Acute Anaphylaxis Clinical Care Standard
New adrenaline formulation
The Commission is aware of the availability of a new intranasal formulation of adrenaline for the treatment of severe allergic reactions. We are considering the need to review relevant content of the Acute Anaphylaxis Clinical Care Standard, including statements relating to adrenaline administration.
Further information is available from:
The goal of the Acute Anaphylaxis Clinical Care Standard is to improve the recognition of anaphylaxis, and the provision of appropriate treatment and follow-up care.
What is anaphylaxis?
Anaphylaxis is a severe form of allergic reaction that is potentially life-threatening, especially if not treated immediately. It is characterised by a sudden onset; however, the clinical presentation is variable. The diagnosis of anaphylaxis is based on clinical findings and takes the patient’s history and physical examination into consideration.
About the Standard
The Acute Anaphylaxis Clinical Care Standard was published in 2021.
The Standard includes:
- six quality statements to improve the recognition, treatment and follow-up of anaphylaxis in acute care
- a set of indicators to support monitoring and quality improvement
We also have resources for clinicians, healthcare services and consumers to support implementation of the Anaphylaxis Clinical Care Standard.
Quality Statements
Quality Statement 1 – Prompt recognition of anaphylaxis
A patient with acute-onset clinical deterioration with signs or symptoms of an allergic response is rapidly assessed for anaphylaxis, especially in the presence of an allergic trigger or a history of allergy.
For clinicians
Assess patients immediately for possible anaphylaxis if they present with rapid development of airway, breathing and/or circulation problems, with or without skin or mucosal changes. The presence of an allergic trigger or a history of allergy should heighten suspicion even if the patient is not in severe distress. Symptoms typically occur in two or more body systems, but this can be variable.
The most common triggers of anaphylaxis are food, insect venom and medicines (Table 1).
After exposure to a trigger, the time until onset of signs and symptoms of anaphylaxis (Table 2) may range from minutes to several hours.
Obtain a history from the patient, noting:
- recent exposure to substances known to cause an allergic reaction
- any known allergies for the patient, including previous reactions and treatment
- any history of anaphylaxis.
Document the time of symptom onset in the patient’s healthcare record.
Consider patient risk factors that potentially contribute to fatal anaphylaxis (for example, older age and cardiovascular and respiratory diseases) and cofactors that are likely to amplify the severity of an allergic reaction (such as exercise or acute infection).
Common differential diagnoses include acute asthma, syncope, panic attacks and septic shock. Rule out other sudden-onset multisystem illnesses. However, a patient who experiences sudden difficulty in breathing, has asthma, and is known to be at risk of anaphylaxis, should be treated as having anaphylaxis.
Monitor patients with allergy symptoms who do not meet the criteria for anaphylaxis, to allow prompt recognition of progression of a mild-to-moderate allergic reaction to anaphylaxis. Reactions can progress to severe involvement of more than one body organ system and rapidly become life-threatening.
Rehearse the anaphylaxis management protocol regularly to ensure prompt recognition of anaphylaxis for patients presenting with allergic reactions.
Consider serial measurements of mast cell tryptase concentrations. Taken during anaphylaxis, results can be useful for identifying the trigger when reviewed after the event, usually by a clinical immunology or allergy specialist.
Table 1: Triggers of anaphylaxis
| Common triggers of anaphylaxis | |
|---|---|
| Food |
|
| Medicines |
|
| Insect settings |
|
| Less common triggers of anaphylaxis | |
|---|---|
| Other foods |
|
| Topical medicines |
|
| Biological therapies |
|
| Physical |
|
| Other |
|
*An allergic reaction where the trigger is in an individual’s hormones.
Table 2: Signs and symptoms of allergic reactions
| Reaction | Signs and symptoms include one or more of the following |
|---|---|
| Mild to moderate allergic reaction |
These signs and symptoms may or may not be present with anaphylaxis. Skin and mucosal changes can be subtle or absent in up to 20% of anaphylaxis cases. |
| Anaphylaxis | Airway
Breathing
Circulation
Gastrointestinal
|
For healthcare services
Ensure that an anaphylaxis management protocol that outlines clinical criteria (consistent with the ASCIA Acute Management of Anaphylaxis guidelines) is available and used consistently by clinicians. Confirm that clinicians have the skills and competency to promptly recognise the signs and symptoms of allergic reactions, including anaphylaxis.
Ensure that systems are in place for the continuing assessment of the patient experiencing an allergic reaction, to monitor for the possible progression of symptoms to anaphylaxis. This may include drills to ensure proficiency of relevant staff in the anaphylaxis management protocol.
Reporting of incidents of delayed recognition of anaphylaxis or missed anaphylaxis as an adverse event should be included in the quality management program – for example, through incident reporting and management.
For consumers
If you have sudden difficulty in breathing, swelling of your face, tightness in your throat, persistent dizziness, hives, or other symptoms that could indicate an allergic reaction, your clinician will assess if you are experiencing anaphylaxis, the most severe form of allergic reaction. Abdominal pain with or without vomiting can also be a sign of anaphylaxis, usually for people allergic to insect bites or stings.
A reaction can occur within minutes or several hours after exposure to a trigger (also called an ‘allergen’). Your clinician will ask about food and drinks in the past few hours, medicines used and any insect bites or stings, as these are the most common triggers of anaphylaxis.
