Assessment to the National Clinical Trials Governance Framework
Accreditation for the provision of clinical trial services located within health service organisations (HSOs) provides assurance to the community that a healthcare service meets expected safety and quality standards. It is a formal program in which trained, independent reviewers assess HSO's evidence of implementation against applicable actions in the National Clinical Trials Governance Framework (NCTGF).
Key messages
- Assessment to the NCTGF began in May 2023
- Initially HSOs were assessed against a maturity scale for the safety and quality of their clinical trial service for their first accreditation cycle only - see Maturity rating for clinical trial service assessment - Fact sheet
- The maturity rating scale period has now been extended - see Advisory CT25/01: Extension of the maturity rating scale for clinical trial service assessments against NCTGF
- Assessments occur alongside an HSOs routine assessment to the NSQHS Standards - see Flow chart of an assessment to NSQHS Standards and Fact sheet 18: Scheduling for short notice assessment
- For advice on not-applicable actions as they relate to the NCTGF see Advisory AS18/01: Advice on not applicable actions
Outline
All public and private hospitals and day procedure services (HSOs) providing a clinical trial service are required to be assessed against applicable actions of the NCTGF relevant to their own level of service delivery. This assessment is conducted in accordance with the Australian Health Service Safety and Quality Accreditation Scheme (AHSSQA Scheme) by an accrediting agency approved by the Commission.
For HSOs already entered into a contractual agreement with an accrediting agency as part of their accreditation to the National Safety and Quality Health Service (NSQHS) Standards, this agency can also undertake an assessment to the NCTGF.
Implementation of the NCTGF will occur in two stages. In the first stage of implementation, the process will involve assessment against a maturity scale. The accrediting agency will:
- Review evidence provided by health service organisations to demonstrate clinical trial services have implemented the relevant actions and award a maturity rating for each action.
- Advise of any actions rated as initial systems at the time of assessment and share strategies for quality improvement.
- Provide an assessment report to the health service organisation within five business days of the assessment.
Accreditation to the NCTGF is conducted via a Short Notice Assessment (SNA) format. The Commission has developed resources to support health service organisations with the process.
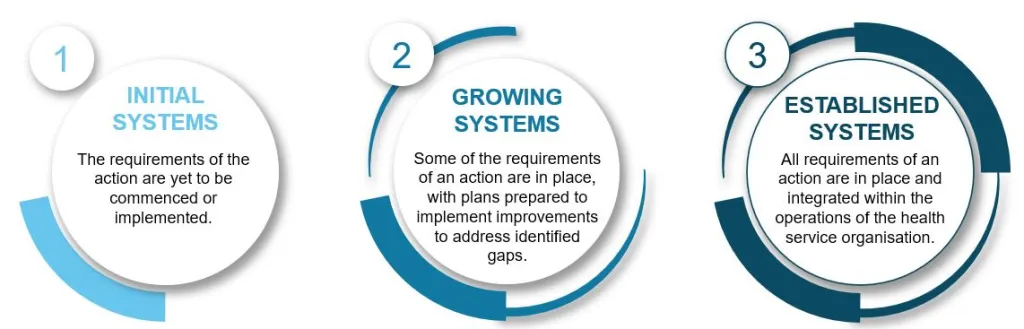
To give HSOs time to implement the NCTGF they will be assessed against a maturity scale. This means HSOs will be assessed as either having Established, Growing, or Initial systems in place to meet applicable actions of the NCTGF for clinical trial service provision. The purpose of a maturity determination as an assessment outcome during this period is to:
- introduce the clinical trials sector to the processes and language of HSO accreditation
- serve as a quality improvement guide for HSOs to receive an external review of the safety and quality of their clinical trial service
- identify gaps (major or minor) and address them by investing in ongoing safety and quality measures against applicable actions of the NCTGF
- revise and/or implement robust systems and processes to deliver safe and high-quality clinical trial services to prepare for full accreditation at the next accreditation cycle according to the NSQHS Standards under the AHSSQA Scheme.
