The Commission has released the latest draft of the National Safety and Quality Medical Imaging Standards (draft Standards), ahead of a regulatory impact assessment.
Use this summary sheet during your appointment to help you Ask, Share, Know.
Ask Share Know is a methodology that will help consumers make shared decisions with their health professionals. The Ask Share Know methodology helps consumers:
This report is a technical highlights report of antibacterials prescribed by MedicineInsight general practices from 2015 to 2024. It is complementary to the Antimicrobial use in the community: 2024 report, which includes analyses of Pharmaceutical Benefits Scheme and Repatriation Pharmaceutical Benefits Scheme (PBS/RPBS) data.
To describe the legislated requirements for health service organisations to report medical device related adverse events.
This issue includes items on mental health, infertility, decarbonizing health care, diagnostic safety, and more. Along with the latest from BMJ Quality & Safety, Australian Prescriber, Public Health Research & Practice, Emergency Medicine Australasia, Health Expectations, Journal of Health Services Research & Policy JBI Evidence Synthesis, and InternationalJournal for Quality in Health Care. Also included is the latest from the World Health Organization (WHO) and the USA’s Agency for Healthcare Research and Quality (AHRQ).
Read the latest news, advisories, fact sheets and guidance on the National Safety and Quality Health Service (NSQHS) Standards.
FAQs have been developed for users of the NPS MedicineWise Drs Bag app and clinicians.
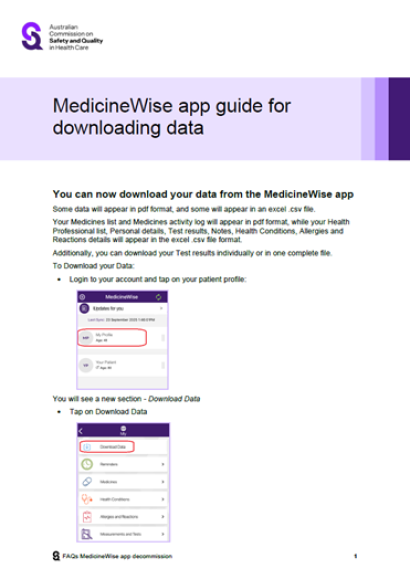
FAQs have been developed for users of the MedicineWise app and clinicians.