A mild or moderate allergic reaction can rapidly become severe (anaphylaxis). Be aware of the symptoms and signs of anaphylaxis so you can recognise if this is happening.
If you have an allergy or have had anaphylaxis before, it is important to let your clinician know. If you have asthma, are at risk of anaphylaxis, and experience sudden difficulty in breathing, this should be treated as anaphylaxis.
Related resources
- Allergy & Anaphylaxis Australia (A&AA) signs and symptoms video
- ASCIA e-training courses for health professionals, developed by ASCIA to provide reliable clinician education including Anaphylaxis e-training for health professionals
Quality Statement 2 – Immediate injection of intramuscular adrenaline
A patient with anaphylaxis, or suspected anaphylaxis, is administered adrenaline intramuscularly without delay, before any other treatment including asthma medicines. Corticosteroids and antihistamines are not first-line treatments for anaphylaxis.
For clinicians
Immediately on diagnosis of anaphylaxis, administer adrenaline via intramuscular (IM) injection into the mid-anterolateral thigh using a needle of appropriate length. Subcutaneous or inhaled routes for adrenaline are not recommended as they are less effective. Pregnant women experiencing anaphylaxis require the same dose of IM adrenaline as other patients. The recommended doses for IM adrenaline are indicated in Table 3.
Table 3: Recommended doses for intramuscular adrenaline
| Age (years) | Weight (kilograms) | Volume (millilitres) of adrenaline 1:1,000 ampoules | Adrenaline injector (for use instead of ampoules) |
|---|---|---|---|
| ~<1 | <7.5 | 0.1 mL | Not available |
| ~1-2 | 10 | 0.1 mL | 7.5-20kg (~<5 years) 150 microgram device |
| ~2-3 | 15 | 0.15 mL | 7.5-20kg (~<5 years) 150 microgram device |
| ~4-6 | 20 | 0.2 mL | 7.5-20kg (~<5 years) 150 microgram device |
| ~7-10 | 30 | 0.3 mL | >20kg (~>5 years) 300 microgram device |
| ~10-12 | 40 | 0.4 mL | >20kg (~>5 years) 300 microgram device |
| >12 and adults | >50 | 0.5 mL | >50kg (~>12 years) 300 microgram or 500 microgram device |
Delayed administration of adrenaline is a risk factor for fatal anaphylaxis. If anaphylaxis is suspected in the presence of an allergy or anaphylaxis history, or following exposure to a potential allergen, it is safer to administer adrenaline early than to wait for progression, which may be hard to reverse. There are no absolute contraindications to adrenaline administration in anaphylaxis.
In most situations, IM adrenaline is preferred and is safer than the intravenous (IV) route. Adverse events have been reported in adult patients who received overdoses of IV adrenaline, but these are rare with IM adrenaline.
An IV adrenaline infusion should only be administered when clinically appropriate, and:
- by clinicians trained in the use of IV adrenaline
- in a critical care setting where there is appropriate haemodynamic monitoring available.
Repeated IM adrenaline injections can be given at five-minute intervals if the patient’s symptoms are not improving. Escalate care as per organisational protocols if the patient’s condition is not improving after two to three doses of adrenaline.
Do not administer corticosteroids or antihistamines first-line, as they are not effective in treating anaphylaxis. Corticosteroids have a delayed effect of four to six hours and are adjuncts in the management of anaphylaxis – they do not replace adrenaline. Antihistamines are only helpful for relieving associated urticaria (hives), angioedema and itch. Do not give promethazine or other sedating antihistamines, as the sedating effect can mask deterioration or a biphasic reaction. Injecting promethazine can worsen hypotension and cause muscle necrosis.
Consider the implications of the treatment provided in the healthcare facility and what this communicates regarding adrenaline use. Avoiding adrenaline use in the case of a severe allergic reaction, or preferentially using corticosteroids, bronchodilators or antihistamines, may inadvertently give a message to patients that they should delay using their adrenaline injector, thus increasing potential risk in a subsequent anaphylaxis.
Include a ‘when required’ (prn) order for IM adrenaline on an admitted patient’s medication chart if they have a known allergy and have been prescribed an adrenaline injector. This can expedite the administration of IM adrenaline if the patient experiences anaphylaxis while in care.
For healthcare services
Ensure that there is a protocol for the management of anaphylaxis in place that supports prompt administration of IM adrenaline by all relevant clinicians, including nurses. The use of protocols can significantly improve IM adrenaline injection rates for anaphylaxis.
Ensure that all clinical areas have access to adrenaline for the treatment of anaphylaxis, and specify access arrangements in the protocol for the management of anaphylaxis. This will ensure that adrenaline is readily accessible to any clinician who may administer it, including prn orders for IM adrenaline.
Ensure that clinicians have training in the management of anaphylaxis and are practised using adrenaline injector or pen devices. Ensure adrenaline injector practise devices are available; the closest adrenaline may be the patient’s own injector device.
The use of ‘anaphylaxis management’ cards for an anaphylaxis event can serve as a cognitive aid when rehearsing the protocol for an event.
