Digital health
Digital health is the knowledge and practice associated with developing and using digital tools and technologies to improve health outcomes. It includes a wide range of technologies such as electronic medical records, electronic medication management systems, virtual care, My Health Record, mobile health applications, wearable sensors, and machine-learning algorithms for clinical decision support.
Digitally enabled care is the appropriate integration and application of digital health in clinical settings to deliver (remotely or face-to-face), augment, or coordinate patient care.
Our work to support digitally enabled care
-
Priorities for high-quality digitally enabled care
Our four priorities for high-quality digitally enabled care helps achieve the appropriate application and integration of digital health tools and technologies in clinical settings.
-
Artificial Intelligence (AI)
Artificial intelligence (AI) is being used in healthcare to support a range of clinical and administrative tasks.
-
Transitions of care
When adopted in a standardised and meaningful way, digital health technology is a strong enabler for high-quality transitions of care.
-
Electronic Medication Management Systems
EMM systems can improve the accuracy, visibility and legibility of medical information, so that the communication between professionals and consumers is clearer.
-
Real-time prescription monitoring (RTPM)
Real-time prescription monitoring (RTPM) provides vital information about a person’s prescribing and dispensing history for certain high-risk medicines.
-
My Health Record
My Health Record is Australia’s national electronic health record that securely stores key health information.
-
Strengthening virtual care quality
Strengthening virtual care quality is one of our four priorities of high quality digitally enabled care.
-
Unique identifiers for medical devices
The Australian Government is improving patient safety by introducing Unique Device Identification (UDI) for medical devices and in vitro diagnostic devices.
Who we work with
We work alongside leading experts and committees to help inform our work in digital health and provide advice and support on providing digital health initiatives that are safe, high quality and appropriate.
Digitally Enabled Care Advisory Committee
The Digitally Enabled Care Advisory Committee (DECAC), established in 2024, provides expert advice to support the safe, high quality and equitable use of digitally enabled care across Australia’s health system.
About the DECAC
Established in April 2024, the DECAC advises on the Commission's priorities for high-quality digitally enabled care, in alignment with Commonwealth, state and territory digital health strategies.
DECAC strengthens the reach of our digital health agenda by identifying opportunities to raise awareness, improve stakeholder engagement and support implementation across the healthcare system.
We monitor emerging opportunities and risks to inform national guidance, standards and accreditation processes, including the National Safety and Quality in Health Service (NSQHS) Standards.
A strong focus is placed on equity, access, including culturally safe design for First Nations people and diverse communities, and the building of enduring digital capability across sectors and regions, particularly rural and remote areas.
The Committee further advises on strengthening clinician and consumer trust through robust governance, evidence-based practice and shared design.
Membership
DECAC is co-chaired by Dr Andrew Hugman, Clinical Director, Digital Health, and Richard Taggart, eHealth NSW Chief Executive. Members include consumers, senior clinicians and representatives from primary care and private hospitals.
A list of the full membership is provided below.
| Member | Organisation |
|---|---|
| Co-Chair – Dr Andrew Hugman | Australian Commission on Safety and Quality in Health Care |
| Co-Chair – Mr Richard Taggart | eHealth NSW |
| Ms Alex Powell | Australian Digital Health Agency |
| Dr Amandeep Hansra | Australian Digital Health Agency |
| Ms Karen Booth | Australian Digital Health Agency |
| Mr Matthew Castle | Department of Health, Disability and Ageing |
| TBA | Department of Health, Tasmania |
| Dr Vineesh Khanna | Department of Health, Victoria |
| Dr Santosh Verghese | Department for Health and Wellbeing, South Australia |
| Professor Peter Sprivulis | Department of Health, Western Australia |
| Dr Rae Donovan | Queensland Health |
| Dr Andrew Bell | NT Health |
| Ms Beelah Bleakley | ACT Health Directorate |
| Dr Rob Hosking | Royal Australian College of General Practitioners |
| Associate Professor Shannon Nott | Australian College of Rural and Remote Medicine |
| Adjunct Professor Naomi Dobroff | Australian College of Nursing |
| Associate Professor Christine Slade | Consumer representative |
| Ms Melissa Cadzow | Consumer representative |
| Mr Matt Malone | Australian Private Hospital CIO Forum |
| Professor Matt Sabin | Healthdirect |
Reporting
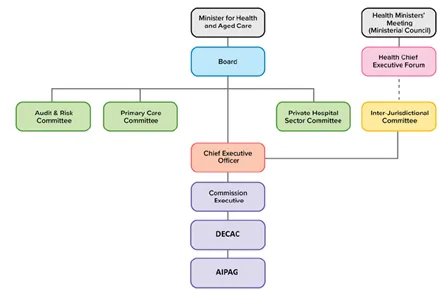
The reporting structure for DECAC is outlined in Figure 1 below.
Figure 1: Chart showing the reporting lines of the Digitally Enabled Care Advisory Committee.
Artificial Intelligence Program Advisory Group
The Artificial Intelligence Program Advisory Group (AIPAG) was established in March 2025 to provide expert advice to support the Commission's work on Artificial Intelligence (AI).
About the AIPAG
The AIPAGs purpose is to help ensure AI is safe, high quality and appropriately integrated into healthcare, including digitally enabled care.
AIPAG advises on our current and emerging AI priorities, informs future strategic directions and operates as a subcommittee of the Digitally Enabled Care Advisory Committee (DECAC).
It provides strategic advice on the safe, effective and responsible use of AI in clinical settings, including how AI can support clinical decision‑making, the risks and opportunities associated with its use, and the clinical governance arrangements needed to ensure appropriate oversight.
Through its membership, AIPAG promotes alignment across Commonwealth, state and territory AI strategies and government policies, shares real‑world experiences and guidance, and raises awareness of our AI initiatives.
By identifying emerging trends and advising on new AI developments, AIPAG helps ensure our AI work remains relevant, supports patient safety and quality care, and maximises benefits from responsible AI adoption.
Membership
AIPAG is chaired by Dr Sharon Hakkennes, Chief Digital Health Officer, Victorian Department of Health. A list of the full membership is provided below.
| Member name | Organisation |
|---|---|
| Chair – Dr Sharon Hakkennes | Victorian Department of Health |
| Ms Sallyanne Wissmann | Health Information Management Association of Australia Ltd. |
| Dr Helen Toyne | Department of Health, Disability and Ageing |
| Professor Clair Sullivan | Australian Council of Senior Academic Leaders in Digital Health |
| Dr Vincent McCauley | Flinders University |
| Dr Marcelle Noja | Therapeutic Goods Administration, Department of Health, Disability and Ageing |
| Mr Timothy Panoho | Digital Solutions Division, Australian Digital Health Agency |
| Professor Farah Magrabi | Australian Institute of Health Innovation, Macquarie University |
| Mr Thomson Delloso | Australian College of Nursing |
| Ms Judith Drake | Consumer representative |
| Dr Farhannah Aly | The Royal Australian and New Zealand College of Radiologists |
| Ms Rachel Griffiths | Australian Health Practitioner Regulation Agency |
| Professor Andrew Milat | NSW Agency for Clinical Innovation |
| Dr Janice Tan | Royal Australian College of General Practitioners |
| Professor Peter Soyer | The Australasian College of Dermatologists |
| Ms Maria Paz | Australian Private Hospital CIO Forum |
| Dr Jacqui Hoepner | Department of Health, Disability and Ageing |
| Dr Rae Donovan | eHealth Queensland |
Contact us
For questions about our work in digital health, please email our Digital Health team.
For updates and insights about our work, sign up to our mailing list.