Resources for the National Clinical Trials Governance Framework
The Commission has developed resources for health services, accrediting agencies and assessors to support implementation of the National Clinical Trials Governance Framework.
Key messages
The Commission are constantly working on delivering new and updated resources to support the sector in implementing the NCTGF. Recent releases and current projects include:
- A new Advisory CT25/01 on Extension of the Maturity Rating Scale for Clinical Trial Service Assessments against the National Clinical Trials Governance Framework (released Sept 2025)
- An update to Advisory AS18/01 Advice on not applicable actions that includes the NCTGF (released Sept 2025)
- An assessment outcomes data dashboard (released July 2025)
- A fact sheet on Incident Management (released July 2025)
- A new webinar series in development for early 2026
- Enhancements to the self-assessment and monitoring tools of the Clinical Trials Portal. Feedback on development opportunities can be shared directly with the Health and Medical Research Team via HMR@safetyandquality.gov.au
Download the Framework and User Guide

National Clinical Trials Governance Framework and User Guide
To support the delivery of high-quality clinical trial services the Commission has developed the National Clinical Trials Governance Framework on behalf of all jurisdictions in collaboration with the Australian Government Department of Health.
Knowledge building resources
Fact sheets and advisories
Fact sheets
These fact sheets provide details on the process and scope of assessment to the NCTGF.
An Advisory is a formal communication issued by the Commission to accrediting agencies and health service organisations. Advisories provide guidance and direction on the interpretation and/or assessment of the NCTGF under the AHSSQA Scheme. Advisories will usually be issued when the Commission identifies that additional support is required to assist with the consistent implementation or assessment of a specific action or topic. An advisory may also be issued where the implementation of an action requires a stepwise process with long implementation timeframes.
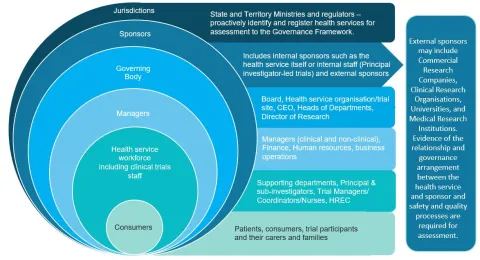
Roles and responsibilities
Understanding the roles and functions of identified positions relating to clinical trial service provision within a health service is an important step in preparing for accreditation to the NCTGF.
For example, Action 1.1 of the NCTGF requires health service organisations to ensure roles and responsibilities are clearly defined for the governing body, management, clinicians and the workforce in relation to the provision of clinical trial services. The following fact sheets are intended to support health service organisations meet this requirement.
Tools to support implementation
The Clinical Trials Portal
The Commission has developed the following web-based tools to assist HSOs monitor their clinical trial service operations and self-assess their capacity to meet the actions within the NSQHS Standards, as provided in the NCTGF.
- The self-assessment tool
- The operational metrics tool.
Across 2025 the Commission will undertake enhancements against the self-assessment and monitoring tools to support HSOs and accreditation agencies prepare for assessment to the NCTGF. Feedback on development opportunities can be shared directly with the Health and Medical Research Team via HMR@safetyandquality.gov.au.
Self-assessment tools
This self-assessment tool assists HSOs assess their capacity to meet the actions in the NCTGF. The tool enables HSOs to:
- Determine whether they meet the actions
- Document the evidence that demonstrates each action has been met
- Create an action plan of any tasks to meet the actions, including allocating a person responsible for completing the tasks.
Actions within the NCTGF are aligned with the NSQHS Clinical Governance Standard and Partnering with Consumers Standard. Speak with your hospital quality officer about the systems and processes currently in place to meet the actions within the NSQHS Standards and use the tool to assess your clinical trial service.
Operational metrics tool
The operational metrics tool enables the workforce within trial units, clinical departments, hospitals and health networks to report on their clinical trial service operations. This information enables HSOs to review their clinical trial services operating in multiple departments using a series of automated reports.
The operational metrics tool is aligned with the reporting requirements for the National Aggregate Statistics (NAS).
The Commission have published a resource to support HSOs navigate the Clinical Trials Portal. View the webinar below.
Victorian Comprehensive Cancer Centre (VCCC) Minimum Data Set (MDS)
The Commission have worked with the VCCC to refine the MDS to support reporting requirements of governing bodies and clinical trial workforce in relation to the NCTGF.
The MDS provides a series of baseline operational metrics (indicators) relevant to national priority indicators and regulatory requirements, such as those provided in Actions 1.1, 1.8 and 1.9 of the NCTGF, in addition to workforce and trial participant experience.
An updated version of the MDS is available at VCCC Alliance Metrics.
Orientation for Assessors
This National Clinical Trials Governance Framework: Orientation for Assessors course aims to support assessors in assessing service providers for implementation of the National Clinical Trials Governance Framework (download this document before commencing the course).