Consider providing access to adrenaline in readily identifiable anaphylaxis kits for emergency use, to reduce the time to administration of intramuscular adrenaline. The anaphylaxis kit may be placed on the resuscitation trolley and should be easily distinguished from the intravenous adrenaline for cardiac emergencies. An anaphylaxis kit also reduces the risk of an inadvertent IV overdose of adrenaline for anaphylaxis.
For consumers
If a clinician believes you are experiencing anaphylaxis, they will immediately give you an injection of adrenaline into the outer mid-thigh muscle.
When you recognise the signs of anaphylaxis, use your adrenaline injector without delay (if you have been prescribed one) and call for help immediately. Give the intramuscular injection of adrenaline into your outer mid-thigh. Using your adrenaline injector when you first have symptoms of anaphylaxis can help reverse the allergic reaction and prevent it from becoming life-threatening. If you are not sure, it is safer to use adrenaline than to wait for your symptoms to get worse. The adrenaline injection should work within minutes. If you do not start to feel better after 5 minutes, use a second adrenaline injector, if you have one.
Adrenaline lessens the effects of anaphylaxis by reducing throat swelling, opening the airways, and maintaining heart function and blood pressure.
Other medicines (including non-sedating antihistamines and asthma medicines) that relieve symptoms such as itchy or red skin and breathlessness should only be used after adrenaline, and will be prescribed and/or given if considered necessary.
Related resources
- Adrenaline injector practise devices are available and commonly sourced from the Allergy and Anaphylaxis Australia website.
- EpiPen and Anapen training videos are available on Allergy & Anaphylaxis Australia website.
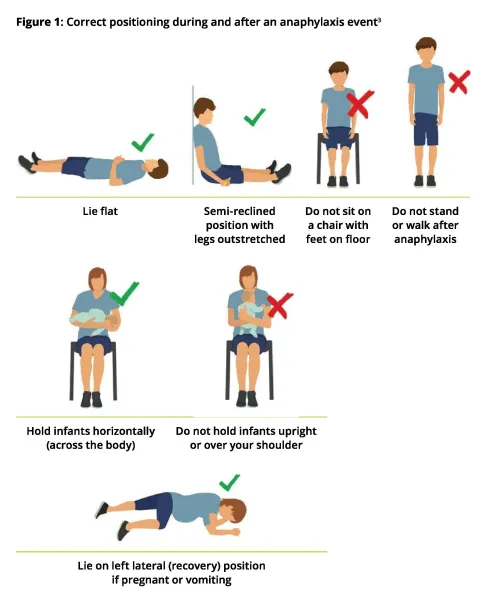
Quality Statement 3 – Correct patient positioning
A patient experiencing anaphylaxis is laid flat, or allowed to sit with legs extended if breathing is difficult. An infant is held or laid horizontally. The patient is not allowed to stand or walk during, or immediately after the event until they are assessed as safe to do so, even if they appear to have recovered.
For clinicians
Ensure the patient is in a supine position; do not allow them to stand or walk. Monitor the patient’s blood pressure and elevate their legs if their blood pressure is low. Fatality can occur within minutes if a patient stands or sits up suddenly while they have inadequate perfusion (see Figure 1).
A semi-reclined position sufficient to relieve respiratory distress is allowed while the patient is monitored carefully for any circulatory collapse. Patients who are pregnant or vomiting should be placed on their side in the left lateral (recovery) position.
Infants should be held horizontally (across the body). They must not be held upright or over a shoulder.
Ensure the patient understands that they are not to stand up suddenly or walk until after they have been treated and assessed, and why this is important. Communicate this to other staff caring for the patient.
Assess for circulatory stability after the patient has been adequately treated and before they are allowed to mobilise. This is usually a minimum of one hour after one dose of adrenaline, and four hours if more than one dose of adrenaline is administered.
For healthcare services
Ensure acute anaphylaxis management protocols are in place to:
- provide guidance on appropriate positioning for patients with anaphylaxis
- specify that patients should not stand or walk until assessed as safe to do so after treatment with adrenaline.
For consumers
When you are experiencing anaphylaxis, you will be advised to lie flat, or sit with your legs outstretched if breathing is difficult. Your legs can be elevated if you feel faint. An infant should be held horizontally (across your body). Do not hold an infant upright or over your shoulder. If you are pregnant you should lie on your left side to ensure continued blood circulation to your baby.
If you stand up too quickly after anaphylaxis, your blood pressure may drop dangerously. You should not stand up after having adrenaline - wait until a clinician assesses it is safe for you to get up. Do not stand or walk anywhere, even to the bathroom, ambulance or into the emergency department. This is usually a minimum of one hour after one dose of adrenaline, or four hours if more than one dose is given.
Quality Statement 4 – Access to a personal adrenaline injector in all healthcare settings
A patient who has an adrenaline injector has access to it for self-administration during all healthcare encounters. This includes patients keeping their adrenaline injector safely at their bedside during a hospital admission.
For clinicians
For adrenaline to be given as soon as possible after the onset of symptoms of anaphylaxis, it is important for the patient (or their carer) to be able to immediately administer their own adrenaline injector, regardless of the setting. A readily accessible adrenaline injector may also be used by a clinician if necessary.
