National Hand Hygiene Initiative (NHHI)
Quick links
Important update - NHHI auditing and LMS resources have moved to the NHHI Knowledge base
NHHI auditing and learning management/eLearning support resources have moved to the Commission’s new dedicated NHHI Knowledge base and are no longer available on the Commission’s website.
The NHHI Knowledge base has a search function to assist you in finding useful resources such as how to guides, promotional materials, implementation guides, auditor training resources, and frequently asked questions. If your enquiry is not addressed through the NHHI Knowledge base, you are welcome to submit a ticket via the NHHI Knowledge base.
Login pages for the NHHI Learning Management System (eLearning) and HHCApp (auditing database) remain the same.
What is hand hygiene?
Hand hygiene is a fundamental part of patient care to prevent the spread of infections and contamination of the healthcare environment.
When performed correctly, hand hygiene results in a reduction of microorganisms on hands and contributes to prevention of healthcare-associated infection and improved patient safety.
Alcohol-based hand rubs
Choosing alcohol-based hand rub products
Selection of alcohol-based hand rub products is the responsibility of individual health care facilities.
It is recommended that alcohol-based hand rub products:
- meet the EN1500 testing standard for bactericidal effect
- have Therapeutic Goods Administration (TGA) approval as a hand hygiene product for a healthcare setting.
Other factors that should also be considered include:
- dermal tolerance
- aesthetic preferences such as fragrance, colour, texture and ease of use
- practical considerations such as availability, convenience and functioning of dispenser, and ability to prevent contamination
- cost.
When to use alcohol-based hand rub
Alcohol-based hand rubs should be used when hands are visibly clean instead of handwashing because they:
- substantially reduce bacterial count on hands
- require less time than handwashing
- are more accessible
- are self-drying
- are gentler on skin and cause less skin irritation and dryness than frequent soap and water washes, since all hand rubs contain skin emollient (moisturisers).
Handwashing is reserved for situations when hands are visibly soiled, or when gloves have not been worn in the care of a patient with Clostridioides difficile.
Alcohol-based hand rubs have excellent antimicrobial activity against gram-positive and gram-negative vegetative bacteria, and good antimicrobial activity against enveloped viruses. Alcohol solutions containing 60-80% alcohol are most effective. It has been well established that alcohols effectively reduce the bacterial counts on hands better than soap and water.
Alcohol-based hand rubs are more effective against most bacteria and many viruses than either medicated or non-medicated soaps.
Alcohol-based hand rub is the hand hygiene product of choice for all standard aseptic procedures. Surgical scrub is required for surgical procedures. For definitions on standard versus surgical aseptic technique refer to the Australian Guidelines for the Prevention and Control of Infections in Healthcare.
Alcohol-based hand rub is also the recommended product for the prevention of intravascular catheter-related infections.
Cutaneous absorption of alcohol
Recent studies have demonstrated minimal rates of cutaneous alcohol absorption such that there should be no concern for healthcare workers. A study suggested that isopropanol might be less likely to be absorbed than ethanol. Healthcare workers who are concerned about absorption for religious or other reasons may elect to use an alcohol-based hand rub that contains isopropanol rather than ethanol.
When implementing a hand hygiene campaign with an alcohol-based hand rub in a healthcare setting, it is important to provide opportunities for healthcare workers to raise concerns about the use of alcohol-based hand rubs for cultural or religious reasons or due to alcohol abuse and help them to understand the evidence underlying its use, and to identify that address their concerns.
Alcohol-based hand rub placement
Ensuring alcohol-based hand rub is available at the point of care improves hand hygiene compliance.
Alcohol-based hand rub should be easily accessible, generally within arm's reach of where patient care or treatment is taking place.
Dispensers act as a visual cue for hand hygiene behaviour. Placement of alcohol-based hand rub needs to be consistent and reliable. Clinical staff should assist with the decision-making process regarding placement of alcohol-based hand rub, as they generally best understand the workflow in their area.
Where possible, alcohol-based hand rub should be placed at the foot of every bed or within each patient cubicle. When designing new healthcare facilities, consideration should be given to appropriate placement of alcohol-based hand rubs so that healthcare workers can easily see and reach them.
Alcohol-based hand rub dispensers should not be placed next to sinks, as this can cause confusion for healthcare workers who may think they need to rinse their hands with water after using alcohol-based hand rub.
The following locations are suggested for placement of alcohol-based hand rub:
- on the end of every patient bed using either a fixed or removable bracket
- attached to mobile work trolleys, for example, intravenous, drug and dressing trolleys, especially in emergency department and recovery areas
- high staff traffic areas, for example, nurses' station, pan room, medication room and patient room entrance
- all other clinical and patient care areas, such as examination rooms and outpatient consultation rooms
- entrances to each ward, outpatient clinic or department
- public areas such as waiting rooms, reception areas, hospital foyers, near elevator doors in high-traffic areas.
Signage about the appropriate use of alcohol-based hand rubs should be prominently displayed in all clinical areas in all health service organisations.