Decision Matrix
| Established systems | Evidence to demonstrate that all requirements of an action are in place and integrated within the operations of the health service organisation |
|---|---|
| Growing systems | Evidence to demonstrate that some of the requirements of an action are in place, with plans prepared to implement improvements to address identified gaps |
| Initial systems | Evidence to demonstrate that the requirements of the action are yet to be commenced or implemented |
Beyond the maturity rating scale period, HSOs will transition fully to the assessment of their clinical trial services under the NSQHS Standards rating scale. They will be assessed as either having met or not met the actions within the NCTGF and receive 60 business days to remediate or risk accreditation of the entire health service. For information on when the maturity scale applies to your HSO see Advisory CT25/01: Extension of the maturity rating scale for clinical trial service assessments against NCTGF.
Assessment procedures, rules, and requirements
Accreditation to the NCTGF
The Australian Health Service Safety and Quality Accreditation (AHSSQA) Scheme provides for the national coordination of accreditation processes and sets out the responsibilities of accrediting agencies in relation to implementation of the NCTGF.
The AHSSQA Scheme outlines the Commission’s policy and processes for obtaining, maintaining and removing approval as an accrediting agency under the AHSSQA Scheme and includes information and guidance on the award of accreditation including:
- accreditation testimonial, certificate, and reports (p 25)
- notification of significant risks (p 23).
Accrediting agencies approved under the Scheme must comply with this Policy.
Award of accreditation
For the initial stage of implementation HSOs are assessed using a maturity scale.
Following this stage, an HSO is accredited if all actions in the NCTGF are rated as 'met' or 'met with recommendations'. See Advisory CT25/01: Extension of the maturity rating scale for clinical trial service assessments against NCTGF.
Accreditation is awarded for a period of three years. The date of commencement of the accreditation period is the date the accrediting agency formally notifies the HSO of the final outcome of the accreditation assessment.
Assessment methodology (Structure, Format, PICMoRS, Sampling)
Structure
The Commission has developed the Assessment Framework for Safety and Quality Systems (the Framework) to improve the effectiveness, rigour and consistency of assessment to the NSQHS Standards. It incorporates a structured assessment method, which can be used when interviewing members of the workforce to comprehensively review the processes that make up the safety and quality systems specified in the NCTGF.
A fact sheet on the process for accreditation for the NCTGF has also been developed by the Commission to support health services prepare for assessment and accreditation.
The format of assessments to the NSQHS Standards and the NCTGF can be on-site, desktop, virtual or hybrid although they are mostly conducted on-site at the HSO.
To provide certainty for HSOs, and facilitate accreditation assessments proceeding where possible, the Commission has developed a process for requests to conduct a hybrid or virtual assessment.
- Further information and guidance for the submission of requests
- NSQHS Standards Guidance on conducting hybrid assessments
PICMoRS
PICMoRS is a mnemonic that was designed to help assessors to undertake assessment for accreditation under the AHSSQA Scheme. Originally developed for assessment to the NSQHS Standards, it stands for:
- Process
- Improvement
- Consumer participation
- Monitoring
- Reporting and Systems
Sampling of clinical trials operated by an HSO is required for all hospitals and day procedure services to be assessed against the actions in the NCTGF.
To ensure consistency and rigor of assessments, the Commission has developed a methodology, to be applied by all accrediting agencies, when assessing the safety and quality performance of an HSO conducting clinical trials.
Applying for not applicable actions
Not all actions within the NCTGF will be applicable to HSOs providing a clinical trial service due to variations in service delivery. Where an HSO provides partial clinical trial services, implementation of the NCTGF should focus on those actions that reflect their level of clinical trial service delivery.
Not applicable actions can be identified by completing a gap analysis against the actions in the NCTGF. Any identified not applicable actions need to be considered and approved with the organisations accrediting agency and the Commission.
Mediation and appeals process
Mediation is the process of an intervention to resolve conflict. Accrediting agencies or HSOs may request the Commission mediate where there is a dispute regarding applications for not applicable actions and assessment outcomes.
If an HSO does not agree with their assessment outcome, an appeal may be submitted to their accrediting agency or directly to the Commission.
Transitioning between accrediting agencies
From time to time, an HSO may wish to change the accrediting agency they have engaged to conduct accreditation assessments. HSOs should not be prevented from transferring between accreditation service providers.
The Commission has provided Advisory AS18/05: Transitioning between accrediting agencies to clarify for accrediting agencies and HSOs their responsibilities and the recommended processes to be followed when HSOs change accrediting agencies.