If a patient has an adrenaline injector, as soon as possible following presentation or admission, assess their capacity to safely use it during the healthcare encounter. This includes:
- checking their capacity, physical capability and willingness to use their personal adrenaline injector and their ability to recognise the symptoms of anaphylaxis
- considering medicines administered during the healthcare visit that may impair the patient’s usual ability to recognise and treat anaphylaxis
- involving a parent, guardian or carer in the assessment when in the paediatric setting
- involving a family member or carer in the assessment, if appropriate, when a patient is cognitively impaired or lives with a disability.
As part of the assessment, identify a safe place for the adrenaline injector to be kept that allows ease of access for the patient, is in an unlocked location, and maximises the safety of others. Before making the adrenaline injector available, ensure it is suitable for use: check the expiry date, check the viewing window to make sure the solution is clear and that the device has not been used, and ask the patient how it has been stored. If in doubt, make arrangements for the patient to obtain a new device.
The adrenaline injector should be:
- kept with the patient’s ASCIA Action Plan for Anaphylaxis
- labelled with the patient’s name.
Notify all staff that the patient has an allergy and has an adrenaline injector with them. This includes at clinical handover in the ward or when the patient temporarily leaves the ward, such as for scans or other tests. Add an allergy alert to the patient’s records, including the electronic medical record (EMR) and electronic patient journey boards.
For healthcare services
Ensure a policy, and the necessary protocols, are in place to allow a patient who has a personal adrenaline injector to maintain easy access to it at all times in a manner that is safe to others. The policy should describe steps for:
- checking the expiry date of the adrenaline injector
- checking the device to make sure the solution is clear, does not appear to have been used, and has been stored appropriately
- obtaining a replacement device if there are doubts about the integrity of the patient’s injector
- assessing the patient’s capacity to administer the injector safely during their hospital admission
- involving the parent, guardian or carer, if appropriate
- identifying a readily accessible location
- ensuring that access does not compromise the safety of others.
Ensure clinical communication processes are in place for alerting staff to the patient’s allergy and anaphylaxis risk (for example, via EMR and patient records) at clinical handover and during transitions of care, and of the need to ensure the patient has access to the adrenaline injector for self-administration at all times, including while in the hospital. In addition, the policy should ensure that nurses are aware the patient has their own adrenaline injector device and that hospital staff can administer the device in an emergency, if this is the closest available adrenaline.
For consumers
If you have a personal adrenaline injector (such as an EpiPen or Anapen) and know how to use it, you should:
- keep it close by while you are being treated in a health service, hospital, ambulance or clinic
- tell your healthcare team that you have an adrenaline injector and arrange with them to keep it near you during your care
- keep the adrenaline injector with your ASCIA Action Plan for Anaphylaxis in an unlocked location that you can easily reach.
If your child is admitted to hospital, their adrenaline injector can be kept at their bedside for you or staff to use if necessary.
Your healthcare team may want to confirm that you know how and when to use your adrenaline injector, and that it is safe to use.
If you believe you are having an allergic reaction and experience symptoms such as breathing difficulties, faintness, swelling of your tongue or tightness of your throat while in health care, lie down (or sit with your legs outstretched if breathing is difficult), use your adrenaline injector without delay and alert a staff member immediately.
Related resources
The Safer Care Victoria change package, Use of a patient’s own adrenaline (epinephrine) autoinjector in hospital, includes an in-hospital checklist for assessing the ability of patients to use their own adrenaline injectors.
Quality Statement 5 – Observation time following anaphylaxis
A patient treated for anaphylaxis remains under clinical observation for at least 4 hours after their last dose of adrenaline, or overnight as appropriate according to the Australasian Society of Clinical Immunology and Allergy Acute Management of Anaphylaxis guidelines. Observation timeframes are determined based on assessment and risk appraisal after initial treatment.
For clinicians
Observe patients for at least four hours after the last injection of adrenaline following anaphylaxis. Reassess the patient after four hours. Consider the severity of the reaction, concomitant conditions and history of anaphylaxis when deciding if more time is needed.
When the initial injection of adrenaline for anaphylaxis is administered in general practice or another primary care location, where observation for four hours is not possible, arrange ambulance transfer to an appropriate facility for clinical observation.
Prolonged and biphasic reactions may occur. Biphasic reactions are estimated to occur following 3–20% of anaphylactic reactions, and cannot be predicted.
As per the ASCIA Acute Management of Anaphylaxis guidelines, a patient should be observed overnight if they:
- had a severe reaction (hypotension or hypoxia)
- required repeated doses of adrenaline
- have a history of severe asthma or protracted anaphylaxis
- have other concomitant illnesses, such as asthma, chest infection or arrhythmia
- live alone or are remote from medical care
- have known systemic mastocytosis
- presented for health care late in the evening
- cannot easily replace their adrenaline injector on discharge and have no other adrenaline injector.
For healthcare services
Ensure protocols align with ASCIA guidelines and that systems and processes are in place for patients to undergo clinical observation for the appropriate length of time.
For consumers
When you have been treated for anaphylaxis, you will be kept under clinical observation for at least four hours after the last injection of adrenaline. Adrenaline has a short duration of action and wears off quickly.
Occasionally, some people have another episode of anaphylaxis without coming in contact with their allergic trigger and require further treatment with adrenaline. This is called ‘biphasic anaphylaxis’. A clinician will review your risk of recurrence of anaphylaxis and re-exposure before you are discharged.