There are 4 modules in this course as listed below. The modules vary in length. An estimated time for completion is provided below:
- Introduction to clinical trials (Completion time: 60 minutes)
- Clinical trials regulation and legislation (Completion time: 30 minutes)
- National Clinical Trials Governance Framework (Completion time: 90 minutes)
- Assessing service providers to the Governance Framework (Completion time: 60 minutes)
Each module includes interactive content, knowledge check activities and ends with a quiz.
The course requires an 80% pass in the quiz to receive a completion certificate.
Clinical Trials Portal FAQs
Who can access the Clinical Trials Portal?
All HSOs and interested stakeholders (for example university staff and students, industry representatives, consumer representatives or state or territory government representatives) can register with the Commission to access the Clinical Trials Portal. Users will be required to provide a professional email domain to register.
Are site contributors able to register new users if there is no site administrator for the health service organisation?
Site contributors do not have any administrative rights so they cannot register other users on behalf of their HSO. If a HSO does not have a site administrator, all site contributors (general users) will have to register and be verified by the Commission to access the Clinical Trials Portal.
User accounts have one of the following levels of access:
- Basic access: users with basic access are only able to view and edit their own submissions and generate basic reports
- Clinical department access: users with clinical department access are able to view and access submissions from other users within the same clinical departments and generate reports at the clinical department level
- Health service organisation access: users with health service organisation access are able to view and access submissions from all users within the same organisation and generate reports at the clinical department level and for the whole organisation.
How secure is the data hosted on the Clinical Trials Portal?
The Clinical Trials Portal is hosted on a secure HealthIT system. All users will be verified either by the Commission or by the designated site administrator for the HSO before they can access the Clinical Trials Portal. All users can be confident that their data is hosted in a secure environment.
The Commission is committed to the protection of personal information in accordance with the Privacy Act 1988 (Privacy Act). The Commission aims to ensure that all personal information is managed in accordance with the Australian Privacy Principles (APPs) contained in the Privacy Act.
Further information can be found at the Commission's Privacy Policy.
Is any commercial-in-confidence information required for the operational metrics tool?
The operation metrics tool does not collect any commercial-in-confidence information. Commercial-in-confidence information relates to the clinical trial protocol, the investigator’s brochure, study materials, early study results and the investigational product.
The clinical trial information required for the operational metrics relates to the project title; the type of study; trial phase and sponsor type; time frame for human research ethics committee approval; local site authorisation and recruitment activity. The investment data section collects information on the expected income such as per patient payment. The financial information should be available in the clinical trial research agreement. Generally, this is the only information a trial coordinator has at hand to contribute to the organisation’s financial and business planning.
Which web-browsers can be used to access the Clinical Trials Portal?
To access the Clinical Trials Portal, users are recommended to use Chrome, Microsoft Edge or Firefox. Internet Explorer is no longer a supported system.
Is there any option to import data from existing clinical trials management systems into the operational metrics tool?
The current system allows for manual data entry only.
Can the self-assessment be completed at a clinical trial unit level?
It is important that clinical trial units become familiar with the actions in the NCTGF. This is because assessment to the NCTGF includes clinical trial units and ensures all actions in the NSQHS Standards have been met by the HSO.
HSOs may wish to nominate someone to coordinate the completion of the self-assessment tool for the organisation. It is intended that this person would coordinate a whole of organisation approach inclusive of the clinical trials work force, clinical and non-clinical managers, human resources, finance and the executive to review the organisation’s readiness to meet the actions in the NCTGF.
Fast facts on the NCTGF
What is a clinical trial?
Clinical trials provide early access to innovative treatments and interventions for patients, and improve the overall standard of medical care provided in Australian hospitals through the uptake of evidence into practice. Data suggest that improved outcomes for patients participating in clinical trials are due in part to the increased clinical surveillance a trial provides and greater clinician adherence with evidence-based care.
A clinical trial is any research study that prospectively assigns human participants or groups of humans to one or more health-related interventions to evaluate the effects on health outcomes.1 Clinical trials include but are not limited to:
- Surgical and medical treatments and procedures
- Experimental drugs and diagnostics2
- Biological products
- Medical devices
- Health-related service changes
- Health-related preventative strategies
- Health-related educational interventions.
All trials undergo rigorous evaluation to make sure that the intervention being tested is effective, safe and feasible for use more broadly in the community.
Clinical trials generate evidence to inform best-practice ways of providing care and treatment to patients. A thriving clinical trials environment is essential for a robust and self-improving healthcare system.
- World Health Organisation - http://www.who.int/topics/clinical_trials/en/
- In the Australian context, this includes novel treatments and interventions
Self–improving health system
What is clinical trial governance?
Governance is the set of relationships and functions established by a health service organisation or trial service between its state or territory department of health, governing body, executive, workforce, patients, consumers, and other stakeholders to ensure good clinical trial service provision.