Health service organisations should clearly document and assign responsibility for replacement of empty alcohol-based hand rub dispensers. For example, workplace agreements or job descriptions may include a requirement for prompt replacement of these dispensers. Alcohol-based hand rub should never be decanted from one bottle to another as this can cause contamination.
Safe placement of alcohol-based hand rub
The benefits to patients and staff of the use of alcohol-based hand rub outweigh the risks. A risk assessment should be undertaken and a management plan put in place for all clinical areas, and particularly for areas that manage patients with alcohol use disorders, patients at risk of self harm and patients such as children who may be at risk of accidental ingestion.
The following factors should be considered in relation to safe placement of alcohol-based hand rub:
- The maximum size of an individual alcohol-based hand rub dispenser should not exceed 500 mls.
- No more than 80 individual alcohol-based hand rub dispensers (each with a maximum capacity of 500 mls) should be installed within a single smoke compartment.
- Dispensers should not project more than 15 cm into corridor egress.
- Wall-mounted brackets should be located at a height of between 92 cm and 122 cm above the floor (avoid placing at eye level)Dispensers should not be located over carpeted areas, unless the area is protected by active sprinkler.
- Dispensers should not be located over or directly adjacent to ignition sources such as electrical switches, power points, call buttons, or monitoring equipment.
- Dispensers should be separated from heat sources and electric motors.
- Dispensers should be installed according to manufacturer’s recommendations and to minimise leaks or spills.
- Regular maintenance of dispensers and brackets should occur in accordance with manufacturer’s guidelines.
- Product usage signs should be clearly visible and laminated.
- Regular monitoring of each area is recommended for misuse or removal of product.
- Each facility should take adequate care with the placement of each dispenser so as to protect vulnerable populations, for example in psychiatric units, drug and alcohol units, paediatric units and units caring for cognitively impaired patients.
- Alcohol-based hand rub bottles should be designed to minimise evaporation due to the volatile nature of alcohols.
- Site-specific instructions should be developed to manage adverse events, such as alcohol-based hand rub ingestion, eye splashes or allergic reactions.
- Bracket design is important, as alcohol-based hand rub placement may be affected if brackets are ill-fitting (for example, varying sizes of bed rails can affect the efficacy of some brackets).
- Brackets that are removable, or product that can be removed from brackets easily, to accommodate short term patient demands.
- Bracket availability and installation costs.
Clinical area placement considerations
The placement of alcohol-based hand rub needs to be aligned with risk assessment of the individual patients or patient populations. For example, clinical areas where ingestion or accidental splashing of alcohol-based hand rub is a particular risk, such as:
- paediatrics – locate with care near children (see below)
- mental health – locate with care near mental health patients, patients undergoing alcohol- or other drug-withdrawal, or where there are cognitively impaired patients
- public areas - place in high traffic areas with clear signage about appropriate use and the need for parents to carefully supervise their children.
Small personal bottles that healthcare workers carry with them may be more appropriate in some of the above areas.
Paediatric setting product placement
Alcohol-based hand rub can be placed in paediatric wards. The placement of alcohol-based hand rub within neonatal intensive care units, special care nurseries, maternity wards and on cots should follow the recommendations for product placement at the point of care.
Placement in general paediatric wards should be at the point of care, except in situations where intellectual disability or alcohol abuse or where the child could unintentionally or intentionally harm themselves. Personal bottles of alcohol-based hand rub should be used in any area where alcohol-based hand rub cannot be placed at the point of care.
Limitations of alcohol-based hand rub
In some situations washing hands with soap and water is preferable to use of alcohol-based hand rub.
Bacterial spores
Alcohol has virtually no activity against bacterial spores. Washing hands with soap and water is preferred in this situation because it is the best method of physically removing spores from the hands. However, the vegetative form of Clostridiodes difficile is highly sensitive to alcohol-based hand rub.
The November 2018 ASID/AICA position statement on Infection Control Guidelines for Patients with Clostridiodes difficile Infection (CDI) in Healthcare Settings recommends the primary use of alcohol-based hand rub in accordance with the WHO 5 Moments for Hand Hygiene when caring for patients with CDI. Gloves should be used during the care of patients with CDI, to minimise spore contamination, and if hands become soiled, or gloves have not been used, then hands must be washed with soap and water.
Non-enveloped (non-lipophilic) viruses
Alcohol has a poor activity against some non-enveloped viruses. (for example: rotavirus, norovirus, polio, hepatitis A). However, there is conflicting evidence suggesting that alcohol-based hand rub is more effective than soaps in reducing virus titres on finger pads. Thus, in norovirus outbreaks it is usually best to reinforce the use of alcohol-based hand rub, unless hands are visibly soiled, in which case handwashing with soap and water is preferred.
Other organisms
Alcohol has a poor activity against tropical parasites, and protozoan oocysts. Handwashing soap and water is preferred.
Safe use of chlorhexidine
We have developed guidance for health service organisations on the appropriate and safe use of chlorhexidine in healthcare settings.