In some cases, you may need to be admitted overnight for observation after having anaphylaxis if:
- you have received more than one dose of adrenaline to treat your anaphylaxis
- you have a history of severe asthma
- you have arrived late in the evening
- you live alone or a long way from healthcare services
- your adrenaline injector cannot be replaced before you get home and you do not have another one.
Quality Statement 6 – Discharge management and documentation
Before a patient leaves a healthcare facility after having anaphylaxis, they are advised about the suspected allergen, allergen avoidance strategies and post-discharge care. The discharge care plan is tailored to the allergen and includes details of the suspected allergen, the appropriate ASCIA Action Plan, and the need for prompt follow-up with a general practitioner and clinical immunology/allergy specialist review. Where there is a risk of re-exposure, the patient is prescribed a personal adrenaline injector and is trained in its use. Details of the allergen, the anaphylactic reaction and discharge care arrangements are documented in the patient’s healthcare record.
For clinicians
Discuss the suspected allergen with the patient and their carer and provide advice about allergy avoidance. Tailor the patient’s discharge care to the suspected allergen and risk of re-exposure to ensure adequate follow-up and preventive measures.
In most cases this will include:
- an ASCIA Action Plan
- advice about the allergen or suspected allergen and how to avoid it
- advice about follow-up visits with their GP and a clinical immunology/allergy specialist
- the Commission Anaphylaxis Discharge Checklist and Discussion Guide
- prescribing personal adrenaline injector(s), where there is a risk of re-exposure.
See Figures 2 & 3 for discharge requirements according to the type of allergen.
Document the allergen or suspected allergen in the patient’s health record, including food, medicine, and sting or bite exposure in the hours before anaphylaxis. For medicine allergies, include details of the adverse reaction. Upload the anaphylaxis event and document their allergy status in the patient’s My Health Record when local health service arrangements allow.
Provide the discharge documents, including the suspected allergen, discharge care plan and the patient’s ASCIA Action Plan to their general practitioner or ongoing clinical provider within 48 hours of discharge. For patients with a medicine allergy, ASCIA provide a template for documenting the reaction - the ASCIA Record for Drug (Medication) Allergy. This, or a local or electronic equivalent, should be completed and provided to the patient to assist patient information and clinical communication.
Recognise the degree of anxiety the patient and/or their family may experience after an anaphylaxis event. Provide the patient with information about support available in the community, such as Allergy & Anaphylaxis Australia (1300 728 000), and the Australasian Society of Clinical Immunology and Allergy.
For healthcare services
Ensure systems, policies and protocols are in place for clinicians to tailor discharge requirements to the patient’s needs and provide relevant documentation, as described in Figures 2 and 3, as appropriate to the local setting. Consider how to incorporate the tools, templates and resources described in this standard into policies, procedures and guidelines. This should include the ability to:
- Provide information on anaphylaxis and allergy management, including appropriate written patient information such as the Commission Anaphylaxis Discharge Checklist and Discussion Guide, and patient information from ASCIA and Allergy & Anaphylaxis Australia.
- Provide tailored action plans including the ASCIA Action Plan for Anaphylaxis and/or ASCIA Action Plan for Drug (Medication) Allergy
- Document the allergic reaction and provide a record to the patient - for medicines allergies, use the ASCIA Record for Drug (Medication) Allergy or local electronic equivalent.
- Supply a personal adrenaline injector at discharge - or provide a prescription where this can be dispensed immediately upon leaving the facility - for patients at risk of re-exposure to the allergen (note: where eligible, up to two injectors may be prescribed with a PBS Authority prescription). Arranging supply of an adrenaline injector after hours may be required in some local settings.
- Provide training to patients and carers on the use of the adrenaline injector when supplied or prescribed, from staff competent to provide this training using an appropriate training device.
Ensure processes are in place for appropriate documentation of the details of the suspected allergen and the allergen exposure in the patient’s record. For medicine allergies processes for documenting and reporting details of the adverse reaction should be adhered to, including accurate documentation in the healthcare record and the discharge plan provided to the GP and other clinicians providing ongoing care. Ensure processes are in place for documenting the anaphylaxis event and their allergy status in the patient’s My Health Record, when local health service arrangements allow.
For consumers
Before you are discharged from hospital or a healthcare service, your clinician will talk to you about the cause or ‘trigger’ for the anaphylaxis (if known), and how to manage your allergy. These triggers are also called allergens. It is important that you know the trigger for your anaphylaxis so you can avoid it. You also need to know how to recognise an allergic reaction and what to do in case of another severe allergic reaction. In some cases, your trigger may not be known and further tests may be needed.
Before you are discharged it is important that you receive:
- information about your allergic trigger and how to avoid it
- an ASCIA Action Plan that includes information about:
- how to recognise an allergic reaction including anaphylaxis. Anaphylaxis may be different each time, so it is important that you can recognise all of the symptoms
- how to use the adrenaline injector, if prescribed.
- advice to see your general practitioner (GP) promptly, within one week
- information on how to arrange an appointment with a clinical immunology/allergy specialist. If this is your first anaphylaxis event, the specialist will help to confirm the cause of your anaphylaxis, and advise you about how to manage your allergy. Ask your GP to refer you to an allergy specialist as soon as possible, if arrangements are not made by the hospital. If you already have a regular specialist, arrange to see them for follow-up
- advice about wearing special jewellery to identify that you have an allergy.
