Assessment outcomes data and lessons learnt - National Clinical Trials Governance Framework
Summary of accreditation outcomes and lessons learnt from the assessment of health service organisations (HSOs) against the National Clinical Trials Governance Framework (NCTGF).
Overview
The purpose of this page is to provide a national aggregation of assessment outcomes against the NCTGF to provide an indicator to the sector on implementation progress.
Assessments have been undertaken within HSOs (including day procedure facilities) that provide a clinical trial service. Performance is measured against a maturity scale.
- For further details on the maturity scale see Maturity rating for clinical trial service assessment - Fact sheet
- For information on when the maturity scale applies to your HSO see Advisory CT25/01: Extension of the maturity rating scale for clinical trial service assessments against NCTGF
Reporting period
The figures below provide information on the number and percentage of HSOs that have either registered or completed assessments to the NCTGF, along with the actions commonly rated initial and established systems across the sector.
The reporting period commenced in May 2023 and data is updated monthly.
Public reporting
Across the maturity scale period individual performance of an HSO against the NCTGF will not be publicly reported.
The intent of a maturity determination during assessments is to provide HSOs with a quality improvement measure for their clinical trial service ahead of formal assessment under the Australian Health Service Safety and Quality Accreditation Scheme.
Maturity scale
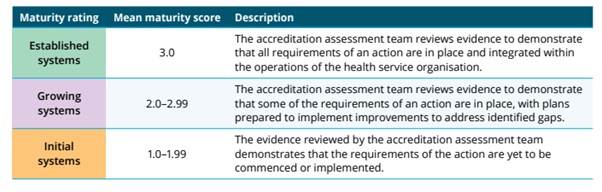
The NCTGF is made up of 27 actions across the Clinical Governance and Partnering with Consumer Standard of the NSQHS Standards. Each action is awarded a rating after assessment. The rating scale is as follows:
Registering for assessment
It is the role of state and territory health departments to identify and register eligible HSOs within their jurisdiction for assessment to the NCTGF, alongside routine processes for registering HSOs for assessment to the NSQHS Standards.
If there is uncertainty regarding accreditation requirements, contact your state and territory health department who regulates accreditation. See state and territory health department contact details.
Reporting period
The figures below provide information on the number and percentage of HSOs that have either registered or completed assessments to the NCTGF, along with the actions commonly rated initial and established systems across the sector.
The reporting period commenced in May 2023 and data is updated monthly.
Public reporting
Across the maturity scale period individual performance of an HSO against the NCTGF will not be publicly reported.
The intent of a maturity determination during assessments is to provide HSOs with a quality improvement measure for their clinical trial service ahead of formal assessment under the Australian Health Service Safety and Quality Accreditation Scheme.
Maturity scale
The NCTGF is made up of 27 actions across the Clinical Governance and Partnering with Consumer Standard of the NSQHS Standards. Each action is awarded a rating after assessment. The rating scale is as follows:

Registering for assessment
It is the role of state and territory health departments to identify and register eligible HSOs within their jurisdiction for assessment to the NCTGF, alongside routine processes for registering HSOs for assessment to the NSQHS Standards.
If there is uncertainty regarding accreditation requirements, contact your state and territory health department who regulates accreditation. See state and territory health department contact details.
Assessment outcomes dashboard
Maturity ratings
During the maturity rating scale period each applicable action of the NCTGF is rated in accordance with the maturity rating scale and the HSO given an overall maturity rating for their clinical trial service provision based on the sum of these scores.
It is important to note that even if a minor part of an applicable action is not met, then that action will be awarded a 'growing' rating and this will impact the overall rating a health service organisation receives against the maturity scale for their clinical trial service. See Maturity rating for clinical trial service assessment - Fact sheet and Advisory CT25/01: Extension of the maturity rating scale for clinical trial service assessments against NCTGF.