How do I perform hand hygiene?
Use an alcohol-based hand rub for all clinical situations where hands are visibly clean.
Wash with soap and water:
- when hands are visibly dirty or contaminated with proteinaceous material
- hands are visibly soiled with blood or other body fluids
- exposure to potential spore forming organisms is strongly suspected or proven
- after using the bathroom.
Glove use and hand hygiene
Wearing gloves does not replace the need for hand hygiene. Gloves do not provide complete protections against contamination. Microorganisms can reach the hands though small, often unnoticed defects in gloves or contamination of the hands during glove removal.
Gloves should be worn:
- when there is a risk of contamination of the healthcare workers hands with blood or body fluids
- where indicated by local infection control policy, for example with transmission-based precautions.
Gloves should be changed:
- between episodes of care for different patients, to prevent transmission of microorganisms
- during the care of a single patient, to prevent cross-transmission of body sites.
Hand hygiene is required with glove use:
- before putting on gloves
- after removing gloves
- during the care for a single patient as indicated by the 5 Moments for Hand Hygiene
- after each use of single use gloves which are discarded.
Hand hygiene products and gloves should be made available inside isolation/contact precaution rooms to allow for appropriate hand hygiene to occur during the care of a patient.
Prolonged and indiscriminate use of gloves should be avoided as it may cause adverse reactions and skin sensitivity.
For further information on glove use refer to the Australian Guidelines for the Prevention and Control of Infection in Healthcare.
Hand care
Intact skin is the body’s first line defence against infection. Damaged skin can not only lead to infection in the host, but can also harbour higher numbers of microorganisms than intact skin and increase the risk of transmission to others.
Early recognition and systematic management of hand care issues associated with the use of hand hygiene products are essential.
There are two major types of skin reactions associated with hand hygiene:
- Irritant contact dermatitis, which includes symptoms that can vary from mild to debilitating, including dryness, irritation, itching, and even cracking and bleeding
- Allergic contact dermatitis, which is rare and represents an allergy to some ingredient in a hand hygiene product. In its most serious form allergic contact dermatitis may be associated with symptoms of anaphylaxis.
Causes and prevention of dermatitis
Most hand hygiene-related skin problems in healthcare workers are caused by irritant contact dermatitis, primarily due to:
- frequent and repeated use of soaps and detergents
- excessive paper towel use causing skin drying.
Some staff may experience stinging when first using alcohol-based hand rubs on damaged skin. However, evidence shows that continued use of emollient=containing alcohol-based hand rubs improves symptoms in approximately 70% of affected healthcare workers.
Using an oil-containing moisturiser or barrier cream three times per shift can significantly reduce dryness, chemical irritation, and skin breakdown.
It is important to ensure that the selected alcohol-based hand rub, soaps, and moisturising lotions are chemically compatible to minimise skin reactions among staff.
Factors that may contribute to dermatitis include:
- fragrances and preservatives, which commonly cause of contact allergies
- washing hands regularly with soap and water immediately before or after using an alcohol-based hand rub, which is unnecessary
- putting on gloves while hands are still wet from either handwashing or applying alcohol-based hand rub, which increases the risk of skin irritation
- using hot water for handwashing
- failure to use supplementary moisturisers
- quality of paper towels.
Strategies for minimising occupational hand dermatitis include:
- use of a hand hygiene product that contains skin emollient to minimise the risk of skin irritation and drying
- educating staff on the correct use of hand hygiene products
- educating staff on caring for their hands, including the regular use of skin moisturisers both at work and at home
- providing a supportive attitude towards staff with skin problems.
Alcohol-based hand rub produces the lowest incidence of irritant contact dermatitis of all the hand hygiene products currently available. True allergy to alcohol-based hand rub is rare, and allergy to alcohol alone has not been reported.
Healthcare workers should be encouraged to notify their hand hygiene lead if skin irritation occurs following the use of alcohol-based hand rub. All hospitals should have access to referral for follow up for healthcare workers with persistent skin problems.
Hand care resources
Materials to support improved hand hygiene in Australia
A range of promotional materials are available to support implementation of the National Hand Hygiene Initiative (NHHI).
Using our resources
Organisations may adapt these resources to meet local requirements. However, if the resources are modified in any way, the NHHI logo should be removed and the resource should be annotated to acknowledge the Commission as the original source.
For materials developed by the World Health Organization, please refer to the WHO copyright, licencing and permissions web page.
WHO hand hygiene posters
The World Health Health Organization (WHO) has developed the following posters on performing hand hygiene:
These posters outline the correct technique and duration for effective hand hygiene.
Hand hygiene training videos are currently accessible via the Commission's YouTube channel.
Hand Hygiene Expert Insights videos
Do you understand the importance of hand hygiene? Watch this video series to hear from leading experts across the healthcare sector highlight key elements of hand hygiene practice.
