Quality Use of Medicines and Medicines Safety was made the 10th National Health Priority Area in 2019 by the Council of Australian Governments (COAG) Health Council. The Commission are developing a National Baseline Report on Quality Use of Medicines and Medicines Safety in response to this initiative. This discussion paper was published to support the public consultation which informed Phase 1 of the National Baseline Report.
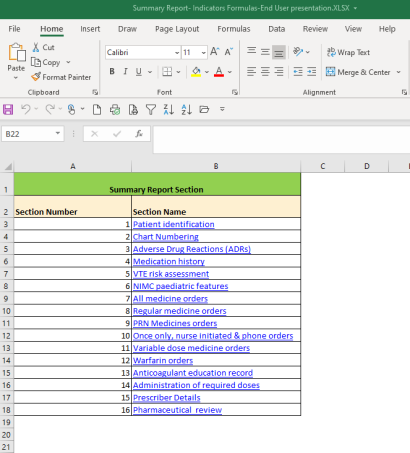
This resource has been developed for hospitals participating in the National NSMC Audit to assist with calculating audit results.
This user guide for the Clinical Trials Portal has been developed to assist with the navigation and use of the self-assessment and operational metrics tools including the registration process.
This fact sheet includes information for clinicians about informed consent in health care.
Released September 2020
Healthcare professionals have ethical, legal and professional requirements to make sure patients give consent before any procedure, test or treatment, and when prescribing medicines.
This report provides the latest update report for data submitted to CARAlert for the reporting period: 1 May 2020 to 30 June 2020, and complements previous analyses of and updates on CARAlert data.
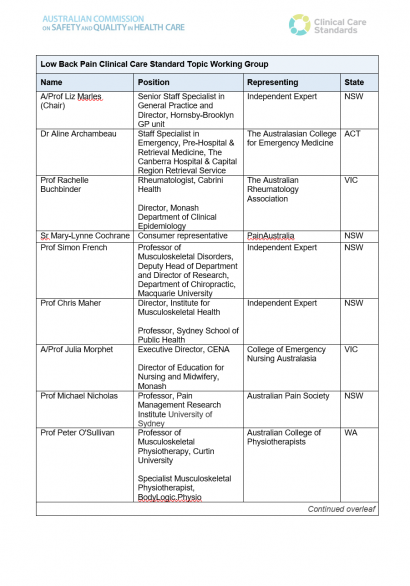
List of Topic Working Group members for the Low Back Pain Clinical Care Standard.
The Corporate Plan 2020-21 outlines the strategic planning activities of the Commission and reports on the key performance indicators and performance targets.
This is the flowchart for the assessment process for the National General Practice Accreditation Scheme.