If there is a risk of re-exposure to the trigger, you will also be given a personal adrenaline injector or a prescription for this medicine. If you are given a prescription, it is very important that you go to a pharmacy to get the adrenaline injector as soon as possible, preferably on the way home. Anaphylaxis could occur at any time and you will need to keep an adrenaline injector with you all the time. You, and your family or carer, should be trained on how to use the adrenaline injector.
If your anaphylaxis was caused by a medicine, you will be given an ASCIA Action Plan for Drug (Medication) Allergy and a record of the details of your drug allergy such as an ASCIA Record for Drug (Medication) Allergy. These will be filled out with your details. In the future, you will need to tell healthcare staff who may prescribe, or provide you with medicines, about your allergy. It is important that you know the medicine’s active ingredient name so that so you can avoid it, and that this is accurately recorded in your healthcare record.
You can also enter or update information about your allergies within your My Health Record. A guide for consumers can be found here.
You can use the ASCIA Event record for allergic reactions to make a record of the anaphylaxis event.
Information for ongoing support services available in the community, such as the Allergy & Anaphylaxis Australia information and advice line (1300 728 000), and Australasian Society of Clinical Immunology and Allergy (ASCIA) information leaflets and website will be given to you.
Related resources
Support services available in the community include:
- ASCIA leaflets and website
- anaphylaxis for patients, consumer and carers (translated resources)
- anaphylaxis fact sheet for parents (English and other languages)
- Allergy & Anaphylaxis Australia (A&AA) leaflets, videos, training, and information and advice line (1300 728 000)
- Report an allergic reaction from food eaten at a restaurant or café at the Food Allergy Aware website
- Getting set up with My Health Record: A guide for consumers
- ASCIA Action Plans for Anaphylaxis and ASCIA Action Plan for Drug (Medication) Allergy
- ASCIA Record for Drug (Medication) Allergy
Information about trigger avoidance
Indicators
Indicators
The Commission has developed a set of indicators to support clinicians and healthcare services to monitor how well they are implementing the care recommended in this Clinical Care Standard. The indicators are intended to support local quality improvement activities. No benchmarks are set for these indicators by the Commission.
When using the indicators, please refer to the definitions required to collect and calculate indicator data which are specified online at METEOR.
You can find a description of each indicator below with links to its individual specifications.
List of indicators
Cultural safety and equity for Aboriginal and Torres Strait Islander peoples
Health outcomes for Aboriginal and Torres Strait Islander peoples can be improved by addressing systemic racism and other root causes that reduce access to care. Historical and current contributing factors include a lack of culturally safe care, culturally appropriate health education and sociocultural determinants such as differences in employment opportunities.
The considerations for improving cultural safety and equity in this Clinical Care Standard focus primarily on overcoming cultural power imbalances and improving outcomes for Aboriginal and Torres Strait Islander people through better access to health care
Cultural safety and equity recommendations in this document have been developed in consultation with Aboriginal and Torres Strait Islander individuals, clinicians and representative health service organisations. However, it is recognised that cultural safety is determined by the Aboriginal and Torres Strait Islander individuals, families and communities experiencing the care.
Recommendations
When implementing this Clinical Care Standard, cultural safety can be improved through embedding an organisational approach such as described in the recommendations below. Specific considerations for cultural safety for people undergoing colonoscopy are provided throughout this Standard.
When providing care for Aboriginal and Torres Strait Islander people, particular consideration should be given to the following recommendations.
Building culturally safe systems
- Ensure systems and processes support people to self-report their Aboriginal and Torres Strait Islander status and to record self-identification.
- Ensure all staff engage regularly in cultural safety training.
- Implement the six actions for Aboriginal and Torres Strait Islander Health from the NSQHS Standards.
Flexible and connected service delivery
- Provide flexible service delivery to optimise attendance and help develop trust with individual Aboriginal and Torres Strait Islander people and communities.
- Establish robust communication channels and referral pathways with primary healthcare providers (including Aboriginal Community Controlled Health Organisations [ACCHOs]).
- Where possible, provide outreach services close to home, on Country or in collaboration with ACCHOs or other community healthcare providers.
Communication and person-centred care
- Take a collaborative approach to ensure that interventions are suitably tailored to the individual’s personal needs and preferences for care.
- Encourage the inclusion of support people, family and kin or the person’s trusted healthcare provider (such as their ACCHO) in all aspects of care, including decision making and planning treatment and management.
- Engage culturally appropriate interpreter services and cultural translators when this will assist the patient.
- Involve Aboriginal and Torres Strait Islander Health Workers or Aboriginal and Torres Strait Islander Health Practitioners as part of a patient’s multidisciplinary team and involve Aboriginal and Torres Strait Islander Liaison Officers in hospital settings.
- Use culturally and linguistically appropriate materials to aid in communication and discussion, accounting for varying levels of health literacy.
Resource hub
Implementation resources are resources developed by the Commission that will assist in implementing and understanding the Clinical Care Standards. They include short guides to the Standards for consumers, clinicians and healthcare services, and other tools and resources to support implementation.
Related resources are other resources that the Commission has identified as relevant and useful. Most often, these come from sources outside the Commission. They may include additional information, guidelines, tools and consumer materials.