National Hand Hygiene Audit Dashboard
The NHHI Audit dashboard allows trend tracking by compliance, audit period, state and territory and public and private sector.
Hand hygiene auditing and monitoring
5 Moments for Hand Hygiene
The 5 Moments for Hand Hygiene approach was designed by the World Health Organization to minimise the risk of transmission of microorganisms between a healthcare worker, the patient, and the environment.
The 5 Moments - infographic
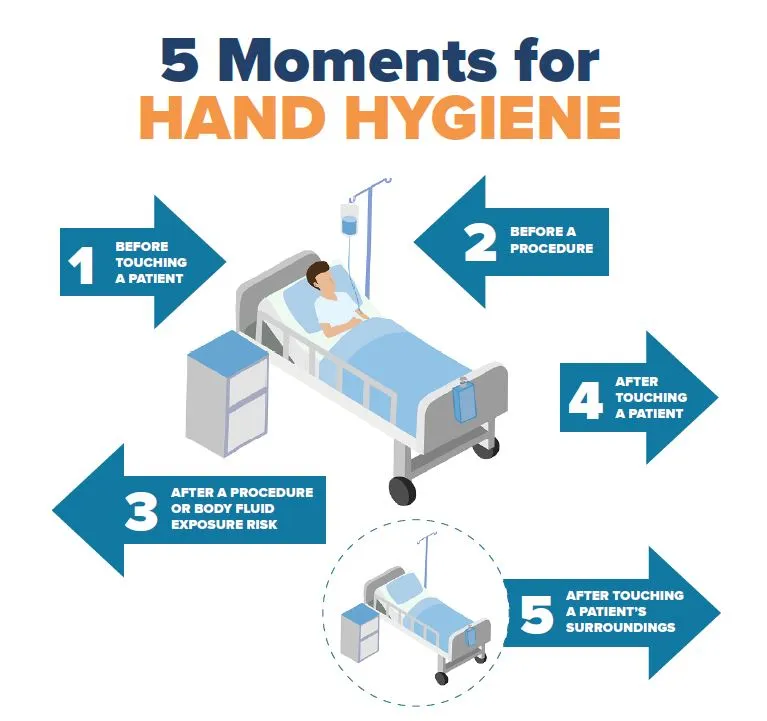
Based on the 'My 5 moments for Hand Hygiene' © World Health Organization 2009. All rights reserved
Moment 1 - Before touching a patient
When
Immediately before touching the patient.
Why?
To protect the patient against microorganisms from the hands of the healthcare worker.
In detail
| Before touching a patient | Shaking hands, assisting a patient to move, allied health interventions, touching any medical device connected to the patient (for example, intravenous line pump, urinary catheter). |
|---|---|
| Before any personal care activities that require physical contact | Bathing, dressing, brushing hair, putting on personal aids such as glasses. |
| Before taking any non-invasive observations | Taking a pulse, blood pressure, oxygen saturation, temperature, chest auscultation, abdominal palpation, applying ECG electrodes, cardiotocography. |
| Before providing any non-invasive treatment | Applying an oxygen mask or nasal cannula, fitting slings/braces, application of incontinence aids (including condom drainage), conducting an oral examination without using a sharp instrument (such as using a mirror probe), performing oral X-ray. |
| Before the preparation or administration of oral medications | Oral medications, nebulised medications. |
| Before providing oral care and feeding | Feeding a patient (excluding feeding via nasogastric tube or percutaneous endoscopic gastrostomy), brushing teeth or dentures. |
Moment 2 - Before a procedure
When
Immediately before a procedure. Once hand hygiene has been performed, nothing else in the patient's environment should be touched prior to the procedure starting.
Why?
To protect the patient against microorganisms from entering the patient’s body, including the patient’s own microorganisms.
In detail
| Before inserting a needle into a patient’s skin or into an invasive medical device connected to the patient | Venepuncture, blood glucose level, arterial blood gas, subcutaneous or Intramuscular injections, intravenous line flush. |
|---|---|
| Before administration of any medications given via an invasive medical device | Intravenous medication, nasogastric tube (NGT) feeds, percutaneous endoscopic gastrostomy (PEG) feeds. |
| Before the preparation of an aseptic field | Dressing trolley set up for an aseptic procedure. |
| Before the administration of medications where there is direct or potentially direct contact with mucous membranes | Eye drop instillation, suppository insertion, vaginal pessary. |
| Before the insertion of, or contact with, an invasive medical device | Procedures involving endotracheal tube, tracheostomy, nasopharyngeal airways, suctioning of airways, urinary catheter, colostomy/ileostomy, vascular access systems, invasive monitoring devices, wound drains, percutaneous endoscopic gastrostomy tubes, nasogastric tube, secretion aspiration. |
| Before any assessment, treatment or patient care where contact is made with non-intact skin or mucous membranes or there is a likelihood of penetration of tissue or a cavity | Wound dressings, surgical procedures, digital rectal examination, invasive obstetric and gynaecological examinations and procedures, digital assessment of newborn palate, invasive dental examinations, dental extractions, dental restoration. |
| Before preparation and administration of any medications or materials for any oral health/dental procedure | Administering topical medication such as fluoride, topical anaesthetic, local anaesthetic or tooth mousse or restorative materials used for restoration procedure. |
Moment 3 - After a procedure or body fluid exposure risk
When
Immediately after a procedure or body fluid exposure risk.
