urticaria.
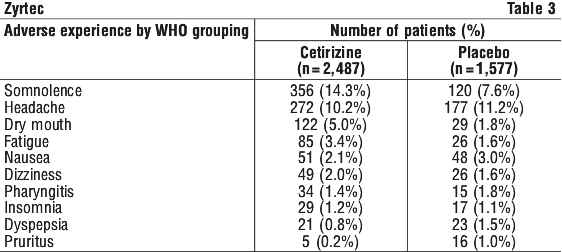
The following events were observed infrequently (less than 1/100), but more than once, in 2,487 patients who received cetirizine in all US and European trials, a causal relationship with cetirizine administration has not been established. Events are listed in order of decreasing frequency within a given body system.
Autonomic nervous system. Increased appetite, anorexia, flushing, increased sweating.
Cardiovascular. Palpitations/ tachycardia.
ENT. Earache, epistaxis, altered sense of taste, tinnitus, tongue disorder.
Vision. Eye abnormality, periorbital oedema, abnormal vision, eye pain, conjunctivitis.
Gastrointestinal. Abdominal pain, diarrhoea, vomiting, constipation, flatulence.
Genitourinary. Polyuria, urinary retention, urinary tract infection.
Musculoskeletal. Back pain, myalgia, arthralgia, bone disorder (fracture), leg cramps.
Neurologic. Nervousness, impaired concentration, confusion, paraesthesia, asthenia, hypertonia, tremor.
Respiratory system. Respiratory disorder, coughing, bronchospasm, upper respiratory tract infection, dyspnoea.
Miscellaneous. Weight increase (see comment below), fever, oedema, chest pain, pain, rigors, dysmenorrhoea, thirst, decreased libido.
Weight gain was reported as an adverse effect in 0.4% of cetirizine patients in placebo controlled trials. In an open study of six months duration, the mean gain in weight was 2.8% after 20 weeks, with no further increase at 26 weeks. This effect has been reported for other antihistamines.
Occasional instances of reversible liver function test (transaminase) elevations have occurred during cetirizine therapy, without evidence of jaundice, hepatitis or other clinical findings.
Adverse events at rates of 1% or greater in children aged from 6 months to 12 years, included in placebo controlled trials are in Table 4.

Post marketing data. Adverse drug reactions (ADRs) identified during Post-marketing experience with cetirizine are included in the Tables 5 and 6. The frequencies are provided according to the following convention: very common ≥ 1/10; common ≥ 1/100 to < 1/10; uncommon ≥ 1/1,000 to < 1/100; rare ≥ 1/10,000 to < 1/1,000; very rare < 1/10,000, not known (cannot be estimated from the available data).


Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at: www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms observed after an overdose of cetirizine are mainly associated with CNS effects that could suggest an anticholinergic effect. Adverse events reported after an intake of at least 5 times the recommended daily dose are confusion, diarrhoea, dizziness, fatigue, headache, malaise, mydriasis, pruritus, restlessness, sedation, somnolence, stupor, tachycardia, tremor and urinary retention.
Should overdose occur, treatment should be symptomatic or supportive, taking into account any concomitantly ingested medications. There is no known specific antidote to cetirizine. Cetirizine is not effectively removed by dialysis, and dialysis will be ineffective unless an agent which is removed by dialysis has been concomitantly ingested.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Cetirizine, a human metabolite of hydroxyzine, is an antiallergic compound; its principal effects are mediated via competitive occupancy of peripheral H1-receptors.
In vitro receptor binding studies have shown no measurable affinity for receptors other than H1-receptors.
Clinical trials. Studies in normal volunteers show that cetirizine at doses of 5 and 10 mg strongly inhibits the skin wheal and flare caused by the intradermal injection of histamine. The onset of activity corresponds with the occurrence of maximal plasma levels, and significant blockade persists for at least 24 hours after a single dose. The effects of intradermal injection of various other mediators or histamine releasers are also inhibited by cetirizine, as is cold-induced urticaria. The late phase recruitment of eosinophils, a component of the allergic inflammatory response, is inhibited by cetirizine following cutaneous antigen challenge.
CNS effects. Autoradiographic studies with radiolabelled cetirizine in the rat have shown very low penetration of the brain. Sedation was observed in animal studies, but only at doses at least 1,000 times greater than those required for antagonism of histamine H1-receptors. Studies in normal volunteers using objective measurements have, most of the time, shown no effect of cetirizine at the recommended dose of 10 mg on sleep latency time, cognitive function or motor performance. However, the occurrence of CNS effects has been observed in clinical trials in some patients (see Section 4.8 Adverse Effects (Undesirable Effects)).
