Assessment outcomes data for clinical trial services
Summary of accreditation outcomes and lessons learnt from the assessment of health services against the National Clinical Trials Governance Framework.
Overview
This page provides a national aggregation of assessment outcomes against the National Clinical Trials Governance Framework (NCTGF) to provide an update to the sector on implementation progress.
Assessments have been undertaken within health services (including day procedure facilities) that provide a clinical trial service. Performance is measured against a maturity scale.
For further details on the maturity scale see Maturity rating for clinical trial service assessment - Fact sheet.
For information on when the maturity scale applies to your health service see Advisory CT25/01: Extension of the maturity rating scale for clinical trial service assessments against NCTGF.
Reporting period
The figures below provide information on the number and percentage of health services that have either registered or completed assessments to the NCTGF, along with the actions commonly rated initial and established systems across the sector.
The reporting period commenced in May 2023 and data is updated monthly.
Public reporting
Across the maturity scale period individual performance of a health service against the NCTGF will not be publicly reported.
The intent of a maturity determination during assessments is to provide health services with a quality improvement measure for their clinical trial service ahead of formal assessment under the Australian Health Service Safety and Quality Accreditation Scheme.
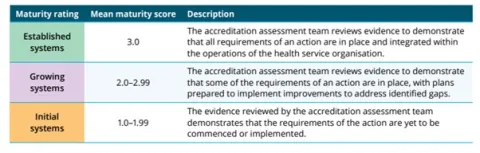
Maturity scale
The NCTGF is made up of 27 actions across the Clinical Governance and Partnering with Consumer Standard of the NSQHS Standards. Each action is awarded a rating after assessment. The rating scale is as follows:
Registering for assessment
It is the role of state and territory health departments to identify and register eligible health services within their jurisdiction for assessment to the NCTGF, alongside routine processes for registering health services for assessment to the NSQHS Standards.
If there is uncertainty regarding accreditation requirements, contact your state and territory health department who regulates accreditation.
Assessment outcomes dashboard
Maturity ratings
During the maturity rating scale period each applicable action of the NCTGF is rated in accordance with the maturity rating scale and the health service is given an overall maturity rating for their clinical trial service provision based on the sum of these scores.
It is important to note that even if a minor part of an applicable action is not met, then that action will be awarded a 'growing' rating and this will impact the overall rating a health service receives against the maturity scale for their clinical trial service. See Maturity rating for clinical trial service assessment - Fact sheet and Advisory CT25/01: Extension of the maturity rating scale for clinical trial service assessments against NCTGF.
Lessons learnt
Initial ratings
Actions most frequently rated Initial Systems during accreditation to the NCTGF are consistent with results observed among health services during accreditation to the NSQHS Standards. The clinical trial workforce should engage with health service safety and quality teams to identify areas of improvement in relation to these actions for the provision of clinical trial services.
Planning ahead of assessment - identification of not-applicable actions
Members of the clinical trial workforce should communicate with safety and quality teams within their health service to identify the accrediting agency that has been procured and confirm the scope of assessment to the NCTGF.
It is important for health services to establish an ongoing dialogue with their accrediting agency to appropriately plan for assessment. Further information is included in Fact sheet 17: Short notice accreditation assessment.
Key topics for discussion during the Planning Phase include:
- Determination of ‘not-applicable’ actions of the NCTGF that may apply to the health service based on their service level context for clinical trials. Refer to Advisory AS18/01: Advice on not applicable actions.
- Processes to regularly share information, including throughout the accreditation cycle and during assessment
- Approaches to conducting on-site assessment including access arrangements to the site, systems and key members of the clinical trial workforce.
Action 1.11 – Incident Management
The aim of incident management systems in health services is to incorporate improved and updated approaches to effectively manage incidents, with the view to proactively reduce preventable patient harm.
It relies on strong governance arrangements to enable the health service to effectively and efficiently identify risk, develop mitigation strategies, including processes to analyse and evaluate risks and uses risk assessments to set priorities for and to improve the safety and quality of clinical trial service provision.
The Incident Management fact sheet describes the intent of Action 1.11 of the NCTGF and provides guidance on how an incident arising from a clinical trial is identified, reported, and managed in health services with relevant examples.
Assessment cycles
Health services providing a clinical trial service must be assessed at least once to the NCTGF.
Assessment to the NCTGF operates under the short notice assessment process (SNAP) format. SNAP commenced in July 2023 and is mandatory for the accreditation of health services to the NSQHS Standards and where applicable, the NCTGF.
A standard accreditation cycle is three-years. Despite the launch of the NCTGF in May 2023, timings as to when a SNAP occurs will be variable across health services and is influenced by:
- When the health service moved from announced to short notice assessments
- When they opened/registered for assessment.
For example, health services that moved to short notice earlier in their own accreditation cycle are likely to have commenced a new cycle in May, which will be different for those who moved mid-way through a cycle.
Fact sheet 18 provides breakdown around the timings of short notice assessment and related cycles.
Members of the clinical trial workforce are also encouraged to engage with their safety and quality teams to understand their health services own accreditation cycle to prepare for assessment to the NCTGF.