Even if you have had gloves on you should still perform hand hygiene after removing them as gloves are not always a complete impermeable barrier. Hands may also have been contaminated in the process of removing the gloves.
Why?
To protect the healthcare worker and the healthcare environment from becoming contaminated with the patient's microorganisms.
In detail
| After performing a procedure |
|
|---|---|
| After a potential body fluid exposure | Contact with a used urinary bottle/bedpan, contact with sputum either directly or indirectly via a cup or tissue, contact with used specimen jars/pathology samples, cleaning dentures, cleaning spills of body fluids from patient surroundings, after touching the outside of a wound or chest drain or oral suction tubing, contact with a used dental instruments or appliances, after touching surfaces that potentially contaminated with body fluid. |
Moment 4 - After touching a patient
When
After touching a patient.
Why?
To protect the healthcare worker and the healthcare environment from becoming contaminated with the patient’s microorganisms.
In detail
| After a Moment 1, except if there has been exposure to body fluids | After touching a patient, after touching an intravenous pump or other medical equipment connected to the patient. |
|---|
Moment 5 - After touching a patient's surroundings
When
After touching anything in the patient's surroundings when the patient has not been touched or is not present.
Always hand hygiene before leaving the patient's room.
Why?
To protect the healthcare worker and the healthcare environment from becoming contaminated with the patient’s microorganisms.
In detail
| After touching any items in the patient zone but the patient has not been touched while the healthcare worker was in the patient zone | Any items in the patient zone include bed, bedrails, linen, table, bedside chart, bedside locker, call bell/TV remote control, light switches, personal belongings (including books, mobility aids), chair, footstool, monkey bar that is attached to a patient’s bed. |
|---|
National audits and HHCApp - NHHI
Data on hand hygiene compliance are collected by states and territories for all public health service organisations, and by many private health service organisations.
Hand hygiene compliance is assessed against a national benchmark, which is currently 80%. There are three national hand hygiene audits conducted each year:
- Audit period 1: 1 November to 31 March.
- Audit period 2 (voluntary): 1 April to 30 June.
- Audit period 3: 1 July to 31 October.
Auditing using the HHCApp
National Hand Hygiene Initiative - Audit Requirements
Hand hygiene compliance auditing is conducted to assess the effectiveness of hand hygiene programs in Australia, as part of the National Hand Hygiene Initiative (NHHI). Hand hygiene compliance is assessed across both public and private Australian hospitals, consistent with AHMAC endorsed benchmark of 80 per cent.
Auditing using the HHCApp
The HHCApp is the Commission's web-based application for use by healthcare organisations to collect, review and report their hand hygiene compliance rates.
For settings such as non-acute, primary care and mental health it is preferable to assess other aspects of a hand hygiene program, such as product placement and availability and participation in education. Routine hand hygiene compliance auditing is not recommended in these settings, as there is a low level of staff/patient activity and interaction that will result in a small number of Moments being observed.
All facilities should be aware of their jurisdictional and organisational requirements when planning measurement of their hand hygiene program. A number of audit tools [ADD LINK] are available that can be used or modified as desired. Hand hygiene compliance audits should only be undertaken by trained and validated Hand Hygiene Auditors or Hand Hygiene Auditor Educators.
Annual audit data submission dates
Health service organisations which are being accredited to the National Safety and Quality Health Service Standards are required to collect hand hygiene compliance data for national hand hygiene audits, unless exempted by the relevant state and territory regulator. Health service organisations should refer to current NSQHS Advisories regarding any changes to this requirement.
Annual audit periods for the NHHI are:
| Audit period 1 | 1 November to 31 March. |
|---|---|
| Audit period 2 | 1 April to 30 June. |
| Audit period 3 | 1 July to 31 October. |
National Audit period 2 became voluntary from 1 April 2023 to provide health service organisations with additional time for quality improvement activities. Organisations should refer to their jurisdictional requirements at Advisory AS23/01: Advice on national hand hygiene audit period 2 (for example, performance or service level agreements) when deciding whether to participate in Audit period 2.
HHCApp Desktop
HHCApp Desktop is used to enter hand hygiene data that has been collected on paper. If you have administrator access, you can also use HHCApp desktop to access reports and administrator functions:
How to enter hand hygiene data using the HHCApp desktop
HHCApp mobile
HHCApp Mobile is used to enter hand hygiene data in real time as you audit. HHCApp Mobile is not a mobile app; it is a webpage that can be accessed via the internet browser of a mobile device. If using this option to enter data, please ensure you login to begin, and when finished, sync your data and use the 'logout' button on completion to minimise errors.