Studies using quantitative EEG recordings and various other tests of cognitive function confirmed that cetirizine does not cause CNS depression.
5.2 Pharmacokinetic Properties
Absorption. Cetirizine is rapidly absorbed after oral administration. In adults, peak plasma levels after a 10 mg dose are approximately 300 nanogram/mL and occur at about one hour. Coadministration with food decreases the rate of absorption by 1.7 hours (lower Cmax and greater Tmax), but does not affect bioavailability as measured by the AUC. Plasma protein binding is 93%. The bioavailability of cetirizine hydrochloride is similar from the different dosage forms of Zyrtec. The mean time taken to reach the peak serum cetirizine concentration (Tmax) was 0.67 hour after a single 10 mg dose of the film coated tablets.
Distribution. The apparent volume of distribution is 0.45 L/kg, suggestive of significant extravascular distribution. The plasma elimination half-life in adults is approximately 8 hours and does not change with multiple dosing. Plasma levels are proportional to the dose administered over the range of 5 to 20 mg.
Metabolism. In contrast to other known antihistamines, cetirizine is less extensively metabolised, and approximately 2/3 of an administered dose is excreted unchanged in the urine. This results in high bioavailability with low inter- or intrasubject variation in blood levels. A study using 14C-labelled cetirizine showed that most of the plasma radioactivity is associated with the parent compound. Only one metabolite has been identified in human plasma, the product of oxidative dealkylation of the terminal carboxymethyl group. The antihistaminic activity of this metabolite is negligible.
Excretion. In children, as with adults, cetirizine is eliminated mostly in the urine. Children over 6 years of age show peak plasma levels and times to peak similar to adults, with slightly more rapid elimination. Children younger than 6 years have more rapid clearance and a shorter half-life relative to adults. The half-life of cetirizine is approximately 6 hours in children aged 6-12 years; 5 hours in children aged 2-6 years; and 3 hours in infants and toddlers aged 6-24 months.
The total body clearance of cetirizine is reduced in subjects with renal dysfunction but below a creatinine clearance of about 30 to 50 mL/minute, little further change occurs. The pharmacokinetics of the drug was similar in patients with mild impairment (creatinine clearance higher than 40 mL/min) and normal volunteers. Moderately renally impaired patients had a 3-fold increase in half-life and 70% decrease in clearance compared to normal volunteers. Plasma levels of cetirizine are essentially unaffected by haemodialysis, and the plasma elimination half-life in dialysis patients is approximately 20 hours. The plasma AUC is increased about threefold in these patients. The clearance of cetirizine is reduced in elderly patients, but only in proportion to the decrease in creatinine clearance. Thus, in 16 patients with a mean age of 77 years, half-life increased to 12 hours. Cetirizine blood levels were monitored in a clinical trial of 59 patients, aged 60 to 82, who received 10 mg of cetirizine daily for three weeks, and no undue accumulation of cetirizine was found.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. Carcinogenicity studies over 24 months showed increased incidences of benign liver tumours in male mice (at the maximum dose of 16 mg/kg/day), but not in female mice or in rats. These benign tumours in mice are commonly found with compounds which cause liver enzyme induction. Since cetirizine does not induce liver enzymes in nonrodents and humans, this may be considered to be a species specific phenomenon. Cetirizine was devoid of mutagenic activity in a series of in vitro and in vivo assays.
Allergy skin tests are inhibited by antihistamines. Wash-out periods vary in individuals due to different rates of metabolism and different antihistamines. For cetirizine, a wash-out period of at least four days is generally recommended before performing the allergy skin tests.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine. See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Zyrtec tablets. Available in blister packs of the following sizes: 5, 10, 30, 50, 60, 70.
Zyrtec oral liquid. Available in a 75 mL and 200 mL bottle.
Zyrtec oral drops. Available in 20 mL bottle.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
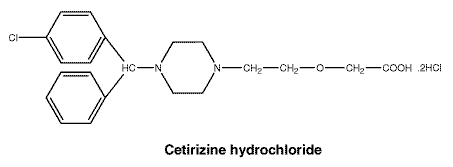
Cetirizine hydrochloride is an orally active, H1-receptor antagonist. The molecular weight is 461.8. Cetirizine hydrochloride is a white, crystalline powder and is water soluble (160 g/100 mL).
Chemical name. 2-(2-(4-(4-chlorophenyl) phenylmethyl)-1-piperazinyl) ethoxy) acetic acid, dihydrochloride.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S2.
Date of First Approval
28 August 1997
Date of Revision
01 May 2019
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.