Clinical trial service governance is an integrated component of the corporate and clinical governance of HSOs. It ensures that everyone, including frontline trial investigators undertaking clinical trials and members of governing bodies such as Boards, is accountable to patients and the community for assuring the delivery of clinical trials is of high quality, integrated into clinical care and continuously improving.
How the NCTGF will assist hospital administrators to drive efficiencies in clinical trial service provision
The cost of delays in trial start-up impact on clinical trial budgets and are frequently related to strategic and operational barriers. Improved organisational governance of trial services enables health administrators to oversight trial operations including (complex) contract negotiation (including financial schedules), use of national forms and standard contracts, determine work force arrangements and optimise organisational strategic planning to deliver clinical trial services. It is anticipated that, accreditation to the actions within the NCTGF will remove barriers to the following clinical trial processes.
- Trial site feasibility assessment
- Timely ethical and site-specific assessment approval timeframes
- Participant screening and recruitment activities
- Trial management
- Workforce planning and staff training
- Trial related financial management and reporting.
Systems and processes currently in place in health service organisations that underpin strategic planning to deliver clinical trial services include: organisational leadership; policies and procedures; risk management; incident management and open disclosure; workforce safety and quality training and infrastructure to deliver clinical trials.
The Commission has developed resources to support health services implement the NCTGF. Once implemented, health service administrators will have visibility of trial related activity within their organisation. Clinical trial operations will be measured and reported by trial; trial site; health service organisation and jurisdiction. Measures of operational efficiency will align with the National Aggregate Statistics (NAS) currently reported only at the jurisdictional level for public health organisations.
How accreditation against the actions in the NCTGF fits with current accreditation processes
HSOs accredited to the National Safety and Quality Health Service (NSQHS) Standards conducting clinical trials will be assessed against the actions in the NCTGF.
For HSOs that are already accredited to the NSQHS Standards, the accreditation process will remain unchanged. However, assessment against the actions in the NCTGF will be included.
HSOs will continue to select their accrediting agency, from among approved agencies, to assess their performance.
How the Commission supports the implementation of the NCTGF
The National Clinical Trials Governance Framework and User Guide provides detailed information and strategies to assist HSOs with implementing the improvements required by the NCTGF. Suggested strategies to meet the actions within the NCTGF, and examples of evidence an HSO might provide to demonstrate they have met the actions are also provided.
The Commission has developed resources (including a self-assessment tool, case studies, fact sheets, video tutorial and online operational metrics tool) to support implementation.
The Commission has a NSQHS Standards Advice Centre available for support and, if required, mediation between HSOs and accreditation agencies during assessment. Support will be provided by telephone 1800 304 056 and email accreditation@safetyandquality.gov.au.
Additional support will be provided by the Commission to health service organisations throughout the first assessment cycle.
What happens if the HSO does not meet all the actions in the NCTGF?
For the first assessment cycle, HSOs will be assessed using a maturity scale for each applicable action to provide an indicator of success. This approach recognises that HSOs are at different stages of maturity in implementation of the actions and provides HSOs with a unique quality improvement opportunity to implement safety and quality enhancements ahead of the transition to the NSQHS Standards rating system. It is expected that improvements should be achievable over a health services standard planning cycle of three years.
Figure 1: Maturity scale for health service organisation assessment
How will the Governance Framework impact investigator initiated-clinical trials?
One of the underpinning principles of health service provision is equity. HSOs are required to provide equitable access to the community as part of routine health care. The NCTGF extends this responsibility to clinical trials (both commercially sponsored and investigator-initiated). HSOs providing a clinical trials service are required to:
- Ensure patients and consumers can access to clinical trials in a timely manner
- Observe cultural safety, competence and respect in providing clinical trial services to meet the needs and priorities of First Nations peoples
- Provide appropriate resourcing to allow patients and consumers from different demographics, geographics and culturally diverse background to participate in all available trials within and across HSOs
- Maintain a skills and reputable clinical trials workforce in accordance with Action 1.20
The NCTGF also provides guidance for HSOs when they act as the trial sponsor. For investigator-initiated trials, the HSO or trial site that is the approving authority may also be the trial sponsor. In such cases, the governing body ensures that its overarching governance and quality management systems separates its responsibilities as a trial sponsor from its responsibilities as a trial site and ensures that the requirements of sponsorship can be met. For example, implementation of organisational strategies to meet Actions 1.7, 1.11, 1.12.
What is the role of clinical trial networks and Advanced Health Research and Translation Centres (AHRTCs) networks in supporting HSOs implement the NCTGF?
Clinical trial networks and ARHTCs could work with their member organisations to build capacity in a number of ways including, but not limited to: clinical trial education and training for the workforce and consumers; providing advice and support to trial investigators and researchers on good clinical trial practice relating to protocol development; data management and analytics; report writing; navigating ethical processes; participant recruitment and project management.
AHRTCs could also support HSOs in the review of clinical trial outcomes and contribute to guideline development and evidence translation into clinical practice.