If you have difficulty syncing your mobile device, you may need to update the shortcut on your device to the new link below, delete your mobile device browser history and remove the previous shortcut you used for the HHCApp login page.
How to enter hand hygiene data using the HHCApp mobile
Guidelines for submission
Submitting data to the National Audit
The HHCApp is used to enter data which must be submitted by the last day of each audit period.
The hand hygiene lead for each organisation is required to press the 'submit for approval' button in the HHCApp to demonstrate that data collection has been completed. Data submission can be completed at any time in the lead up to the last day of each audit period.
Please note: by pressing the 'submit for approval' button you are closing off the audit for your organisation, which does not allow for further data entry for that audit period. Please read the guidance on how to validate and submit a completed audit prior to submitting your organisation's audit data.
Guidelines for data submission
Guidelines that support submission of hand hygiene audit data by hospitals and some specific clinical settings are listed. These guidelines are intended to ensure all hand hygiene compliance data collected and submitted, as part of the NHHI, is an accurate and reliable representation of a participating organisation's hand hygiene compliance. All organisations submitting data as part of the NHHI are required to follow these guidelines.
Use of Audit data for quality improvement and improved patient safety
The health service organisation must also demonstrate that it uses the results of audits to improve hand hygiene compliance and patient safety.
Guidelines for submission of data for specific settings
The HHCApp has guidelines for hospitals, day hospitals, dialysis/oncology, dental facilities, data validation as well as audit tools and resources.
Data validation
Data validation must be completed by hand hygiene leads before final submission of data to the NHHI.
While an audit is active in HHCApp, changes can be made to data if errors are found. Once an audit has been submitted and the status in HHCApp is “pending approval”, then changes can only be made after discussion with your jurisdictional coordinator, or by contacting the NHHI Helpdesk.
Final submission of data by the hand hygiene lead provides confirmation that the data has been reviewed and accepted by the organisation.
For more information on data validation in HHCApp please refer to the NHHI Knowledge base.
HHCApp audit tools and resources
There are a number of tools available to support hand hygiene auditing in acute and non-acute healthcare settings.
Hand hygiene programs should have a range of processes to monitor and report healthcare workers’ hand hygiene practices, including:
- Direct observational auditing of hand hygiene practices
- Auditing of hand hygiene product availability
- Conducting staff surveys on their knowledge of hand hygiene practices
- Conducting consumer surveys on staff hand hygiene practices
- Monitoring staff hand hygiene education uptake.
Tools and resources
National hand hygiene audit requirements
Auditing using the HHCApp
The HHCApp is the Commission's web-based application for use by healthcare organisations to collect, review and report their hand hygiene compliance rates.
For settings such as non-acute, primary care and mental health it is preferable to assess other aspects of a hand hygiene program, such as product placement and availability and participation in education. Routine hand hygiene compliance auditing is not recommended in these settings, as there is a low level of staff/patient activity and interaction that will result in a small number of Moments being observed.
All facilities should be aware of their jurisdictional and organisational requirements when planning measurement of their hand hygiene program. A number of audit tools are available that can be used or modified as desired. Hand hygiene compliance audits should only be undertaken by trained and validated Hand Hygiene Auditors or Hand Hygiene Auditor Educators.
Annual audit data submission dates
Health service organisations which are being accredited to the National Safety and Quality Health Service Standards are required to collect hand hygiene compliance data for national hand hygiene audits, unless exempted by the relevant state and territory regulator. Health service organisations should refer to current NSQHS Advisories regarding any changes to this requirement.
Annual audit periods for the NHHI are:
| Audit period 1 | 1 November to 31 March |
|---|---|
| Audit period 2 | 1 April to 30 June |
| Audit period 3 | 1 July to 31 October |
National Audit period 2 became voluntary from 1 April 2023 to provide health service organisations with additional time for quality improvement activities.
Submitting data to the National Audit
The HHCApp is used to enter data which must be submitted by the last day of each audit period.
The hand hygiene lead for each organisation is required to press the 'submit for approval' button in the HHCApp to demonstrate that data collection has been completed. Data submission can be completed at any time in the lead up to the last day of each audit period.
- Please note: by pressing the 'submit for approval' button you are closing off the audit for your organisation, which does not allow for further data entry for that audit period. Please read the guidance on how to validate and submit a completed audit prior to submitting your organisation's audit data.
Guidelines for data submission
Guidelines that support submission of hand hygiene audit data by hospitals and some specific clinical settings are listed. These guidelines are intended to ensure all hand hygiene compliance data collected and submitted, as part of the NHHI, is an accurate and reliable representation of a participating organisation's hand hygiene compliance. All organisations submitting data as part of the NHHI are required to follow these guidelines.
Use of Audit data for quality improvement and improved patient safety
The health service organisation must also demonstrate that it uses the results of audits to improve hand hygiene compliance and patient safety.