For clinicians and healthcare services
Related resources
Action plans
Training
ASCIA e-training courses for health professionals developed by ASCIA to provide reliable clinician education
Training and information about allergies and anaphylaxis
Adrenaline autoinjectors
- Adrenaline injector practise devices are available and commonly sourced from the Allergy & Anaphylaxis Australia website: allergyfacts.org.au/shop/training-accessories
- EpiPen and Anapen training videos are available on Allergy & Anaphylaxis Australia website: allergyfacts.org.au/resources/videos-from-a-aa/how-to-give-epipen
Change policy for use of a patient’s own adrenaline autoinjector in hospital
Use of a patient’s own adrenaline (epinephrine) autoinjector in hospital includes an in-hospital checklist for assessing the ability of patients to use their own adrenaline injectors.
For consumers
Implementation resources
Consumer Guide - Acute Anaphylaxis Clinical Care Standard
Steps to stay safe
It is important for people to know what to do after experiencing anaphylaxis. Our resources will help educate people who are at risk of anaphylaxis to manage their condition safely.
If you are at risk, have a conversation with your healthcare professional to ensure you are kept safe and feel equipped to manage your allergy. This includes knowing the signs and symptoms of anaphylaxis, knowing your allergy triggers and knowing what to do if you have symptoms. It is also vital to keep your adrenaline injector with you at all times, if you have been advised by your clinician to have one.
Five steps to stay safe after anaphylaxis
If you have had anaphylaxis, it is important to know what you need to do to manage your allergy and stay safe, especially after a hospital visit for anaphylaxis.
- Know the signs and symptoms – they can differ each time
- Manage your allergy and avoid triggers – to prevent future episodes
- Have an ASCIA Action Plan – so you, or others, can act fast in an emergency
- Always keep your adrenaline injector close by – and know how to use it
- Follow-up with your GP and specialist – to know how to best manage your allergy
Talk to your healthcare professional if you have questions.
Poster
Our poster outlines what you need to know after an episode of anaphylaxis and where to find more information.
Health services can print this poster and display in their hospital, clinic or practice waiting room, to increase awareness of how to stay safe after anaphylaxis.
5 Steps to Stay Safe after Anaphylaxis - poster
Anaphylaxis discharge checklist and discussion tool
This practical checklist explains what you need to do when you leave hospital to go home after anaphylaxis.
Before you leave hospital, your healthcare professional will use this checklist to have a conversation about how you can manage your allergy and reduce your future risk of anaphylaxis, and make sure you know what you can do to stay safe when you go home.
Related resources
Allergy & Anaphylaxis Australia (A&AA)
- A&AA provide information and advice about living with allergy and anaphylaxis including leaflets, videos, training
- Call the information and advice line 1300 728 000 or visit www.allergyfacts.org.au
Australasian Society of Clinical Immunology and Allergy (ASCIA)
- Patient information
- Anaphylaxis for patients, consumer and carers (translated resources)
- Anaphylaxis fact sheet for parents (English and other languages)
National Allergy Strategy
Training and information about allergies and anaphylaxis
Positioning of a person having anaphylaxis
- National Allergy Strategy animation: nationalallergystrategy.org.au/resources/anaphylaxis
After an anaphylaxis event, you may want to:
- Keep a record of the anaphylaxis event to give your doctor or other health professional, using ASCIA's Anaphylaxis Event Record form
- Add allergy information to your My Health Record, following guidance from the National Allergy Strategy
- Report an allergic reaction from food eaten at a restaurant or café, via Food Allergy Education reporting.
Related resources are also provided within the relevant quality statement.
Communication resources
The Commission has a suite of promotional resources to assist with implementation of the national Acute Anaphylaxis Clinical Care Standard.
This infographic outlines key data highlights on anaphylaxis in Australia, to support the release of the new Standard.
National launch webcast
The Acute Anaphylaxis Clinical Care Standard was launched in November 2021. Watch the launch recording to see experts in paramedicine, emergency medicine, allergy and general practice discuss and explain the new Standard.
Listen to the host Associate Professor Amanda Walker and the panel of experts as they discuss key areas for improvement addressed by the standard including prompt recognition of anaphylaxis, appropriate treatment, and safe discharge and follow-up care.
More about the Standard
What is the background to the Standard?
Over four million Australians live with allergies. Food allergy, for example, occurs in around 10% of infants, 4-8% of children, and 2% of adults in Australia.
While not everyone with an allergy is at risk, recent studies show increasing incidence of all-cause anaphylaxis in Australia, the United Kingdom and the United States. In Australia, in the five years to 2019-20:
- anaphylaxis presentations to emergency departments in public hospitals grew by 51% - to more than 11,594 in 2019-20.
- anaphylaxis hospital admissions increased by 35% - from 9,042 in 2015-16 to 12,179 in 2019-20.
Despite well-recognised guidelines, care is not always provided as recommended. In a study in eight Australian emergency departments, 27% of reactions consistent with anaphylaxis were not given adrenaline. Analysis of fatalities recorded by the Australian Bureau of Statistics between 1997-2013 highlighted gaps between guideline recommendations and patient care, such as delayed treatment with adrenaline.
Read more about the scope and goal of this Standard or see further background in the Acute Anaphylaxis Clinical Care Standard.
Where does the Standard apply?
