Endorsements and Topic Working Group - Acute Anaphylaxis Clinical Care Standard
The standard has been endorsed by a number of key health organisations. The development of the standard was guided by an expert Topic Working Group.
Endorsing organisations
The Acute Anaphylaxis Clinical Care Standard was endorsed by the following organisations in 2021:

List of endorsing organisations:
- Allergy & Anaphylaxis Australia (A&AA)
- Australasian College of Paramedicine
- Australasian Society of Clinical Immunology and Allergy (ASCIA)
- Australasian College for Emergency Medicine (ACEM)
- Australian College of Nursing (ACN)
- Australian College of Rural and Remote Medicine (ACRRM)
- Australian and New Zealand Anaesthetic Allergy Group (ANZAAG)
- Australian and New Zealand College of Anaesthetists (ANZCA)
- Australian Primary Health Care Nurses Association (APNA)
- College of Emergency Nursing Australasia (CENA)
- Council of Remote Area Nurses plus (CRANAplus)
- National Allergy Strategy (NAS)
- Rural Doctors Association of Australian (RDAA)
- Pharmaceutical Society of Australia (PSA)
- Society of Hospital Pharmacists of Australia (SHPA)
- The Royal Australasian College of Physicians
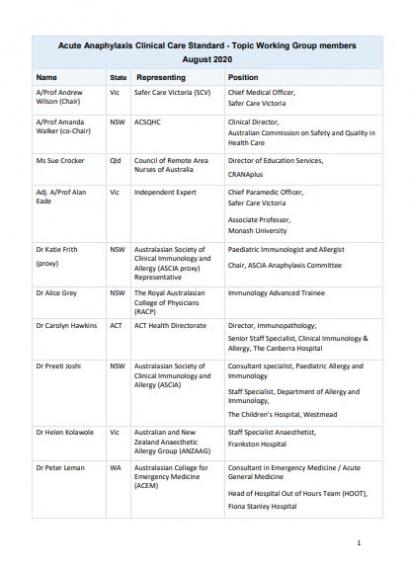
Acute Anaphylaxis Clinical Care Standard Topic Working Group
The Acute Anaphylaxis Clinical Care Standard Topic Working Group was established to provide clinical advice, expertise and consumer input during the development of the Clinical Care Standard. The group included individuals with relevant expertise and experience in the Australian health care system and representatives of some key organisations.
The role of the Topic Working Group was to:
- Advise on the scope and key components of care for the Acute Anaphylaxis Clinical Care Standard
- Advise on key sources of evidence including clinical practice guidelines, standards and empirical literature to build upon the body evidence supporting the existing model
- Advise on the formulation of quality statements and supporting indicators
- Recommend strategies to support the implementation of the Acute Anaphylaxis Clinical Care Standard
- Actively support raising awareness of the Acute Anaphylaxis Clinical Care Standard.
All members are required to disclose financial, personal and professional interests that could, or could be perceived to, influence a decision made, or advice given to the Commission.
Disclosures are updated prior to each meeting and managed in line with the Commission’s Policy on Disclosure of Interests.
Membership of the Topic Working Group
-
2020Other resource
A Topic Working Group was convened to provide advice to the Commission on this project. The group comprised individuals with relevant expertise in anaphylaxis, representatives of key stakeholder organisations as well as a consumer representative.
Updated August 2020.