Hospitals
| Number of acute inpatient beds | Minimum Total number hand hygiene moments per audit |
|---|---|
| >400 | 2,450 |
| 301 to 400 | 2,100 |
| 201 to 300 | 1,750 |
| 151 to 200 | 800 |
| 101 to 150 | 600 |
| 51 to 100 | 200 |
| 25 to 50 | 100 |
| <25** | 50 |
*** Auditing in hospitals with <25 beds is dependent on jurisdictions. See table below.
| Jurisdiction | Auditing required in hospitals <25 beds |
|---|---|
| New South Wales | Yes |
| Victoria | Yes |
| Queensland | No |
| South Australia | No – refer to jurisdictional representative for options |
| Western Australia | No – further information here |
| Tasmania | Yes |
| Northern Territory | Yes |
| Australian Capital Territory | Yes |
Day hospitals
Guidelines have been developed for stand-alone day hospitals regarding the collection of representative hand hygiene compliance as part of the National Hand Hygiene Initiative.
Day hospitals – guidelines for data collection
To ensure the collection of representative hand hygiene compliance data for the National Hand Hygiene Initiative from freestanding day hospitals, the following is recommended:
Day hospital size:
| Peer Group | Definition |
|---|---|
| Large | Stand-alone facility performing >5,000 procedures per annum |
| Medium | Stand-alone facility performing 2,000–5,000 procedures per annum |
| Small | Stand alone facility performing <2,000 procedures per annum |
| Solo practice, solo practitioner or very small settings | Stand-alone facility that is operated by a single medical professional and has a total clinical staff of five or less. Applicable to endoscopy and cosmetic surgery day procedure services. |
Target number of Moments for day hospitals:
| Day hospital size | Required number of hand hygiene audits per year | Required number of hand hygiene observations per facility each audit |
|---|---|---|
| Large | 2 | 350 |
| Medium | 2 | 200 |
| Small | 2 | 100 |
| Solo practice, solo practitioner or very small settings | Direct observational hand hygiene auditing is not recommended - use other strategies to assess hand hygiene performance in these settings. Surgical hand hygiene should be undertaken in all theatre settings | |
Organisations should refer to their jurisdictional requirements at Advisory AS23/01: Advice on national hand hygiene audit period 2
Recommendations:
- Facility Hand Hygiene Project Coordinator to determine facility size, according to peer grouping guidelines above
- Target number of Moments to be collected as per table above, dependent on facility size
- Collection of target number of Moments will be comparable to hospitals of the same peer grouping collecting data in their Day Procedure Unit
- Compliance data to be collected and submitted as per the National Hand Hygiene Initiative website.
Guideline for Data Submission - National Hand Hygiene Initiative
Dialysis/oncology
In the dialysis and oncology settings the risk of transmission of infection for all patients and healthcare workers is high due to repetitive invasive procedures and blood handling. It is extremely important to meet the requirements for optimal hand hygiene, despite the high number of opportunities for hand hygiene.
Dialysis/oncology centres – guidelines for data collection
To ensure the collection of representative hand hygiene compliance data for the National Hand Hygiene Initiative from stand-alone/satellite dialysis/oncology centres*, the following is recommended:
Dialysis/oncology centre size:
| Peer Group | Definition |
|---|---|
| Large | Stand-alone facility performing >5,000 procedures per annum |
| Small | Stand-alone facility performing <5,000 procedures per annum |
*adapted from the Australian Hospital Peer Groupings, AIHW 2008
Target number of Moments for dialysis/oncology centres:
| Dialysis/oncology centre size | Required number of hand hygiene audits per year | Required number of hand hygiene observations per facility each audit |
|---|---|---|
| Large | 2 | 200 |
| Small | 2 | 100 |
Organisations should refer to their jurisdictional requirements at Advisory AS23/01: Advice on national hand hygiene audit period 2
Recommendations:
- Facility Hand Hygiene Project Coordinator to determine facility size, according to peer grouping guidelines above.
- Target number of moments to be collected as per table above, dependent on facility size.
- Collection of target number of Moments will be comparable to dialysis centres of the same peer grouping
- Compliance data to be collected and submitted as per the National Hand Hygiene Initiative website.
* A standalone or satellite dialysis or oncology centre is one which is physically separate to a healthcare facility with other areas requiring HHC data collection.
# A healthcare facility with a dialysis or oncology ward is required to collect HHC moments for that ward as per the Guidelines for Data Submission Hospitals.
Several resources specific to these settings have been developed to provide guidance on hand hygiene practices as well as guidelines regarding the collection and submission of data as part of the NHHI.
Dental facilities
Where sites deem hand hygiene auditing to be appropriate, the following information provides guidance regarding the collection of representative hand hygiene compliance data by solo, group and hospital based dental services as part of the National Hand Hygiene Initiative.