This Standard applies to care provided in the following care settings:
- hospitals, including public and private hospitals, subacute facilities, day procedure services and outpatient clinics
- emergency services, such as ambulance services
- radiology and imaging services
- general practices
- other primary healthcare settings, such as Aboriginal Controlled Health Services and community pharmacies.
Not all quality statements in this Standard will be applicable to every healthcare service or clinical unit. Healthcare services should consider their individual circumstances in determining how to apply each statement.
When implementing this Standard, healthcare services should consider:
- the context in which care is provided
- local variation
- quality improvement priorities of the individual healthcare service.
In rural and remote settings, different strategies may be needed to implement the standard. For example, the use of:
- hub‑and‑spoke models integrating larger and smaller health services and ACCHOs
- telehealth consultations
- multidisciplinary teams including allied health involvement where clinically appropriate.
The Standard relates to the care provided to adults, children and infants when they are experiencing anaphylaxis – from initial presentation to a healthcare setting or first clinical contact in the community, through to discharge and planning for follow-up care. It also applies to many patients who experience anaphylaxis while in a healthcare facility.
National Safety and Quality Health Service Standards
Monitoring the implementation of Clinical Care Standards helps healthcare services to meet some of the requirements of the:
- National Safety and Quality Health Service Standards (NSQHS Standards) for acute services
- National Safety and Quality Primary and Community Healthcare Standards (Primary and Community Healthcare Standards) for services that deliver health care in a primary and/or community setting.
Find out more about how healthcare services are expected to implement the national standards in How to use the Clinical Care Standards.
How does the Standard support cultural safety and equity?
The Commission is committed to supporting healthcare services to provide culturally safe and equitable healthcare to all Australians.
Person-centred care recognises and respects differences in individual needs, beliefs, and culture. The Commission:
- is committed to supporting healthcare services to provide culturally safe and equitable healthcare to all Australians
- acknowledges that discrimination and inequity are significant barriers to achieving high‑quality health outcomes for some patients from culturally and linguistically diverse communities.
Culturally safe service provision and environments are those where the places, people, policies and practices foster mutual respect, shared decision making, and an understanding of cultural, linguistic and spiritual perspectives and differences. Cultural safety is supported by organisations and individuals that recognise cultural power imbalances and actively address them by:
- ensuring access to and use of interpreter services or cultural translators when this will assist the patient and aligns with their wishes
- providing visual or written information in a language that the patient, their family and carers will understand
- providing cultural competency training for all staff
- encouraging clinicians to review their own beliefs and attitudes when treating and communicating with patients
- identifying variation in healthcare provision or outcomes for specific patient populations, including those based on ethnicity, and responding accordingly.
Which key organisations have endorsed the Standard?
The Acute Anaphylaxis Clinical Care Standard has been endorsed by 16 key organisations:
- Allergy & Anaphylaxis Australia (A&AA)
- Australasian College of Paramedicine
- Australasian Society of Clinical Immunology and Allergy (ASCIA)
- Australasian College for Emergency Medicine (ACEM)
- Australian College of Nursing (ACN)
- Australian College of Rural and Remote Medicine (ACRRM)
- Australian and New Zealand Anaesthetic Allergy Group (ANZAAG)
- Australian and New Zealand College of Anaesthetists (ANZCA)
- Australian Primary Health Care Nurses Association (APNA)
- College of Emergency Nursing Australasia (CENA)
- Council of Remote Area Nurses plus (CRANAplus)
- National Allergy Strategy (NAS)
- Rural Doctors Association of Australian (RDAA)
- Pharmaceutical Society of Australia (PSA)
- Society of Hospital Pharmacists of Australia (SHPA)
- The Royal Australasian College of Physicians
Who was consulted on the Standard’s development?
The Commission develops Clinical Care Standards taking into account:
- advice from multidisciplinary topic working groups which include clinicians, consumers, and researchers
- consultation with key stakeholders including consumer bodies, professional organisations, and state and territory health departments.
The Acute Anaphylaxis Clinical Care Standard Topic Working Group provided expert advice on the review of the Standard. In addition, a public consultation process was conducted with key stakeholders.
Acute Anaphylaxis Clinical Care Standard - Topic Working Group
The main roles of the Topic Working Group were to:
- advise on the scope and key components of care for the Standard
- advise on key sources of evidence including clinical practice guidelines, standards and empirical literature to build upon the body evidence supporting the existing model
- advise on the formulation of quality statements and supporting indicators
- recommend strategies to support the implementation of the Standard
- actively support raising awareness of the Standard.
All members are required to disclose financial, personal and professional interests that could, or could be perceived to, influence a decision made, or advice given to the Commission. Disclosures are updated prior to each meeting and managed in line with the Commission’s Policy on Disclosure of Interests.
What was the evidence base for this Standard?
The quality statements in the Standard are based on the best available evidence and guideline recommendations at the time of development.
Further information is available on the evidence sources underpinning the Standard.
Evidence Sources – Acute Anaphylaxis Clinical Care Standard
Key sources that underpin the Standard are:
- Current clinical guidelines from the Australasian Society of Clinical Immunology and Allergy (ASCIA), including Acute Management of Anaphylaxis (2021)
- The Safer Care Victoria standard, Anaphylaxis Clinical Care Standard (2019).