Dental service description:
| Peer group | Definition |
|---|---|
| Solo practice, solo practitioner or very small oral health service | An oral health/dental practice with a single dentist or an oral health service with a single dental chair/surgery |
| Small oral health service/dental practice | Oral health/dental practice with a total of 2–5 dental chairs/surgeries in one or more locations |
| Medium-sized oral health service/dental practice | Oral health/dental practice with between 6 and 10 dental chairs/surgeries in one or more locations |
| Large oral health service/dental hospital | Any dental oral health services/dental hospitals with more than 10 dental chairs/surgeries in one or more locations |
Suggested target number of Moments for oral health/dental services:
| Peer group | Recommended number of hand hygiene audits per year | Recommended number of hand hygiene observations per facility per audit |
|---|---|---|
| Solo practice | HHC Auditing not appropriate | |
| Small | 2 | 50 |
| Medium | 2 | 100 |
| Large dental hospital | 2 | 200 |
Organisations should refer to their jurisdictional requirements at Advisory AS23/01: Advice on national hand hygiene audit period 2
Recommendations:
Facility Hand Hygiene Project Coordinator to determine facility size, according to peer grouping guidelines above. If a dental service makes the decision to perform hand hygiene compliance auditing by direct observation, the target number of Moments to be collected is provided in the table above, dependent on facility size. Collection of target number of Moments will be comparable to oral health services of the same peer grouping. Compliance data is to be collected and submitted as per National Hand Hygiene Initiative website.
Data validation
Data validation must be completed by hand hygiene leads before final submission of data to the NHHI.
While an audit is active in HHCApp, changes can be made to data if errors are found. Once an audit has been submitted and the status in HHCApp is “pending approval”, then changes can only be made after discussion with your jurisdictional coordinator, or by contacting the NHHI Helpdesk.
Final submission of data by the hand hygiene lead provides confirmation that the data has been reviewed and accepted by the organisation.
Please review the NHHI HHCApp FAQs for detailed guidance on how to conduct validation of your data. In summary, hand hygiene leads should review the following:
- Correct number of Moments reported for the organisation
- Correct number of Moments reported for each eligible department
- Data were collected by currently validated Hand Hygiene Auditors only
- Auditor hand hygiene compliance is appropriate for the area audited
- Observations were conducted in clinical settings
- Moments observed were appropriate for healthcare worker type
All outlier data should be investigated by the hand hygiene lead prior to submission.
National Hand Hygiene Initiative Helpdesk
The National Hand Hygiene Initiative (NHHI) operates a Help Desk to support users of the NHHI Learning Management System (LMS) and the Hand Hygiene Compliance Application (HHCApp).
The Help Desk team endeavours to respond to your enquiry as quickly as possible.
Before contacting the Help Desk
Before contacting the NHHI Help Desk, please take the following steps to resolve your enquiry:
- Go NHHI Knowledgebase.
- Contact your Organisation or Region Administrator (hand hygiene lead) within your health facility, local health district/network or corporate group.
- Contact your Jurisdictional Coordinator for your state/territory if your organisation is based in the public sector.
The relevant contact details below:
| New South Wales | CEC-HAI-IPAC@health.nsw.gov.au |
|---|---|
| Victoria | handhygienevictoria@safercare.vic.gov.au |
| Northern Territory | safetyandquality.doh@nt.gov.au |
| Queensland Health | QLD Health Hand Hygiene Program Support Contact Us Form |
| South Australia | Hand hygiene in the healthcare environment - SA Health |
| Western Australia | handhygienewa@health.wa.gov.au |
| Australian Capital Territory | ACTHealthCMO@act.gov.au |
Contact form
If your query has not been answered by any of the above and you require further assistance, please submit a ticket to contact the NHHI Help Desk.
- NOTE: When you submit this form you will receive an auto-response email to let you know we have received your enquiry. Please ensure to check your junk or spam for this email and for any further NHHI Help Desk responses to your enquiry. Please do not create multiple enquiries within this timeframe.
Helpdesk phone service
In addition to the email support service, the Commission operates a NHHI Help Desk phone line at 02 9126 3511 between 10am - 1pm (AEDT) Tuesday to Thursday.
To avoid the issue of multiple tickets for your enquiry, please do not call more than once, or send a follow up email within 5 business days of your initial enquiry.
During peak busy periods, you may experience difficulty accessing support by phone. As an alternative, please access support via email. To ensure your enquiry is dealt with promptly and effectively, please include: your full name, organisation name, contact email address and a brief outline of your inquiry. Please also state whether your inquiry relates to the online learning modules within the Learning Management System (LMS), or the Hand Hygiene Compliance Application (HHCApp).
Privacy
Your privacy is important to the Australian Commission on Safety and Quality in Health Care (the Commission). The Commission only collects and uses information about you (name, employer, training/education provider, email address) to implement the National Hand Hygiene Initiative.
Your information may be accessed by authorised officers of the Commission to assist you with resolving any issues you raise with the National Hand Hygiene Initiative Help Desk. Authorised Organisation and Region Administrators from your registered organisation or training/education provider may also access your information and training records. Your state/territory Jurisdictional Coordinator (public health service organisations only) may also access your information for local Hand Hygiene program implementation purposes and to support you in using the NHHI systems.